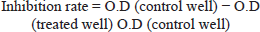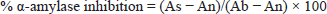Abstract
Background
Odontonema strictum is an herbaceous plant, indigenous to Central America and Central Africa, and has been used traditionally by the local community against various ailments. The current piece of work was carried out to evaluate the cytotoxic potential of O. strictum by using brine shrimp lethality and the in vitro cytotoxicity/viability assay (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide [MTT]), as well as the in vitro enzyme inhibitory potential by using alpha-amylase, protein kinase enzymes and phytochemical analysis of the different extracts of aerial parts of O. strictum.
Materials and Methods
Extracts of aerial parts of O. strictum were prepared through the successive extraction method using solvents with the order of polarity as n-hexane, dichloromethane, ethyl acetate, methanol and water. Cytotoxicity was assessed through brine shrimp lethality assay and in vitro cell viability assay (MTT), whereas enzyme inhibition potential was evaluated through Alpha-amylase and protein kinase assay. Phytochemical analysis was done by assessing the total phenolic and total flavonoid contents of both leaves and flowers.
Results
All the extracts exhibited various phytochemical and biological activities. Remarkable toxicity profiles against the brine shrimp lethality assay (LD50:11.5 µg/mL) were shown by the leaf extracts, whereas both leaves and flowers exhibited promising anti-proliferative activity, assessed by the MTT assay. Significant alpha-amylase and protein kinase inhibition potential were manifested by leave extracts with IC50:195.6 µg/mL and minimum inhibitory concentration (MIC): 16.3 µg/mL respectively. The phytochemical analysis of the plant showed that it was sufficiently rich in phenolic compounds with a total phenolic content and total flavonoid content (96.8 µg GAE/mg and 65.1 µg QE/mg, respectively).
Conclusion
It is plausible to believe that the cytotoxic and enzymatic activity of the aerial parts of O. strictum aerial parts might be, at least partly, to the presence of phenolic chemicals and could be a possible source of cytotoxic agent and an enzyme inhibitor.
Introduction
Odontonema strictum is a herbaceous plant, indigenous to Central America and Central Africa. It is commonly known as fire spike/scarlet flame and can be grown in a variety of soil conditions. If grown in a container, the plant will only reach a height of one and a half metres. The waxy flowers have 12-inch spikes and bloom in the fall, luring hummingbirds and butterflies to their native land. The genus Odontonema belongs to the family Acanthaceae. The family contains more than 4,000 species, which are mostly herbs, shrubs or climbers and rarely trees. 1 The family is rich in secondary metabolites that have been used traditionally by the local community against various ailments such as anti-hypertensive properties. 2 The phytochemical family has many biological properties such as antiplasmodial, anti-HIV, 3 antihypertensive, 4 antifungal 5 and anticancer. 6 Phytochemical analysis of the leaves shows that they are enriched with flavonoids, tannins and saponins.
The brine shrimp lethality assay is used to conduct a preliminary toxicity evaluation on sample extracts. The assay is based on the sample’s ability to destroy Artemia nauplii. 7 It is employed as a general toxicity indicator and as a cytotoxicity indicator for the initial assessment of samples’ antitumor potential. 8 Cytotoxicity is quantified using the LD50 value, which is the lowest concentration of the sample that kills or inhibits 50% of the nauplii population. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) is a colorimetric test for reduction that is most frequently employed in high-throughput screening and for determining the metabolic activity of cells. The assay is based on the idea that living cells convert MTT to its insoluble, purple-colored formazan product with the aid of NADH. Non-viable cells, on the other hand, lack this capability. As a result, the amount of formazan (color) shows the viability of the cells. The conversion, however, is time-dependent. It is, therefore, the assay that is used to determine the cytotoxic effect of chemicals or extracts. 9
Our body contains many naturally occurring enzymes. Enzyme inhibition can also help to cure certain diseased states. So, it is important to focus on the enzyme inhibition capacity of medicinal plants. 10 Enzyme inhibition assays are in vitro assays that help in the assessment of the enzyme-inhibiting potential of the extracts. As enzymes are involved in almost all of the physiological as well as pathological states of the body, inhibiting such enzymes can help in the reversal of diseased states.
The phenolic group of compounds makes up one of the most common plant metabolites and can be found in many food plants. Antioxidant activity is one of the many notable properties of phenolic compounds. 11 This family of chemical compounds has anti-inflammatory, anti-apoptotic, anti-atherosclerosis and anti-angiogenic activities, among other biological qualities. 12 Promising phenolic content and antioxidant activity have been found in the genus. 13 It is, therefore, this study was carried out to evaluate the cytotoxic potential of O. strictum by using brine shrimp lethality and the in vitro MTT cytotoxicity/viability assay, as well as the in vitro enzyme inhibitory potential by using alpha-amylase and protein kinase enzymes phytochemical analysis of the different extracts of aerial parts of O. strictum.
Materials and Methods
Reagents and Chemicals
All chemicals (Doxorubicin, Acarbose, Surfactin, Folin-Ciocalteu reagent, Gallic acid, aluminium chloride) were purchased from Sigma-Aldrich (Dorset, UK). Solvents (DMSO) were obtained from Fischer Scientific (Loughborough, UK). All cell culture reagents (MTT dye, fetal bovine serum) were obtained from Biosera (Nauaille, France).
Collection of Plant Material
Leaves and flowers of O. strictum were collected in March 2017, from the “Traditional Pharmacopeia Medicine and Pharmacy department garden” in Ouagadougou (Burkina Faso), with GPS coordinates: 12°- 22′ 9.67″ N, 01°- 29′ 05.43″ W. Dr. Amadé OUEDRAOGO made the plant identification at Joseph KI-ZERBO University. A voucher specimen with the identifier N° 6900 was deposited in the Joseph KI-ZERBO University Herbarium (Ouagadougou, Burkina Faso). O. strictum flowers were meticulously cleansed after harvesting before being lyophilized and pulverized into a fine powder in a blender.
Extraction and Preparation of Samples
The successive extraction method was carried out using solvents with the order of polarity as; n-hexane (FON), dichloromethane (FOD), ethyl acetate (FOA), methanol (FOM), and water (FOW). The powdered flowers and leaves of O. strictum (20 g) were macerated successively in 300 mL of solvents such that the plant residue was dried at room temperature and macerated in the next solvent and the filtrate was concentrated using a rotary vacuum evaporator. 14
Cytotoxicity: Brine Shrimp Lethality Assay
The cytotoxic effect was assessed using a modified brine shrimp lethality assay. 14 A 20 mg/mL stock solution was made by dissolving 20 mg of extract in 1 mL DMSO. It was tested at 100, 50, 25, and 12.5 (µg/mL). About 38 g of sea salt and 6 mg of dried yeast were dissolved in 1 L distilled water while stirring. A magnetic stirrer aerated it for 2 h. The pH was kept at 7. An egg-hatching tank with two small compartments separated by a perforated wall was used to hatch brine shrimp eggs. The tank held fake seawater. The huge compartment was covered with aluminium foil, and the little section was lit. To incubate eggs, pour them into a big container. The phototropic nauplii were extracted using a Pasteur pipette and put into a beaker. Doxorubicin (4 mg/mL) was employed as a positive control at concentrations of 100, 50, 25, and 12.5 (µg/mL). The negative controls were DMSO, nauplii, and seawater. The 96-well plates were filled with 10 nauplii and 300 L seawater from each sample concentration. For 24 hrs, the plates were inverted to allow oxygen flow. A water dish was placed inside the incubator to keep it damp. Dead nauplii were counted in each well after 24 h of incubation, and the LD50 was computed using able curve software.
In vitro MTT Assay
MTT assay was performed for the in vitro screening of antitumor activity of the extracts, that is, to determine the cell viability.
15
The assay was repeated in triplicate. The human colorectal carcinoma cell line was grown in 24-well plates containing McCoy’s medium, supplemented with antibiotics (1%) and fetal bovine serum (10%). The cell suspension (105 cells/mL) was then seeded in wells and incubated for 48 h at 37°C. For the formation of a confluent monolayer, a 5% CO2 incubator was used. The cells were then exposed to different concentrations of extracts. The cell control was also maintained throughout the protocol, and optical density was measured using a microplate reader at 595 nm with, 655 nm as a reference. The IC50 value of the fractions was also calculated. The growth inhibition rate was calculated by the following equation:
Enzyme Inhibitory Assay: Alpha-amylase Inhibition
Starch-iodine assay with slight modification has used the determination of the inhibition capacity of the extracts against alpha-amylase.
16
Plant extract (4 mg) was dissolved in 1 mL of DMSO with a final concentration of 4 mg/mL. Acarbose (5–200 µg/mL) was employed as a positive control. Blank was prepared using equal quantities of a buffer as a substitute for the amylase enzyme and extracts. A volume of 10 µL of plant extracts along with 15 µL phosphate buffer, 40 µL starch solution and 25 µL alpha-amylase enzyme were added to the microplate and incubated for 30 min at 50°C. In order to stop the reaction, 20 µL of HCl was introduced, followed by 90 µL iodine reagent. Absorbance was measured at 540 nm after color development. The activity was performed in triplicate and was stated as percentage inhibition of the enzyme per milligram of the extract, calculated as follows:
where,
Protein Kinase Inhibition
The assay was performed in triplicate in order to observe hyphae formation. 17 Purified isolates of Streptomyces 85E strain were used. The concentration of plant extract was such that 20 mg of extract was dissolved in 1 mL of DMSO (20 mg/mL). Surfactin was used as a positive control at a concentration of 1 mg/mL. DMSO was used as a negative control. A bacterial lawn was formed by spreading the mycelia fragments (spores) on the ISP4 medium containing sterile plates. About 5 µL of the test extract was loaded on the sterile filter paper discs and was directly applied to the plates. This was followed by incubation at 30°C for a period of 72 h (optimum time for hyphae formation). The results were interpreted as bald zones of inhibition.
Phytochemical Analysis
Total Phenolic Content
The total phenolic content of the extracts was determined using a Folin-Ciocalteu reagent with a slight modification in the procedure by Astill et al. 18 The stock solution of extract was prepared by dissolving 4 mg of crude extract in 1 mL of DMSO, that is 4 mg/mL. Gallic acid at a concentration of 5 to 25 µg/mL was used as a positive control. The plant extracts, at a volume of 20 µL, were transferred into the 96-well plates. This was followed by the addition of 90 µL of Folin-Ciocalteu reagent. After incubation of the plate for 5 min, 90 µL of sodium carbonate was introduced into the mixture. The absorbance of the reaction mixture was taken at 630 nm using a microplate reader. The assay was triplicated and results were expressed as microgram gallic acid equivalent to per milligram of extract (µg GAE/mg extract).
Total Flavonoid Content
The total flavonoid content of the extracts was determined using the colorimetric aluminium chloride method with a slight modification in the procedure Almajano et al. 19 The stock solution of extract was prepared by dissolving 4 mg of crude extract in 1 mL of DMSO, that is 4 mg/mL. Quercetin (at final concentrations of 2.5, 5, 10, 15, 20 and 40 µg/mL) was taken as standard for the assay. Test samples 20 µL and 10 µL of aluminium chloride and potassium acetate were added to 96-well plates along with 160 µL of distilled water. The mixture was kept for 30 min at room temperature. Absorbance was then measured at 415 nm using a plate reader. The assay was performed thrice and results were expressed as microgram quercetin equivalent to per milligram of extract (µg QE/mg extract).
Statistical Analysis
The data were presented as mean values with standard error of the mean from three parallel repetitions. The graph was created using nonlinear regression and Microsoft Excel version 2013. Analysis of variance with Tukey’s range test was used to differentiate the means at a confidence level of p ≤ 0.05.
Results and Discussion
Cytotoxicity: Brine Shrimp Lethality Assay
The LD50 value calculated in the case of leaves was lowest for FO fraction, being only 24.3 µg/mL while the highest was for FOW being only 55.6 µg/mL summarized in Table 1. For the extracts of flowers, OSN showed an LD50 value of 22.5 µg/mL while OSW, OSM, OSA and OSD calculated value was 11.5 µg/mL. The brine shrimp assay exhibits the lethal potential of the extracts. The LD50 value of the different extracts of leaves and flowers was determined. The results indicated that the flowers of O. strictum are comparatively toxic, with an LD50 range of 11.5 µg/mL to 22.5 µg/mL. The LD50 value decreased as the polarity was increased. The results are in correspondence to the previously conducted studies in which the lethal dose of the ethanol fraction was 8.12 µg/mL when compared to the standard vincristine, which exhibited a value of 6.76 µg/mL.20, 21 In the case of the extracts of leaves, the LD50 value increased as the polarity of the solvent was increased. The range of LD50 values for leaves was 24.3 µg/mL to 55.6 µg/mL. The results are consistent with previously conducted results on different plants such as Aloe vera whose LD50 value was calculated as 3.59 mg/mL for the ether extract.22, 23 In comparison to the studies, our flower extracts are toxic whereas the leaf extracts are moderately toxic.
Brine Shrimp Lethality Assay Potential of Aerial Parts of O. strictum.
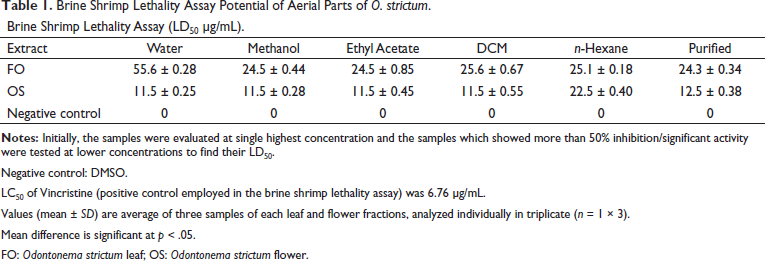
Negative control: DMSO.
LC50 of Vincristine (positive control employed in the brine shrimp lethality assay) was 6.76 µg/mL.
Values (mean ± SD) are average of three samples of each leaf and flower fractions, analyzed individually in triplicate (n = 1 × 3).
Mean difference is significant at p < 0.05.
FO: Odontonema strictum leaf; OS: Odontonema strictum flower.
In vitro MTT Assay
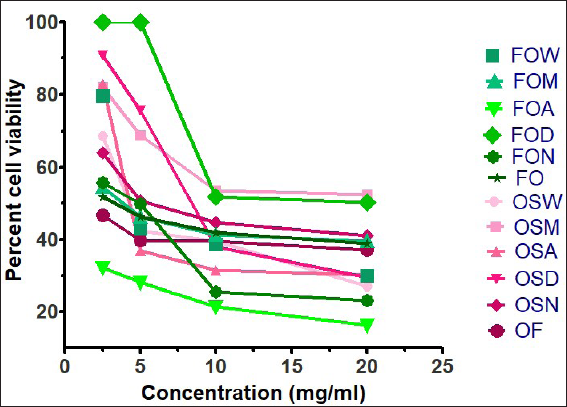
Ethyl acetate extract of leaves (FOA) showed the lowest percent cell viability of 32.26% at 2.5 mg/mL whereas the highest percentage of cell viability was given by FOD of 100%, summarized in Figure 1. The purified fraction of flower (OF) exhibited the lowest percent cell viability of 46.62% at 2.5 mg/mL and the highest viability was shown by OSD of 90.72%. MTT assay is a cytotoxic assay that is used to determine cell viability. 24 Programmed cell death or apoptosis is a major strategy to kill or eliminate cancerous cells. Apoptosis acts as a protective mechanism that eliminates the damaged cells before they develop into malignant cells. 25 Among the different solvent extracts of flower, the OF exhibited the lowest percentage of cell viability of 46.62% at 2.5 mg/mL and the highest viability was shown by OSD of 90.72% using the MTT assay against the HCT-116 cell line. Our results are different from a previously conducted study on the ethyl acetate fraction of Aegle tamilnadensis where a dose-dependent inhibition of the cells was observed. The study is consistent with our results of leave extracts where the FOA showed the lowest percentage of cell viability of 32.26% at 2.5 mg/mL whereas the highest percentage of cell viability was given by FOD 100%. This result is different from a study in which the methanolic fraction of Plumbago zeylanica exhibited weak anti-proliferative property, with a percentage cytotoxicity of 42.1%. 26
Cytotoxic Potential of Aerial Parts of O. strictum.
Alpha-amylase Inhibition Assay
In the case of leaves, the highest inhibition of the enzyme was depicted by the FOA (66.76%), whereas the least inhibition was shown by the FOD (11.7%). For the flower extracts, OSA extract showed the highest inhibition of 48.34% while OSM showed the lowest inhibition of 12.2%, results are exhibited in Table 2. The IC50 value for the FOA extract was calculated to be 195.6 µg/mL. The alpha-amylase inhibition property of the extracts was determined in order to identify the part of a plant and its fraction that can inhibit the enzyme. No significant results were shown by the different extracts of a flower. In the case of leaves, the results were also non-significant except for the FOA, which showed an IC50 value of 195.6 µg/mL. The results are in correspondence to a study conducted by Sallam and Galala, 27 in which the ethyl acetate fraction of Gymnocarpos decandrus showed the most significant and highest inhibition of the alpha-amylase enzyme by 87.6%. In another study conducted by Wickramaratne, 28 FOA of Adenanthera pavonina inhibited the enzyme with an IC50 value of 59.93 µg/mL. The alpha-amylase inhibition property of our extract was comparatively weak.
Alpha-amylase Inhibition and Protein Kinase Inhibition Potential of Aerial Parts of O. strictum.
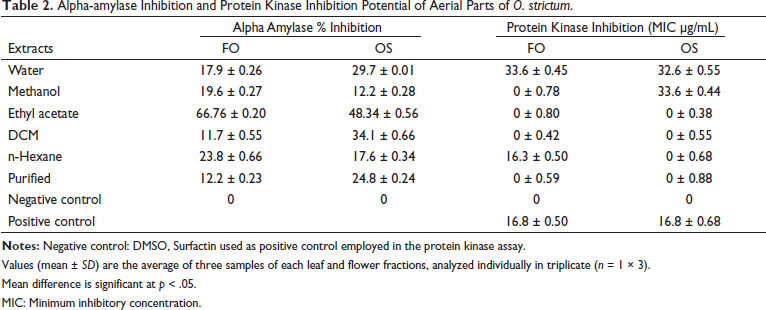
Values (mean ± SD) are the average of three samples of each leaf and flower fractions, analyzed individually in triplicate (n = 1 × 3).
Mean difference is significant at p < 0.05.
MIC: Minimum inhibitory concentration.
Protein Kinase Inhibition Assay
Only two extracts of leaf inhibited the enzyme, and results are shown in Table 2. FON showed inhibition of 16 mm whereas FOW showed inhibition of 12 mm. Similarly, only two flower extracts showed protein kinase inhibition activity. OSW with a zone of inhibition of 14 mm and OSM with a zone of 13 mm. Surfactin which was used as the standard exhibited a zone of 17 mm. The MIC for the values above 15 mm of inhibition was calculated. MIC calculated for FON was 16.3 µg/mL, whereas, for Surfactin, it was 16.8 µg/mL. For the formation of aerial hyphae in Streptomyces, protein kinase plays a significant role. 14 Inhibitors of kinases in Streptomyces give rise to bald colonies indicating aerial mycelia inhibition.29–30 Among all the fractions of leaves and flowers, only the n-hexane extract of leaves (FON) showed potential and promising results with a bald zone of inhibition of 16 mm. The MIC value calculated for this extract was 16.3 µg/mL when compared to the standard Surfactin, whose MIC value was 16.8 µg/mL. Our results show that the n-hexane extract of leaves can be used as an agent for eukaryotic protein kinase inhibition as other similar studies have also indicated potential inhibitory agents. 29 The results obtained showed promising findings.
Phytochemical Analysis
Total Phenolic Content
The highest phenolic content was present in the purified methanolic fraction of the flowers (OF, 96.8 µg GAE/mg). The lowest concentration was present in the OSD, being only 30.8 µg GAE/mg, summarized in Table 3. Among the extracts of leaves, the highest phenolic content was shown by the FOW was 67.4 µg GAE/mg and the lowest value by the FOD was 10 µg GAE/mg. Collectively, the highest phenolic content was found in the purified methanolic fraction of the flower while the lowest was present in the dichloromethane extract of leaves. O. strictum constitutes an excellent source of bioactive compounds, especially phenolic components. The phenolic contents reported in the current study for leave extracts are highest in the most polar extract, that is FOW, with a value of 67.4 µg GAE/mg. In the case of the O. strictum flowers, the purified methanolic fraction (OF) showed the highest phenolic content of 96.8 µg GAE/mg. Current findings are in agreement with a study conducted on the leaves of guava, where the polar extracts constituted the highest phenolic content 31 such that the total flavonoid content of the water fraction of leaves was 148 mg GAE/g of extract. Similarly, for the methanolic extract, the phenolic content was 91 mg GAE/g of extract. Also, the results are consistent with the previous studies conducted. 32 Therefore, the polar extracts of plants contain more phenolic compounds and hence greater total phenolic content.
Total Phenolic Content (mg GAE/g) and Total Flavonoid Content (mg QE/g) of Aerial Parts of O. strictum.
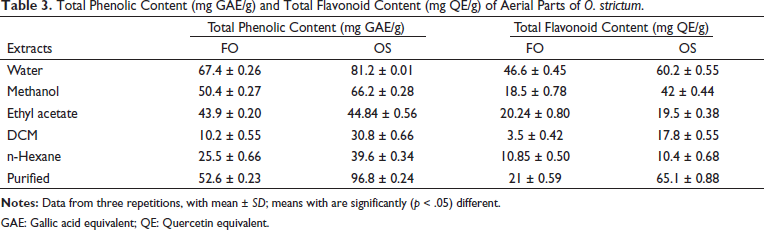
GAE: Gallic acid equivalent; QE: Quercetin equivalent.
Total Flavonoid Content
For the flower extracts, the highest flavonoid value was obtained from the purified methanolic fraction (OF) 65.1 µg QE/mg and the lowest was observed in the n-hexane extract (OSN) 10.4 µg QE/mg, summarized in Table 3. In the case of leaf extracts, the highest flavonoid content was found in the FOW 46.6 µg QE/mg, and the lowest was observed in the FOD 3.5 µg QE/mg. Collectively, the highest value for flavonoid content was found in the purified methanolic fraction of flowers whereas the lowest was observed in the dichloromethane extract of leaves. Similar to the total phenolic content results, the FOW and the OF showed the highest total flavonoid content of 46.6 µg QE/mg and 65.1 µg QE/mg, respectively. The results are consistent with the previously conducted studies using polar solvents for extraction where the methanolic fraction exhibited a total flavonoid content of 10.28 mg QE/g. 33 A study conducted by Nyirenda et al. 34 reported that phenolic compounds are more soluble in polar solvents rather than organic solvents. High phenolic concentration in the plant protects its chloroplast from the harmful effects of ultraviolet radiation. 35
Conclusion
The outcomes of the current investigation support the assumption that the use of diverse solvent extracts truly retrieves the whole phytochemical and biological profiling of plants. In the current study the cytotoxic potential, enzyme inhibition ability and phytochemical evaluation of different solvent extracts of leaves and flowers of O. strictum were carried out. All the extracts showed various phytochemical and biological activities. Different solvents extract showed a considerable level of total phenolic content and total flavonoid content of both leaves and flowers. It is plausible to conclude that the cytotoxic and enzymatic activity of O. strictum aerial parts is attributed, at least partly, to the presence of phenolic compounds and could be a potential course of enzyme inhibitor. The current study could be expanded to target the isolation of secondary metabolites responsible for the observed activities.
Abbreviations
Footnotes
Acknowledgment
The authors would like to acknowledge the editing support of Dr. Khezar Hayat, Lecturer, Institute of Pharmaceutical Sciences, University of Veterinary and Animal Sciences, Lahore, Pakistan.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Summary
OS leave extracts exhibited remarkable toxicity profile against the brine shrimp lethality assay.
OS leave and flower extracts leaves and flowers exhibited promising anti-proliferative activity, assessed by the MTT assay.
OS leave extract manifested significant alpha amylase and protein kinase inhibition potential.
OS extract showed sufficiently rich in phenolic compounds.