Abstract
Background
Tuberculous meningitis (TBM) is a severe form of tuberculosis (TB) that affects the meninges, frequently causing major neurological problems. Timely diagnosis and care are crucial to avoid unfavourable consequences, particularly in vulnerable populations.
Purpose
This case report aims to highlight the diagnostic problems and management complications of TBM, especially when combined with rare neurological events such as acute ischaemic stroke (AIS).
Methods
An 84-year-old female presented with disorientation, seizures, aphasia and hemiparesis. The initial imaging revealed normotensive hydrocephalus and bilateral delta activity on the electroencephalogram (EEG). Despite anti-fungal treatment, CT imaging revealed widespread inflammatory alterations. Sputum and cerebrospinal fluid (CSF) tests were done and revealed the presence of Mycobacterium tuberculosis. MRI indicated several acute lesions, which are symptomatic of TBM.
Results
The patient developed AIS, a relatively rare complication of TBM. Treatment was initiated with a combination of anti-TB drugs and corticosteroids. While corticosteroids reduced inflammation and mortality risk, their potential to contribute to long-term disability was noted as an area of concern.
Conclusion
This case underscores the critical role of MRI in the early detection of TBM-related complications. Although corticosteroids improve short-term outcomes, their impact on long-term disability necessitates further research. Effective management strategies tailored to vulnerable populations are essential to improve the prognosis of TBM patients.
Introduction
Tuberculosis (TB) is a life-threatening infection caused by the bacterium Mycobacterium tuberculosis. While it mainly targets the lungs, TB can also affect other organs such as the kidneys, spine and brain. Although it is both preventable and treatable, TB continues to be a major global health issue, particularly in developing countries. It is a primary infection transmitted via airborne aerosol droplets. Airborne nuclei released when an infected individual coughs or sneezes pose a significant risk of spreading the infection, particularly during close contact. The infection is often asymptomatic initially due to the innate and cellular immune response. However, it becomes active when the immune system is compromised. TB has symptoms like fever, weight loss, night sweats, malaise, non-productive cough, etc. 1 It can present with some complications such as hydrocephalus, cranial nerve (CN) palsies, Visual impairments, Stroke (ischaemic or haemorrhagic), seizures and epilepsy, sero-fibrinous pleural effusion, epituberculosis, bronchial compression or bronchial perforation by lymph nodes. Complications such as stroke and seizures primarily occur in tuberculous meningitis (TBM), affecting 15%–57% of patients, especially in advanced stages. 2 Tremors and abnormal movements are more common in children, and conditions such as monoclonus and cerebral dysfunction may also occur. Additionally, the majority of strokes can be asymptomatic. There are various types of strokes; for instance, most strokes associated with TBM are typically multiple, bilateral and located in the basal ganglia, particularly in the ‘tubercular zone’, which includes the caudate nucleus, anterior thalamus, anterior limb and genu of the internal capsule. Stroke is a serious complication of TBM and might contribute to long-term disability.2, 3 TB continues to be a global health challenge, with approximately 10.4 million cases and 1.8 million deaths reported in 2015. TBM, the deadliest and most debilitating form of TB, accounts for over 100,000 new cases annually. There are several studies regarding the location of the infarction due to TBM; for instance, a study done by F.Y. Hsieh and colleagues reported that 75% of infarctions occurred in the ‘TB zone’, which is supplied by medial striate and thalamoperforating arteries. 4 Only 11% occurred in the ‘IS zone’ supplied by lateral striate, anterior choroidal and thalamogeniculate arteries whereas the research conducted by Mei-Ling Sharon Tai which was published in scientific reports contradicts by stating that infarcts that infarcts involved mainly perforators and cortical branches, rather than ‘TB zone’ versus ‘IS zone’.4, 5 They also mentioned that normally, during atherosclerotic infarction, there is a complete blockage of the major intracranial arteries, but in the case of TBM-induced infarction, there is an involvement of perforators and terminal cortical branches, which helps to distinguish between atherosclerosis and TBM. 5 For early prediction of cerebral infarction in TBM, there is a significant increase in the intracranial pressure. The golden standard for the detection of infarction in TBM is MRI. 6
Method
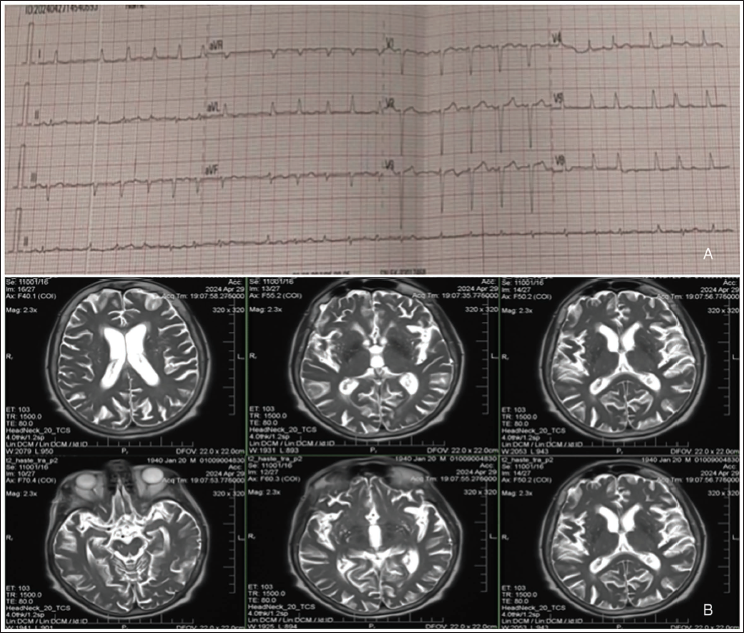
An 84-year-old woman presented with disorientation, a single episode of loss of consciousness, and three seizures per month. Her medical history included arterial hypertension, rheumatoid arthritis (four years on methylprednisolone), and Type 2 diabetes (treated with metformin 500 mg daily). After admission, an ECG revealed atrial fibrillation. An electroencephalogram (EEG) indicated bilateral delta activity in the right frontal lobes, resulting in a diagnosis of secondary epilepsy owing to dementia, for which oral levetiracetam was prescribed. Atrial fibrillation was observed after admission, as shown in Figure 1.
Initial Neuroimaging of the Patient Suggests Acute Infarction. Additionally, an ECG Taken Upon Admission Shows Atrial Fibrillation. (A) ECG Showing Atrial Fibrillation. (B) Axial Brain MRI With Contrast Showing Small, Multiple Acute Lesions in Both Hemispheres and Near the Cortex.
On the second day of hospitalisation, she developed a 39°C fever. Although her CBC was normal, her CRP levels were high. A CT scan of the chest indicated diffuse inflammatory alterations in the upper lobes, indicating a fungal or tuberculous cause. Fluconazole (100 mg/day) was administered to treat the fungal infection. However, sputum and cerebrospinal fluid (CSF) testing revealed the presence of M. tuberculosis.
The CSF study showed a cytosis of 18, a specific gravity of 1.015, a pH of 7.5, high protein levels (0.591 g/L) and low glucose levels (1.71 g/L). A CT scan indicated normotensive hydrocephalus, whereas brain imaging, including MRI with contrast, revealed tiny acute lesions in both hemispheres close to the cortex. Axial brain MRI with contrast revealed many tiny hyperintense lesions on T2-weighted and FLAIR sequences, primarily in the cortical and subcortical regions of both cerebral hemispheres, indicating acute ischemia infarctions caused by TB meningitis-related vasculitis. Increased signal intensity was observed in the periventricular white matter, indicating possible ischaemic changes, as well as hyperintense areas in the basal ganglia and thalamic regions, both of which are commonly affected in tuberculous vasculopathy. Post-contrast sequences revealed leptomeningeal growth, particularly around the basal cisterns, Sylvian fissures and interpeduncular cisterns, confirming the diagnosis of TBM. Mild ventricular enlargement was seen, indicating communicating hydrocephalus, a known TBM complication induced by CSF fluid obstruction. Furthermore, perilesional cerebral oedema near the infarcts indicated an ongoing inflammatory process. Vascular changes, such as possible narrowing of minor arteries, were also identified, notably in the middle cerebral artery (MCA) region, which is consistent with TBM-related vasculitis. These MRI findings strongly supported the presence of acute ischaemic infarctions in the context of TB meningitis, necessitating close monitoring for infarction progression, increased hydrocephalus and secondary infections. Throughout her hospitalisation, the patient received a comprehensive medical regimen that addressed both his underlying disorders and acute problems. Digoxin (0.25 mg/mL) and metoprolol (25 mg) were prescribed to treat atrial fibrillation and hypertension, respectively. Antibiotic therapy for bacterial and fungal infections consisted of cefepime (1 g) and fluconazole (100 mg/50 mL IV). The first anti-TB medications were isoniazid (300 mg daily) and rifampicin (300 mg twice daily). To prevent thromboembolic complications, anticoagulation therapy included the administration of Fraxiparine (0.4 mL subcutaneously). Pantoprazole (40 mg IV) and omeprazole (20 mg IV) provided gastrointestinal protection. Ringer’s solution (500 mL), sodium chloride (0.9% IV), potassium chloride (4% solution) and glycerine suppositories were used to control electrolytes, fluids and bowel function. To treat pain and symptoms, 2 mL of Metamizole (50% Analgin) was combined with 1 mL of Diphenhydramine (1% solution) and 2 mL of Lidocaine. Dexamethasone (4 mg/mL IV) was administered as an anti-inflammatory. Cardiomagnil (150 mg) was added for cardiovascular protection, while Dominal (40 mg/50 mL IV) was administered to alleviate symptoms. This regimen was intended to treat the patient’s acute ischaemic stroke (AIS), TB meningitis, atrial fibrillation and other hospital-related problems.
Results
The patient continued to experience febrile episodes despite antifungal treatment, and although the CRP levels were still high, they were lower than when they were initially measured. Antifungal therapy was stopped, and anti-TB treatment was started after TB was confirmed.
The patient experienced deteriorating left hemiparesis and aphasia before beginning the anti-TB treatment. Multiple acute ischaemic lesions, an uncommon consequence of TBM, were confirmed by imaging. The aggressive character of TBM and its propensity for serious neurological consequences are shown by this trend. Although long-term disability remained a concern, the primary goal of treatment with the anti-TB regimen and corticosteroids was to reduce mortality risks. Upon discharge, the patient received a comprehensive pharmacological regimen to address his various comorbidities and ongoing treatment needs. His treatment regimen comprises Pantoprazole (40 mg IV) for gastrointestinal protection, Rivaroxaban (20 mg) for anticoagulation and Metformin (500 mg controlled-release) for glycaemic control. Dexamethasone (4 mg/mL IV/IM) was used to alleviate inflammation and probable TB complications. Levetiracetam (500 mg) was given to manage seizures, while Meropenem (IV) was given to treat infections. Metoprolol (52 mg) was given for cardiovascular support, while Rifampicin (20 mg) and Isoniazid (200 mg) formed the basis of TB treatment. Ademetionine (540 mg enteric-coated tablets) was also given to help with hepatic function, and Sodium Ferrous Citrate (Soferi, IV) was used to treat anaemia. The patient was initially admitted to the hospital in an unstable condition. However, with appropriate treatment and supportive care during his stay, his condition improved. After stabilisation, he was transferred to the National Centre for Tuberculosis and Lung Diseases for specialised TB care.
Discussion
TBM is where the pathogen Mycobacteria tuberculosis infects the cranial meninges, leading to symptoms like fever and headache and progressing to serious neurological impairments, with one of the rare but fatal complications being AIS. It most commonly affects people from resource-poor regions and immunocompromised individuals. TB in the central nervous system can be classified into three clinical forms. These are TBM, Intracranial tuberculoma and Spinal tuberculous arachnoiditis. TBM is the most common form in the West, which shows symptoms including fever, headache, malaise, altered mentation and focal neurologic deficits. It may present with acute to chronic meningitis syndrome. Intracranial tuberculoma is marked by the formation of tuberculous granulomas as the primary lesion in the brain. This may lead to a series of symptoms like headaches, seizures and focal neurologic deficits. Spinal Arachnoiditis is identified by the inflammation of the arachnoid mater in the spinal cord, which leads to certain symptoms like back pain, neck pain, radicular pain and even neurological deficits, which depend on the level of spinal involvement. 2
In patients who already have chronic conditions like diabetes, it can act as a potential risk factor by acting as a link between TB and coronary heart diseases and further progress to atherosclerosis and other cardiovascular diseases. 2
A small subset of patients present without the characteristic prodrome and subacute progression. In other patients, atypically, varied presentations of TBM are observed, such as slowly progressive dementia with changes in personality, social withdrawal and memory deficits. In Acute Rapidly Progressive Meningitis, patients show symptoms that are very similar and almost indistinguishable from pyogenic bacterial infections. As mentioned above, Focal neurological deficits include CN palsies, hemiparesis and seizures. In some cases, Hydrocephalus is also observed with symptoms such as headache, papilledema, diplopia (double vision) and visual disturbances. 6 An Encephalitic syndrome has been described to occur mostly in children and occasionally in adults, which present with stupor, coma and convulsions, but no classic meningeal signs or CSF abnormalities are observed. Patients also present with other neurological signs, like Basal meningitis enhancement, which occurs in about 38% of the cases. It is strongly suggestive of TBM when combined with hydrocephalus, which occurs in approximately 15%–30% of patients. Cerebral infarcts, which are associated with cerebral vasculitis, a catastrophic complication of TBM, increase the risk of infarcts in the basal ganglia. An indicator of inflammation and damage to the brain tissue is seen through cerebral oedema and infarction. 7
The inflammatory response in TBM leads to the formation of a thick exudate in the base of the brain. This particularly affects the optic chiasm, pons and cerebellum. This can encase and cause inflammation of the blood vessels, resulting in cerebral vasculitis. This inflammation mainly involves small and medium-sized arteries, which leads to a granulomatous formation and adventitial inflammation. This process causes reactive intimal proliferation, which can occlude the vessel and cause infarction in the supplied brain regions. The MCA territory is frequently affected due to its anatomical location at the base of the brain, where the inflammatory response is the most intense. 8 Cerebral vasculitis in TBM can lead to vessel constriction, spasm, thrombosis and occlusion, resulting in multiple small, bilateral infarcts, which are often located in the periventricular regions, basal ganglia, thalamus and internal capsule. These infarcts can cause stroke, affecting various brain regions, including the cerebral cortex, basal ganglia, pons and cerebellum. 9 Moreover, the inflammatory exudate can also obstruct CSF flow, leading to hydrocephalus, which may be communicating or non-communicating. These immune responses contribute to the complications and chronic sequelae associated with TBM. In some cases, cerebral vasculitis in TBM may result from immunologic injury, where the deposition of tuberculoprotein immune complexes in small vessel walls leads to inflammation. This mechanism is similar to how other infections, such as hepatitis B and Epstein–Barr virus, can cause vasculitis. Understanding these inflammatory processes is crucial in the management of TBM, which significantly contributes to the disease morbidity and mortality.9, 10
Several biomarkers and clinical predictors have been identified among patients with TBM that increase the risk of AIS. Elevated levels of inflammatory markers, such as C-reactive protein and pro-inflammatory cytokines like interleukin-6 (IL-6), are associated with a higher risk of AIS in TBM patients. 11 These markers reflect the elevated inflammatory state contributing to vascular injury and subsequent ischaemic events. The presence of vascular risk factors (VAFs) like hypertension, diabetes mellitus and dyslipidaemia further increases the likelihood of AIS in TBM patients. When combined with elevated inflammatory markers, these VAFs significantly enhance the predictive accuracy for the occurrence of AIS. Neuroimaging data can also be used as crucial predictions. Meningeal enlargement and hydrocephalus found in imaging investigations are closely linked to an elevated risk of AIS in TBM patients. These imaging markers show severe meningeal inflammation and CSF flow blockage, which both lead to cerebral ischemia. Additionally, clinical variables such as advanced age and increased disease severity at presentation are independent predictors of AIS in TBM patients. Older patients and those with more severe illness symptoms are more likely to experience ischaemia problems. Identifying these biomarkers and clinical indicators is critical for risk classification and implementation of tailored therapies for preventing AIS in TBM patients.11–13
Age, disease severity, meningeal enhancement and hydrocephalus were identified as independent predictors of AIS in TBM patients. 11 Inflammation markers, when combined with VAFs, improve the prediction of AIS in TBM patients. The key inflammation-associated neuroimaging markers are found to be meningeal enhancement and hydrocephalus.11, 12
There is a poor correlation between neurological findings and focal lesions observed in imaging studies which are associated with TBM and their prognosis. CT scans are used for detecting hydrocephalus, basal meningeal enhancement and cerebral infarcts, while MRI is used for evaluating brain stem, midbrain and basal ganglia lesions, especially in children. Laboratory studies may also include CSF findings in CSF protein levels, WBC counts and positive bacilli culture.2, 7
For a better understanding, the symptoms of TBM are classified into three stages, as mentioned in Table 1. If we compare the consciousness state, stage 1 is when the patient is conscious and rational, stage 2 is when the patient shows slight lethargy and appears confused and stage 3 is the advanced stage of the disease. 7 In general, thalamic contractures are caused by lesions in the posterior thalamus, whereas in this case, they were seated in the anterior nuclei. 2
Symptoms of TBM in Different Stages. 7

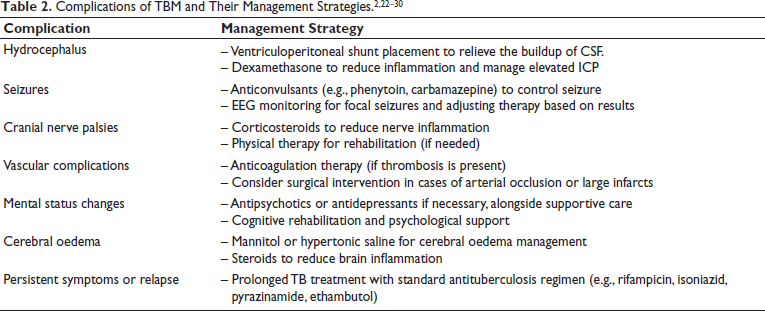
Treatment of TBM using antituberculous drugs alone has a poor prognosis. However, there are other treatment options using corticosteroids. These drugs are often used in addition to antituberculous drugs to enhance their effect in reducing the inflammation of the brain’s surface and associated blood vessels, decreasing the pressure inside the brain, and hence reducing the risk of death. Some clinicians have raised concerns regarding the use of corticosteroids—even if they improve survival, they result in more severely disabled survivors. Even though brain damage causing disability is uncommon, a slightly increased risk is noted with the use of corticosteroids, but not as significant compared to the reduction of deaths. While corticosteroids are frequently administered in conjunction with antituberculous medications to reduce inflammation and intracranial pressure, their usefulness is debated due to potential side effects, including greater impairment among survivors. Alternative treatments try to minimise inflammation and the risk of AIS while avoiding these downsides. Studies have shown that supplementary aspirin medication, which has anti-inflammatory and antithrombotic qualities, can reduce the incidence of new cerebral infarctions in TBM patients. Linezolid, an antibacterial antibiotic with high central nervous system penetration, has shown promise as an alternative. Furthermore, research into host-directed therapies (HDTs), such as thalidomide and other immunomodulatory medicines, is ongoing, concentrating on modifying the host’s immune response to improve treatment efficacy and reduce inflammation.14–18 In patients with HIV-associated TBM, there is insufficient data to confirm if the benefits of corticosteroids are preserved.19–21 Kindly refer to Table 2 for understanding the complications of TBM and their management strategies.
Conclusion
This case report highlights the rare but significant complication of AIS in a patient with TBM. The 84-year-old woman was admitted with neurological symptoms, including disorientation and seizures, which progressed to aphasia and hemiparesis, underscoring the severe impact of TBM on the central nervous system. Imaging revealed multiple acute lesions, and the diagnosis was confirmed with positive sputum and CSF tests for M. tuberculosis. Despite timely initiation of anti-TB and adjunctive corticosteroid therapy, the patient developed AIS, illustrating the challenging management and prognosis of TBM in elderly, immunocompromised patients. It also emphasises the importance of early MRI imaging for the detection of cerebral infarction in TBM, as well as the need for careful monitoring and intensive management of complications like AIS. Future research should focus on optimising treatment strategies to balance the benefits of corticosteroids in reducing mortality with the potential risk of long-term neurological disability.
Footnotes
Abbreviations
AIS: Acute ischaemic Stroke, CSF: Cerebrospinal Fluid, CT: Computed Tomography, EEG: Electroencephalogram, HIV: Human Immunodeficiency Virus, MRI: Magnetic Resonance Imaging, TB: Tuberculosis, TBM: Tuberculous Meningitis, VAF: Vascular Risk Factors, WBC: White Blood Cells.
Acknowledgements
We would like to express our sincere gratitude to S. Khechinashvili University Hospital in Tbilisi, Georgia, and Dr Ann Dzagnidze for their tremendous support and direction during the production of this case report. Understanding the intricacies of this case and guaranteeing a thorough study were made possible by Dr Dzagnidze’s knowledge and guidance during our clinical observations. Her contributions and the chance to study under her guidance are greatly appreciated.
Authors’ Contribution
Original draft writing, review and editing, supervision, corresponding author: HSW, Original draft writing, review and editing: HV, Original draft writing, review and editing: PG, Original draft writing, review and editing: SC, Clinical case and Records, mentorship: AD.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
ICMJE Statement
All authors have contributed substantially to the conception, design and drafting of the manuscript. Informed consent was obtained from the patient (or guardian) for publication of the case details and images. All authors take full responsibility for the integrity of the work and have approved the final manuscript for submission.
Patient Consent
The consent and confidentiality form to use the patient data for scientific study and research have been obtained from the institution, and informed consent for publication of this report was also obtained from the patient. Thus, the authors certify that they have obtained all appropriate patient consents.
Statement of Ethics
Ethical approval from the local teaching hospital has been granted. Institutional Review Board approval was obtained for this study. Patient information, such as sex and age, has been anonymised in the case section to prevent the identification of individuals.