Abstract
Background
Transsynaptic degeneration of retinal ganglion cells (RGCs) likely plays a role in progressive loss of vision in glaucoma. Carrageenan (Carr), obtained from seaweed (Rhodophyceae), a sulphated polysaccharide extracted from a species of red algae (Chondrus crispus), is a reversible gel mostly used in eye-drop preparation due to its hydrocolloid nature. But Carr-induced inflammation causing oxidative stress in animals has not yet been evaluated. It was reported that amlodipine (AML) can inhibit inflammatory cytokines by lowering intracellular calcium concentration and increasing antioxidant defences.
Purpose
In this study, we examined the impact of AML administration on Carr-induced oxidative stress, causing free radial–mediated neurodegeneration and behavioural alteration in zebrafish.
Methods
We aimed to evaluate the behavioural alteration in zebrafish using the novel tank diving test (NTDT) and the light-dark preference test (LDPT) as invaluable tools for analysing visual functions. To understand the role of the antioxidant defence system, oxidative stress biomarkers such as lipid peroxidation and CAT activity were analysed. RGCs of the retina of the eye and neurons of the optical tectum (TeO) in the brain were investigated by histopathological studies.
Results
Carr-treated groups swam in random patterns and showed an increased number of transitions and spent more time in alter zones, whereas controls prefer perceived motion. AML significantly attenuates the increased lipid peroxidation level and decreased CAT activity with Carr administration. Histopathological analysis showed an increase in the percentage of pyknotic cell counts after Carr treatment, whereas AML supplementation significantly reduced the pyknotic RGC cell count in the retina and TeO of the brain.
Conclusion
Findings showed the effectiveness of AML as an antioxidant and neuroprotective agent by reducing pyckonsis in developmental RGCs and possible repair mechanisms against Carr-induced oxidative stress. This study may be useful to know the underlying mechanisms of weakening and eliminating central neurons for synaptic connections in the visual system of zebrafish.
Introduction
Glaucoma causes characteristic damage in retinal ganglion cells (RGCs), causing vision loss and blindness by damaging the optic nerves. RGCs are the binding neurons connecting the retina with the visual processing centre within the central nervous system (CNS) with restricted regeneration capacity.1, 2 Moreover, the RGC axon extends from the retina to the brain and is most vulnerable to free radicals. An increase in free radical generation contributes to DNA and protein damage, leading to apoptosis. Free radicals attack neural cells and contribute to an increase in wall pressure in the eye cup that may trigger the degeneration of RGCs.3, 4 RGCs and their trans-synaptic neuronal protection have become an emerging topic in the control of glaucoma.5, 6 Axonal degeneration of RGCs in glaucoma contributes to progressive damage to the visual field, including the optic tectum (TeO), that is, due to transsynaptic neuronal damage.7–9 However, the pathophysiologic mechanisms of increased oxidative stress in RGCs and its association with the development and progression of glaucoma are not completely understood.
Carrageenan (Carr), the red edible seaweed, is a common emulsifying agent used as an additive in different types of food and beverages and also as an ingredient in personal hygiene products. Carr is highly resistant to microbial and enzymatic degradation in the digestive system and, therefore, can easily pass through the gastrointestinal system without any alteration. 10 Previous studies have linked Carr-like food additives to inflammatory bowel disease (IBD), leading to behavioural aberrancy.11, 12 Carr can trigger neurotoxicity through the cytotoxic effect of reactive oxygen species (ROS) production, which is associated with increased oxidative stress 13 during metabolic transformation in an organism.11, 14 Previous literature suggested that Carr administration can alter tight junction protein expression, leading to an alteration in blood–brain barrier xenobiotic permeability.14–16
Antioxidant defence systems chiefly contribute to the protective mechanism against ROS and consist of CAT and reduced glutathione (GSH).17, 18 The main reducing system in the eye is the glutathione system, which includes reduced GSH and enzymatic antioxidants like CAT that regulate the oxidative balance. The enhanced lipid peroxidation, the production of ROS and the decrease in antioxidant enzyme activities contribute to the development of oxidative stress through the production of hydroxyl ions. In the eye, oxidative stress can alter the homeostasis of cells and initiate a protective cell response, which is most relevant in intrinsically photosensitive RGCs. Mostly, the endogenous oxidants are harmful to the retinal epithelial cells and transsynaptic neurons.19, 20
Amlodipine (AML) is a known store-operated chain-breaking type of antioxidant and calcium channel antagonist that could be a powerful therapeutic tool acting as an effective chemo-preventive agent in preventing glaucoma and glaucoma-related retinal degeneration. 21 As AML contains the dihydropyridine ring, which is responsible for antioxidant properties, it can protect the RGCs from getting degraded. 22 The synaptogenesis process is very important for retinal development, where the conveying of visual signals among neurons depends mostly on normal synaptic functions. AML administration plays a major role in the regulation of CAT enzyme levels and the lowering of lipid peroxidation in mice.23, 24 Earlier reports highlighted the possible protective role of AML against chlorpromazine-induced oxidative stress and behavioural deficits. 25
The zebrafish (Danio rerio) model has the potential to elucidate the developmental toxicity pathways, as system toxicology can provide a means for risk assessment specific to developmental toxicity in human health risk assessments. 26 Zebrafish have also been used to model several human ocular diseases, such as cataracts and glaucoma. The zebrafish, being a relatively simple vertebrate, is used as one of the leading models for conducting research on visual systems. 27 Recent studies in the zebrafish model state that this will mitigate the need to understand the pathological changes and also provide medically relevant insights into the human eye.28–30 Glaucoma is a blinding neurodegenerative disease that strikes at RGCs, the output neurons of the retina. Many previous reports have suggested that antioxidants could be a promising treatment strategy,23, 31 where Carr-induced oxidative stress and immune responses causing inflammation may be a risk factor for glaucoma.32, 33 We hypothesised that zebrafish could be a model organism for Carr-induced oxidative stress–linked disorders, focusing on the antioxidant properties of AML for the protection of glaucoma. Hence, this study aimed to evaluate the antioxidative and neuroprotective properties of AML for the hypothesis that it might play an important role in Carr-induced oxidative damage to protect the axons of RGCs, the only neuronal cell type in the retina, the primary visual centres and the brain in zebrafish.
Methods
Chemicals Required
The Carr used in experimentation is basically ‘I-Carrageenan’ procured from Hi Media. Amlodipine (AML) was purchased from Sigma Aldrich, India.
Experimental Animals and Treatment Process
Zebrafish (Danio rerio) has become a popular model organism to study retinal dystrophies. 23, 31, 34 At the start of the study, six- to eight-month-old zebrafish of either sex were procured in bulk from the fish hatchery of the Central Institute of Fresh Water Aquaculture (ICAR), Odisha, India, and kept in laboratory conditions. The zebrafish were divided into five different experimental groups (n = 10), namely naïve (no treatment), control [vehicle treated, 1 µl/L dimethyl sulphoxide (DMSO)], AML, Carr and Carr + AML, and maintained in 3 L glass containers with continuous aeration, and each experiment was repeated three times to reduce experimental error. All these experimental protocols for the use of animals were already approved by Ravenshaw University, Cuttack (Reg. No. 1927/Go/Re/S/16/CPCSEA dt. 12.07.2017) animal ethical committee guidelines and reported in accordance with ARRIVE guidelines.
Dose Standardisation
Zebrafish were allowed waterborne exposure to Carr directly at a concentration of 600 mg/L, followed by minor modifications. 33 Similarly, for AML, an acute toxicity test was conducted at 0.0001%, 0.001%, 0.01% and 0.1% concentrations to evaluate LC50. We found half of the fish were dead at 0.1% concentration; hence, a lower concentration of 0.01% was considered for the dose administration (data not shown).
Behavioural Test Measures
Light and Dark Preference Test
The light and dark preference test (LDPT) was conducted to quantify the scototaxis behaviour (anxiety-like behaviour). LDPT was conducted after seven days post-exposure of Carr and AML treatment to different groups of experimental zebrafish accordingly.31, 34 All the video tracking was done by the ANY-Maze video-tracking system, and the data were analysed using a software (Stoelting Co., USA).
Novel Tank Diving Test
The novel tank diving test (NTDT) was performed seven days post-waterborne exposure to Carr and AML.34–36 The zebrafish model explains the observation obtained by the behavioural test paradigm that normally animals tend to spend the maximum time duration in the bottom zone of a novel tank (bottom dwelling). Fish behaviour was examined by measuring the following variables: the total time spent in the top zone, the number of entries to the top zone and the latency to enter the top zone by using ANY-Maze software (Stoelting Co., USA, Version 6.1) to record the time spent in the zone, the latency towards the top-to-bottom zone and the number of transitions.
Oxidative Stress Marker Analysis
Following the behavioural studies, biochemical assays were performed to check for any changes in oxidative stress biomarkers. Samples (n = 10) containing brain and eye were homogenised separately, and as per the protocol, each sample was pooled in an ice-cold 50 mM phosphate-buffered saline (PBS) and then processed as per the protocol in Tris-HCl buffer, pH 7.4, for the preparation of 10% tissue homogenate.37, 38
Lipid Peroxidation Assay
In brain and eye tissues, lipid peroxidation was measured by monitoring the formation of thiobarbituric acid reactive substances (TBARS) as per Ohkawa et al. 39 In brief, after adding 3.8 mL of thiobarbituric acid reagent (TBA) to 100 µL of crude sample (with triple aliquots), it was heated at 95°C in a hot water bath for about 60 minutes and then centrifuged at 10,000×g for 10 minutes at room temperature. The pink-coloured chromogen formed by the reaction was spectrophotometrically measured at 532 nm by using tetramethoxypropane (TMP) as a standard. The findings of the results were expressed as nMoles of TBARS complex formed per milligram protein.
Catalase Assay
Catalase estimation of brain and eye samples was carried out according to the protocol. 40 Catalase activity was then assessed by measuring the rate of decrease in H2O2 absorbance at 240 nm at every 15-second interval until 2 minutes. Values are expressed as nkatals per mg of protein, where 1 kat = 1 mol of H2O2 consumed per second, and the results were evaluated as nanokatals per milligram of protein produced, where 1 kat = 1 mol of H2O2 consumed per second.
Histological Study by Cresyl Violet
For histopathological studies, zebrafish were euthanised by submersion in ice water (5 parts ice/1 part water, 0–4ºC) for at least 10 minutes following cessation of opercular (i.e., gill) movement. 38 The brains and eyes (n = 3) were collected for histopathological studies after dissection through proper procedures for histological analysis.35, 36, 41 Cresyl violet staining (CVS) was commenced to observe the neuromorphological observations.34, 35 The intact brain and eye samples of zebrafish were isolated and stored at a temperature of 4°C and then immerged with 30% sucrose solution. Cryosectioning was carried out serially with a 10 µm thickness, and CVS was performed to check histopathological changes in neuronal ganglionic cells and RGCs.
The slides were observed under a bright field microscope, and images of brain and eye sections were taken with the requisite magnification (Olympus, BX43, Japan). Cells showing an irregular staining pattern of typical pyknosis were counted in the telencephalic (TeO) region of the brain and ganglionic retinal layer (RGC) of the eye in zebrafish using Image J software. The results were expressed in percentages showing the pyknotic cell count and compared with the control.
Statistical Analysis
All the experimental data were presented as mean ± SEM. The results were compared using one-way analysis of variance (ANOVA) with the Newman–Keuls post hoc test performance for comparisons between naïve, control, AML, Carr and AML + Carr. A two-way ANOVA with Tukey’s multiple comparison test was also conducted for the histogram analysis of pyknotic ganglionic cell count in the retina (in %). All the experiments were repeated on triplication to minimise equipment and handling errors. In all the experimental groups, the significance level was mostly p < .0001.
Results
Behavioural Study
Light and Dark Preference Test Analysis
The representative track plots of LDPT showed behavioural alterations in zebrafish after the Carr and AML treatments (Figure 1a). LDPT results exhibited that the Carr-treated group mostly spent significantly more time in the light zone (F4,145 = 106.6, p < .0001) when compared with the naive, control and AML co-supplemented groups (Figure 1c). The number of transitions to the light zone was found to be significantly decreased (F4,145 = 59.30, p < .0001) in the Carr + AML group as compared to the Carr-treated group (Figure 1a). Also, a significant decrease concerning latency towards the light zone was observed in comparison to control groups, and in the Carr-treated group, it was further shown to be restored with AML co-supplementation (Figure 1d) (F4,145 = 34.64, p < .0001). The AML-supplemented group showed similar behavioural responses as the naïve and control groups.
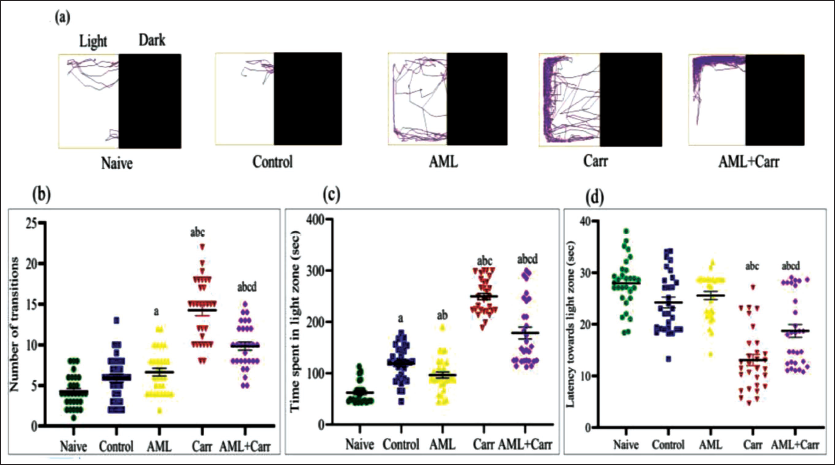
(a) The representative track plots show the scototaxis pattern of behaviour in different treatment groups of zebrafish, that is, naïve, control, AML, Carr and AML + Carr. (b) The number of transitions (line crossings). (c) Stay time in the light zone (in seconds). (d) Latency towards the first entry to the light zone in each experimental group in LDPT.
Novel Tank Diving Test Analysis
The track plots for the NTDT represent the alteration in behavioural responses after AML administration to Carr-treated groups (Figure 2a). The NTDT data showed a significant increase in stay time (F4,145 = 56.21, p < .0001) in the top zone of the diving tank in comparison to the control group (Figure 2c). The latency to enter the top zone of the tank significantly decreased in the Carr-treated group as compared with the naïve and control groups, which was significantly restored in the AML co-supplemented group (F4,145 = 39.93, p < .0001) (Figure 2d). A significant increase (F4,145 = 81.62, p < .0001) concerning the number of transitions to the top zone was also observed in Carr-treated groups with AML administration in comparison to the Carr-treated group (Figure 2b). Similar to LDPT, NTDT data also showed the groups administered with AML only showed no deflection in bottom dwelling responses like a normal control.
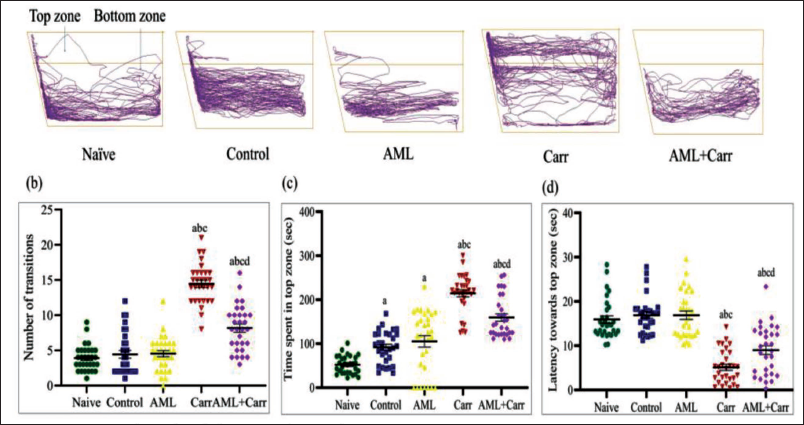
The representative track plots of NTDT, displaying the bottom dwelling pattern of behaviour in different groups. (b) The number of transitions (line crossings). (c) Time spent in the top zone (in seconds). (d) The latency to enter the top zone (in seconds).
Lipid Peroxidation and Catalase Levels Reconciled on AML Treatment
Waterborne exposure to Carr for seven days in zebrafish significantly increased the TBARS complex production in the brain as well as in the eye of zebrafish, a biomarker of lipid peroxidation. The increased levels of TBARS production in the brain were found to be attenuated after AML co-supplementation, whereas the effective rate of amelioration in the eye was slower in comparison to the brain (F4,10 = 203.00, p < .0001 (Figure 3a). AML co-supplementation groups showed a significant reduction in production of TBARS by lipid peroxidation test (F4,10 = 92.33, p < .0001) in the eye of zebrafish as compared to Carr-treated groups (Figure 3b). Carr-treated groups showed a significant depletion in the catalase enzyme in the brain tissues of zebrafish compared to other naïve and control groups. The CAT enzyme level after AML co-supplementation was significantly restored in Carr-treated AML co-supplemented groups, implicating that AML reduced free radical production in the brain (F4,10 = 185.5, p < .0001) (Figure 3c). The eye tissues of zebrafish showed a significant decrease in CAT level in comparison to the control group. However, the CAT level was significantly decreased in the zebrafish eye in the Carr-treated group when compared with the Carr-treated with AML co-supplemented group (F4,10 = 155.1, p < .0001) (Figure 3d), where the effective restoration rate of the CAT level was found to be more significant in the AML co-supplemented groups.
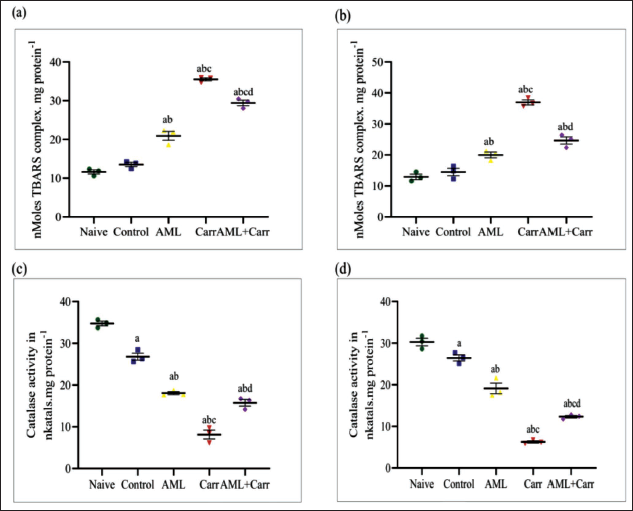
The measurement of oxidative stress biomarkers in assessed lipid peroxidation and antioxidant enzymes like catalase after co-supplementation with AML showed a significant decrease in lipid peroxidation level and increased expression of catalase enzyme (p < .0001).
Histopathological Analysis
CVS of the optic tectum (TeO) of the periventricular grey zone (PGZ) in the brain and ganglionic cell layer (GCL) and inner nuclear layer (INL) of RGC sections of the eye of zebrafish was shown in comparison to naïve and control. The pyknosis process in RGCs and the TeO brain region was significantly restored on co-supplementation of AML against Carr-induced neurodegeneration. The brain sections of Carr-treated groups showed a significant (F4,10 = 219.9, p < .0001) increase in percentage change of pyknotic cell counts in comparison with the control group (Figures 4a, 4b). Representative images (Figure 4a) on 40× magnification and histogram (Figure 4b) showed clear indication towards deeply stained pyknotic neuronal cells in the TeO region of Carr-treated groups, whereas on AML co-supplementation, the number of pyknotic cells was restored in the zebrafish brain. The percentage of pyknotic cell count was found to be significantly high in the Carr-treated group, which reflects nuclear loss in zebrafish RGCs. However, AML co-supplementation with Carr-treated groups significantly reduced the percentage change in pyknotic cell count in INL and GCL (F4,1 = 35.99, p < .0001) retinal ganglionic regions in the retina of zebrafish eyes in comparison to the Carr-treated group (Figures 5a, 5b).
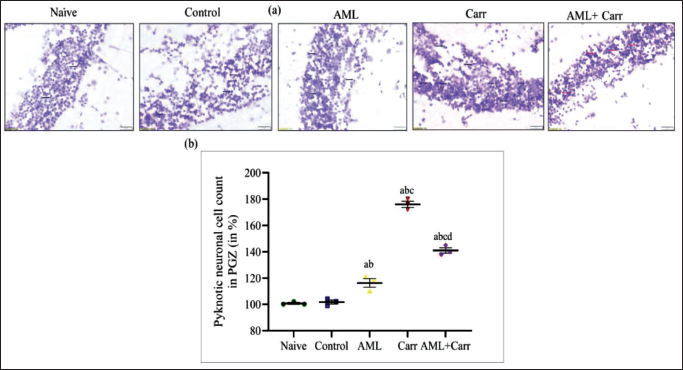
(a) Cresyl violet staining of the optic tectum (TeO) of the zebrafish brain region showing representative micro-images of pyknotic cell count in naïve, control, AML, Carr- and AML + Carr-treated groups with 40× magnification. The black arrow mark indicates the loss of neuronal cells, and the red arrow mark indicates a reduction in changes in pyknotic neuronal cell count, showing restoration of pyknosis processed by co-supplementation of AML.
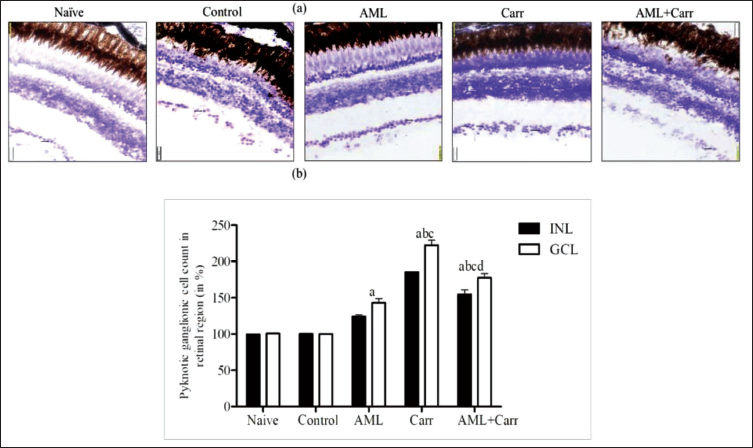
(a) CVS of RGCs of zebrafish eye showing pyknotic cell counts in naïve, control, Carr, AML, Carr co-supplemented with AML groups, with 40× magnification. The black arrow mark indicates the increased number of pyknotic cells in inner nuclear layer (INL), and the white arrow head marks the pyknotic cells in the ganglionic cell layer (GCL) and RGC layer of the adult zebrafish retina. (b) Histogram showing percentage changes (%) in a number of pyknotic cells in the INL and GCL of the RGC layer of the zebrafish eye region of different treatment groups: naïve, control, AML, Carr and AML + Carr-exposed groups. Two-way ANOVA with Tukey’s multiple comparison tests.
Discussion
In this study, the findings support the hypothesis and suggest that AML has an antioxidant property that may have an effective neuroprotective role against Carr-induced oxidative stress. Free radical production can affect the zebrafish eye due to cytotoxicity, in which the ganglionic cells, the only neuronal cells, have their cell bodies in the inner retina and axons travel via the optic nerve to the optic chiasm, which may suffer functional or sensory loss, leading to glaucomatous neurodegeneration.42–44 The highly prevalent neurodegenerative condition of the optic nerve in glaucoma is the leading cause of blindness worldwide.45, 46 RGCs fail to regenerate their axons, resulting in permanent and irreversible blindness. 2 Studies also showed the relationship between glaucoma and neurodegenerative diseases through transsynaptic neurodegeneration, which occurs between the eye and brain connection.47, 48
Carrageenan, a linear sulphated polysaccharide, is a straight-chain galactan sulphate of high molecular weight, mostly extracted from red seaweed, and has a broad spectrum of applications in the biomedical and biopharmaceutical fields. 49 Carr is typically present in the formulation of eye drops as an ophthalmic vehicle (about 0.1% to about 3.0% by weight/volume), such that the composition is administrable as a drop and gels upon instillation into the eye. No adverse effects have been detected in chronic toxicity studies with Carr in rats up to 7,500 mg/kg bw per day. 50 The dose of Carr (600 mg/L) was used in the present study through waterborne exposure and was found to affect behavioural changes in zebrafish.33, 51, 52 Our findings advocate the hypothesis that Carr-induced ROS production can lead to functional and morphological impairments in RGCs and the transsynaptic progression of neurodegeneration in the visual system, leading to behavioural alteration. Previous findings from our laboratory have also suggested that the periventricular grey zone of the TeO region is the largest visual brain area that regulates the visual behaviour of zebrafish.38, 53, 54 The increased time spent in the light zone in LDPT found in the present study could indicate an increased aversion towards illumination due to the Carr treatment. The zebrafish was also found to spend more significant time in the top zone, swim towards the perceived motion and gather at the upper zone in NTDT. Carr could have produced potent oxidative stress, which led to a neurobehavioural alteration in zebrafish. In the behavioural test measures, the normal behaviour of zebrafish was restored when a therapeutic dose of AML was co-supplemented with Carr, which could be due to the neuroprotective activity of AML against oxidative stress caused by Carr treatment.
Recent studies on calcium function confirmed the role of calcium channel blockers (CCBs), and blocking calcium channels showed a decrease in oxidative stress.53, 55 In the current study, AML, as a CCB, was evaluated for oxidative damages induced by Carr, restoring the behavioural responses of zebrafish by protecting the TeO from getting degenerated due to free radical (ROS) generation. The present study showed a significant increase in lipid peroxidation, producing more amounts of TBARS in the eye and brain in the Carr-treated group, which might be due to the production of ROS, causing oxidative stress. A significant alteration in antioxidant enzyme status was also observed in the zebrafish eye and brain. A depletion in catalase level was observed in the Carr-treated group in comparison to the control group. Furthermore, AML treatment showed a decrease in lipid peroxidation and significantly increased CAT activity. Previous studies have shown that AML is known to ameliorate the Carr-induced oxidative stress effects, causing a reduction in free radical generation.56–58
The present findings demonstrated that AML protects Carr-induced free radical generation that causes degeneration to RGCs and cells of TeO of the visual cortex in the zebrafish model. The RGC layers and the PGZ of the brain are most vulnerable to free radical generation 59 The histopathological observations revealed that Carr-induced effects can be restored by co-supplementation of AML, causing a reduction in neuronal chromatin condensation (pyknosis) in the brain and RGC degeneration by modulating the mechanism of oxidative stress. The behavioural responses in zebrafish have been affected due to neuronal damage in the TeO of the brain region. The increased pyknotic cell count in RGCs’ inner nuclear layer (INL) and ganglionic cell layer (GCL) of the zebrafish eye and optic tectum (TeO) of the PGZ region of the zebrafish brain after Carr treatment suggests some Carr-induced oxidative stress and cellular activation of inflammatory action. The present findings revealed the possible protective effect of AML and its capacity to scavenge Carr-induced free radicals, which could be due to the free radical quenching property and the antioxidant activity, showing the possibility for safer use in the treatment of glaucoma.
Conclusion
The Carr treatment showed damage to the retina of the eyes as well as the visual centres of the central nervous system. The synergistic action of AML against Carr-induced oxidative stress showed a dramatic decrease in oxidative stress and neuronal cell damage with altered neuro-behavioural phenotype, antioxidant stress marker levels replicating reduced pycknotic nuclei in zebrafish. The present findings reveal the synergistic effects of AML in designing and evaluating drug combinations for the biomedical application of Carr-like bioactive agents.
Footnotes
Acknowledgement
The authors are thankful to Dr (Mrs) Monalisa Mishra, assistant professor, Department of Life Sciences, NIT Rourkela, Odisha, for her kind help and technical support for photographic data analysis.
Authors’ Contribution
MP conceived, designed and supervised the experiments with overall mentorship. AN and SA carried out the investigations and analysis with the majority of the experiments and wrote the original draft of the manuscript. RNS performed part of the experiments and literature review. All authors contributed to the data analysis and commented on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Manorama Patri received financial support from DRDO, New Delhi, No. O/o DG (TM)/81/48222/LSRB-294/PEE & BS/2017 and the Department of Science and Technology (DST), Odisha (No. 27562800402014/20-665), for the research, authorship and/or publication of this article.
Statement of Ethics
All the experimental procedures were performed according to the institutional animal ethical committee guidelines at Ravenshaw University, Cuttack, Odisha (Reg. No. 1171/C/08/CPCSEA dt. 12.07.2017) and were consistent with the Association for Research in Vision and Ophthalmology (ARVO) statement for the care and use of laboratory animals in ophthalmic and vision research. The study was reported in accordance with ARRIVE guidelines.
