Abstract
Background. To date, no specific therapy is available for optic glioma (OG)–induced visual loss. Objective. To evaluate the effects on visual function of murine nerve growth factor (NGF) eye drop administration in children with severe visual impairment due to low-grade OGs. Methods. Five patients with OGs and advanced optic nerve atrophy were assessed before and after a single 10-day course of 1 mg murine NGF topical administration by clinical evaluation, visual evoked potentials (VEPs), and brain magnetic resonance imaging (MRI). VEPs, the main functional outcome measure, were recorded at baseline and 1, 30, 45, 90, and 180 days posttreatment. MRI examinations were performed at baseline and at 180 days after NGF treatment. Six untreated control patients with OGs also underwent serial VEPs, clinical testing, and MRI assessments. Results. After NGF treatment, median VEPs amplitude showed a progressive increase from the baseline values (P < .01). VEPs reached a maximum amplitude at 90 days (170% increase) and declined at 180 days, still remaining above the baseline level. Perception of spontaneous visual phosphenes was noted in all patients after NGF administration. MRI showed stable tumor size. In controls, clinical findings and VEPs did not show any significant change over the observation period. Conclusions. The findings from the study show that NGF administration may be an effective and safe adjunct therapy in children with optic atrophy due to OGs. The beneficial effect on optic nerve function suggests a visual rescuing mechanism exerted by murine NGF on the residual viable optic pathways.
Keywords
Introduction
In children, several neoplastic diseases, such as craniopharyngiomas and optic gliomas (OGs), can compromise visual function involving both anterior and retrochiasmatic optic pathways. No specific therapy is currently available for OG-induced visual loss. Therefore, any improvement that might be obtained is of major clinical and socioeconomical value both to the patients and their relatives.
Nerve growth factor (NGF) is the first discovered neurotrophin involved in the development and survival of sympathetic, sensory, and forebrain cholinergic neurons. 1 In experimental animal models, NGF promotes nerve terminal outgrowth and neuron recovery after inflammatory, ischemic, and toxic injuries. 2,3 Given its actions favoring neuronal survival, NGF has been proposed for the treatment of some traumatic, ischemic, and neurodegenerative brain diseases. 4-6 Intraventricular NGF administration ameliorates symptoms in adults with Parkinson and Alzheimer disease and improves cerebral blood flow in infants with hypoxic-ischemic brain injury. 7-10 Exogenous NGF showed neuroprotective effects also on the visual system 11 due to the presence of NGF receptors on the conjunctiva, cornea, as well as in the retinal pigment epithelium, photoreceptors, and retinal ganglion cells (RGCs). 12 The effects of NGF and other neurotrophins, such as brain-derived neurotrophic factor (BDNF), are mediated via uptake by RGCs, anterograde transport along the optic nerve, and release to the postsynaptic geniculate neurons. 13
In experimental animal models, intraocular NGF administration keeps RGCs from degeneration after optic nerve transection and ocular ischemia and leads to the rescue of axotomized forebrain cholinergic neurons, 14-16 while conjunctivally applied NGF proved to be effective in patients with corneal ulcers and severe glaucoma. 17,18 Indeed, it has been reported that NGF administered to the conjunctiva reaches rapidly the retina and the optic nerve where this neurotrophin exerts its biological activities through the uptake by RGCs. 19
The aim of this study was to evaluate the safety and efficacy of NGF eye drops administration in improving optic nerve responses in advanced optic pathways damage induced by OGs. We report the treatment results of a pilot, open-label, longitudinal study in a group of 5 pediatric patients suffering from severe visual impairment associated with OG involving the chiasm and retrochiasmatic pathways. Results were compared with those obtained from an untreated control group of OG patients matched for age and disease severity.
Methods
Study Population
This open-label study was conducted in children with OGs without or with neurofibromatosis type I (NF1) by genetically confirmed diagnosis, admitted to the Division of Paediatric Oncology at the “Agostino Gemelli” Hospital in Rome, Italy. Five patients (3 males and 2 females) with severe impairment of visual acuity and visual field from optic nerve atrophy due to the presence of the tumor were included. None of the children enrolled had concomitant ocular diseases (Table 1). The median age was 9.5 years (mean age, 11 years; range, 4.6-18.5 years). Three patients had a diagnosis of NF-1. Before entering the study, 3 patients underwent 1 or more courses of chemotherapy (with an induction cycle of carboplatin and vincristine as suggested by Packer), 20 followed by maintenance treatment with the same drugs alternated with vincristine, 1-[2-chloroethyl]-3-cyclohexyl-1-nitrosurea] CCNU/procarbazine/desametasone.
Clinical and Demographic Data of Patients Treated With Nerve Growth Factor and Controls
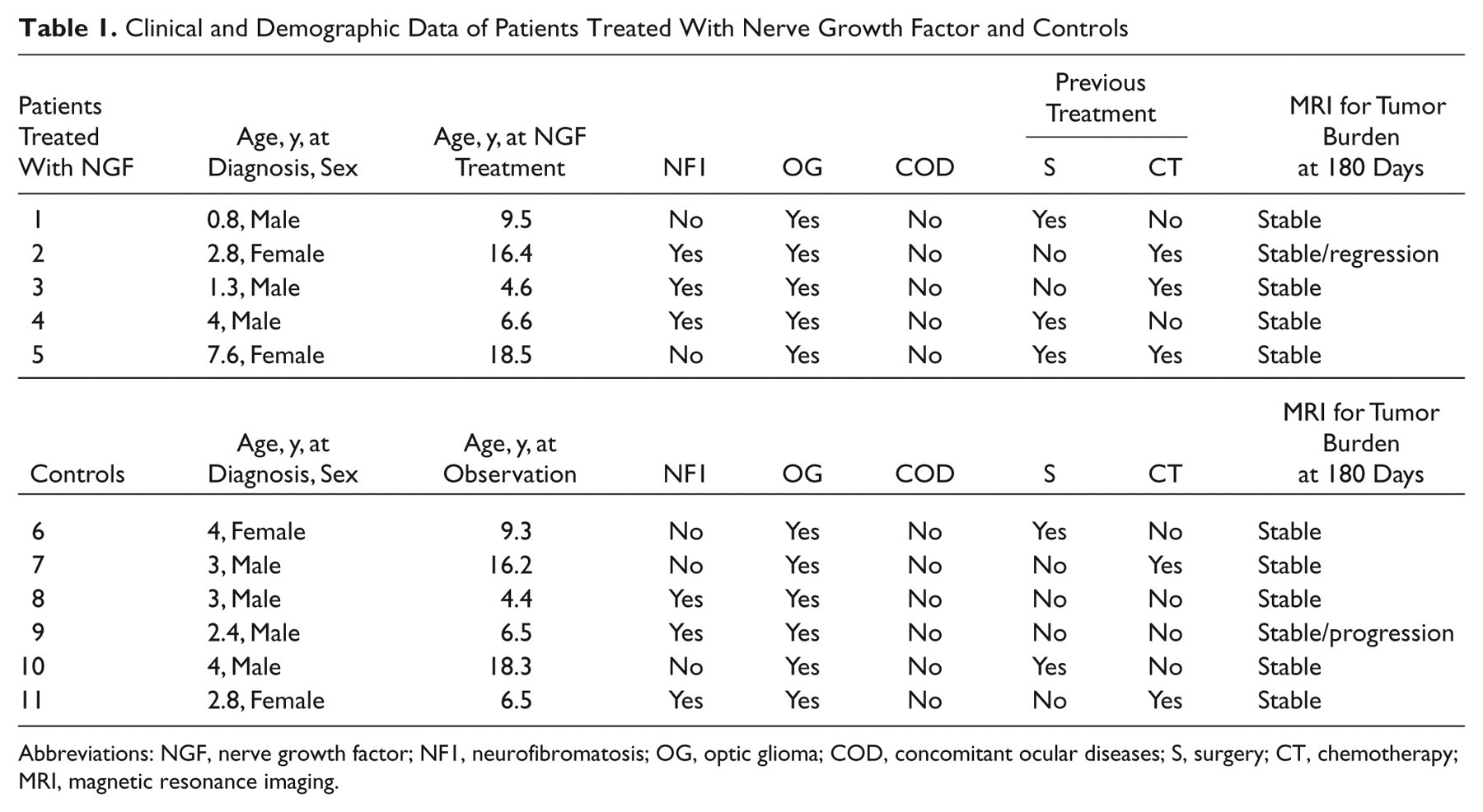
Abbreviations: NGF, nerve growth factor; NF1, neurofibromatosis; OG, optic glioma; COD, concomitant ocular diseases; S, surgery; CT, chemotherapy; MRI, magnetic resonance imaging.
At enrollment, all children were not receiving chemotherapy and the time interval between the last chemotherapy and the inclusion into the study was more than 24 months (mean time, 59.8 months; range, 25-131 months).
Six untreated control patients with OGs were also included in the study. They were matched for age and sex distribution, as well as for disease severity and residual visual function, to the treatment group.
Clinical and demographic features of all patients are reported in Table 1. Before starting treatment, children underwent a careful general and neuro-ophthalmologic examination. The latter included intrinsic ocular motility testing, voluntary conjugate eye movements assessment, anterior segment biomicroscopy, direct and indirect ophthalmoscopy, and clinical assessment of very low visual acuity ranging from light perception to hand motion. Given the very low vision condition of our patients, the visual acuity pretreatment and posttreatment was considered as an unreliable outcome parameter, because of a possible bias in the examination. We therefore established, at the beginning of the study, to consider the visual evoked potentials (VEPs), recorded in response to steady-state flicker stimulation following a published technique, 21,22 as our main outcome measure in the evaluation of potential drug efficacy. All patients had clear optical media and no concomitant disorders that could have affected the electrophysiological assessment.
Cranial and orbital magnetic resonance imaging (MRI) scans were performed using standard imaging parameters and gadolinium enhancement. Initial and follow-up scans were reviewed by one neuroradiologist (CC) to evaluate the extent and location of tumor and to record any changes after the treatment. He did not know whether patients belonged to the control group or to the treatment group. OG location was determined as involving the chiasm and retrochiasmatic pathways in all patients. Changes in tumor size/volume were assessed by measuring the 3 largest diameters of the optic/hypothalamic gliomas. Sagittal and craniocaudal largest diameters were electronically calculated on sagittal T1-weighted postcontrast images; the largest transverse diameter was calculated on axial postcontrast T1-weighted images. In patients where the optic hypothalamic tumors showed partial contrast enhancement, the final values of the 3 largest diameters were defined using also unenhanced T2-weighted images. Moreover, the diameters of the intra-orbital optic nerves tumors were assessed on axial and coronal images. Finally—if present—the leptomeningeal metastatic deposits were also measured in terms of maximum thickness of the cisternal/leptomeningeal metastases.
All patients, both treated and controls, had participated in several repeated clinical, VEP, and MRI examinations before inclusion in the study. The clinical and VEP repeated sessions took place 2 to 4 months apart, and MRI evaluation 8 to 12 months. All patients were familiar either with the examination techniques that were employed in the study or with the personnel involved. This allowed reasonably good cooperation of all patients throughout the study. In addition, the data collected from the patients before inclusion in the study served as reference (eg, VEP test–retest variability) for comparison with the actual study data and provided a detailed picture of disease natural history for every patient.
All the enrolled patients (and/or their parents) were fully informed as to the nature and goals of the study. Written informed consent was obtained. The study followed the tenets of the Declaration of Helsinki and was approved by the ethics committee of the institution.
Visual Evoked Potential Methods
In all patients, electrophysiological testing was always performed within 1 week of the clinical and neuroimaging examinations by an independent examiner who was masked as to the patients’ clinical and MRI findings or to the study group of NFG-treated or controls.
The stimulation/recording technique has been described in detail by Trisciuzzi et al. 21 Briefly, VEPs were recorded in response to the sinusoidal luminance modulation of a uniform field (temporal frequency = 8 Hz; modulation depth = 98%; mean luminance = 100 cd/m2) generated by an array of 8 LEDs (peak emission wavelength 580 nm) presented monocularly in a mini-ganzfeld. During monocular stimulation (the right and left eyes were stimulated in a random order), the nonstimulated eye was kept adapted at the same mean luminance level as the stimulus. VEPs were recorded by using surface electrodes, placed on the scalp 3 cm above the inion and referenced to the right mastoid. The left mastoid was grounded. Interelectrode resistance was kept less than 5 kΩ. Signals were amplified (100 000 times), filtered (band-pass, 1-100 Hz), sampled with 12-bit resolution on a 100-µV AC range (2 kHz sampling rate), and averaged at the stimulus period (125 ms sweep) with automatic artifact rejection (±30 µV amplitude window). For each recording, 8 blocks of 200 sweeps were collected. A discrete Fourier series of the resulting grand means was performed off-line to isolate the fundamental (1F) component of the response, whose peak-to-peak amplitude (in µV) and phase angle (in degrees) were measured. 23 Since the response energy was mostly concentrated at this Fourier component, it may well represent the main VEP waveform for every record. The standard deviations (SDs) of individual blocks were measured to assess signal reliability. Typically, SDs were less than 30% for amplitude and 60° for phase. Responses were also averaged at a frequency 1.1 times the stimulus frequency to measure the residual “noise” level after averaging. Provided that noise spectrum is sufficiently smooth this gives a good estimate of noise amplitude at stimulus frequency. A “noise” evaluation was made using a control response at the same temporal frequency by presenting a subthreshold stimulus (ie, 0.001 modulation depth) whose mean luminance was the same as the actual stimulus. In both cases, the averaged noise at the 1F did not exceed 0.12 µV during all experimental sessions. At this noise level also baseline measurements that fall in the sub-microvolt region may be considered as reliable.
In both NGF-treated and control patients, potential retinal toxicity by concomitant chemotherapeutic drugs (or by NFG treatment in the former group) was excluded by ganzfeld scotopic and photopic electroretinogram testing performed according to ISCEV standards 24 at least twice during the observation period.
Nerve Growth Factor Isolation
The drug used was 2.5S NGF, which was purified and lyophilized from male mouse submandibular glands and prepared according to the method of Bocchini and Angeletti. 25 Briefly, the submandibular glands of adult male mice were explanted under sterile conditions and the tissues were homogenized, centrifuged, and dialyzed. This aqueous gland extract was then passed through subsequent cellulose columns, thereby separating NGF by adsorption. The first step was gel filtration at pH 7.5, in which most of the active NGF was eluted in the 80 000 to 90 000 molecular weight range. The samples obtained were analyzed by spectrophotometry at a wavelength of 280 nm to identify NGF-containing fractions. Specificity of fractions was determined by Western blot analysis. NGF purity (>95%) was estimated by high-performance liquid chromatography, the column equipped with a guard column calibrated with 40 mg of purified and bioactive murine 2.5S NGF standard. The NGF obtained was then dialyzed and lyophilized under sterile conditions and stored at −20°C until used. Biological activity of purified NGF was evaluated by in vitro stimulation of neurite outgrowth in rat pheochromocytoma PC12 cells over a period of 7 to 14 days. Subsequently, NGF was dissolved in 0.9% sterile saline solution in concentrations of 200 µg/mL. The concentration of NGF in this solution was stable for the entire treatment time.
Nerve Growth Factor Eye Drop Administration
NGF eye drop was administered after at least 2.5 years from the diagnosis of OGs, if the patients showed loss or severe impairment of visual acuity. A total of 1 mg of NGF diluted in 5 mL of saline solution was administered onto the conjunctiva of both eyes for 10 consecutive days 3 times a day. This amount is considered sufficient to reach and stimulate NGF receptors in most cerebral cholinergic areas of the brain and optic pathways, as previously reported in the literature. 7 We preferred to use murine NGF, instead of human-recombinant NGF, because contrasting results have been reported on the efficacy of the latter, mainly due to a lack of in vivo studies. Clinical studies using human NGF have been performed only in patients with sensory polyneuropathy reporting either positive effects 26 or no significant effects on this condition. 27 Currently, there are no studies on conjunctivally applied human NGF, unlike that reported for murine NGF demonstrating a therapeutic effect for corneal pathologies 17 or supporting a potential therapeutic effect for retinal degenerations. 18 In addition, there is no evidence showing that the human NGF is able to reach and affect the optic nerve and brain areas, such as the septum and Meynert basal nuclei, as reported for murine NGF. 19,28
Follow-up and Testing Schedule
In both groups of patients (the NGF treated and the controls), clinical, VEP, and MRI examinations were performed at the beginning of the follow-up period. In the treatment group, clinical and VEP testing were repeated at 10, 30, 45, 90, and 180 days after the beginning of the NGF treatment. MRI was repeated at 180 days. In the control group, clinical and VEP examinations were repeated at the same time points throughout the observation period. MRI was repeated at 180 days.
Ocular and Systemic Complications Potentially Related to NGF Administration
During the entire period of assessment (180 days; see below) particular attention was paid to detect ocular and/or systemic side effects. Potential ocular complications included inflammation (external or on the iris and/or ciliary body), pain, development of lens opacities, and increased intraocular pressure. Systemic complications included acutely increased intracranial pressure and development of acute intratumor hemorrhagic infarction, as well as allergic reactions. Progression of the OG mass, as reflected by a volumetric increase of more than 25% from baseline at brain MRI, was regarded as a potential adverse outcome event. Other potential side effects related to NGF administration were systemic pain as well as weight loss, as previously reported in the literature. 7
Statistical Analysis
VEP amplitude and phase results were evaluated in both treated and control patients by nonparametric statistics. Data obtained over the follow-up period were statistically analyzed using a method that takes into account the circular distribution of phase space after conversion of amplitude and phase data into cosine and sine values. 29 Longitudinal changes (baseline vs end of follow-up) of VEPs from OGs patients were evaluated by statistically comparing (by means of nonparametric analysis of variance [ANOVA]) the amplitude and phase values across the recording sessions. Percentage amplitude difference (ie, 2nd − 1st test/1st test × 100) and phase difference in degrees (taking into account circular phase distribution) between test results were calculated for each patient and the median, 5th, and 95th percentiles of the resulting distributions established. In some analyses, VEP amplitudes were also converted to log 10 values to better approximate normal distribution. In all the analyses, a P value <.05 was considered as statistically significant.
Results
In treated patients, no local or systemic adverse events related to NGF treatment were observed either during treatment or over the 180-day period of follow-up.
At baseline, in all patients the visual acuity was extremely low, ranging from light perception to hand motion perception at the distance of 10 cm. Pupils were normal in shape and size (mean pupil size, 4 mm; range, 3.5-5 mm). Ganzfeld electroretinograms (ERGs) were normal in both amplitude and implicit time. This allowed us to exclude any potential retinal damage due to the previous chemotherapeutic treatment.
After NGF treatment and during the 180-day period of follow-up, patients and/or parents on their behalf reported some visual improvements during and after the treatment. All of them reported phosphene perception at night, and some figures’ shapes instead of light only in the daylight. In particular, a patient (case 2 in Table 1) started watching TV using an anomalous head posture. Subjective symptoms of visual improvement were never reported in the untreated controls during the follow-up. No changes in pupil size or pupillary light response were detected after NGF treatment.
The flicker VEPs results are shown in Figures 1 to 3. In Figure 1, representative VEP waveforms recorded in an NGF-treated patient at baseline, 10, 90, and 180 days are shown.
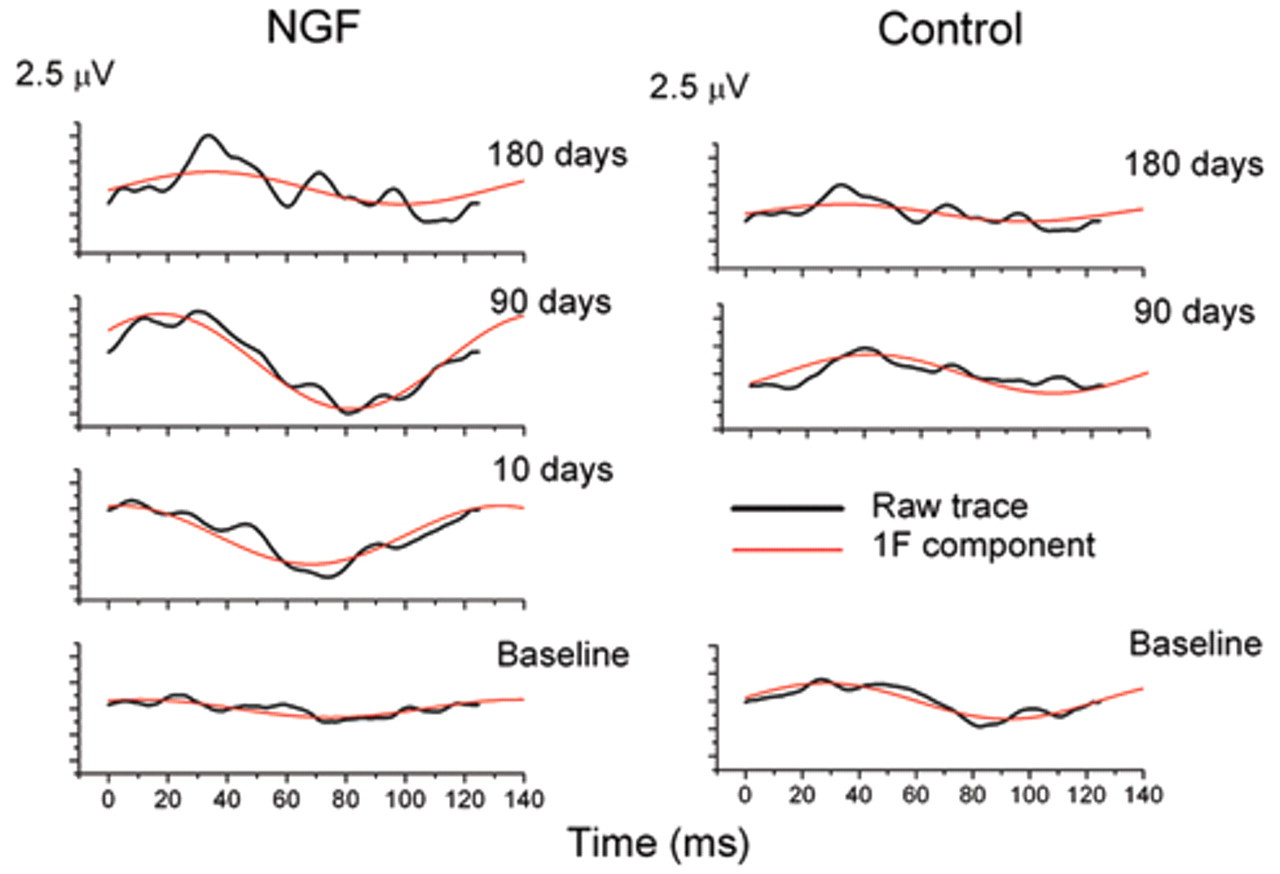
Representative visual evoked potential waveforms recorded in a nerve growth factor (NGF)–treated patient at baseline, 10, 90, and 180 days. For each record, the Fourier-analyzed fundamental harmonic component is also shown (in grey). Data from a control patient are shown at baseline, 90, and 180 days of follow-up. For each record, the sweep duration is equivalent to 1 stimulus cycle (8 Hz, 125 ms).
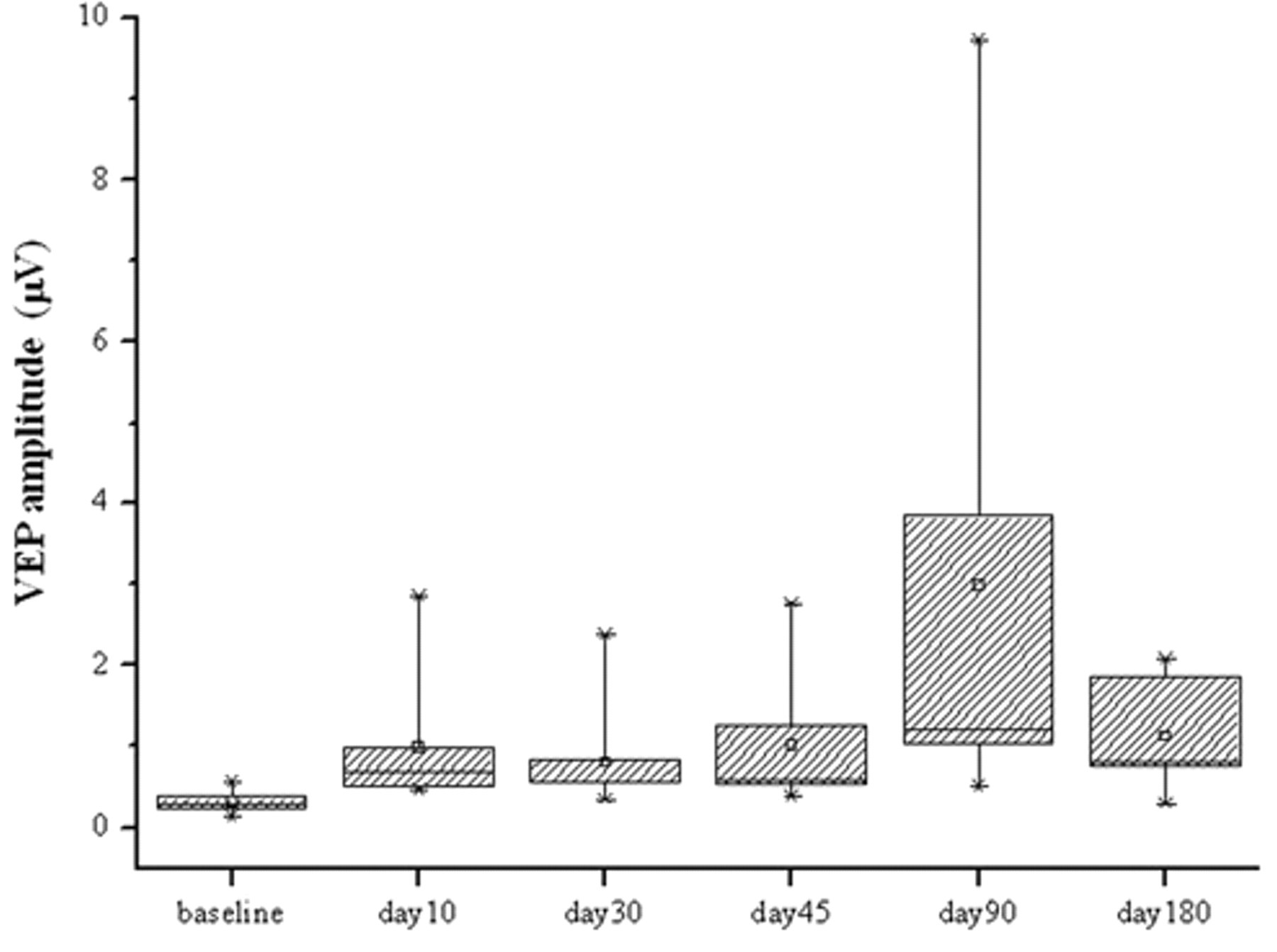
Box plots showing the distribution of visual evoked potential (VEP) 1F amplitude values recorded in nerve growth factor (NGF)–treated patients at baseline and at various times following NGF treatment. Each box shows 75th, 50th, and 25th percentiles. The point inside the box indicates the mean. Error bars show 99th and 1st percentiles.
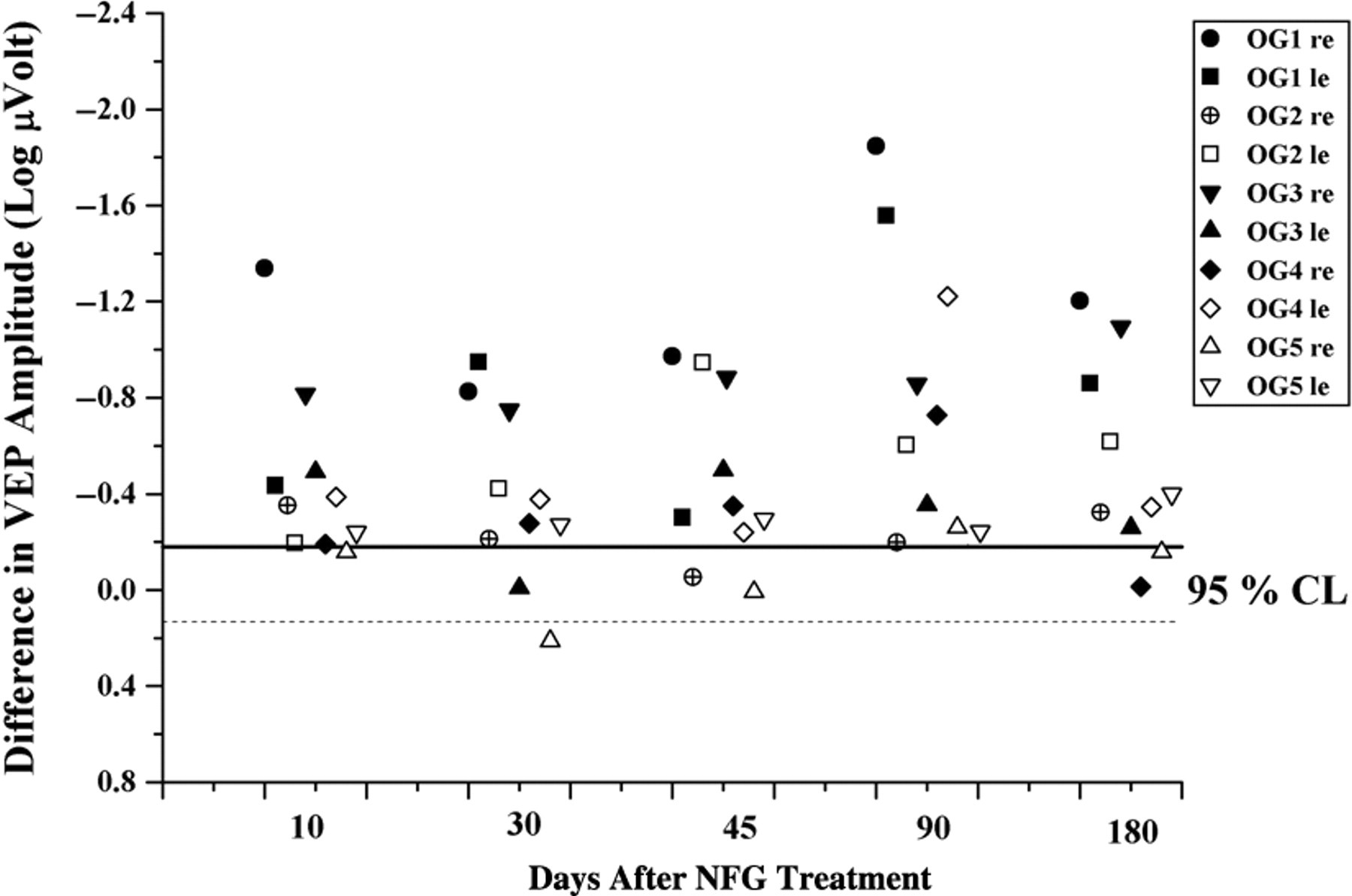
Scatterplots showing the individual visual evoked potential (VEP) log amplitude changes from baseline (ie, log amplitude at a given recording session minus log amplitude at baseline) observed in each eye of all participants at the different follow-up times. Horizontal lines in the plot indicate the 95% confidence limits for test–retest amplitude changes observed in the same patients before inclusion in the study. NGF, nerve growth factor.
The response fundamental component, shown in grey, isolated by Fourier analysis, is also reported in the figure for each record. Data from a control patient are shown at baseline, 90, and 180 days of follow-up. It can be noted that in the treated patient, but not in the control, the VEP response showed a clear increase in amplitude at both 90 and 180 days compared with baseline.
In Figure 2, box plots showing the distribution of VEP amplitude values recorded in NGF-treated patients at baseline and at various times following treatment are reported. Each box shows 75th, 50th (median), and 25th percentiles. The point inside each box indicates the mean. Error bars show 99th and 1st percentiles. In all treated children, median VEP amplitude, severely attenuated at baseline compared with normal values, 22 showed a sharp increase immediately after the end of the treatment. This improvement lasted throughout the entire follow-up period, progressively reaching a maximum after 90 days (170%) and declined afterward, but still remained above the posttreatment values. The changes shown in Figure 2 were statistically significant by nonparametric, repeated-measures ANOVA (P < .01). No statistically significant changes in the VEP phase of the treated patients were found throughout the follow-up. In the untreated controls, no significant changes in VEP amplitude and phase were found throughout the follow-up. The magnitude of VEP amplitude changes found during the follow-up in the treated patients was significantly greater than that observed in the test–retest assessment either in the untreated controls or in the treated patients before inclusion in the study (see Methods).
Table 2 reports the variability of VEP measurements obtained in 2 VEP recording sessions recorded 6 months apart in the untreated controls. It can be noted that the difference within tests is smaller and divided between changes above or below the baseline level, as compared with the changes observed in treated patients after NGF treatment.
Test–Retest Variability Measurements for VEP 1F Component Recorded in Control Patients Before Inclusion in the Study

Abbreviation: VEP, visual evoked potential.
Percentage difference in amplitude (µV) between second and first tests.
These last findings are shown in Figure 3, which depicts, as scatterplots, the individual VEP log amplitude changes from baseline (ie, log amplitude at a given recording session minus log amplitude at baseline or log amplitude ratio) observed in each eye of all participants at the different follow-up times. Horizontal lines in the plot indicate the 95% confidence limits for test–retest amplitude changes observed in the same patients before inclusion in the study. VEP amplitude increased following treatment in most of the recordings, indicating that the general trend toward an improvement was observed in all individual patients. In addition, in most of the recording sessions the VEP amplitude increase was higher than the 95% confidence limits established for test–retest variability observed in the same patients before the study. MRI evaluation, performed at baseline and after 180 days in the treated and untreated children, did not show any significant change in tumor size and volume after NGF treatment. Both morphometric and morphological evaluations on the optic pathways after 180 days did not show any changes compared with the baseline (Table 1).
Discussion
We evaluated safety and efficacy of NGF, administered via the conjunctiva, as a potential neuroprotective factor for severely damaged optic pathways in children suffering from OGs. Although spontaneous regression of optic pathway gliomas associated with significant visual improvement may occur in rare patients, 30 we are unaware of any cases in which children with vision in the range of our patients spontaneously improved. Thus, we do not believe that the clinical and electrophysiologic improvement observed in our patients were spontaneous, but on the contrary, we suppose that this improvement was related to the signaling effect on damaged but still viable RGCs exerted by NGF. This effect resulted in an increased visual response from these cells.
Our study demonstrated that in treated patients the functional electrophysiological results showed a significant improvement of the flicker-evoked VEP amplitudes following NGF administration. The magnitude of such improvement was far greater than the test–retest variability observed either in treated patients before their enrolment or in untreated controls. The prolonged and sustained time course of VEPs amplitude changes following a short NGF treatment course (10 days) suggests a sequence of molecular events into the surviving RGCs leading to a recovery of their physiological responses.
It is known that RGCs express NGF receptor (TrkA) and that NGF, binding to TrkA, upregulates Bcl-2 protein, which protects cells from apoptosis by preventing caspase activation. 31,32 Furthermore, intravitreal NGF delivery to the retina and optic nerve is crucial to the survival of RGCs and for functional recovery of the retina following ocular ischemia and hypertension in experimental animal models. 15,33 Last, conjunctivally applied NGF has been shown to reach sharply the retina and optic nerve where this neurotrophin exerts its biological activities. 19
Ten days of eye drop NGF administration in 5 blind or severely visually impaired children resulted in a significant improvement in VEP responses. Despite any attempt to control the effects of tumor progression on optic pathways by medical and chemotherapy treatment, these patients had progressive visual loss and severe abnormalities in their VEP responses. These electrofunctional abnormalities reflected a dysfunction of the innermost retinal layers, delay in visual cortical responses, and delay in neural conduction along postretinal visual pathways related to RGCs and optic nerve degeneration. The significant improvement of VEPs observed in NGF-treated patients suggests a functional recovery of RGCs and an improvement of neural conduction along the postretinal visual pathways. These effects confirm the key role of neurotrophins in modulating RGCs function and visual cortical neuronal activity reflected by receptive field size and response latency. 34-36
Improvement of VEP amplitude persisted for about 180 days after discontinuation of treatment, indicating that changes induced by NGF had a prolonged duration. This prolonged and stable NGF effect may be related not only to a protective activity against neural apoptosis but also to the formation of new neural pathways, since it is known that NGF promotes neuronal repair and axonal regeneration. 37-39 NGF acts on different levels to promote neuronal recovery following ischemic, inflammatory, and traumatic injuries: through a neosynaptogenetic mechanism, by directly affecting precursor cells, and/or by induction of other growth factors, such as BDNF, whose neuroprotective effects on visually evoked RGC responses after optic nerve section has been reported. 13,40-43 These different neuroprotective mechanisms exerted by NGF may cause the significant improvement of VEP response observed during and after NGF treatment.
In our patients, the electrofunctional changes of VEPs after NGF administration were also followed by some clinical ameliorations, without any systemic or ocular side effects. Reporting on subject improvements was difficult to obtain in these subsets of patients since their residual sight was almost null. Nevertheless, we asked them or their parents to report on any new visual sign or behavior attributable to the improved visual function. Different from untreated controls, who never reported subjective symptoms of visual improvement during the follow-up period, all children treated with NGF had phosphenes perception at night, reporting figures’ shapes instead of light only, and, in 1 case, attempting watching TV. In this last case, we assumed the child was trying to involve a part of the peripheral temporal visual field that was still sensitive to visual stimuli. All these signs could be considered as indicators of partial visual recovery after NGF administration, supporting the hypothesis that conjunctivally applied NGF can reach easily the retina and the optic nerve where this neurotrophin exerts its neuroprotective effects on residual viable optic pathways, as previously reported both in experimental animal models and in adult patients with severe glaucoma. 18,19
In conclusion, this is a first step toward the development of a large clinical project aimed at evaluating the potential effectiveness of NGF eye drop administration for improving visual function in patients with low-grade OGs affecting the optic pathways. The current preliminary findings and the ease of administration of the drug make it worthwhile to be investigated further, mainly in OG patients with better baseline visual functions, in order to explore more thoroughly the benefits of NGF on visual function recovery. Although further controlled, randomized, double-blind studies are needed for a better understanding of the neuroprotective mechanisms of this neurotrophin, eye drop NGF administration appears to be a promising rescuing strategy for the treatment of children with different neurodegenerative diseases that involve the optic pathway.
Footnotes
Benedetto Falsini and Antonio Chiaretti contributed equally to this study.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
