Abstract
Background
Osteoporosis is one of the detrimental effects of spinal cord injury (SCI), leading to bone loss. It has already been established that superparamagnetic nanoparticles when exposed to an external magnetic field (MF) show strong magnetisation and promote locomotor recovery.
Purpose
The aim of the present study is to explore the role of magnetised nanoparticles in ameliorating SCI-induced osteoporosis.
Methods
The rats were divided into Sham, SCI, SCI+MF, SCI+V, SCI+NP and SCI+NP+MF groups. A complete transection was performed at the T13 level, followed by iron oxide nanoparticle implantation along with MF exposure for 7 or 14 days.
Results
A significant increase in locomotor score was evident at day 5 in all groups except in the SCI+V group, and at day 7, all groups showed a significant increase in Basso, Beattie and Bresnahan score as compared to the pre-surgery score at week 1 of the study period. A significant decrease in bone volume/total volume ratio and trabecular thickness and increase in trabecular separation were observed in all groups as compared to Sham. A significant increase in trabecular thickness in the SCI+NP+MF group as compared to the SCI+MF group was observed after one week. After two weeks, the SCI+MF group showed a significant increase in locomotor scores at days 5 and 13 as compared to the SCI, SCI+V and SCI+NP groups. Bone loss was significantly observed in all groups except SCI+MF, as compared to Sham. Cortical bone showed no significant change at both time points. On histopathological examination of the spinal cord, we observed significant improvement in lesion volume in SCI+MF and SCI+NP+MF groups after one week, whereas only the SCI+NP+MF group showed a significant decrease after two weeks.
Conclusion
Electromagnetic field stimulation partially restored bone architecture after superparamagnetic nanoparticle implantation, which may be due to reduced lesion volume and improved locomotor behaviour.
Keywords
Introduction
Spinal cord injury (SCI) initiates a cascade of systemic effects that disrupts the normal neural, vascular, hormonal, mechanical and molecular balance, resulting in bone loss. In SCI, rapid and sustained bone loss occurs below the level of injury. The outcome is more severe in a complete transection than in partial injury to the cord. In complete SCI, bone loss proceeds at a rate of 1% per week for the first 6–12 months, a rate that is substantially greater than that observed during microgravity (0.25% per week) or prolonged bed rest (0.1% per week). 1 Rapid bone loss persists for 3–7 years after acute SCI, depending on the bone region and parameters studied. The loss of bone tissue varies according to the anatomic region; the most frequently fractured sites are the distal segment of the femur and the proximal segment of the tibia, where the bone tissue is predominantly trabecular. Osteoporosis after SCI mainly results from the loss of trabecular bone quality, which is characterised by decreased mechanical properties, altered chemical composition and disarrangement of the bone tissue microarchitecture. 2
SCI induces an increase in bone turnover, with a bone resorption rate exceeding that of bone formation, resulting in a significant reduction of bone mass and deterioration of bone microarchitecture.3–5 These studies showed that SCI caused a significant decrease in the tibial bone mineral density (BMD) within three weeks. This bone loss was illustrated at the trabecular level by a significant decrease in bone volume/total volume (BV/TV) ratio, the number of trabeculae and the trabecular thickness in the secondary spongiosae. The SCI-induced increase in bone cellular activities was associated with both increased eroded surfaces and bone-formation parameters. A large body of evidence indicates that pulsed electromagnetic field (PEMF) is a safe and non-invasive method to treat osteoporosis. In SCI models, sinusoidal electromagnetic fields (SEMFs) inhibited the osteoblast proliferation and promoted the differentiation and mineralisation potentials of osteoblasts in an intensity-dependent manner, with peak activity at 1.8 and 3.6 mT. 6 Researchers observed that pulsed EMF can reduce trabecular bone loss in the proximal femur resulting from disuse atrophy in SCI. 7 The efficacy of PEMF on osteoporotic bone microarchitecture, bone strength and bone metabolism, as well as its associated signalling pathway mechanism, was systematically investigated in hindlimb-unloaded (HU) rats. In these rats, PEMF ameliorated the deterioration of trabecular and cortical bone microarchitecture and mitigated the HU-induced reduction in femoral mechanical properties, including maximum load, stiffness and elastic modulus. HU also increased trabecular Structural Model Index (SMI), revealing a reduction of plate-like structure, whereas PEMF exposure for four weeks was able to significantly inhibit the deterioration of trabecular bone microarchitecture and decrease in cortical bone thickness. 8 Manjhi et al. 9 observed a statistically significant decrease in bone mineral content, total calcium, phosphorus and bone mineral density levels in the tibia and femur of SCI rats as compared with the Sham group, which was significantly attenuated following MF exposure for eight weeks. Electron microscopic analysis revealed facilitation of microstructural composition and increased compactness in cortical and trabecular parts of MF-exposed bones. These results suggest that chronic (2 h day−1 × 8 weeks) MF exposure to SCI rats is effective in limiting SCI-induced osteoporosis. 9
Magnetic nanoparticles and the use of external magnetic fields are novel technologies to combat traumatic SCIs. Liu et al. proposed that magnetic nanoparticles, which are superparamagnetic in nature, show strong magnetisation in the presence of a magnetic field (MF) and retain no permanent magnetisation upon removal of the field. 10 Pal et al. used a combinatorial approach of superparamagnetic iron oxide nanoparticle administration along with exposure to external electromagnetic fields to promote functional recovery and axonal regeneration in complete transected rats. 11 Utilising this approach, they observed abolition of collagenous scars, attenuation of lesion volume, demyelination and reduction in oxidative stress. The aim of the present study was to investigate the effect of iron oxide nanoparticle (IONP) implantation along with magnetic field exposure on bone microarchitecture at one and two weeks after complete SCI in rats. We hypothesised that this combinatorial approach will restore/modulate bone morphology after a complete spinal cord transection.
Methods
Animals
72 adult male Wistar rats (250–270 g) were obtained from an institutional (All India Institute of Medical Sciences, New Delhi, India) central animal facility. They were housed in a temperature-controlled room (24 ± 2°C) with a light/dark cycle of 14:10 hours. All the rats were equally divided into two batches of 36 rats, corresponding to study periods of 7 or 14 days. In each batch, the rats were randomised into six groups, and a few rats from each group were assessed simultaneously: Sham (only laminectomy); SCI; SCI with MF exposure; SCI administered with vehicle; SCI injected with IONPs and SCI with IONPs along with MF exposure. The rats were provided ad libitum food and water.
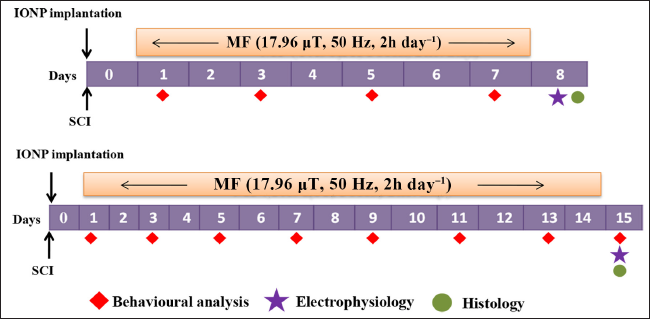
Surgery
Rats were anaesthetised by intraperitoneal injection of thiopentobarbitol (50 mg kg−1 of body weight). Prior to anaesthesia administration, glycopyrrolate injection (30 mg kg−1 of body weight) was given intramuscularly to reduce oral secretions. The anaesthetised rats were fixed in a stereotaxic apparatus (Model 1404, David Kopf, USA). Under aseptic conditions, an incision was made using a scalpel blade from the upper thoracic area to the upper lumber region. Fascia and muscle were removed, the floating ribs corresponding to the T13 vertebral level were located and ack-counting for T11 vertebrae was done to locate the T13 spinal segment. Laminectomy and complete transection of the cord were done at this level with the help of a micro-scissor. A glass seeker was used to ensure that no connection remained between the two ends of the transected spinal cord. 11 A 2 mm piece of IONP-embedded agarose gel was implanted at the site of injury in each of the rats in SCI+NP and SCI+NP+MF groups. In SCI+V rats, only agarose gel was implanted at the site of injury. The skin and underlying tissue were sutured with 1-0 silk sutures (Ethicon, non-absorbable surgical suture, Johnson & Johnson Ltd., Mumbai, India), and antibiotic powder (Neosporin Antibiotic Powder, GlaxoSmithKline Pharmaceuticals Ltd., Bangalore, India) was applied locally. Gentamycin (20 mg kg−1) was administered intramuscularly for five days following surgery. The injured rats’ urinary bladders were manually evacuated four times daily, until the onset of spontaneous evacuation. To prevent dehydration, 0.9% saline was injected subcutaneously as per the requirement.
Synthesis of a Superparamagnetic Iron Oxide Nanoparticle
Formation of a Magnetic Nanoparticle
A citrate-capped superparamagnetic IONP was synthesised by co-precipitation method. Ferric chloride (FeCl3.6H2O) and ferrous sulphate (FeSO4.6H2O) were mixed in deionised water under vacuum conditions. Ammonium hydroxide (NH4OH) was then added slowly dropwise under continuous stirring and heated at 90◦C. Sodium citrate was added, and the reaction was continued for 30 minutes to obtain magnetic nanoparticles. The solution was washed with anhydrous ethanol and re-dispersed in deionised water for further use. To prepare IONP-embedded agarose gel, 25µg ml−1 IONPs were dissolved in ultrapure agarose (30 mg ml−1). The hot gel containing IONPs was then poured into a glass tube of 2 mm diameter and left to solidify at room temperature.
Characterisation of IONPs by Transmission Electron Microscopy
The average size of citrate-capped IONPs synthesised by chemical method was calculated from TEM images. It showed that the average size of nanoparticles was 30 nm. TEM image showed that all nanoparticles had a spherical shape (Figure 1).
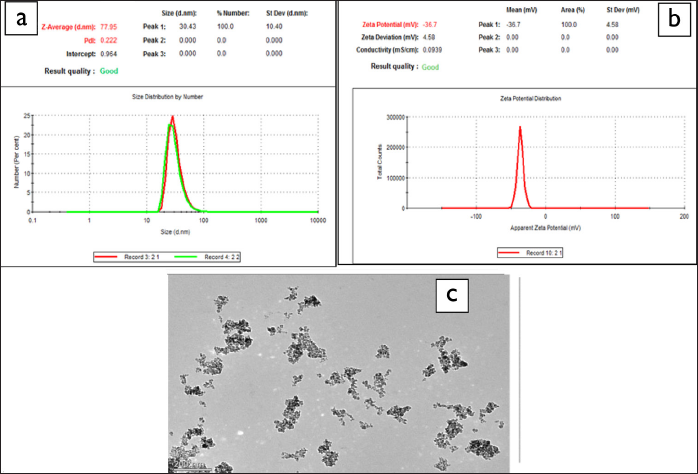
A: Size Distribution of IONP.
Assessment of Locomotion
Locomotor function was assessed using a 0–21 points open field locomotor scale, wherein the rat was left in the open field and its movement was recorded in the camera for four minutes for its ability to use hind limbs simultaneously by two observers blinded to the rat group identity. The movement of three joints, that is, knee, ankle and hip joints; paw placement; weight support and forelimb/hind limb coordination were judged according to a 0–21 point scale. It was used to evaluate the functional motor recovery after SCI, and the data was scored and analysed offline as per the scoring method. 12
Micro-computed Tomography of Tibia
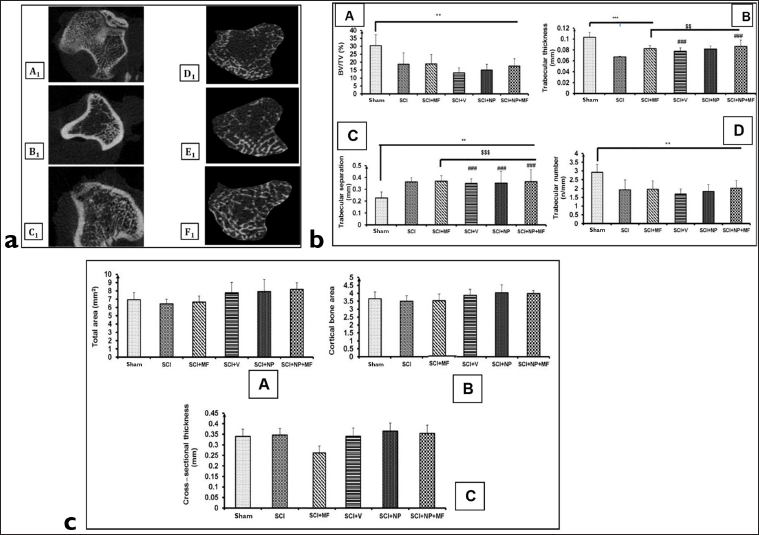
Micro-computed tomography (micro-CT) is a standard and essential tool for quantifying structure–function relationships as well as an imaging tool for high-resolution three-dimensional images. Tibia bone was kept in 4% paraformaldehyde after transcardial perfusion in rats for scanning by the micro-CT method. Tibia was scanned in SKYSCAN instrument using X-ray voltage of 70 kV with 140 vA current, where the rotation step was 0.6◦ and aluminium filter was 1.00 mm. Scanned images (.tiff files) were then reconstructed using NRecon software, which generates transverse images of bones. These images were used for quantitative analysis by drawing regions of interest for trabecular and cortical parts individually using CTAn software (Figure 2). Using the region of interest, trabecular parameters (BV/TV, trabecular thickness, trabecular number and trabecular separation) and cortical bone parameters (total area, cortical area and cross-sectional thickness) were measured and analysed using BATMAN software. 13
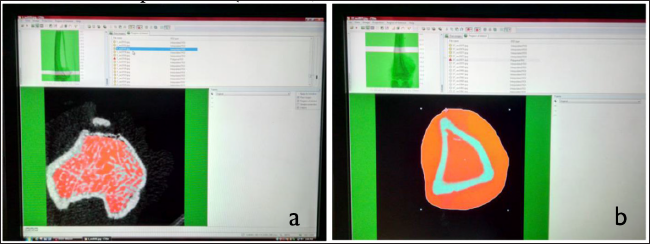
A: For Trabecular Bone Analysis.
Processing for Spinal Cord Tissue
On the 8th or 15th day of the study, rats were transcardially perfused with 4% paraformaldehyde under deep anaesthesia. The spinal cord was taken out and post-fixed in 4% paraformaldehyde for 24 h at 4˚C. A 12 mm long tissue block (with the centre of the lesion as the epicentre) was taken and processed for histology. Tissue blocks were cryoprotected in 15% and 30% sucrose serially for 48 h at 4˚C for cryoprotection. Every alternative longitudinal section (16 µm thick) was taken on the poly-
Assessment of Lesion Volume in the Spinal Cord
The cresyl-violet-stained sections/slides of each rat were scanned at 2X magnification by Nikon NIS software. The total damaged area of the lesion in these sections was traced using Nikon ECLIPSE 80i microscope software (Japan) and measured using NIS-Element software. To calculate the total volume of damaged tissue, the total area was multiplied by the number of sections per spinal cord and the thickness of the single section.
Statistical analysis was performed using SPSS software version 24. The data were presented as mean ± standard deviation. The data were compared between the groups using one-way analysis of variance (ANOVA), followed by Bonferroni post hoc test. Within-group data were assessed by Student’s paired t-test. Repeated-measures ANOVA was used to compare BBB scores within group data. A p value less than or equal to .05 was considered as the level of significance.
Results
One-week Inter-group Comparison
BBB Locomotor Score
In all the rats in all groups, the pre-injury BBB score was 21. After SCI, all the rats in each group exhibited a complete absence of hindlimb movement and moved in the cage with the help of their forelimbs, dragging their hindlimbs.
Post-surgery One-week Inter-group Comparison
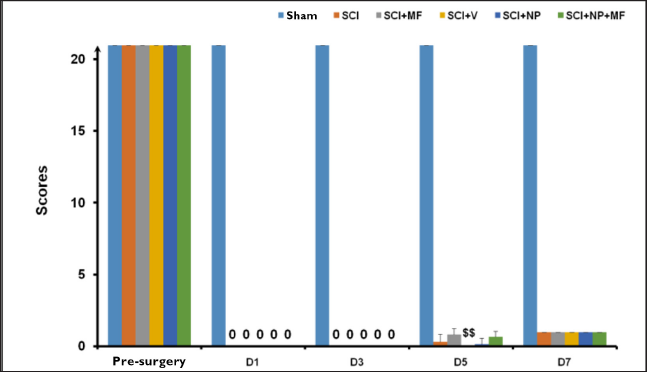
Repeated measures ANOVA determined that the mean BBB score differed significantly between the time points [F(1.005, 35.56) = 174.39, p ≤ .001] among all the groups. Post hoc analysis by Bonferroni correction revealed a statistically significant improvement (p ≤ .001) in hindlimb movement among all the groups at day 7 (D7) as compared to day 5 (D5). The score was significantly increased (p = .005) at day 5 in all groups except the SCI+V group as compared to its pre-surgery score. On day 7, all groups showed a significant increase (p = .005) in the BBB score as compared to the pre-surgery score.
Inter-group comparison by one-way ANOVA followed by Bonferroni correction showed a significant decrease (p ≤ .001) in score in all groups as compared to the Sham group at different time points. The SCI+V group also showed a significant decrease (p ≤ .001) in score at day 5 as compared to the SCI+MF group (Figure 3).
Bone Parameters
The representative images of micro-CT scanning of the tibia after one week of surgery for various groups are shown in Figure 4. As is evident from the images, the trabecular region of tibia showed a significant decrease in the BV/TV ratio [F(5,30) = 8.23; p ≤ .001] among the groups. A significant (p ≤ .05) decrease was evident in BV/TV ratio in all groups as compared to the Sham group. Trabecular thickness also decreased significantly (p ≤ .01) in SCI and SCI+MF groups as compared to the Sham group. However, in SCI+NP+MF and SCI+V groups (p = .001), a significant increase was observed in comparison to SCI. There was also an increase in thickness in the SCI+NP+MF group (p = .004) as compared to the SCI+MF group (Figure 4 II).
Trabecular separation increased significantly (p ≤ .05) in all the groups as compared to the Sham group. SCI+MF and SCI+NP+MF groups revealed no significant difference as compared to the SCI group or among themselves.
Cortical bone parameters—total area, cortical bone area and cross-sectional thickness—showed no significant difference among the groups (Figure 4 II).
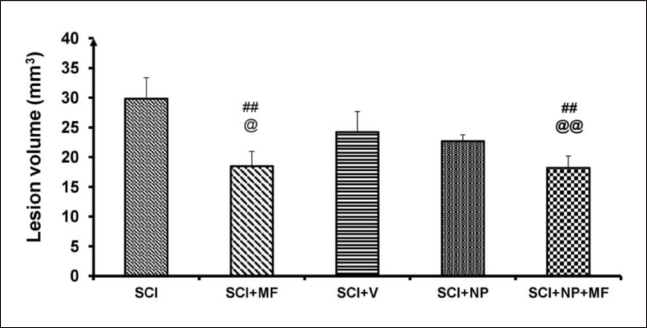
Lesion Volume
Histological analysis of the spinal cord showed lesion of tissue on both dorsal and ventral sides from the site of injury. Cresyl violet staining of the spinal cord revealed cyst formation, gliosis, microcysts, scarring and dead motoneurons indicative of neurodegeneration.
A significant difference was observed among all the groups [F(25,4) = 18.90, p ≤ .001]. Post hoc analysis revealed a significant increase in lesion volume in the SCI group as compared to all other groups (p ≤ .05). The lesion volume also decreased significantly (p ≤ .01) in SCI+NP+MF and SCI+MF groups as compared to the SCI+NP group (Figure 5).
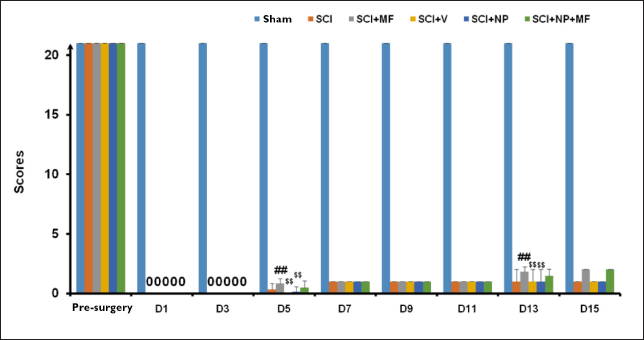
Two-week Inter-group Comparison
BBB Locomotor Score
Repeated measures ANOVA showed a statistically significant difference among all the groups [F(1.005, 35.56) = 174.39; p ≤ .001]. Post hoc analysis by Bonferroni correction revealed a significant improvement (p ≤ .001) in BBB score at days 5 and 7 in all groups as compared to its pre-surgery score.
Inter-group comparison by one-way ANOVA followed by Bonferroni correction showed a significant decrease in score in all groups as compared to the Sham group (p ≤ .001) at all time points. On post hoc analysis, the SCI+MF group showed a significant increase (p ≤ .001) in score at days 5 (D5) and 13 (D13) as compared to the SCI group. SCI+V and SCI+NP groups showed a significant decrease (p ≤ .001) in score at days 5 and 13 as compared to the SCI+MF group (Figure 6).
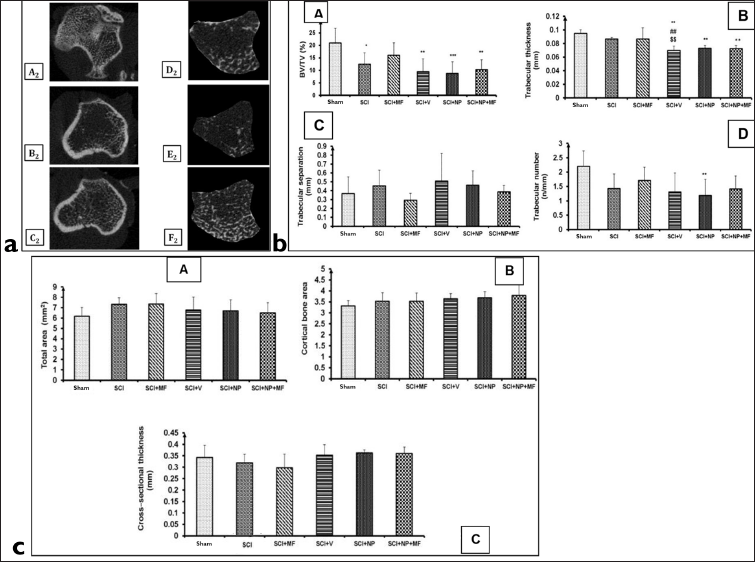
Bone Parameters
The representative images of micro-CT scanning of the tibia after two weeks of surgery for various groups are shown in Figure 7. Trabecular thickness decreased significantly (p = .001) in SCI+V, SCI+NP and SCI+NP+MF groups as compared to the Sham group. There was also a significant (p ≤ .01) decrease in trabecular thickness in the SCI+V group as compared to SCI and SCI+MF groups. The BV/TV ratio significantly decreased in all groups (p ≤ .05) except the SCI+MF group as compared to the Sham group (Figure 7). Trabecular separation and number did not show any significant difference among the groups.
Cortical bone parameters (total area, cortical bone area and cross-sectional thickness) showed no significant difference among the groups (Figure 7).
Lesion Volume
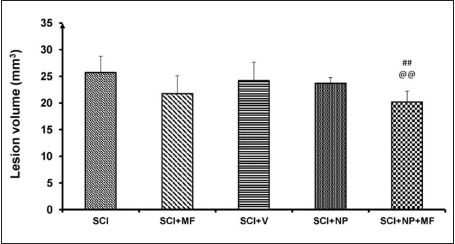
A significant decrease in lesion volume was observed in the SCI+NP+MF group (p = .002) after two weeks as compared to SCI and SCI+NP groups (p = .005). Though a decrease was evident in the SCI+MF group as compared to the SCI group, it was not significant. No significant difference was observed among all the other groups (Figure 8).
Correlation and Regression Analysis
One-week Groups
Stepwise multiple regression analysis showed a significant correlation between BV/TV (%) and trabecular number, trabecular separation and BBB score in one-week study groups (Tables 1 and Table 2) (p < .05 considered as significant).
Multiple Regression Analysis of Bone Parameters (BV/TV and Trabecular Number) for the One-week Study Group.

Correlation coefficient (R) = 0.664.
Multiple Regression Analysis of Bone Parameters (BV/TV, Trabecular Separation and BBB Scoring) for the One-week Study Groups.
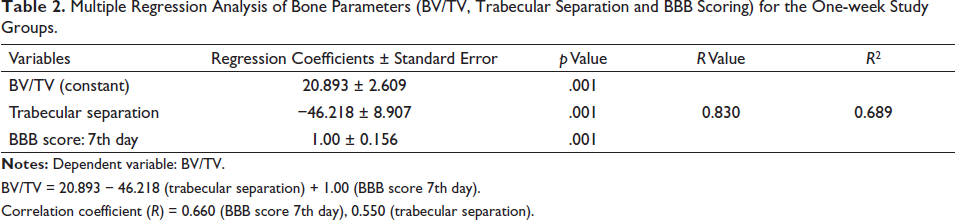
BV/TV = 20.893 − 46.218 (trabecular separation) + 1.00 (BBB score 7th day).
Correlation coefficient (R) = 0.660 (BBB score 7th day), 0.550 (trabecular separation).
Two-week Groups
Stepwise multiple regression analysis showed the most significant and strong correlation between trabecular number, thickness and lesion volume with the dependent variable BV/TV (%) after two weeks (Tables 3–5).
Multiple Regression Analysis of Bone Parameters (BV/TV and Trabecular Number) for a Two-week Study Group.

Correlation coefficient = 0.965.
Multiple Regression Analysis of Bone Parameters (BV/TV, Trabecular Number and Trabecular Thickness) for the Two-week Study Groups.
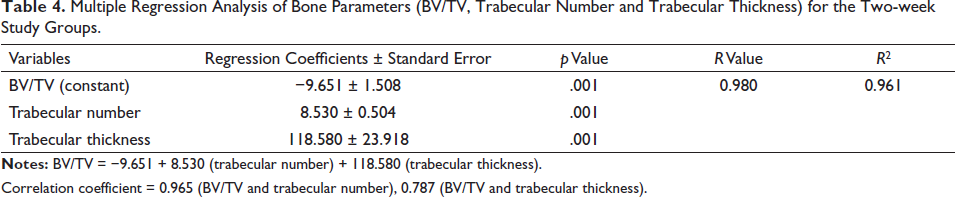
Correlation coefficient = 0.965 (BV/TV and trabecular number), 0.787 (BV/TV and trabecular thickness).
Multiple Regression Analysis of Bone Parameters (BV/TV and Trabecular Number, Trabecular Thickness and Lesion Volume) for the Two-week Study Groups.
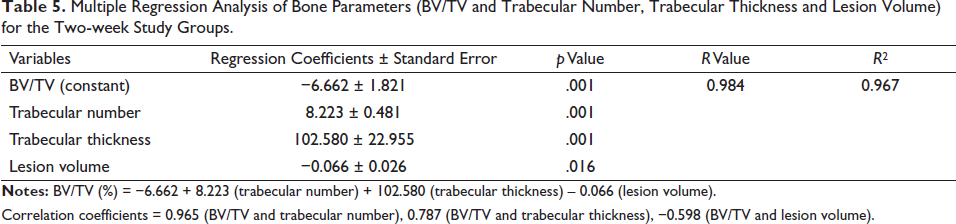
Correlation coefficients = 0.965 (BV/TV and trabecular number), 0.787 (BV/TV and trabecular thickness), −0.598 (BV/TV and lesion volume).
Stepwise regression analysis showed a significant correlation between bone loss [BV/TV (%)] and trabecular number, trabecular separation, thickness and lesion volume after two weeks. It has been clearly interpreted that trabecular number, separation, thickness and lesion volume showed a significant correlation with the dependent variable BV/TV (%) and can be used as prediction parameters.
Therefore, the regression equation is:
BV/TV (%) = −6.662 + 8.223 (trabecular number) + 102.580 (trabecular thickness) – 0.066 (lesion volume)
The dependent variable is BV/TV (%).
Discussion
Bone architecture after SCI has been very well studied and shown to cause severe osteoporosis. Up to 70% of all individuals with SCI undergo fractures in response to minimal trauma at some point following injury. In the present study, we report a significant decrease in the bone volume fraction ratio (BV/TV) of the tibia at both study time points. BV/TV represents the fraction of a given volume of region of interest—total volume of bone or tissue volume that is occupied by bone. A study revealed that BV/TV increased significantly in oxytocin-treated ovariectomised rats as compared to only ovariectomised rats, where it was reduced, suggesting it to be a more sensitive marker for predicting early osteoporosis. 14 The decrease in ratio is associated with a significant decrease in trabecular number and thickness and an increase in trabecular separation. In the present study, a significant decrease in thickness and number and an increase in separation after one week was evident. A similar trend in trabecular parameters was observed after two weeks, but it was not significant. In agreement with our findings, the literature suggests increased bone resorption after SCI due to a significant increase in osteoclastic activity in rats, leading to trabecular deterioration. 15 Though osteoclast activity is more affected than osteoblast activity following spinal cord transection, after 10 days of contusion injury, both osteoclast dysfunction and suppression of osteoblast activity are shown to be responsible for the development of osteoporosis. 16 A dense network of sensory and sympathetic nerve fibres exists within the marrow, periosteum, cortical and trabecular compartments. Immunohistochemical analysis of rodent long bones has shown neuropeptide expression within these nerve fibres as well as on both osteoclasts and osteoblasts. Nerve fibre distribution has been shown to change in response to neurological injury, and sensory and sympathetic denervation also dramatically alters the distribution of immunoreactive fibres within rat bone.17, 18 Bone resorption and formation are regulated through the RANK/receptor activator of the NF-κB ligand (RANKL)/osteoprotegerin (OPG) axis. Increased osteoblast expression of RANKL stimulates osteoclast recruitment and activity and, hence, increases bone resorption. The expression of OPG by osteoblasts, however, antagonises the effects of RANKL and decreases bone resorption. SCI seems to change the osteoblast phenotype in such a way as to promote bone resorption. The change of OPG and RANKL in response to SCI 19 stimulates osteoclastogenesis and osteoclastic bone resorption 20 and induces bone loss. 21 A similar loss of cancellous bone volume and total bone mass is also observed in SCI rats. Thus, the change in RANKL and OPG expression associated with SCI may contribute to the gradual loss of bone that occurs after the injury and the development of osteoporosis. However, the development of osteoporosis after SCI is a complicated process involving both systemic and local changes. The changes in the expression of RANKL and OPG following SCI could be a consequence of systemic changes in growth factors such as insulin-like growth factor (IGF-1), leptin and sex steroids.22, 23 Sensory and sympathetic neuropeptides such as calcitonin gene-related peptide (CGRP), substance P (SP), vasoactive intestinal peptide (VIP) and neuropeptide Y (NPY) are present in bone tissues or bone marrow,24–26 and these neuropeptides are known to be involved in RANKL and OPG production.27, 28 Therefore, the changes in the expression of RANKL and OPG following SCI could also be a result of the loss of these neuropeptides in the bone marrow microenvironment. It appears that mechanical strain inhibits RANKL production and up-regulates OPG production in the marrow stromal cells in vitro. 29 Hence, a lack of mechanical strain after SCI may be another reason for an enhanced RANKL/OPG ratio, leading to increased bone loss. SCI is shown to accompany an inherent change in osteoblast phenotype, resulting in an increase in the expression of RANKL and a decrease in the expression of OPG in osteoblast-like cells. These changes favour increased osteoclast activity over osteoblast activity and may explain the imbalance in bone formation and resorption following SCI. Sahbani et al. showed that inhibition of TGF-β signalling in mice protects trabecular bone integrity after SCI by balancing bone remodelling. 30 However, we observed no change in cortical bone parameters after SCI at both time points. Cortical bone is thick, dense and solid and surrounds the marrow space and mainly serves a mechanical function. This is the part of the bone to which ligaments and tendons attach. It forms the hard exterior (cortex) of bones. Trabecular bone is more active and prone to bone turnover and remodelling as it contains osteoblasts and osteoclasts. Cancellous or trabecular bone is much more porous and, therefore, weaker than cortical bone. Trabecular bone, due to its lesser density when compared to cortical bone, provides only about 20% of the skeletal bone mass, but it is responsible for most of the turnover. Thus, the bone loss in early osteoporosis is mainly trabecular bone loss, as is evident in the present study.
No significant improvement following MF stimulation on trabeculae as well as cortical parameters was evident. Shen and Zhao and Jing et al. studied bone parameters in a hindlimb disuse-induced osteoporosis rat model following magnetic field exposure.7, 31 They observed amelioration of trabeculae and cortical bone microarchitecture after four weeks of stimulation. Zamarioli et al. showed partial restoration of the deleterious effects of SCI-induced bone loss in paraplegic rats using low-intensity pulsed ultrasound after 10 days. 32 However, in the present study, we did not observe any beneficial effects of MF stimulation, which may be due to the short duration of the intervention with respect to bone tissue.
The SCI+NP+MF group showed a significant decrease in BV/TV, trabecular number and increase in trabecular separation as compared to the Sham group. Although we did not observe any statistically significant improvement in the BV/TV ratio, the SCI+NP+MF group showed a significant increase in trabecular thickness and a decrease in trabecular separation as compared to the SCI+MF group after one week, suggesting partial restoration of bone loss. We propose that SCI leads to bone loss through an increase in the expression of RANKL and a decrease in the expression of OPG in osteoblast-like cells, favouring increased osteoclast activity and an imbalance in bone formation and resorption following SCI.15, 16 In the SCI+NP+MF group, partial restoration of bone loss may be due to attenuation of lesion volume, muscle unloading and decreased osteoclast activity over osteoblast activity.
We observed a significant correlation between BV/TV, indicating bone loss and trabecular number, trabecular separation, locomotor score and lesion volume. Stepwise multiple regression analysis explained that bone loss is correlated with trabecular number, separation and locomotor score at one week. However, at week 2, a significant correlation was observed between BV/TV and trabecular number, trabecular thickness and lesion volume.
In a study done by Pal et al., IONPs (25 µg ml−1) were embedded in 3% agarose gel and implanted at the site of transection. 33 The rats were subsequently exposed to an electromagnetic field (50 Hz, 17.96 µT for 2 h daily for five weeks). Locomotor and sensorimotor assessment, along with histological analysis, confirmed a significant reduction in lesion volume and functional recovery observed in rats with IONP implantation and electromagnetic field exposure. No collagenous scar was seen, and IONPs were confined in the immediate vicinity of the lesion intracellularly. Further, in vitro experiments also confirmed no cytotoxic effects of IONPs on cell survivability. However, a significant decrease in H2O2-mediated oxidative stress was evident in the medium containing IONPs, indicating their free radical–scavenging properties. These findings indicated a therapeutic role of IONPs and MF in SCI and possibly other neurodegenerative disorders mediated by reactive oxygen species. 11 Further, Pal et al. showed a significant increase in the expression of the growth-associated protein GAP-43, increased sprouting from mature neurons and axons, significantly less demyelination and the presence of more myelinated fibres at the lesion site. These findings highlight the potential of IONPs, along with MF exposure, in promoting neuroregeneration after SCI. 33 In the present study, however, only partial restoration of bone architecture was observed after giving the combinatorial interventions, which may be due to the shorter duration of interventions of one or two weeks after SCI. We propose that longer-duration studies for at least five weeks, along with analysis of OPG, RANKL and related neuropeptides, would add substantially in elucidating the role of the combinatorial intervention on bone microarchitecture after complete SCI in rats.
Conclusion
This is a novel study wherein the beneficial effect of IONP implantation in combination with exposure to electromagnetic fields on bone loss in complete SCI rats has been shown. The present study has opened new avenues for providing further mechanistic experimental evidence, as this combinatorial approach seems to have immense therapeutic potential.
Footnotes
Authors’ Contribution
AC: Planning the work, analysing and interpreting the data and writing the manuscript.
KP: Helping in micro-CT scanning of the tibia.
SR: Formulation and characterisation of superoxide iron oxide nanoparticles.
SCY: Formulation and characterisation of superoxide iron oxide nanoparticles were performed in his laboratory under his guidance.
SV: Performed all statistical analysis.
KPK: Interpretation of data.
SJ: The main idea of this work was conceived by her. The whole work was done under her supervision, and she helped in writing the manuscript.
Acknowledgement
We would like to thank Mr. Sanjeev Beniwal for helping in post-operative care of rats and Mr Purushottom Samal for histological staining at the Department of Physiology, All India Institute of Medical Sciences, New Delhi, India and the Electron Microscope Facility for EM characterization of nanoparticles.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The micro-CT of bone was done in the Central Drug Research Institute, Council of Scientific and Industrial Research, Lucknow, Uttar Pradesh, India. Spinal cord injury and iron oxide nanoparticle work were funded by All India Institute of Medical Sciences, New Delhi, India.
Statement of Ethics
The study was approved by the Institutional Animal Ethics Committee (Ethical number: 936/IAEC/2016) and was performed in accordance with the Laboratory Animals Welfare Act, the Guide for the Care and Use of Laboratory Animal (National Institutes of Health, Bethesda, MD, USA) and EU Directive 2010/63/EU for animal experiments.
