Abstract
Background:
Magnetic field (MF) stimulation has the potential to reduce secondary damage and promote functional recovery after neural tissue injury. The study aimed to observe the effect of very low intensity (17.96µT) MF on general body condition, secondary damage, pain status, and locomotion.
Methods:
We exposed rats to MF (2 h/day × 3 weeks) after 6.25 mm contusion spinal injury. Locomotor behavior was evaluated by BBB score, pain assessment was done by recording threshold for tail flick, expression of voltage-gated calcium channels and extent of secondary damage in the spinal cord was assessed by immunofluorescence and Cresyl violet staining, respectively.
Results:
A significant (p ≤ .001) improvement in bladder function as well as BBB score was observed after MF exposure in comparison with sham and SCI over the observation period of 3 weeks. SCI group showed an increase in the threshold for vocalization after discharge, which decreased following MF exposure. Cresyl violet staining showed significantly higher tissue sparing (73%) at the epicenter after MF exposure when compared to SCI group. This was accompanied with a significant decrease in calcium channel expression in MF group as compared to SCI.
Conclusion:
The results suggest facilitation of sensory-motor recovery after MF exposure, which could be due to attenuation of secondary damage and calcium-mediated excitotoxicity in a mild contusion rat model of SCI.
Abbreviations
BBB: Basso, Beattie, and Bresnahan
BMR: basal metabolic rate
Ca: calcium ion
CpG: central pattern generator
DAPI: 4,6-diamidino-2-phenylindole
FITC: fluorescein isothiocyanate
K: potassium ion
mA: milliampere
MF: magnetic field
Na: sodium ion
PBS: phosphate buffer saline
PBSTx: phosphate buffer saline triton
SCI: spinal cord injury
TTF: tail flick threshold
VAD: vocalization after discharge
VDCCs: voltage-dependent calcium channels
µT: micro Tesla
Introduction
Spinal cord injury (SCI) initiates cascade of events, which include significant electrolytic shifts, involving monovalent (Na+, K+) and divalent (Ca2+) cations imbalance, that contributes to excitoxicity, oxidative stress, and degeneration of neuronal tissue due to secondary injury progression. 1 This leads to loss of sensory, motor, and autonomic functions in body parts below the level of lesion. It also results in intense chronic or neuropathic pain due to central sensitization. 2 Ionic imbalance, specifically mitochondrial calcium overload immediately after SCI, seems to be one of the key players for the neuronal death and glutamate-mediated neurotoxicity. 3
Transcranial as well as external magnetic field (EMF) stimulation has been shown to improve locomotor deficits, osteoporosis, tonic pain, general body conditions, lesion volume, density of serotonergic fibers at injury site,4–8 and attenuate inflammation and microglial activity 9 in complete transection, hemisection, and compression rat models of SCI. A weight drop contusion rat model is one of the most reproducible and reliable animal model for studying effectiveness of any therapeutic strategy in SCI as it closely mimics human injuries.10, 11 Exposure of severely contused SCI rats to pulsed EMF facilitates functional recovery by reducing oxidative stress, inflammation, and increasing expression of HSP70. 12 However, the effect of MF on calcium-mediated cytotoxicity is not clear. Thus, objective of the study was to observe the low intensity MF stimulation effect on general body condition, locomotion, pain, and calcium imbalance in mild contusion SCI rats.
Methods
Adult female Wistar rats weighing between 200 and 250 grams were used for the study. Animals were obtained from Central Animal Facility (AIIMS, New Delhi, India) and kept in a room having controlled temperature (25 ± 2 °C) and light: dark cycle (14:10 h). Ad libitum water and food pellets (Ashirwad Industries Ropar, India) were provided. Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA, Animal Welfare Division, Ministry of Environment, Forest and Climate Change, India) and Institutional Animal Ethics Committee (ethics no 966/IAEC/16) approved the study. It was a 21 day protocol and after recording all the baseline behavioral recording, the animals were divided into three groups randomly: Sham (n = 6), only laminectomy was performed after anesthesia (Pentabaritol 50 mg/kg b.wt); SCI (n = 6): laminectomy was followed by contusion SCI; SCI + MF (magnetic field exposure) (n = 6): one day after the contusion injury of spinal cord, the rats were exposed to MF (EMF, 50 Hz, 17.96µT, 2 h/d for 3 weeks). After 21 days of injury, all the rats were sacrificed (euthanatized) by transcardial perfusion under deep anesthesia.
Food intake, water intake, body weight, and BBB scoring were recorded on the 1st, 5th, 7th, 9th, 13th, and 21st day of the study. The threshold for tail flick and vocalization after discharge was quantified on 21st day. SCI + MF group of rats were kept in a polypropylene cages and placed in MF chamber for 2 h/day for 21 days. After transcardial perfusion, all the spinal cord tissues were stored for histological analysis.
MF Exposure Chamber
The MF exposure chamber (modified form of Helmholtz coil) used has 18 and 8 turns of wire wound on circular formers of two outer and inner electromagnetic coils, respectively. Rats were kept in a polypropylene cage (50 × 20 × 15 cm3) on central area of the chamber that provides uniform MF of 17.96µT, 50Hz.6, 13
Procedure for Inducing Spinal Cord Injury
Under anesthesia (thiosol; 50 mg/kg body weight, i.p.), the dorsal surface of thoracic area was shaved and cleaned with povidone-iodine. An incision was made from the upper thoracic region to the upper lumbar region. Fascia and muscles were removed from T11–T13 vertebral level (laminectomy) to expose spinal T13 level of the cord (sham group). In SCI and SCI + MF groups after laminectomy, mild contusion injury (height of 6.25 mm) was done by New York University (NYU) SCI contusion device. The body temperature was maintained during surgery by controlled heating pads. Suturing of muscle, fascia, and skin was done, and rats were kept back in their home cages. To compensate for blood loss, ringer lactate solution (5 mL, i.p.) was provided to rats along with antibiotics (50 mg/kg b.wt Gentamycin, im; neosporin ointment applied locally) after surgery. We manually expressed bladder 3 times/day until bladder control restored. The day of return of bladder function to normal was noted for each rat of all the groups.
Assessment of Locomotor Functions
The rat was placed in open field for 4 minutes to assess hind limb movements using Basso, Beattie, and Bresnahan scoring scale, 10 which is based on movement of three hind limb joints (hip, knee, and ankle). Video recording of the locomotor behavior of rat was done to perform off-line BBB scoring between 0 and 21 scale by a blind observer.
Assessment of Sensory Functions
Threshold for tail flick is a segmental response, which is mediated via spinal cord. TTF was determined by applying AC current to tail of the rat. The rat was habituated in a Plexiglas restrainer for 30 minutes, before starting the experiment. Two needle electrodes (0.5″ × 27G) were inserted intradermally, approximately 2 mm deep and 2 cm apart into the middle portion of the tail (rostrally and caudally) and were fastened with adhesive tape. In this test, noxious electrical stimulation was applied to stimulate nociceptive afferents (biphasic square wave pulse with frequency 40 Hz, 1.5 ms pulse width and varying strength of current from 0.1 to 10 mA (AD Instruments and Lab Chart 8 [MLS060]). The current at which tail flick occurred was considered as the threshold for tail flick. 14
Vocalization After Discharge
Electrical stimulus strength was increased beyond tail flick threshold in the same rat. A loud vocal sound of higher pitch that outlasted the duration of stimulus was noted as VAD. It represents emotional component of pain, which is organized in limbic structure. 14
Histology
After three weeks of surgery, the rats were deeply anesthetized by injecting thiosol (i.p., 50 mg/kg of body weight) and perfused transcardially with cold saline (4 °C) and 4% paraformaldehyde. The spinal cord along with the vertebral column was isolated from T5 to L5 and post fixed with 4% paraformaldehyde at 4 °C. A 6 mm long spinal cord tissue containing lesion epicenter was taken, cryosectioned (16µm thick), and mounted on poly-L-lysine coated slides. Cresyl violet staining of the sections was done using standard protocol to assess the lesion area extent and volume. The total area of injury in one section was tracked by light microscope and calculated using NIS-Element software (Nikon).
Immunohistochemistry for L-Type Calcium Channels
Sections were collected on poly-L-lysine coated slides and washed with PBSTx three times each for 5 minutes. After that for 2 hours, 10% normal goat serum was added on to the sections for blocking. Then, they were incubated in primary antibody (anti-L-type calcium channel antibody C1353 at concentration 1:200) overnight at 4°C. It was again washed with PBSTx five times each for 3 minutes and then incubated with secondary antibody (fluorescein isothiocyanate conjugated at concentration of 1:400) for 2 hours at room temperature. Finally washing with PBSTx five times for 3 minutes was done. Slides were mounted in 4,6-diamidino-2-phenylindole (DAPI) containing mounting media.
Statistical Analysis
Statistical analysis was performed using Statistical Package for Social Sciences version 15 software (SPSS Inc, Chicago, IL, USA). The data are presented as the mean ± standard deviation. Data of each time point between the groups was compared by the Kruskal–Wallis test with post hoc analysis done by using Dunn’s test with the Bonferroni correction. Statistical significance was accepted at p ≤ .05.
Results
General Body Condition
A total of 18 rats were taken and after surgery, randomly divided into three groups (n = 6 rats in each group). A total of 16 rats (sham = 6, SCI = 5, SCI + MF = 4) survived the total study period of 21 days. The behavioral and histological data of these rats were used for analysis and are presented in the results.
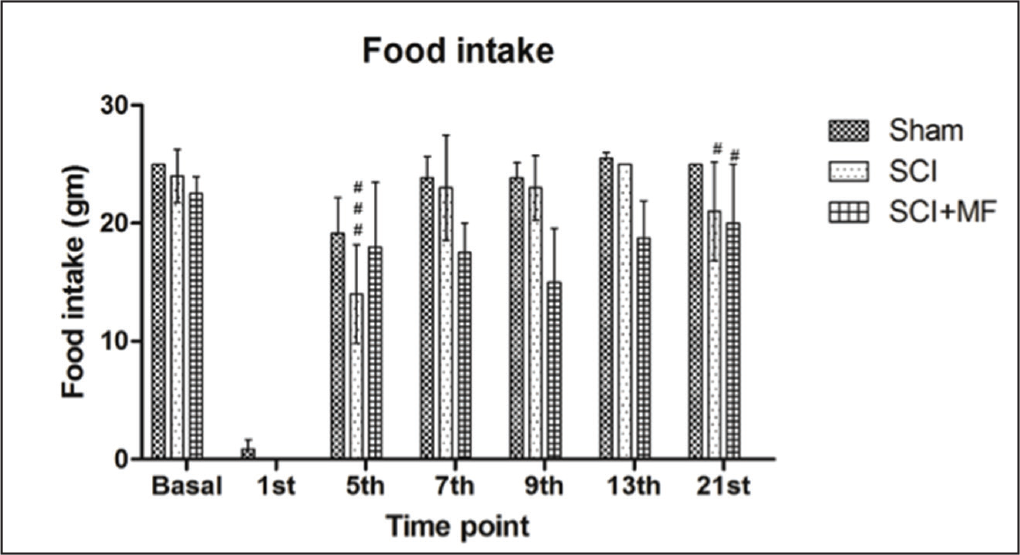
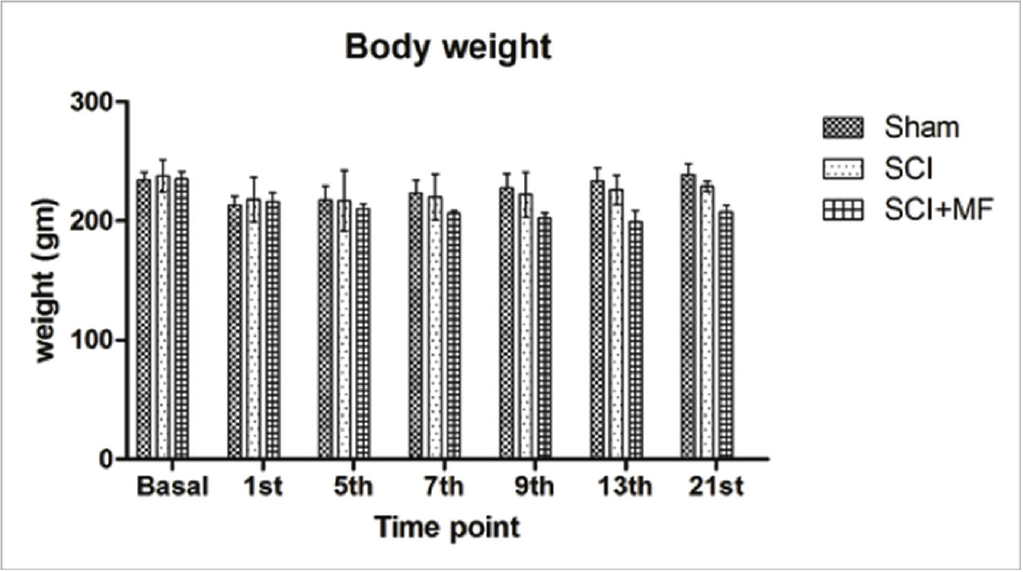
There was no significant difference in baseline presurgery food intake of all the groups, whereas following SCI food intake decreased significantly in all the groups (sham, p = .001; SCI, p = .001; SCI + MF, p = .033). Food intake was nadir immediately after surgery and gradually increased over the days. In SCI group this decrease was maintained till day 5 (p = .039), whereas in SCI + MF group, no statistically significant difference in food intake on any of the postoperative days was observed when compared with basal. Intergroup analysis revealed a significant decrease in food intake in SCI (p = .02) and SCI + MF (p = .03) groups as compared to sham (Figure 1) on day 21 and significant decrease in SCI (p = .001) as compared to SCI + MF on day 5. Body weight did not change significantly among all the groups after surgery (Figure 2).
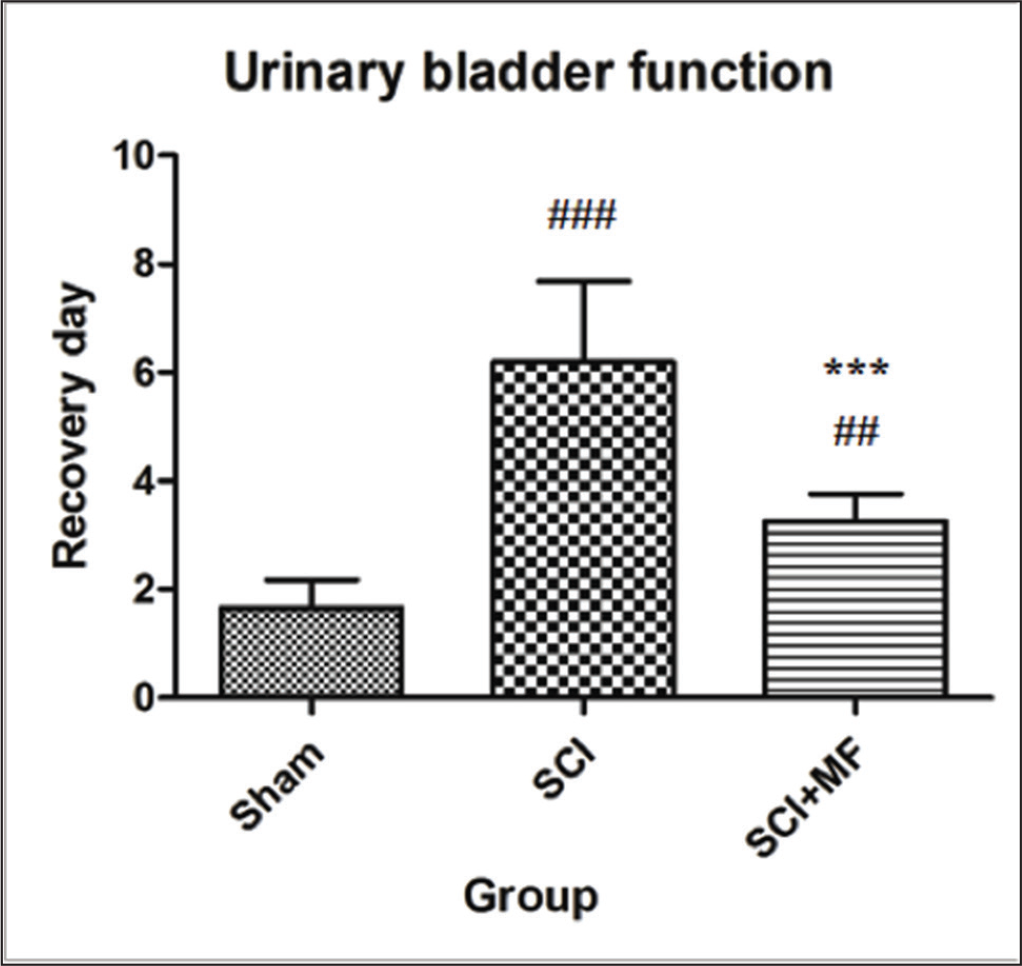
After SCI, there was loss of urinary bladder function in both SCI and SCI + MF groups. Manual evacuation was done eight hourly to survive the rats. A significant effect of intervention was observed on the day of restoration of spontaneous bladder function (χ2 = 12.54; p = .001). In SCI group, post hoc analysis showed that the day of spontaneous evacuation of bladder was achieved significantly later (day 6.2 ± 1.48, p = .0001) in comparison to sham (day 1.6 ± 0.5). MF exposure had significant effect and in SCI + MF group, bladder function recovered significantly (day 3.25 ± 0.5; p = .0002) earlier in comparison to SCI group (Figure 3).
Locomotor BBB Score
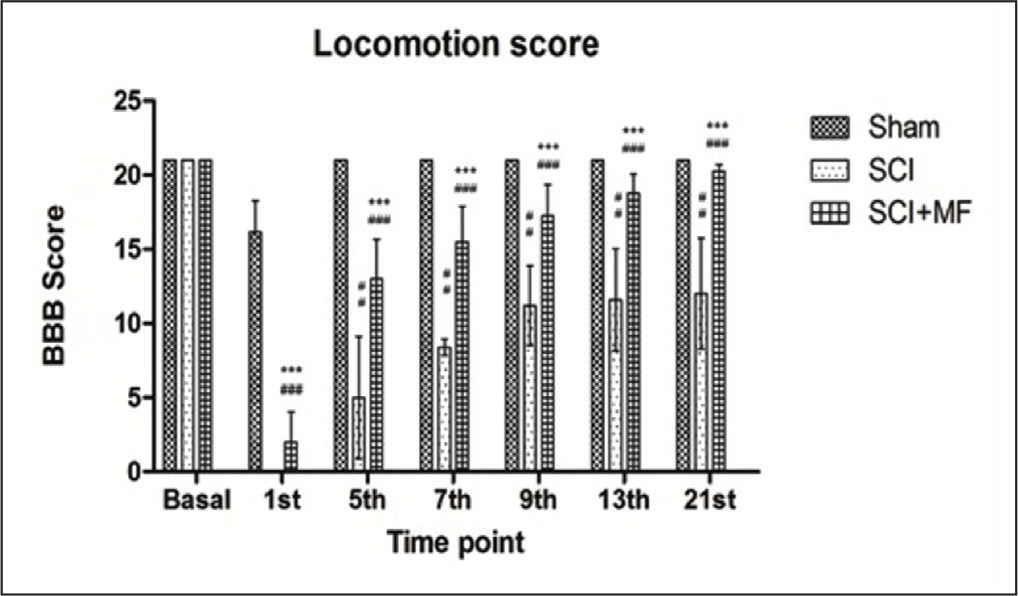
Bilateral hind limb paralysis occurred in all the rats of all the groups after spinal cord contusion injury at thoracic level. BBB score was significantly different (p ≤ .001) among all the groups following surgery. In SCI + MF and SCI groups, a significant drop of BBB score was observed after injury from the basal as well as in comparison to sham group. Over the observation period of 3 weeks, the score increased gradually in both the groups but was maintained at a lower level (p ≤ .001) in comparison with sham. MF stimulation significantly improved hind limb function starting from day 1 of surgery. In MF group, the BBB score was significantly higher (p ≤ .001) at all time points during 3-week study period as compared to SCI group, suggesting beneficial effect of intervention (Figure 4).
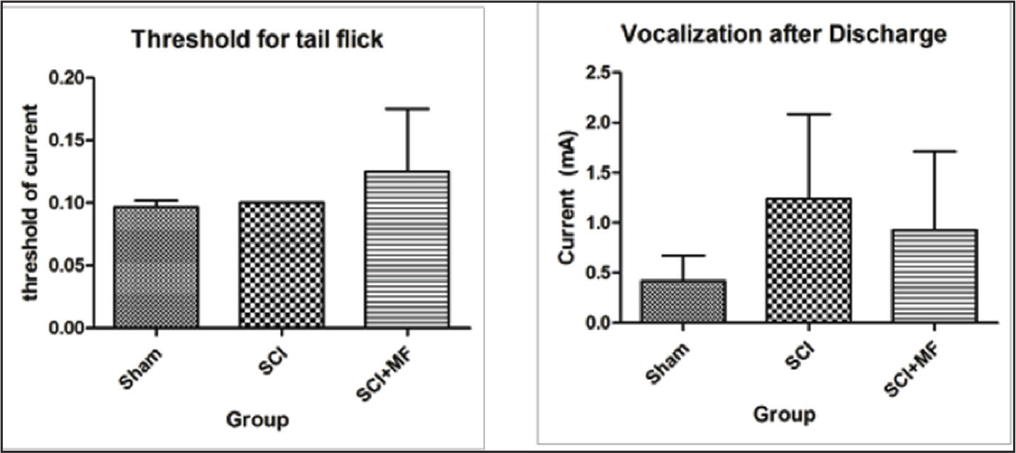
Pain Threshold
The sensory as well as emotional components of pain were assessed by recording threshold for tail flick and vocalization after discharge. There was no significant (χ2 = 4.05, p = .13) difference in the threshold for tail flick among all the three groups (Figure 5). However, on post hoc analysis, on day 21, a significant increase in SCI + MF group (p = .03) was observed as compared to sham. When vocalization after discharge threshold was compared between groups, there was a trend for an increase of the threshold in SCI group and a decrease in MF exposure group, but it was not statistically significant (χ2 = 2.65, p = .26).
Histology and Immunohistochemistry
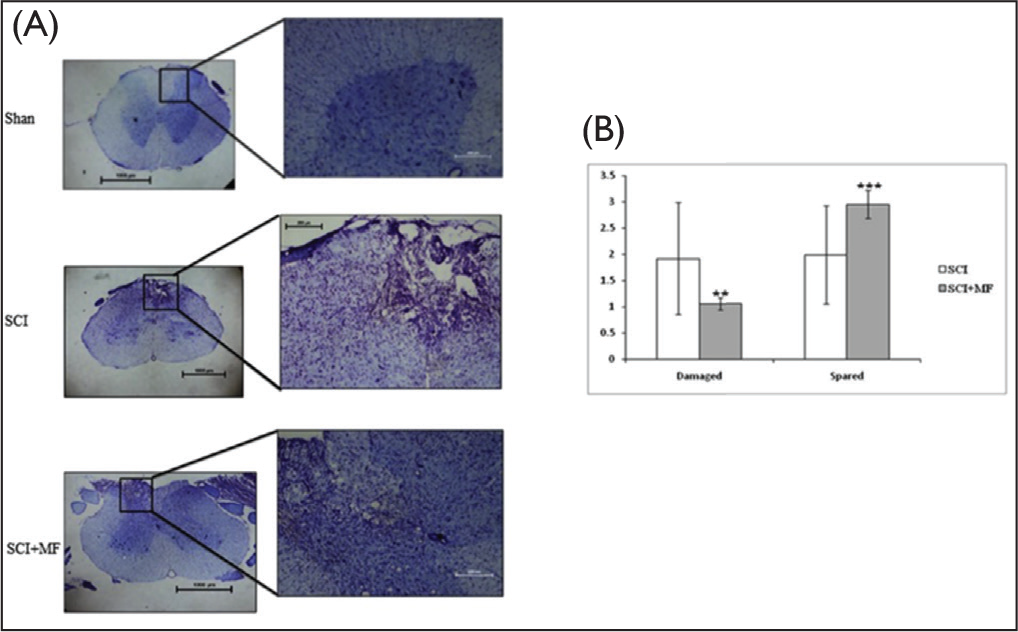
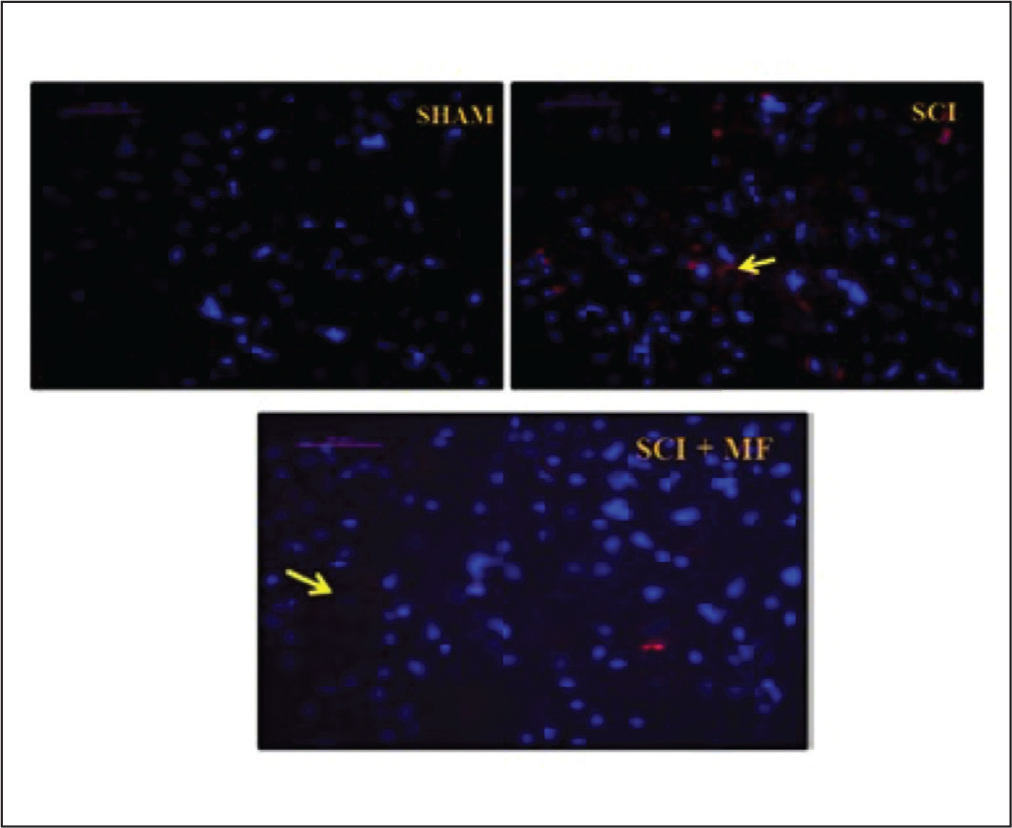
Morphological analysis of the spinal cord revealed presence of lesion in the center and sparing at the periphery of the tissue. In Cresyl violet staining, the central lesion area depicted cyst formations, gliosis, zones of partial demyelination, microcysts, scarring, and was mostly localized to dorsal horn. In SCI group, 51% fibers were spared, whereas following intervention of MF exposure in SCI + MF group, it increased to 73%. A statistically significant (χ2 = 8.43, p = .004) difference in total damage area of spinal cord was observed when comparison was done between SCI + MF and SCI groups. A significant decrease in the damaged area following MF exposure for 3 weeks was evident (Figure 6). Immunohistochemistry for L-type voltage-dependent calcium channels (VDCCs) in spinal cord tissue sections was performed to assess role of calcium in excitotoxicity. DAPI staining revealed calcium localization adjacent to the nucleus, on the cell membrane. A significantly higher expression of L-type VDCCs in SCI group was evident in comparison to sham. However, we observed that in the SCI + MF group, the expression decreased in comparison to SCI (Figure 7).
Effect of magnetic field stimulation on voltage-gated calcium channel expression in spinal cord tissue. The expression was decreased in MF group as compared to SCI.
Discussion
SCI leads to intolerable chronic pain besides severe deficits in motor, autonomic, and sensory functions.15, 16 Chronic pain is an unavoidable part of secondary cascade, which ensues after spinal cord injury subsidies and is resistant to analgesics. This gives a reason to find an intervention that could effectively treat chronic pain. In the recent years, magnetic field has shown to improve sensorimotor function, tonic pain and hyperalgesia in complete transection/hemisection models of SCI.4, 5, 17 The current study was designed to understand low intensity electromagnetic stimulation effect on locomotion, chronic pain, and on calcium signaling in mild contusion rat model of SCI. We used a mild thoracic contusion injury model in our study as it mimics best the human condition among all the other SCI models available. SCI is generally followed by a cascade of events including generation of free radical, ionic imbalance and alteration of cellular events which lead to degeneration of cord both rostrally and caudally and thereby disruption in sensory and motor function. We also observed bilateral hind limb paralysis and loss of bladder function immediately after contusion at T13 in all the rats.
Food intake was significantly decreased in spinal cord injured rats. Consistent decrease in food intake was observed till fifth day of injury in SCI group, following which it recovered spontaneously. Since SCI is a type of neurotrauma, thereby a stressful event, initial decrease in feeding behavior in our study was expected. The decrease in body weight and feeding behavior may also be secondary to alterations in gastric motility and emptying.18, 19 During the initial weeks after SCI, even the patients have been shown to have a tendency to lose weight due to muscle atrophy and bone mineral loss resulting in fat free mass loss and lower body mass index. 20 Reduced fat-free mass, sympathetic blunting, cardiopulmonary dysfunction, work capacity reductions, and diminished anabolic hormones also sustain the BMR decrement after the acute phase of SCI. 21 In SCI rats, urinary bladder function remained disrupted for significantly higher time as compared to sham in the present study. Animal studies have exhibited that reflex micturition is mediated by a spinobulbospinal pathway, whereas urethral relaxation is brought about by excitation of the pontine micturition center, which in turn activates descending pathways. This relaxation eventually is followed by an activation of sacral parasympathetic outflow (L6–S1 in rats), which leads to contraction of the bladder, an increase in intravesical pressure and the flow of urine. 22 Thus, SCI at the thoracic level eliminates voluntary and supraspinal control of voiding, leading to an initial phase of areflexic bladder and complete urinary retention. Finally, there is a slow development of automatic micturition and bladder hyperactivity mediated by spinal reflex pathways.23, 24 Thoracic SCI also can cause complete paralysis or motor deficits in the hind limbs depending on the severity of the injury. Paraplegia was observed immediately following mild thoracic injury in the present study, which gradually partially recovered over the observation period of 21 days postinjury, though it was significantly less than sham group, as assessed by BBB score. This gradual recovery in locomotor function is possibly because of the sparing of central pattern generators, which is located at L1–L2 segment in rat, 25 and we gave lesion at T13 spinal segment. Further, 51% tissue was spared in the SCI rats, sprouting from them may have contributed to the spontaneous recovery of BBB score. Spared area after SCI is a prominent marker for tissue integrity and behavioral recovery. 26
Chronic pain is among the most prominent characteristic feature of SCI; however, mild injury to the cord could not alter pain parameters significantly in our study. Spinothalamic pathway, which is mainly located in the ventral spinal cord, is majorly responsible for the pain perception. 27 In mild SCI, there is major damage to dorsal part, mainly consisting of gracile fasciculus and cuneate fasciculus. These are the first-order sensory neurons that relay proprioceptive, fine touch, and vibration sensations to the thalamus, thereby the pain sensation remains unaffected. Under Cresyl violet staining of the lesioned spinal cord tissue, we also observed major damage to the both fasciculi and sparing of dorsal horn as well as ventral and lateral parts of the spinal cord.
MF exposure for 5–8 weeks has been shown to ameliorate locomotor deficits, osteoporosis, tonic pain, and general body conditions in completely transected or hemisected spinal cord rat models.5, 6, 9, 17 MF exposure, for 3 weeks to rats with mild spinal contusion injury, was able to improve general body condition like food intake and urinary bladder function. This is in agreement with previous literature, wherein MF exposure has been shown to improve general body condition and facilitate early recovery of urinary bladder function. 28 There was also a significant improvement in BBB locomotor scale as compared to SCI, though it was lower than sham group. This may be due to 73% sparing of neural tissue, which under the influence of magnetic fields was able to regenerate and compensate for the lost tissue. MF exposure has been shown to activate regenerative processes under both in vitro and in vivo conditions.29, 30 It also has proved beneficial in restoring locomotion, muscle contraction properties, and limiting muscle degeneration in SCI rats. In total, 5%–10% fibers at the epicenter of contusion (T7–9) and clip compression (T1) injuries of spinal cord and 1%–5% descending axons have been shown to be sufficient to restore the segmental local circuits necessary for locomotion.31, 32
After SCI due to ionic imbalance and excitotoxicity of glutamate receptor, calcium concentrations reach critical level, which leads to neuronal loss. We studied L-type VDCCs, which is a marker of excitotoxicity in neuronal cells to unravel the underlying cellular mechanism of the beneficial effects of MF. A significantly higher expression was observed in SCI as compared to sham and SCI + MF groups. High levels of glutamate cause excitotoxicity by facilitating entry of high levels of calcium ions (Ca2+) to enter the cell. 33 Voltage-gated calcium channels mediate calcium influx into living cells upon membrane depolarization and have a critical role in calcium signaling. Increase in alpha-2-delta subunit of voltage sensitive calcium channel in SCI rats has been shown to correlate with development of tactile allodynia of hind paw. 34 MF stimulation (17.96µT, 50 Hz, 2hrs/day for 3 weeks) of spinal cord of injured rats, in the present study, reduced the calcium channel expression. In literature, there are contradictory reports on the effects of EMF on calcium channels. Low frequency-EMF, 1–25 Hz for <180 ms, with 1–2 T intensity reduced the calcium transit from endoplasmic reticulum in F-11 cells. 35 Sun et al. (2016) reported enhanced calcium channel expression of all the subtypes at presynaptic terminal in situ brain slice preparation, thereby enhancing vesicle endocytosis and synaptic plasticity following exposure to low frequency MF (uniform 1 mT MF with 50 Hz frequency) for 8–10 days. 36 Low frequency electromagnetic field (1 mT, 50 Hz) stimulation also enhanced neurogenesis via expression of calcium receptor (Cav1). 37 It is therefore suggested that the expression of calcium channels following MF stimulation may depend upon the model used (in vivo or in vitro), magnetic stimulation parameters, and the design of the study protocol.
Conclusion
The present study shows beneficial role of short-term MF exposure in the contusion SCI rat model on locomotor behavior by preventing calcium-mediated excitotoxicity.
Footnotes
Acknowledgments
We would like to thank Mr Pursottam Samal for histology and Department of Biotechnology, AIIMS, for all the support during this research work. We acknowledge the support rendered by Dr Anushree Gupta, Associate Professor, AIIMS, Delhi.
Author Contributions
S. J.: Conceived the project and majority of the laboratory work has been performed in her laboratory.
S. B. and S. S.: Performed all the experimental works.
S. B.: Wrote the manuscript also.
S. K.: Standardized and helped in performing behavioral experiments.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The study was approved by the Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA), New Delhi, Animal Welfare Division under Ministry of Environment, Forest and Climate Change (Govt. of India) and approved by the Institutional Animal Ethics Committee (Ethics no 966/IAEC /16).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
