Abstract
Background:
Whether ceftriaxone (CEFT) has any added advantage other than its antibiotics effect in stroke as a neuroprotective agent is not known, and this forms the base of this study.
Purpose:
We tried to assess the predictive role of the use of CEFT with respect to outcome in stroke patients.
Methods:
A retrospective chart review was conducted from a stroke registry over consecutive stroke patients admitted at a tertiary teaching institute from January 2017 to December 2018. Patients were categorized into three groups on the basis of antibiotics they received; patients without antibiotic treatment (NAB), piperacillin plus tazobactam treatment, and the CEFT treatment group. The outcome was assessed by the modified Rankin Scale at three months in good (0–3) and poor outcomes (4–6).
Results:
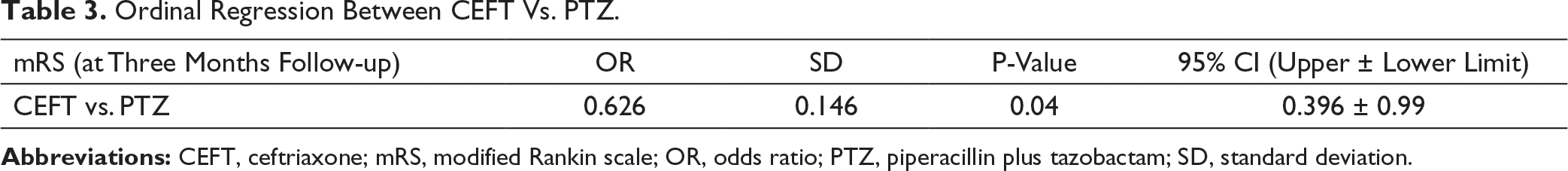
A total of 390 stroke patients were analyzed with ages ranging between 20 and 95 years and 151 of them were females. It was found that the severity at three months was significantly lower in those patients who were given CEFT antibiotic (P = 0.04; OR = 0.626; 95% CI [0.396, 0.990]).
Conclusion:
Stroke patients in CEFT-treated group have a better outcome compared to piperacillin–tazobactam therapy or without antibiotics use at three months. This study indicates the possibility of an additional neuroprotective effect of CEFT apart from its antibacterial property.
Introduction
Infection frequently complicates the acute phase of stroke and has adverse effects on its morbidity and mortality. 1 Pneumonia is the most common infection in acute stroke. 2 While high-income countries (HIC) have implemented evidence-based approaches for improving acute stroke care, low- and middle-income countries (LMIC) are struggling to implement the same. The use of empirical antibiotics in stroke has not been adequately studied in LMIC. In the Preventive Antibiotics in Stroke Study (PASS), the authors concluded that preventive ceftriaxone (CEFT) did not improve the functional outcome at three months in acute stroke patients. 3 A recent systemic review by Vermeij et al. concluded that the use of preventive antibiotics did not have any significant effect on the functional outcome of stroke but did reduce the risk of overall infections. 4
None of the trials or the studies includes patients from the LMIC, and hence, the role of antibiotics on the functional outcome of stroke has not been studied in these nations. This study tries to evaluate whether treatment by CEFT can improve the functional outcome in stroke patients who have infectious diseases during hospitalization compared to piperacillin plus tazobactam (PTZ) or no antibiotic (NAB) use.
Methods
This was a retrospective chart review conducted at the Department of Neurology, Institute of Medical Sciences, Sir Sunderlal Hospital, Banaras Hindu University, Varanasi, India. Consecutive stroke patients admitted between January 2017 and December 2018 were included in the study based on the inclusion and exclusion criteria.
Inclusion Criteria
The patients with age ≥18 years having clinical features of a stroke (ischemic or hemorrhagic) with the onset of symptoms within 48 h and modified Rankin Scale (mRS) score ≥1 at admission were included. The diagnosis of patients was based on the clinical history and neuroimaging evidence (computed tomography and diffusion-weighted imaging for hemorrhagic and ischemic strokes, respectively).
Exclusion Criteria
Patients with any of the mentioned conditions such as denying the consent for participation, requiring intensive care unit (ICU) management, pregnancy, use of antimicrobials less than 24 h before admission, requiring multiple antibiotics therapy, hypersensitive to penicillin and cephalosporins, or diagnosed cases of recurrent ischemic stroke, subarachnoid hemorrhage, traumatic brain injury, vascular aneurysm, arterial malformation, infective endocarditis, central nervous system (CNS) infections or chronic liver, and kidney diseases were excluded.
The included stroke patients received antibiotics if they had any of the features such as continuous fever at least for two days after admission with neutrophilic leukocytosis, clinical features qualifying the severe inflammatory response syndrome (SIRS) criteria, 5 patients having colony count threshold ≥10 2 bacteria/mL in females and ≥10 3 bacteria/mL of urine in males with symptoms of urinary tract infection, 6 tachypnea with basal crepitations on the chest auscultation or neutrophilic leukocytosis with neurological deterioration unexplained by other relevant etiology. Patients were not administered any antibiotics if they did not have any of the above-mentioned features.
Antibiotics were initiated within 48 h of the onset of symptoms. Only patients who received CEFT or piperacillin–tazobactam combination were included. Other antibiotics like ceftazidime, meropenem, etc. were used only for a few patients, and these patients were not included here because of statistical issues. Patients who required other antibiotics as per culture sensitivity reports or ICU care were excluded from the study. If rescue antibiotics were used in patients, they were very few and have been excluded from the study. Hence, patients were grouped into three categories: Group 1 patients were without antibiotic treatment (NAB), Group 2 included patients on piperacillin plus tazobactam group (PTZ), and Group 3 patients were on ceftriaxone treatment.
The detailed clinical information including particulars of the subjects, chief complaints, stroke onset, family, personal, dietary history, past history, addiction, severity assessment using National Institutes of Health Stroke Scale, intracerebral hemorrhage score, and mRS score was noted.7–9 General physical examination, cardiological, respiratory, abdominal, and neurological examination findings were also noted. Findings of 2D ECHO (by transthoracic echocardiography), vascular imaging (duplex imaging of carotid arteries, magnetic resonance angiography of extracranial and intracranial arteries), and 24-h in-patient ECG monitoring and serology for vasculitis/infections were recorded. Furthermore, complete blood count, liver function test, kidney function test, thyroid function test, lipid profile, fasting and postprandial blood sugar, glycosylated hemoglobin, urine routine and microscopy, and blood and urine cultures reports were also recorded. These tests/examinations were repeated during the course of antibiotic treatment to improve the objectiveness of antibiotic-related decisions.
The primary outcome variable was the mRS score, which ranges from 0 (no symptoms) to 6 (death) at three months from the day of admission. 9 Outcome was divided into two categories: good (mRS 0–3) and poor (mRS 4–6) outcome.
Statistical Analysis
The baseline data among all three groups were analyzed by using the analysis of variance for continuous variable and chi-square test for categorical data. The outcome of stroke patients in the three groups was calculated using the chi-square test. The comparative outcome of treatment was assessed using ordinal regression with cut-off P-value < .05 and at 95% confidence interval using SPSS software version 25 (IBM SPSS Statistics for windows, version 25.0. Armonk, NY: IBM Corp).
Results
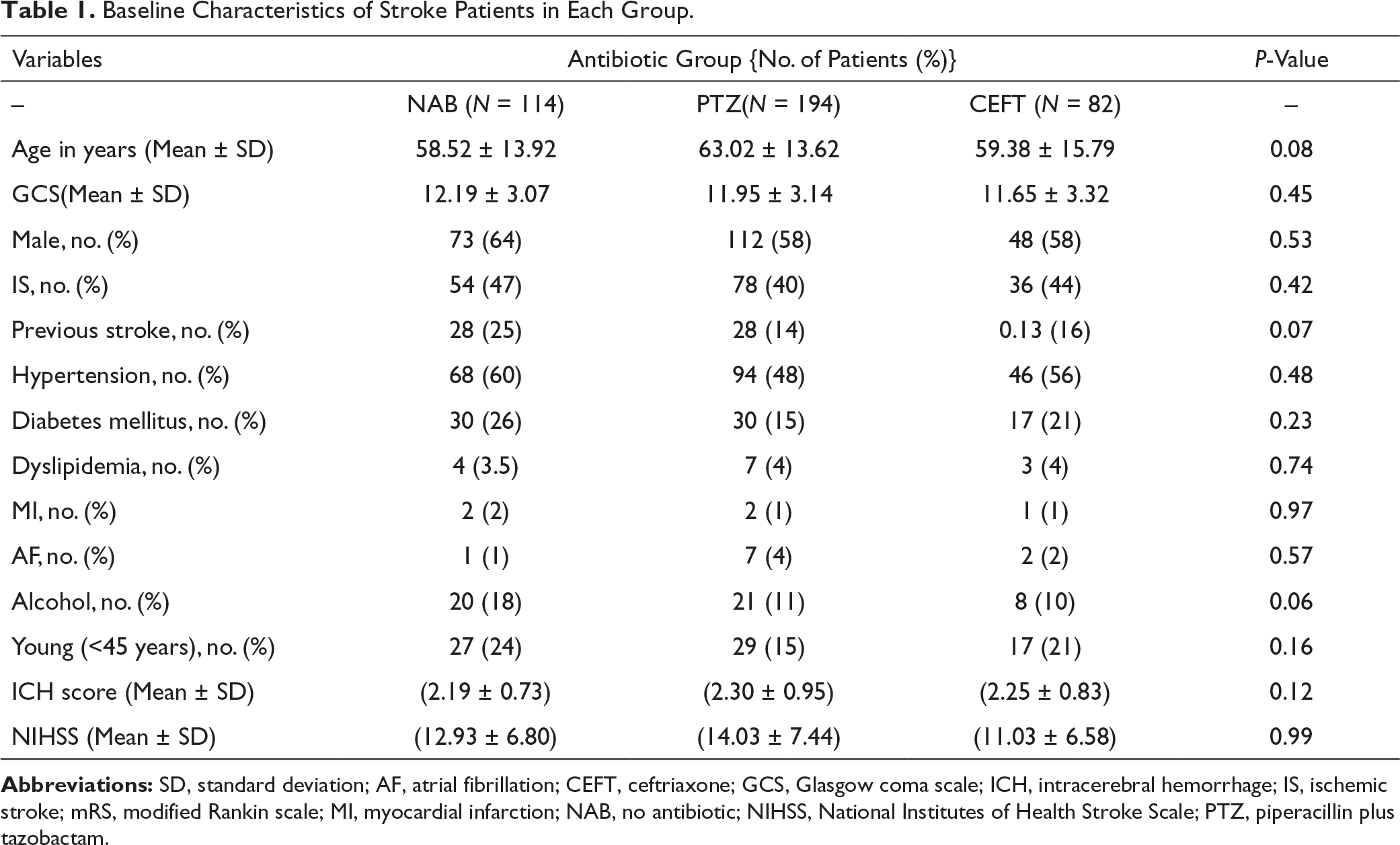
Baseline Characteristics of Stroke Patients in Each Group.
Outcome of Stroke Patients at Three Months in Each Group.

Ordinal Regression Between CEFT Vs. PTZ.
Discussion
This study showed that patients receiving CEFT had better outcomes than patients receiving PTZ or no antibiotics (OR = 0.626). Statistical analysis, performed separately in the ischemic and hemorrhagic stroke, showed that the outcome was nonsignificant. A previous animal study hypothesized that CEFT might have a positive neuromodulation effect in stroke. 10 There is a postulated mechanism that CEFT has an effect on glutaminergic channels and does improve the motor functions in rat model of stroke.11, 12 This result is contrary to the only published randomized trial PASS, where the authors showed that the patient receiving CEFT did not have any improvement in the outcome of the stroke patients, while a posthoc analysis from the same study did show that there was better outcome in CEFT-treated patients who received intravenous thrombolysis. 13 However, the trial did not have any participation from the Asian countries and the CEFT was given just for five days empirically. In our study, CEFT was given for a mean period of 10 days, which might explain the difference in the outcome in our patient population. Several randomized studies along with systematic reviews and meta-analysis studying the role of prophylactic antibiotics in stroke patients have been published and did not show any benefits in the outcome of patients, although studies did treat the poststroke infection. 14 Our study suggests that CEFT, a beta lactam, may have a class effect in the better outcome of stroke patients at three months. However, on subgroup analysis, the outcome of the stroke vis-à-vis class of antibiotics did not reveal significant results which might be because of the small number of sample size in each of the groups.
The strength of the study is that it is one of the first studies investigating the class effect of antibiotics in the outcome of stroke patients at three months to the best of our knowledge.
However, there were certain major limitations. First, this was a retrospective study conducted at a single center. Second, the sample size was small and the external validity of the study was required to be proven in a larger population prospectively. This study does not show any significant difference in the ischemic and hemorrhagic stroke in the subgroup analysis. Individual study separately studying each subgroup of stroke with a larger population would be prudent to draw any further conclusion. Hence, the findings of this study need to be confirmed in a randomized controlled trial in a larger population in hemorrhagic and ischemic stroke separately.
Conclusion
The use of CEFT in stroke patients has a better outcome in comparison to piperacillin–tazobactam and no-antibiotics use. This study indicates the possibility of additional neuroprotective effects of CEFT apart from its antibacterial property, though a larger randomized controlled trial is needed to confirm the finding.
Footnotes
Acknowledgment
We would like to acknowledge the Institute of Medical Sciences for allowing us to conduct this study.
Authors’ Contribution
Manuscript writing, Data Collection, PD; Manuscript Editing, VKS, AK, RNC, NAS, VNM, DJ; Data collection, PG; Supervision of the study, manuscript editing, Statistical Analysis, AP.
Statement of Ethics
Ethical approval was taken from institutional ethical committee. The present research complies with the guidelines for human studies and should include evidence that the research was conducted in accordance with the World Medical Association Declaration of Helsinki. Informed consent was obtained from the participating patients and/or their relatives.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
