Abstract
E-cigarette and vaping-associated lung injury (EVALI) has emerged as a significant public health concern, leading to severe respiratory complications and fatalities worldwide. This systematic review analyses the medico-legal implications of vaping-related deaths, emphasizing postmortem toxicology, autopsy findings, and medico-legal challenges. A comprehensive literature search was conducted across multiple databases, including PubMed, Scopus, Web of Science, Embase, and Google Scholar, to identify relevant studies from 2010 onward. A total of 280 studies were identified, of which 11 met the inclusion criteria, covering 156 cases across multiple countries. The findings indicate that tetrahydrocannabinol (THC) and nicotine exposure vary geographically, with vitamin E acetate being a primary suspect in EVALI-related toxicity. Autopsy results commonly reveal bilateral lung opacities, diffuse alveolar damage, and lipid-laden macrophages, often leading to misdiagnoses such as COVID-19 pneumonia. Forensic investigations face challenges in establishing standardized diagnostic criteria, determining the exact cause of death, and addressing regulatory gaps in e-cigarette formulations. Despite corticosteroid therapy and vaping cessation being the mainstay of treatment, fatalities still occur, underscoring the need for stringent regulatory policies and improved forensic assessment techniques. This review highlights the importance of postmortem toxicology in differentiating vaping-related deaths from other pulmonary conditions, providing critical insights for forensic pathologists, toxicologists, and policymakers. Medico-legal experts must adopt a standardized investigative protocol to accurately document and analyze EVALI-related cases to guide future public health planning. The protocol for this systematic review was registered prospectively with International Prospective Register of Systematic Reviews (PROSPERO) (CRD420251041017).
Keywords
Introduction
The rise of e-cigarette use has sparked global concerns regarding its health consequences, particularly the emergence of e-cigarette and vaping-associated lung injury (EVALI). Since its initial recognition by the Centers for Disease Control and Prevention in 2019, EVALI has been implicated in numerous hospitalizations and deaths, highlighting the potential lethality of vaping-related complications. 1 The exact pathophysiology remains under investigation, but studies suggest that chemical exposure from vaping devices, particularly vitamin E acetate and other toxic substances, plays a critical role in the onset of lung injury. 2
From a forensic perspective, vaping-related deaths present significant challenges in terms of autopsy findings, toxicological analysis, and determination of the cause and manner of death. Unlike traditional cases of respiratory failure or pulmonary toxicity, EVALI-related fatalities exhibit distinct pathological characteristics, including diffuse alveolar damage, lipid-laden macrophages, and acute lung injury. 3 Additionally, forensic toxicologists must navigate the complexities of analyzing various vaping-related compounds, including nicotine, tetrahydrocannabinol (THC), and heavy metals, which may contribute to fatal outcomes. 4
Beyond the pathological and toxicological dimensions, forensic investigations must also consider legal and policy implications. The rapid evolution of the vaping industry, coupled with inconsistencies in regulatory frameworks, complicates efforts to establish clear forensic guidelines. 5 Moreover, distinguishing between accidental, suicidal, and homicidal cases involving vaping-related deaths adds another layer of complexity for forensic pathologists and medical examiners. 6
While EVALI was first recognized in the United States, cases have since been reported globally, including in Canada, the United Kingdom, India, and Japan. Variations in vaping habits, product formulations, and surveillance affect the reported prevalence.
This systematic review aims to provide a comprehensive analysis of the forensic implications of vaping-related deaths, synthesizing available literature on postmortem findings, toxicological profiles, and investigative challenges. By consolidating current knowledge, this review will contribute to improved forensic protocols, enhance the understanding of EVALI’s impact on mortality, and inform regulatory policies aimed at mitigating vaping-related health risks.
Objectives
To analyze postmortem findings in vaping-related deaths.
To review toxicological profiles associated with EVALI fatalities.
To identify forensic challenges in determining cause and manner of death in vaping-related cases.
To propose forensic guidelines related to vaping-related deaths.
Forensic Relevance of this Review
Given the increasing complexity of vaping-related fatalities, this review holds critical forensic relevance by offering insights that can aid in accurate postmortem diagnosis, toxicological interpretation, and the determination of the cause and manner of death in suspected EVALI cases.
Methods
Search Strategy
A comprehensive and systematic literature search was conducted using the following electronic databases: PubMed (for medical and forensic pathology studies) Scopus (for interdisciplinary research, including toxicology and forensic science) Web of Science (for high-impact forensic and medical literature) Embase (for pharmacological and toxicological aspects) Google Scholar (for gray literature, policy documents, and relevant conference proceedings)
Search Terms and Boolean Operators
A structured search strategy was applied using Medical Subject Headings and free-text terms. The search will include variations and synonyms of the following: EVALI-related terms: “EVALI,” “e-cigarette or vaping-associated lung injury,” “vaping-related deaths,” “vaping lung disease.” Toxicological aspects: “Electronic cigarette toxicity,” “nicotine poisoning,” “THC toxicity,” “vitamin E acetate poisoning,” “heavy metal exposure in vaping.” Forensic implications: “Forensic pathology,” “autopsy findings,” “postmortem toxicology,” “cause and manner of death,” “sudden unexplained deaths in vaping.”
A combination of these terms was applied using Boolean operators: (“EVALI” OR “vaping-related deaths” OR “electronic cigarette toxicity”) AND (“forensic pathology” OR “autopsy findings” OR “toxicology” OR “cause of death”).
Search Limits
Timeframe: Studies published from 2010 to present (considering the recent emergence of vaping-related lung injuries)
Language: English
Study Types: Case reports, case series, retrospective studies, systematic reviews, meta-analyses, and forensic investigations
Manual Search and Citation Tracking
Reference lists of selected articles were reviewed to identify additional relevant studies.
Key forensic and toxicology journals were manually searched.
Government and regulatory reports on EVALI-related forensic cases were examined.
Inclusion Criteria
Studies reporting postmortem findings in vaping-related deaths.
Studies discussing forensic toxicology in EVALI cases.
Case reports, case series, retrospective studies, systematic reviews, and meta-analyses.
English language publications.
Exclusion Criteria
Studies without forensic relevance.
Experimental or animal model studies.
Non-English language articles.
Data Extraction and Analysis
Two independent reviewers extracted relevant data, including: Case demographics (age, sex, and region) Cause of death Toxicological findings (nicotine, THC, vitamin E acetate, heavy metals, etc.) Pathological findings (lung histopathology, systemic effects) Legal and forensic challenges in interpretation
PROSPERO Registration
The protocol for this systematic review was registered prospectively with the International Prospective Register of Systematic Reviews (PROSPERO). Details are as follows: Available from:
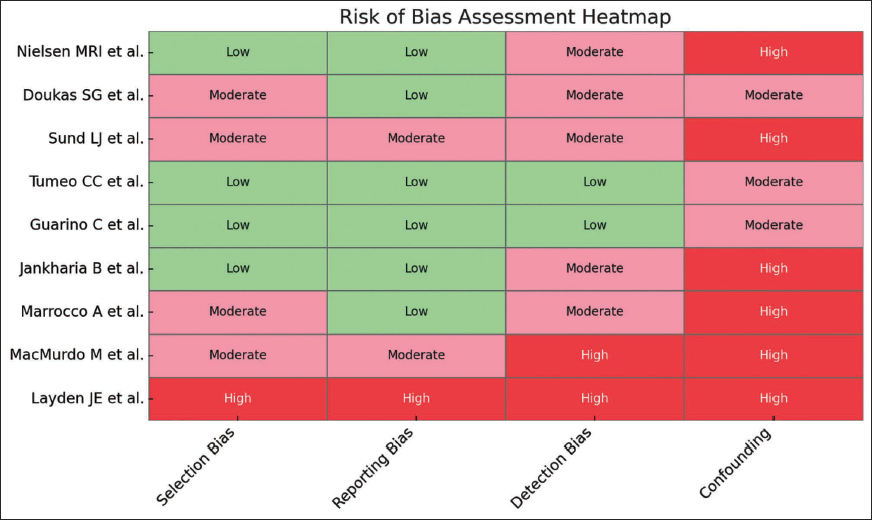
Quality Assessment and Risk of Bias Assessment (Figure 2)
The Joanna Briggs Institute (JBI) Critical Appraisal Checklist was used to assess the quality of the included articles. Since our review encompasses observational studies, case reports, and case series, the JBI checklist is the most appropriate tool for evaluation. The stacked bar chart, as displayed in Figure 1, illustrates JBI checklist scores across five countries, namely Denmark, India, Italy, the UK, and the USA, based on six assessment criteria: Patient demographics, pre-intervention condition, intervention clarity, post-intervention reporting, adverse event consideration, and takeaway message. Variations in stacked segments highlight differences in study rigor and adherence to JBI quality standards. In our review articles, all JBI checklist criteria were satisfied, ensuring methodological rigor and reliability in our findings.
Stacked Bar Chart of JBI Checklist by Country.
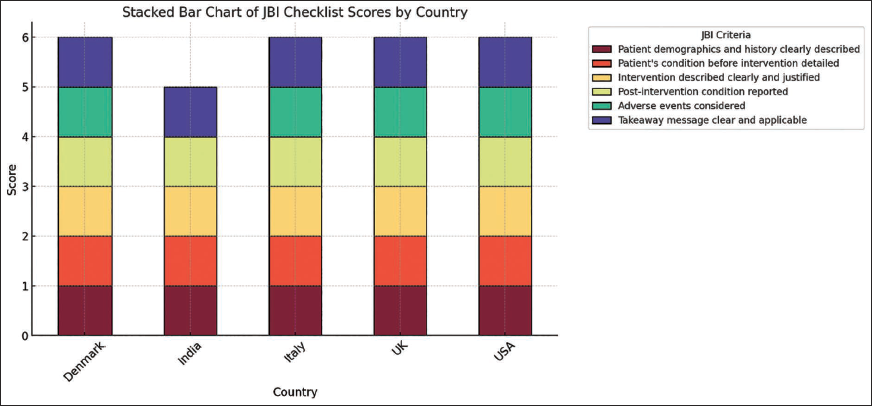
Results
A total of 280 studies were identified through database searching and other sources, with 230 remaining after duplicates were removed. Following title and abstract screening, 110 studies were excluded due to lack of forensic relevance, non-English language, or experimental models. 120 full-text articles were assessed, of which 11 studies met the inclusion criteria, covering 156 cases across multiple countries, including the USA, UK, Italy, Denmark, and India. The studies consisted of four case reports, four case series, two website reports, and one original research study, highlighting geographical differences in vaping exposure and forensic challenges.
Table 1 and the PRISMA flowchart (Figure 3) illustrate the study selection process and key findings. Toxicological findings varied, with THC detection ranging from 0% 7 to 100%, 8 while nicotine-based exposure was more common in non-USA cases. Vitamin E acetate, a suspected EVALI toxicant, was absent in some cases. Pathological findings included bilateral lung opacities, diffuse alveolar damage, and lipid-laden macrophages, with some cases misdiagnosed as COVID-19 pneumonia or infectious lung disease. Chronic EVALI cases displayed organizing pneumonia patterns, complicating forensic differentiation from other pulmonary conditions. Key forensic challenges included diagnostic inconsistencies, a lack of standardized forensic criteria, and systemic involvement beyond lung pathology.
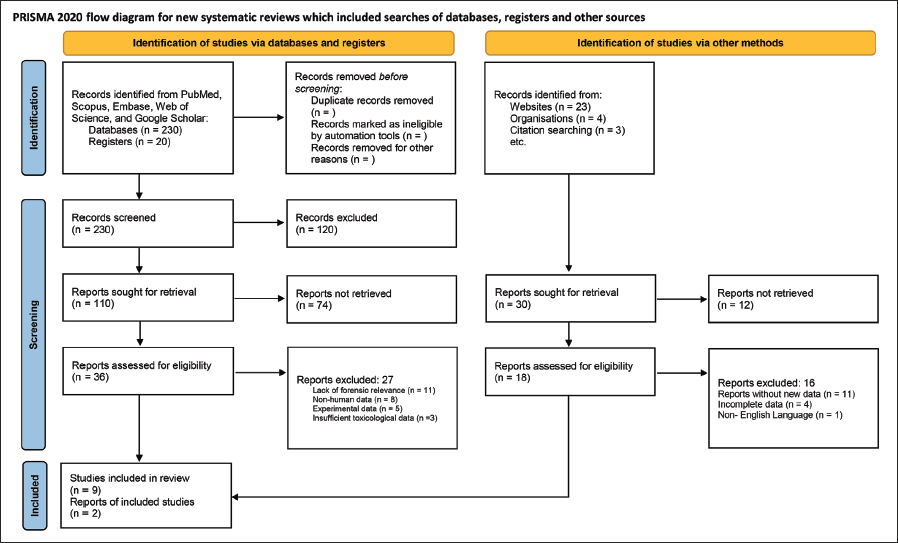
Legal complexities emerged due to off-label THC use, regulatory gaps, and difficulty in attributing the cause of death solely to vaping products. 95% of cases survived with corticosteroid therapy and vaping cessation, but two fatalities 1 were linked to severe lung injury and multi-organ failure, underscoring the forensic importance of postmortem toxicology in determining the cause of death in vaping-related fatalities.
Due to the heterogeneity in study design, sample size, diagnostic criteria, and reporting methods, a meta-analysis was not feasible. Therefore, a systematic review with descriptive synthesis was conducted.
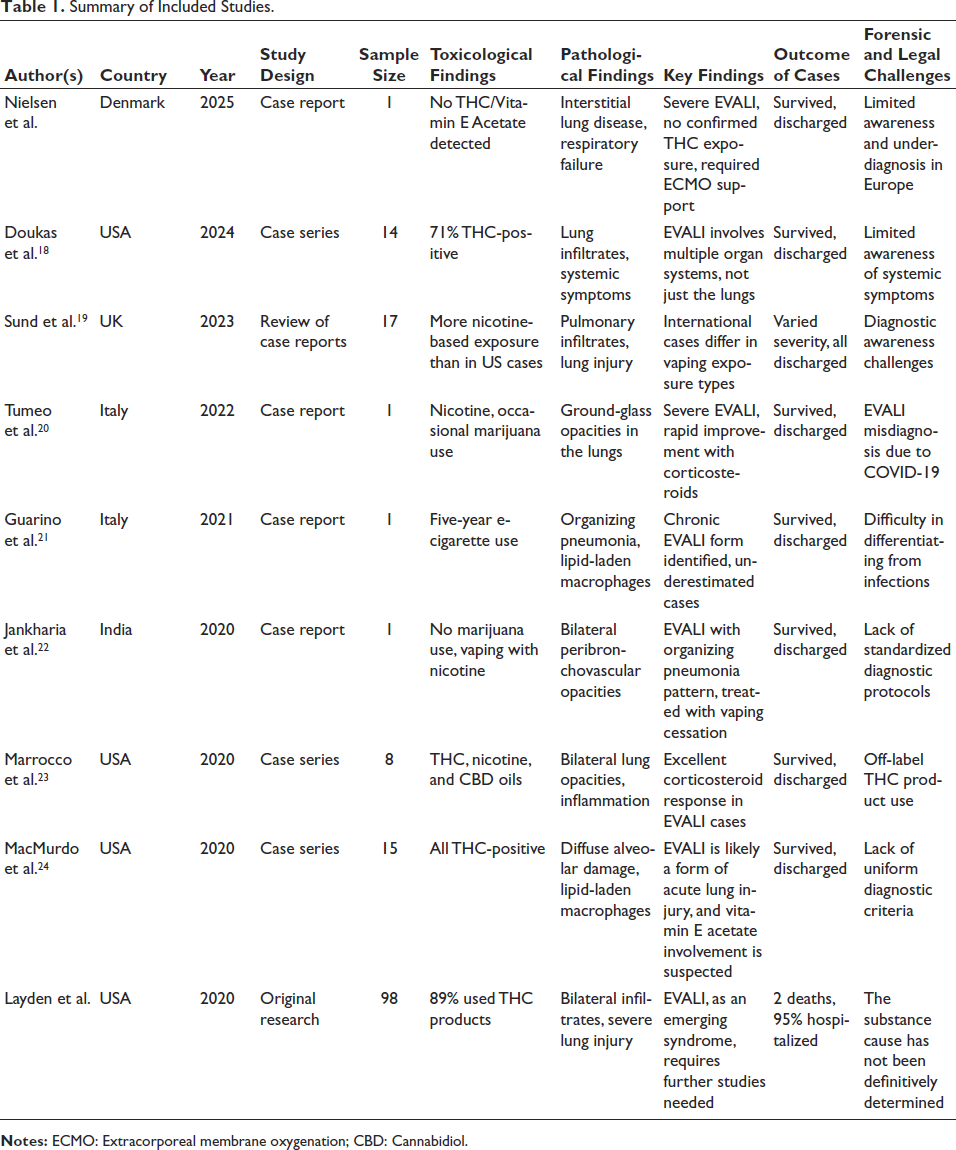
Summary of Included Studies.
Discussion
The findings from this systematic review highlight the global burden of e-cigarette or vaping product use-associated lung injury and the varying patterns of exposure, clinical manifestations, and diagnostic challenges. EVALI remains a multifactorial condition, predominantly reported in the United States, where the adulteration of THC-based vaping products with vitamin E acetate has been widely implicated.1–2 However, international case reports and case series suggest that nicotine-only e-cigarettes can also lead to lung injury under certain conditions. 9 This global variability underscores the complexity of vaping-related lung injuries and the necessity for comprehensive regulatory frameworks.
Toxicological Concerns and Exposure Variability
One of the primary toxicological concerns is the composition of vaping products, including nicotine, THC, and flavoring additives. Studies in the USA have directly linked illicit THC products containing vitamin E acetate to lung toxicity,2, 10 whereas cases from India and Europe are more frequently associated with nicotine-based products.4, 11 This distinction raises concerns about country-specific vaping regulations, the presence of unregulated products in markets, and the need for stringent product labeling and quality control measures. The presence of various chemical additives, including metal particles from heating coils and diacetyl, further complicates risk assessments. 5
Pathological and Clinical Manifestations
From a pathological perspective, most EVALI cases exhibit features such as diffuse alveolar damage, lipid-laden macrophages, and organizing pneumonia.3, 12 Radiological findings frequently include ground-glass opacities and bilateral lung infiltrates, resembling other conditions such as infectious pneumonia or COVID-19-associated lung injury.13, 14 This similarity has contributed to delayed or missed diagnoses, particularly during the COVID-19 pandemic, when EVALI was often misdiagnosed as viral pneumonia. 15
The symptomatic presentation of EVALI is heterogeneous, with patients often experiencing respiratory symptoms such as dyspnea, cough, and chest pain, along with gastrointestinal and systemic symptoms. 1 In severe cases, patients present with acute hypoxemic respiratory failure requiring intensive care. 8 Given these diverse presentations, the diagnosis of EVALI remains largely one of exclusion, necessitating a thorough patient history and toxicological screening.
Forensic and Legal Challenges
The forensic and legal challenges surrounding EVALI are significant. Many countries lack clear regulations on vaping products, leading to underreporting and misclassification of EVALI cases. 4 In the USA, illicit and counterfeit vaping products played a major role in the 2019 outbreak, prompting calls for stricter regulatory oversight. 16 Conversely, in Europe and India, the lack of standardized diagnostic protocols has led to inconsistencies in recognizing and reporting EVALI cases. 9 Forensic investigations into vaping-related fatalities require comprehensive toxicological screening and standardized postmortem protocols to identify vaping-related lung injuries accurately.
Patient Outcomes and Treatment Considerations
Patient outcomes varied across studies, with most cases responding well to corticosteroid therapy. 17 Early recognition and intervention significantly reduced morbidity, with corticosteroids demonstrating efficacy in resolving inflammation and improving lung function. 13 However, severe cases required intensive care, mechanical ventilation, and prolonged hospital stays, with fatalities occurring primarily in patients with delayed diagnoses or extensive lung damage. 12
In terms of treatment, cessation of vaping is a crucial first step, followed by supportive care, oxygen therapy, and corticosteroids in moderate-to-severe cases. 10 Given the evolving nature of vaping-related lung injuries, there is a need for further research on long-term pulmonary outcomes, potential steroid-sparing therapies, and rehabilitation strategies.
Public Health Implications and Future Directions
The widespread use of e-cigarettes among adolescents and young adults raises concerns about future health implications. Public health interventions should focus on increasing awareness regarding the risks associated with vaping, particularly the dangers of illicit THC products and unregulated nicotine e-cigarettes. School-based education programs, stricter advertising restrictions, and public health campaigns can help mitigate the growing trend of e-cigarette use among youth populations. 5
Future research should aim to establish standardized diagnostic criteria, identify biomarkers for early detection, and evaluate the long-term pulmonary consequences of EVALI. Moreover, international collaborations are needed to monitor trends in vaping-associated lung injuries and develop regulatory policies that ensure product safety across different regions.
Limitations of the Review
This review has several limitations. The majority of included studies were case reports and small case series, which introduces selection and publication bias. Significant heterogeneity was observed in diagnostic criteria, toxicological testing methods, and pathological findings, limiting comparability. Sample sizes were generally small, and regional variations in vaping practices, particularly differences between THC and nicotine exposure, restricted the generalizability of the results. Furthermore, incomplete toxicological data and evolving diagnostic practices over time may have influenced findings. The lack of standardized forensic protocols for investigating vaping-related deaths across different countries further complicates interpretation. Due to these factors, a meta-analysis was not feasible, and only a descriptive synthesis could be performed.
Recommendations
Based on the findings of this review, several regulatory changes are urgently needed. Mandatory disclosure of all vaping product ingredients and standardized pre-market toxicological testing should be enforced. Harmful additives such as vitamin E acetate must be banned, and robust age verification mechanisms must be strengthened. Prominent health warnings regarding EVALI risks should be mandated on all vaping products. Establishing centralized post-market surveillance systems and standardized forensic protocols for vaping-related deaths is essential. Additionally, THC-containing vaping products from unregulated sources should be restricted, and public education campaigns must be intensified. Global harmonization of vaping regulations would further ensure product safety and facilitate international monitoring of vaping-associated health risks.
Conclusion
This systematic review underscores the global burden of EVALI, revealing significant regional differences in exposure patterns and diagnostic challenges. While THC-containing products adulterated with vitamin E acetate remain a predominant cause in the USA, international cases suggest that nicotine-based vaping products can also lead to lung injury. Early recognition, standardized diagnostic criteria, and regulatory measures are essential to mitigate the risks associated with vaping. The forensic and legal challenges surrounding EVALI highlight the need for robust postmortem protocols and toxicological investigations. Ultimately, a combination of public health interventions, clinical vigilance, and regulatory oversight is necessary to address this emerging public health crisis. Future research should focus on long-term pulmonary outcomes, risk stratification, and harm-reduction strategies to mitigate EVALI incidence worldwide.
Footnotes
Acknowledgements
We would like to acknowledge the support provided by the librarian and other staff of Tripura Medical College in accessing journals.
Authors’ Contribution
Nani Gopal Das: Conceptualized the study, designed the methodology, and supervised the overall research.
Nirmalendu Das, Amitava Baidya, and Monica Debbarma: Independently conducted the literature search, data extraction, and quality assessment.
Nirmalendu Das and Amitava Baidya: Performed the initial data analysis.
Monica Debbarma: Contributed to data synthesis and interpretation.
Nani Gopal Das and Monica Debbarma: Led the manuscript drafting and revisions.
All authors contributed to the review and final approval of the manuscript.
Data Availability Statement
All data analyzed in this systematic review were obtained from previously published studies, which are publicly available and cited within the manuscript. No new primary data were generated or collected for this study. Any additional information can be provided upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not required for this systematic review as it is based solely on publicly available data from previously published studies and does not involve human participants, animal subjects, or direct data collection.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Since this study is a systematic review of existing literature and does not involve direct interaction with human participants, obtaining informed consent was not applicable.
