Abstract
Prostate cancer is the most commonly diagnosed cancer in UK men excluding non-melanoma skin cancer, with over 64,000 new cases and 12,000 deaths annually. This disparity between incidence and mortality has fuelled debate about over-diagnosis and the value of screening. The UK National Screening Committee has advised against population prostate-specific antigen (PSA) screening, citing uncertainty regarding benefit and harm. However, a risk-based PSA screening algorithm that adjusts PSA levels to multiples of the median for the same age and incorporates age-specific risk demonstrated good screening performance (detection rate of 90% for a false-positive rate of 2%). The European Randomized Study of Screening for Prostate Cancer shows a 16% reduction in prostate cancer mortality with PSA screening, comparable to reductions achieved in bowel cancer screening. The addition of pre-biopsy multi-parametric magnetic resonance imaging (MRI) screening reduces unnecessary biopsies and over-diagnosis while maintaining detection of clinically significant disease. Together, risk-based PSA testing combined with multi-parametric MRI would prevent an estimated 13 prostate cancer deaths per 1000 men screened every 5 years from age 55, with 9 deaths prevented for every man treated unnecessarily.
From age 50, the risk of a diagnosis of clinically significant prostate cancer in men is 9.5%. 1 In the UK, prostate cancer is now the most commonly diagnosed cancer excluding non-melanoma skin cancer. In 2022, the most recent year UK data are available, there were just over 64,000 new prostate cancer diagnoses in the UK2–5 and just over 12,000 deaths6–8 (Table 1), both accounting for about 15% of cancer diagnoses and cancer deaths in men.
Prostate cancer diagnoses and deaths, UK 2022.
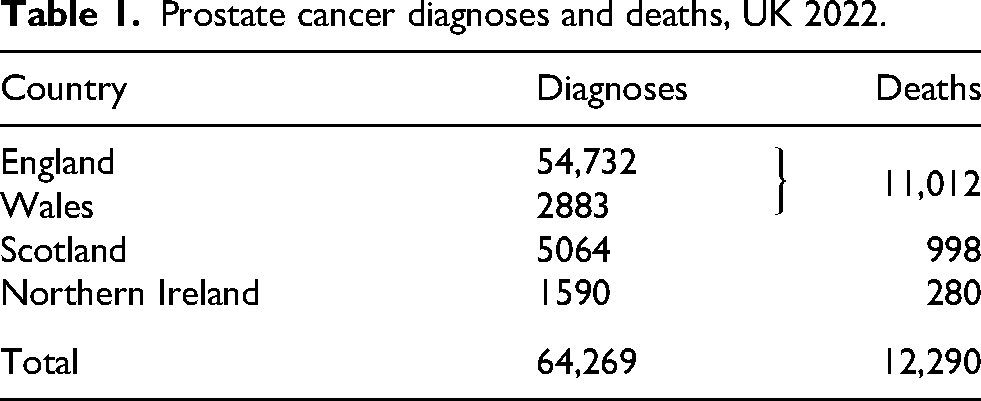
The five-fold difference between prostate cancer diagnoses and deaths from prostate cancer poses a dilemma that has not been resolved. Is the difference due to the fact that most diagnosed cases are relatively indolent or does it reflect the result of effective treatment following a positive diagnosis, or both? This dilemma is illustrated by prominent public figures being diagnosed with prostate cancer assuming that early diagnosis has saved their life while experts, notably those advising those on the UK National Screening Committee, concluded in their advisory report in November 2025 that there is uncertainty over the efficacy of prostate cancer screening, stating that such screening ‘is likely to cause more harm than good’; 9 the apparent good prognosis is simply a reflection of most diagnosed cases being indolent.
The National Screening Committee, seeking to find a middle path, recommended that prostate cancer screening based on prostate-specific antigen (PSA) measurement should be limited to men with a confirmed BRCA1 or BRCA2 variant, and among such men PSA measurement performed every 2 years, from age 45 to 61. This effectively regards such variant analyses as a first-line screening test leading to PSA-based screening, although the National Screening Committee did not state a recommendation for population screening for these variants – not a satisfactory compromise. BRCA1 and BRCA2 variants are rare, present in about 1 in 250 of the general population. 10 This means that as a first-line screening test, even if the BRCA status of all men were known or could be determined by genetic testing, which is anyway questionable, almost all affected cases would be missed.
Are there available data that can determine whether PSA-based screening for prostate cancer is worthwhile? A nested case-control study showed that PSA (total PSA) has a high detection rate (sensitivity) and a low false-positive rate (high specificity) 11 if both the age-related increase in PSA and age-related prostate cancer risk are accounted for and used to estimate the risk of prostate cancer (rather than using a single arbitrary PSA cut-off as has been adopted in clinical trials of screening for prostate cancer using PSA). In the risk-based algorithm, PSA levels are converted into multiple of the median (MoM) values to take into account that PSA levels naturally increase with age. This method of converting screening marker levels into MoM values is used worldwide, in antenatal screening programmes for Down's, Edward's and Patau's syndromes, to take account of naturally changing marker levels during pregnancy. PSA MoM values are then combined with the age-specific risk of prostate cancer to yield a prostate cancer risk estimate. Converting PSA levels into MoM values improves the screening performance over PSA levels expressed in mass units (e.g. ng/ml) and the addition of age-specific risk improves screening performance over PSA MoM values alone; 90% of men who died of or with a history of prostate cancer (case definition) could be detected for a 2.2% false-positive rate with the test performed every 5 years or less; a high screening performance. The false-positive rate could be reduced to 1.2% for the same 90% detection rate with the addition of human kallikrein 2 (hK2) to the screening algorithm; 11 however hK2 is not currently a widely available screening test. The nested case-control study design is the most appropriate in determining the screening performance of prostate cancer screening, being based on stored serum samples that had not initiated intervention (samples were collected between 1975 and 1982, 12 before PSA measurement was used clinically). The study results indicate that the view that PSA screening is an inaccurate screening test is misplaced.
Of course, high screening performance is not enough to justify screening. There needs to be evidence that treatment for screen-detected cancers is effective in preventing clinical and fatal disease. The results of the European Randomized Study of Screening for Prostate Cancer (ERSPC) 13 provide the only reliable evidence of the effect of PSA screening on prostate cancer mortality due to major issues around contamination and adherence in the other trials. 11 Based on an intention-to-treat analysis, the reduction in prostate cancer mortality was 13% (95% confidence interval (CI), 5% to 20%). 13 An on-treatment analysis correcting for non-attendance showed a 16% reduction (95% CI, 8% to 24%). These results are similar to the 13% (intention-to-treat) and 18% (on-treatment) reductions in mortality for bowel cancer screening observed after 20 years of follow-up using the faecal occult blood test, 14 but somewhat lower than the 31% reduction in breast cancer mortality for mammographic screening, 15 all assuming that the effect of screening in non-attendees is similar to that in those who attended screening.
Even if the risk-based screening algorithm using PSA and age that yields a high detection rate and low false-positive rate were used, there would still be concern about ‘over-diagnosis’. Using pre-biopsy multi-parametric magnetic resonance imaging (MRI) after a positive PSA reduces over-diagnosis by shifting biopsies towards MRI-targeted sampling of suspicious lesions and away from clinically insignificant (Gleason Score 6 or lower) disease, while maintaining detection of clinically significant cancer. For example, in the PRECISION trial, 500 men with a PSA above 3 ng/ml were randomised to MRI with or without targeted biopsy (n = 252) or to standard transrectal ultrasonography (TRUS)-guided biopsy (n = 248). Biopsies were avoided in 28% of men in the MRI group. Clinically significant cancer was detected in 38% in the MRI group compared with 26% in the standard-biopsy group. Also fewer clinically insignificant cancers were diagnosed in the MRI group (9% vs. 22%). 16 In the PROMIS (PROstate MRI Imaging Study), 576 men with clinical suspicion of prostate cancer (elevated serum PSA (up to 15 ng/ml) within previous 3 months, suspicious digital rectal examination, suspected organ-confined stage T2 or lower on rectal examination, or family history) underwent multi-parametric (mp)MRI followed by both TRUS-biopsy and transperineal prostate mapping biopsy. 17 The results showed mpMRI works well as a triage test, detecting 93% of clinically significant prostate cancers and leading to about 27% of men avoiding a biopsy.
Figure 1 summarises the evidence in a flowchart of the effect of screening 1000 men aged 55 or more every 5 years using the risk-based PSA algorithm, followed by mpMRI among men with screen-positive results. This shows that amongst the 95 (9.5%) men who are expected to receive a diagnosis of clinically significant prostate cancer, 80 would be screen positive after the PSA algorithm and mpMRI, and after biopsy and treatment 13 prostate cancer deaths would be prevented. Among the 10 false-positives (men who do not develop clinically significant prostate cancer but are screen positive following the PSA algorithm and mpMRI) 1.5 would be unnecessarily treated after being biopsy positive (i.e. they are diagnosed with prostate cancer, but it is not clinically significant). Figure 1 therefore shows that about nine prostate cancer deaths will be prevented for every unnecessarily treated man.
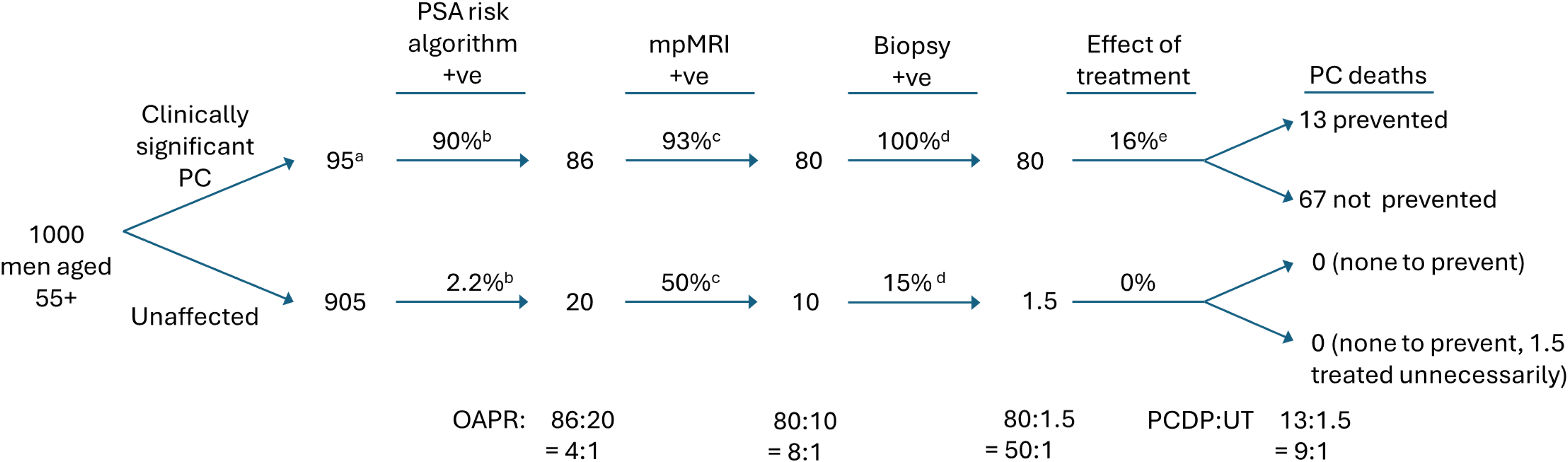
Flowchart outlining the efficacy of 5-yearly screening for prostate cancer from age 55 using PSA risk-based algorithm followed by mpMRI. PSA: prostate-specific antigen; PC: prostate cancer; mpMRI: multi-parametric magnetic resonance imaging; OAPR: odds of being affected given a positive result; PCDP: prostate cancer deaths prevented; UT: unnecessarily treated; PROMIS: PROstate MRI Imaging Study. aLifetime risk of clinically significant PC 95 based on 9.5% from age 50. 1 The remainder (905) are unaffected which includes non-clinically significant PC. bDetection rate and false-positive rate for screening every 5 years from age 55. 11 cDetection rate from PROMIS trial, false-positive rate of 50% assumed slightly lower than observed in PROMIS (59%) due to superior performance of PSA risk-based algorithm over PSA alone. 17 dBiopsy detects 100% of clinically significant PC and also identifies some non-clinically significant PC among men with positive mpMRI results 16 eEffect of treatment from ESPRC trial. 13
Although the National Screening Committee recommended against population screening for prostate cancer using PSA, an updated recommendation is due in Spring 2026 18 and the decision on whether to implement such a screening programme is reported as resting with the Secretary of State for Health and Social Care, Wes Streeting. 19 The decision needs to be made on the scientific and medical grounds for recognising that the effect of PSA-based screening in preventing fatal prostate cancer is similar to the reduction in mortality from bowel cancer from the screening programme for bowel cancer. Policy-makers will need to ask: If screening for bowel cancer is worthwhile, then why not screening for prostate cancer?
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
