Abstract
Many factors need to be considered when planning and managing a screening programme for the early detection of cervical cancer (CC). A non-systematic international review of the organisation of CC screening using high-risk human papillomavirus (HPV-HR) testing, aimed at identifying the organisational methods of these programmes, was conducted with a view to supporting the future of the French system in the context of the transition to HPV-HR testing. In countries where HPV testing has been implemented or planned, the initial reflection process has provided an opportunity to rethink the previous (cytological) screening organisation. Despite considerable differences between countries, a nationally or regionally centralised organisational model appears to be the preferred option in most countries. This model is based on a national/regional structure tasked with all invitations, reminders, follow-up and coordination, centralised laboratories integrating both biology and pathology laboratories, and a unified information system integrated with routine health management tools used by health practitioners and nurses. Besides quality considerations, grouped purchasing makes it possible to implement a public procurement policy that includes price negotiations with suppliers. Discussions around the introduction of HPV testing have resulted in most countries reviewing or creating information systems and quality assurance processes. While the WHO seems to recommend the systematic use of vaginal self-sampling, very few countries have considered this option. More and more countries are planning to implement vaginal self-sampling, but no clear organisational model has emerged from the countries where it has been implemented to date.
Context
The French cervical cancer (CC) screening programme was implemented as part of the 2014–2019 National Cancer Plan, with the aim of reducing inequalities in respect of CC treatment and lowering the incidence of the disease. In France, CC prevention is based on vaccination and voluntary CC screening (IndScr) of women aged 25–65 years. Between 25 and 29 years of age, a cytology test sequence of two cytology tests 1 year apart is recommended,
1
with further testing after 3 years if the results of the first two are normal. From the age of 30 to the age of 65, a high-risk human papillomavirus (HR-HPV) test is recommended: 3 years after the last normal cytology test or from the age of 30 in the absence of previous cytological tests. The interval between two HPV tests is 5 years, as long as the tests are negative. While CC prevention remains mainly based on voluntary screening, an organised screening programme for CC was implemented by the Ministerial Order of 4 May 2018. It includes:
- A process of invitations and reminders limited to women who have not undergone opportunistic screening within the recommended time intervals, and not based on anniversary dates. - Follow-up of all women with abnormal or positive screening test results between the ages of 25 and 65, whether they participated spontaneously or received invitations to attend screening. - Streamlining of screening practices and improvement of professional practices (intervals between two tests, follow-up of women with abnormal/positive results, etc.). - The implementation of specific actions or complementary strategies (support for screening, health mediation, self-testing, mobile units, etc.) for vulnerable populations. - Diversification of healthcare workers involved: general practitioners, gynaecologists, midwives and other health professionals, through sampling training and quality assurance, as well as information campaigns targeting professionals and women. - Organisational methods of these programmes. - Approaches used to transition from cytology-based screening to HPV-HR testing in these programmes. -Organisation and methods for the use of self-collected vaginal sampling if this screening method was already in use.
Systematic HPV-HR testing was generalised following a new Ministerial Order of 30 July 2020. The French National Cancer Institute (INCa) sought to examine the organisation of CC screening programmes using HPV-HR testing and self-sampling in various countries, to identify the most cohesive and cost-effective scenarios to support the future of the French system in the context of the transition to HPV-HR testing. Part of this undertaking was based on a non-systematic international review of the organisation of CC screening using HPV-HR testing, aimed at identifying:
Methods
For a panel of countries where organised CC screening using HPV testing has been implemented, a literature review was conducted to identify:
- The organisational methods used in these programmes - The CC screening transition procedure from cytology alone to HPV testing for these programmes - Self-sampling methods used, if available.
Organised screening consists of an explicit screening policy, defined target population, implementation team, healthcare team for clinical care delivery, quality assurance infrastructure and method for identifying cancer outcomes. Implementation of an organised screening programme offers opportunities to systematically assess the success of the program and develop interventions to address identified gaps in an effort to optimise CC outcomes.
The literature review was supplemented by direct interviews with local officials involved in the most advanced countries in terms of setting up organised CC screening using HPV testing. The methodology was therefore based on a two-pronged approach combining a review of the literature and interviews with foreign correspondents or experts, as needed.
A preliminary list of countries known to INCa as having adopted, or considering adopting in the short term, a CC screening strategy based on the use of HPV testing was considered. In addition, the countries and their population or epidemiological characteristics had to be similar to the situation in France or geographically close to France. This list was completed following the analysis of review articles and subsequently, as the project progressed, on the discovery of international publications referring to countries that had not been previously identified. Conversely, some countries that had been identified for inclusion in the analysis were ultimately excluded in part, in light of the actual progress of their screening programmes in terms of introducing HPV-based screening or difficulties identifying and/or interviewing a contact person. In the end, 13 countries were selected (Australia, Denmark, Germany, Finland, France, Ireland, Italy, the Netherlands, Norway, Sweden, Slovenia, England and Turkey; Supplementary 1).
Keywords and search equations were used to meet the objectives, accounting for the fact that the documents searched are generally found in grey literature, and therefore do not systematically appear in scientific literature databases. A first search was conducted on PubMed®, Embase® and Science Direct®. This search was completed by consulting general or specialist websites along with websites of institutions and/or international organisations capable of providing additional relevant information, published or not (grey literature), on the topic of CC screening using HPV testing. For example, the websites of the main institutions tasked with screening in the selected countries were consulted, as well as the websites of relevant learned societies or expert committees where identified as being of interest. Only existing programmes were considered, excluding simple local trials.
The PubMed® research equation was: (cervical cancer screening [Title/Abstract]) AND (papillomavirus [Title/Abstract]) AND ((COUNTRY1[Title/Abstract]) OR (COUNTRY2[Title/Abstract])) resulting in the review of 301 articles. A similar search equation used in Science Direct® resulted in the identification of 199 more papers. Most of the articles identified using this approach did not discuss screening organisation in the selected countries, but discussed any of the following topics:
- Results obtained in pilot programmes - Studies on the prognostic value of HPV testing - Economic modelling of test use - Surveys on the social acceptability of HPV testing
Therefore, most of the data collected on the organisation of CC screening with HPV testing was retrieved from websites in the area, and through direct interviews of key leaders in each country of interest.
A list of informative dimensions was established to describe each country/programme (Table 1).
Questions addressed during the review of cervical cancer (CC) screening programmes.

Results
Organisational aspects
The screening strategies implemented in the different countries studied vary somewhat, whether in terms of the ages chosen for the use of HPV testing, the time interval between two successive screening tests or the type of investigations performed in the event of an unconfirmed or positive primary test. HPV testing is generally conducted every 5 years in women aged 30 and older (Tables 2 and 3; Supplementary 2).
Main organised cervical cancer (CC) screening programmes using HPV testing.
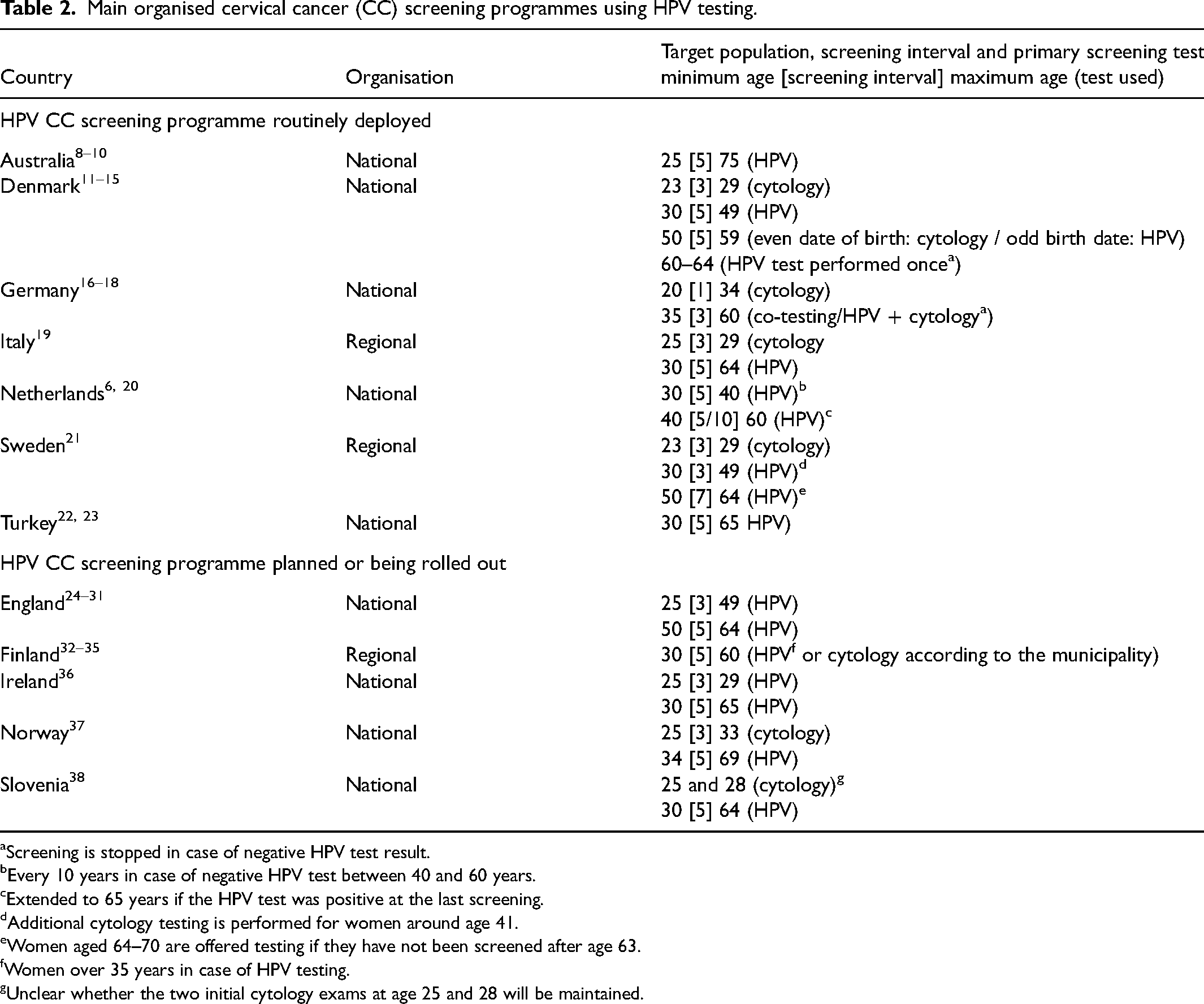
Screening is stopped in case of negative HPV test result.
Every 10 years in case of negative HPV test between 40 and 60 years.
Extended to 65 years if the HPV test was positive at the last screening.
Additional cytology testing is performed for women around age 41.
Women aged 64–70 are offered testing if they have not been screened after age 63.
Women over 35 years in case of HPV testing.
Unclear whether the two initial cytology exams at age 25 and 28 will be maintained.
Main organisational methods for cervical cancer (CC) screening programme by country.

In countries where HPV testing has been implemented or planned, the initial reflection process has provided an opportunity to completely rethink the previous (cytological) screening organisation.
The decisions made have often adopted a nationally or regionally centralised approach. Centralisation has led to a significant reduction in the number of laboratories licensed to perform HPV tests, and in some cases to the selection of only one or two national laboratories (Turkey, Slovenia) or one laboratory per region (the Netherlands, England). However, some countries, such as Australia, have chosen to retain a wide range of services, requiring participating laboratories to undergo accreditation.
Centralised processes are also frequently implemented in relation to invitation strategies. Countries where HPV testing has been implemented generally use national (Australia) or regional (the Netherlands, Sweden) registries, sometimes managed by health insurance funds (Germany). In Turkey, invitations are managed by primary care professionals but using national support software that includes features to simplify invitation recall and editing. It is interesting to note that most countries use age-based invitation schemes, and do not only issue invitations to women not attending spontaneous screening within the recommended intervals. Most programmes in other countries send screening invitations to people with a cervix who are registered as ‘female’ at fixed ages.
The level of centralisation also affects the choice of consumables and technologies used for sampling and the type of tests performed. Besides quality considerations, grouped purchasing makes it possible to implement a public procurement policy that includes price negotiations with suppliers.
Discussions around the introduction of HPV testing have also made it possible to review or create information systems and quality assurance processes. In most cases, this has involved adapting or improving existing systems for CC screening using cytology. However, in Turkey, for example, the entire system was reviewed, and a new software system was implemented to manage the entire screening process, linked with software already in use for primary care or hospital care management. In the Nordic countries and Estonia, a transnational information system has been developed, NordScreen, with the objective of ensuring common monitoring of screening programme quality indicators.
While the WHO recommends that HPV self-sampling should be made available as an additional approach to sampling in CC screening services for women aged 30–60 year, 2 very few countries have considered the systematic use of vaginal self-sampling (VSS). VSS is sometimes proposed and often considered as a future option. Where HPV testing is available as part of the national programme, self-sampling offers an additional option for improving CC screening uptake. The few countries that offer VSS use very different methods. The Netherlands is probably the country with the most experience in using VSS. Dutch women between the ages of 30 and 60 receive an invitation every 5 years. This letter explains how the woman can attend screening, stop being invited for screening, or request a deferral, and also offers her the option of requesting a self-collection kit. If the woman chooses to use the self-collection kit, it will be sent to her home. She will then send the collected material directly to the designated screening laboratory using the pre-printed reply envelope and material container included with the kit. In Australia, the use of VSS by women aged 30 and over who have undergone screening in the previous two years is discussed between the healthcare provider and the woman to determine the most appropriate sampling method. If the woman opts for VSS, the kit is provided by the healthcare provider, and the women carries out sampling directly at the healthcare facility, where a private area is available for sample collection.
In all countries where VSS has been implemented, questions remain as to the ability of women screened using VSS to undergo additional or follow-up testing and treatment following a positive result.
Transition to HPV testing
The literature review found that it takes quite a long time to implement new CC screening methods (a 5-year period is often planned), but the process sometimes takes less time (2.5 years in Turkey). Programmes often lag behind their original ambitions and are staggered in time. The recent period marked by the COVID-19 pandemic has obviously impacted the implementation of HPV screening further.
Some countries have favoured a gradual approach, developed initially in certain geographic areas and then expanded gradually. The existence of great regional variability (including for countries with a national programme), in terms of participation in screening and methods for monitoring the quality of practices and diagnoses, is often cited as a major difficulty during the transition to HPV testing. The use of HPV testing in CC screening does not seem to escape this observation.
The importance of training for providers is mentioned in all countries, although training methods can vary greatly (mandatory in-person training programmes, online training, etc.), even within the same country (e.g. Sweden and the United Kingdom have always had a good record in this area). The lack of training for healthcare providers and information for the population can lead to non-acceptance of the programme, as is the case in Australia, where opposition movements have arisen due to a lack of information. These training/information exercises are especially important as, despite the broad scientific consensus, some misperceptions or scientific publications continue to question HPV testing 3 for a variety of reasons: weak data on optimal triage strategies for HPV-positive women, 4 cost of testing and available infrastructures, development of vaccine prevention and uneven performances of HPV tests depending on whether they are conducted using PCR or a signal amplification method. 5
The financial and human resources required to deal with the additional steps arising from the implementation of HPV testing are sometimes mentioned (Sweden, Italy, the Netherlands). In the Netherlands, in the first 5 years after implementation, the new HPV-based screening programme was expected to be more expensive than cytology-based screening as increases in costs of triage tests, colposcopy referrals and cervical intra-epithelial neoplasia and CC treatments outweigh the decrease in the costs of the primary screening test. 6 After five years, the costs of primary screening tests would decrease further due to the extension of screening intervals for HPV-negative women aged between 40 and 50, resulting in a need for fewer screening tests.
Institutional opposition is infrequent but not non-existent. In Germany, gynaecologists have asked for the new screening programme to be deferred, citing supply issues. In Turkey, anatomic pathologists briefly had concerns about the effects of the new organisation on their professional activity.
Australia has experienced other issues relating to
- Implementation of self-sampling due to regulatory requirements: despite intensive training in self-sampling and enthusiasm among health professionals, self-sampling was not made available in this country until January 2018, and has not been fully implemented to date. This delay is due to an unforeseen regulatory issue: because self-collection is not listed as an ‘intended use’ by HPV test manufacturers, the authorities required each laboratory to have the specific collection device used for self-collected HPV tests validated. Only one laboratory undertook this process in a timely manner. - An unexpected increase in the number of colposcopies due to the very high sensitivity of HPV tests, but probably also due to substandard practices in public laboratories.
7
This has led to long colposcopy waiting times in public clinics, and has led to a risk of inequality in women's access to appropriate follow-up in a timely manner.
Finally, the determinants of women's lack of screening uptake have been the subject of various studies in several countries (Slovenia, Australia, etc.). The findings are frequently the same: women over 50, women with low levels of education and income, single women, immigrant women and women who have never been pregnant are less likely to attend screening. This subject is developed in the report written by INCa, based on several interviews conducted with different representative countries.
The transition to the use of HPV testing for screening may provide an opportunity to increase access for these social groups through the development of VSS, as shown in a study in Slovenia and in a real-life study in the Netherlands.
However, other strategies are undoubtedly necessary, ranging from improving financial coverage in countries where some tests are not fully covered, to systematically translating programme documents into several languages or rallying the community around screening. The latter helps to overcome cultural or religious barriers observed in specific social groups (Turkey, Australia). In some cases, it is important to ensure that women understand that their privacy and confidentiality will be guaranteed. Mobile and outreach services have also been effective in involving women from remote areas in cervical screening, with flexible appointments, group transportation and/or transportation assistance (Australia). France has developed a similar outreach strategy with specific measures targeting specific communities, such as remote communities.
Conclusion
The experience in countries where national and regional HPV-based screening has already been implemented is generally very positive. A wide variety in the type of organisation is observed across countries in terms of CC screening with HPV testing. This is due to multiple factors, including the political system (national, federal, highly decentralised or centralised), the healthcare system, local CC screening history, etc.
Despite these differences, the literature seems to show that a nationally or regionally centralised model is the option adopted in most countries. This model is characterised by a national/regional structure tasked with all invitations, reminders, follow-up and coordination, centralised laboratories integrating both biology and pathology laboratories, and a unified information system integrated with routine health management tools used by healthcare professionals. In many countries, CC screening is performed by primary care practitioners, including nurses.
HPV self-sampling has high sensitivity and specificity, similar to that of HPV testing conducted by a physician or nurse. 1 More and more countries are planning to implement VSS, but no clear organisational model has emerged from the few countries where it has been implemented to date.
Supplemental Material
sj-docx-1-msc-10.1177_09691413231158932 - Supplemental material for Overview of organisational methods of primary cervical lesion screening programmes that use human papillomavirus testing
Supplemental material, sj-docx-1-msc-10.1177_09691413231158932 for Overview of organisational methods of primary cervical lesion screening programmes that use human papillomavirus testing by Agathe Marchadier, Laura Bezannier, Stéphanie Barré-Pierrel, Antoine Manceau, Audrey A Abadie and Bruno Detournay in Journal of Medical Screening
Footnotes
Acknowledgements
We would like to express our gratitude to all the experts for taking the time to answer our many questions. Without them, we would not have been able to complete this research. Ahti Anttila – Research Director of the Mass Screening Registry, Finnish Cancer Registry and Tytti Sarkeala – Director of Mass Screening, Finnish Cancer Registry (Finland). Paola Armaroli – Epidemiologist, Centro di Riferimento per l’Epidemiologia e la Prevenzione Oncologica in Piemonte (CPO) (Italy). Karen Canfell – Director of Cancer Research, Cancer Council NSW (Australia). Louise Dunk – Head of Programme, Cervical Screening Wales, Public Health Wales (Wales). Miriam Elfström – Epidemiologist, Karolinka Institute and Regional Cancer Institute of Gotland (Sweden). Murat Gultekin – Gynaecological oncologist, Hacettepe University Medical School (Turkey). Ruth Stubbs – National Cervical Screening Programme Manager and Anne Mackie – Director of Programmes, UK National Screening Committee (England, UK). Ivanuš Urška – Head of Cancer Screening Department, National Cervical Cancer Screening Programme (Slovenia). Sandra van Dijk-de Bruin – Programme Manager, National Institute for Public Health (the Netherlands).
Authors’ contribution
All authors contributed equally to the research and writing of this manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LB and BD are employed by CEMKA, a consulting firm providing health research and consulting services to private and public organisations. BD has received professional fees for speeches, conferences or participation in scientific committees from AstraZeneca, Merck Sharpe & Dohme Corp, Novo Nordisk, Pfizer Inc., Lilly Inc and Sanofi-Aventis. AMar and SB are employed by INCa (French National Cancer Institute). AMan and AA are employed by PwC Advisory – Healthcare & Pharmaceuticals Life-Sciences.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was self-funded by the French national cancer institute.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
