Abstract
Objectives
Mammography screening is generally accepted in women aged 50–69, but the balance between benefits and harms remains controversial in other age groups. This study systematically reviews these effects to inform the European Breast Cancer Guidelines.
Methods
We searched PubMed, EMBASE and Cochrane Library for randomised clinical trials (RCTs) or systematic reviews of observational studies in the absence of RCTs comparing invitation to mammography screening to no invitation in women at average breast cancer (BC) risk. We extracted data for mortality, BC stage, mastectomy rate, chemotherapy provision, overdiagnosis and false-positive-related adverse effects. We performed a pooled analysis of relative risks, applying an inverse-variance random-effects model for three age groups (<50, 50–69 and 70–74). GRADE (Grading of Recommendations Assessment, Development and Evaluation) was used to assess the certainty of evidence.
Results
We identified 10 RCTs including 616,641 women aged 38–75. Mammography reduced BC mortality in women aged 50–69 (relative risk (RR) 0.77, 95%CI (confidence interval) 0.66–0.90, high certainty) and 70–74 (RR 0.77, 95%CI 0.54–1.09, high certainty), with smaller reductions in under 50s (RR 0.88, 95%CI 0.76–1.02, moderate certainty). Mammography reduced stage IIA+ in women 50–69 (RR 0.80, 95%CI 0.64–1.00, very low certainty) but resulted in an overdiagnosis probability of 23% (95%CI 18–27%) and 17% (95%CI 15–20%) in under 50s and 50–69, respectively (moderate certainty). Mammography was associated with 2.9% increased risk of invasive procedures with benign outcomes (low certainty).
Conclusions
For women 50–69, high certainty evidence that mammography screening reduces BC mortality risk would support policymakers formulating strong recommendations. In other age groups, where the net balance of effects is less clear, conditional recommendations will be more likely, together with shared decision-making.
Introduction
Breast cancer (BC) is the second most common malignancy in the world. 1 In the European Union, 404,920 women were diagnosed with BC and 98,755 died during 2018. 2 Over the last 20 years, BC mortality has decreased due to improvements in treatment, services delivery and implementation of population screening. However, the role of population screening has been under debate over the last three decades due to conflicting systematic reviews and recommendations.3,4
Randomised clinical trials (RCTs) carried out during the 1970s and 1980s showed that mammography screening is associated with a reduction in BC mortality. 5 However, screening a healthy population is also associated with undesirable effects such as recalling women with a false-positive result for additional imaging. 6 Overdiagnosis (BC cases that would not have clinically surfaced in the absence of screening) is another downside of screening, and its magnitude is controversial. 7
Numerous organisations have issued screening recommendations. The WHO recommended in favour of screening starting at 40 years of age in well-resourced settings. 8 The Canadian Task Force recommended screening only in women over 50, due to the risk of overdiagnosis and unnecessary biopsies in younger women. 9 The American Cancer Society, including evidence from RCTs and observational and modelling studies, recommended initiating annual screening at 45. 10
In 2015, the (ECIBC) was launched to develop the European Guidelines on Breast Cancer Screening and Diagnosis. This systematic review informed the recommendations about mammography screening for early detection of BC in asymptomatic women at average risk. During the guideline development, 11 the Guidelines Development Group (GDG) made detailed considerations on the evidence of effects as well as values and preferences, equity, acceptability and feasibility. Readers are welcome to refer to these considerations in the published recommendations and on the ECIBC website (https://healthcare-quality.jrc.ec.europa.eu/european-breast-cancer-guidelines/screening-ages-and-frequencies).12,13
Methods
Structured question and outcome prioritisation
The clinical question prioritised by the GDG, ‘Which is the optimal age range in which to carry out screening for breast cancer?’, followed the Population, Intervention, Comparison and Outcomes format (Box 1). Three sub-populations were pre-defined: women under 50, 50–69 and 70–74 years old at the moment of invitation to screening.
Structured clinical question.
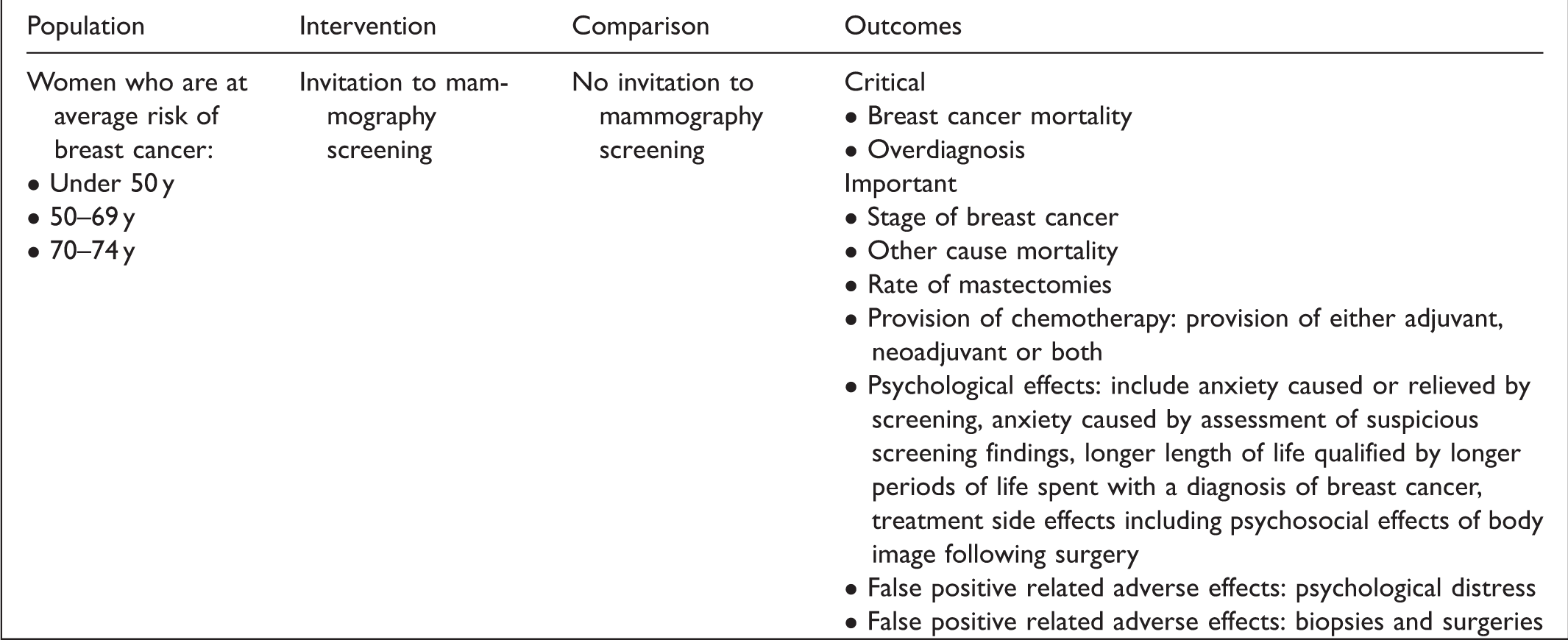
During the development of the recommendations, 11 the GDG decided to split the sub-group of women under 50 into two sub-groups: 40–44 and 45–49. The outcomes were prioritised by the GDG using a 1–9 scale as suggested by the GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach. 14
Data sources and searches
We searched MEDLINE (April 2016), EMBASE (April 2016) and CENTRAL (March 2016) databases using pre-defined algorithms for both systematic reviews and individual studies. We adapted the search terms to each database (see Supplemental material 1). We also reviewed lists of references of the included studies, and members of the GDG were consulted about potentially missing studies.
During June 2018, we performed a new search in MEDLINE as part of the ECIBC’s guideline updating process. The results were assessed by the GDG and, as no relevant studies were identified that could potentially change the recommendations, they decided to not update the review. In November 2019, the GDG met and considered that, to the best of their knowledge, there were no relevant new publications.
Study selection
We included RCTs of women at average risk of BC (without family history of BC or inherited changes of BRCA1 and BRCA2 genes), comparing invitation to mammography screening versus no invitation. If no RCTs were identified, we included systematic reviews of observational studies. For overdiagnosis, we included only trials in which, after completing the study phase, neither women in the control nor the intervention group were offered mammography screening. We excluded studies conducted outside the context of screening programmes or not published in English.
Initially, at the title and abstract level, two reviewers after calibration assessed the eligibility of the references retrieved. Two reviewers independently reviewed the full text of the selected references. Discrepancies were solved either by consensus or with the help of a third reviewer.
Data extraction and risk of bias assessment
Details of the study design, population, follow-up and results were extracted by one reviewer and confirmed by a second reviewer. If needed, we requested additional data from authors of the included studies. We assessed the risk of bias (RoB) of RCTs using the Cochrane RoB Assessment tool 15 , and systematic reviews with the AMSTAR (A Measurement Tool to Assess Systematic Reviews) checklist (see Supplemental material 2). 16
Data analysis
To estimate the effect of mammography screening on BC mortality, we used two methods. The ‘short case accrual’ method includes only BC deaths among BC cases diagnosed during the screening intervention phase.17,18 The ‘long case accrual’ method considers all BC deaths irrespective of the date of diagnosis, accrual time being equivalent to the follow-up of the study.17,18
We estimated overdiagnosis as the difference in the cumulative number of BC in the groups invited and not invited to screening, expressed: (a) as a percentage of the number of cancers in the screening group (population perspective) or (b) as a percentage of the cancers diagnosed during the screening phase of the trial in the invited to screening group (individual perspective). We pooled data as relative risks (RR) using a random-effects model (Review Manager v5.3). We assessed the presence of heterogeneity among studies using the Cochrane chi-square test and the I2 statistic. Additionally, we provided subgroup analysis based on the risk of bias assessment and a post-hoc sensitivity analysis excluding RCTs with a substantial concern for breaking the concealment at randomisation.
To estimate risk differences, we used the baseline risk from the control arms of the RCTs; we also provide estimates using baseline risks proposed by the GDG members considering European population surveillance data. All results are expressed as by 100,000 women invited to mammography screening.
Certainty of the evidence
We rated the certainty of the evidence for each outcome taking into consideration the standard GRADE domains,19,20 described in the evidence profiles (see Supplemental material 4).
Results
Search results
From 2393 unique citations, we selected 57 to be appraised as full text. At this stage, we excluded seven reviews, and 13 observational studies of mammography screening reporting outcomes available from RCTs. Additionally, we excluded the Edinburgh trial because of important baseline differences between the screening and control groups, suggesting suboptimal randomisation (see Supplemental material 5).21,22
We included 30 publications from nine RCTs: the Health Insurance Plan (HIP) of Greater New York trial,5,23–26 the Canadian Breast Cancer Screening Study (CNBSS-1 and CNBSS-2),27–32 the United Kingdom Age trial,33–35 the Stockholm trial,36–38 the Malmö Mammographic Screening Trial (MMST I and MMST II),39–41 the Göteborg trial,42,43 the Swedish Two-County trial (Östergötland and Kopparberg counties),44–49 one publication that reported results for the five Swedish mammography trials, 18 and updated results of the UK Age Trial 50 and the Göteborg Trial (Table 1). 51 We also obtained additional age-stratified results for BC mortality from the authors of the CNBSS trial.
Overview of included randomised clinical trials.
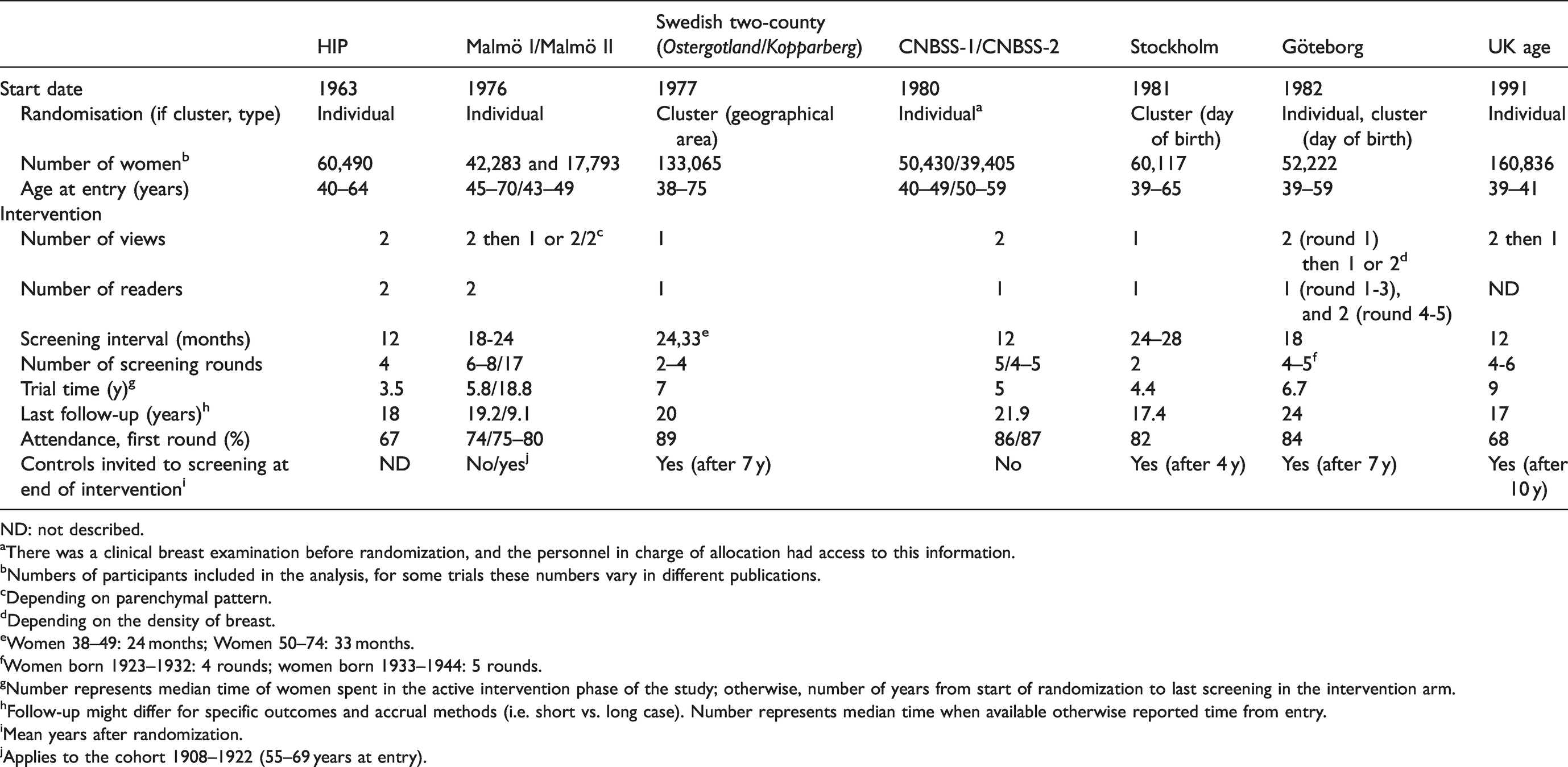
ND: not described.
aThere was a clinical breast examination before randomization, and the personnel in charge of allocation had access to this information.
bNumbers of participants included in the analysis, for some trials these numbers vary in different publications.
cDepending on parenchymal pattern.
dDepending on the density of breast.
eWomen 38–49: 24 months; Women 50–74: 33 months.
fWomen born 1923–1932: 4 rounds; women born 1933–1944: 5 rounds.
gNumber represents median time of women spent in the active intervention phase of the study; otherwise, number of years from start of randomization to last screening in the intervention arm.
hFollow-up might differ for specific outcomes and accrual methods (i.e. short vs. long case). Number represents median time when available otherwise reported time from entry.
iMean years after randomization.
jApplies to the cohort 1908–1922 (55–69 years at entry).
Four systematic reviews of observational studies fulfilled the eligibility criteria (Figure 1).52–55 Brett et al. 53 assessed the adverse psychological impact of mammography screening in the general population. Salz et al. 55 examined the effects of false-positive mammogram results. Bond et al. 52 evaluated the psychological effects of false-positive screening mammograms in the UK, and one review assessed the cumulative risk of false-positive results leading to an invasive procedure (needle biopsy or surgery). 54
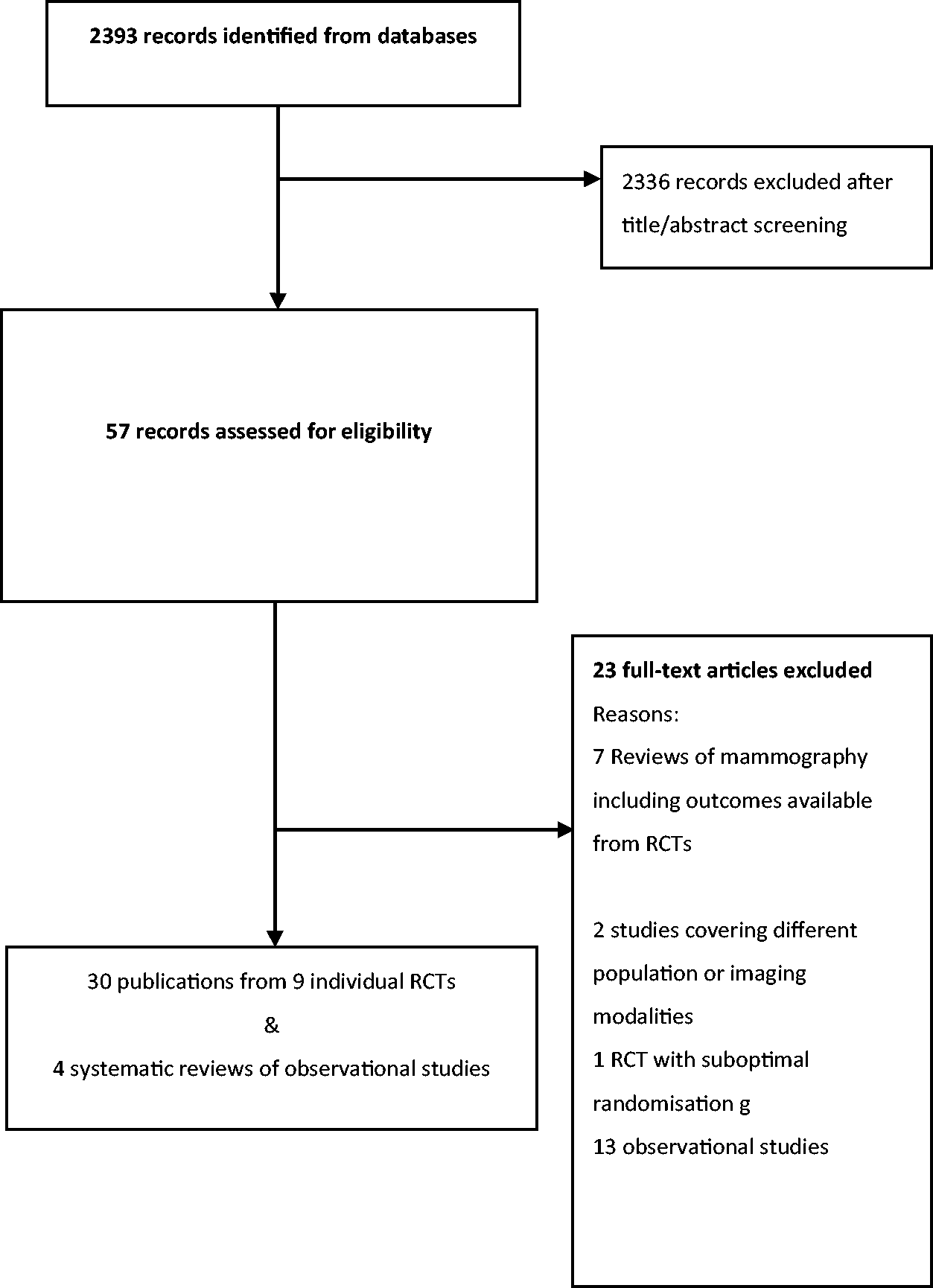
PRISMA flowchart.
BC mortality
Eight trials included 348,478 women less than 50,24,26,28,29,36,37,39,40,43,50,51 six trials 249,930 women aged 50–69,18,23,24,26,27,29,36,37,43,47,48,51 and two trials 18,233 women aged 70–74.18,39,40,47,48 The trial time ranged from 3.5 to 18.8 years, the median short case accrual follow-up time from 9.1 to 24 years, and the median long case accrual follow-up time from 13 to 21.9 years, depending on the age strata (Table 1).
In women under 50, invitation to mammography screening probably reduces the risk of BC mortality (RR 0.88; 95% CI (confidence interval) 0.76–1.02; I2 = 20%; short case accrual) (moderate certainty).18,24,26,28,50,51 Comparable results were obtained using a long case accrual follow-up time (RR 0.92; 95%CI 0.83–1.02; I2 = 6%)18,24,28,43,45,50,51 (Figure 2). In women aged between 50 and 69, mammography screening reduced the risk both for short (RR 0.77; 95%CI 0.66–0.90; I2 = 49%)18,24,26,27,47,51 and long case accrual time18,24,27,43,45,51 (high certainty) (Figure 3). For women aged 70–74, the Malmö I reported short case accrual follow-up and the Swedish Two-County reported long accrual follow-up time; mammography screening reduced the risk of BC mortality (RR 0.77; 95%CI 0.54–1.09; I2 = 0%)18,47 (high certainty) (Figure 4).
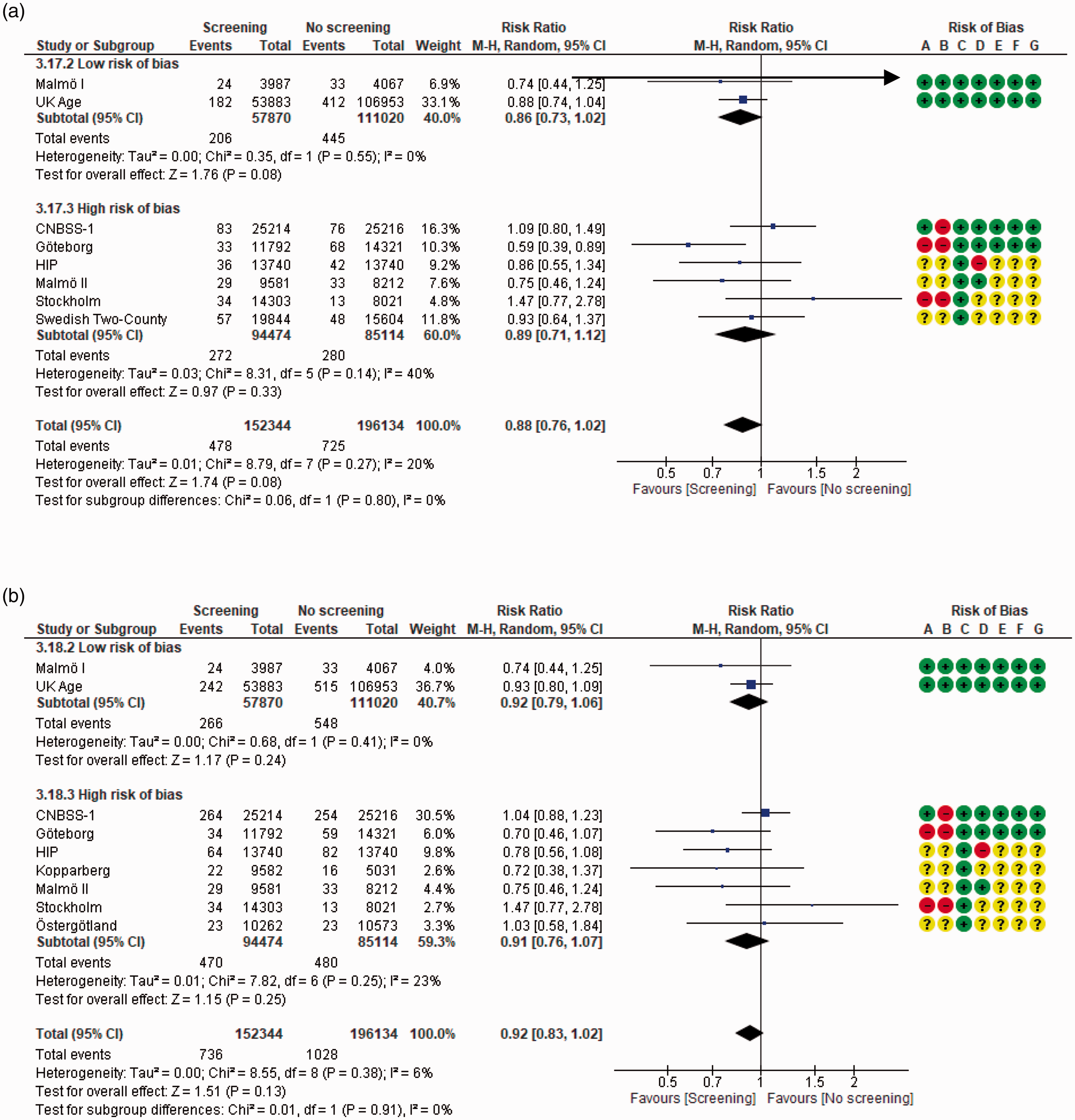
Effect on breast cancer mortality of mammography screening (women under 50 years of age): (a) short case accrual, mean follow up across studies 16.8 years; (b) longest case accrual, mean follow-up across studies 15.2 years. Risk of bias legend: (A) Random sequence generation, (B) allocation concealment, (C) blinding of participants and personnel, (D) blinding of outcome assessment, (E) incomplete outcome data and (F) selective reporting, (G) other bias.
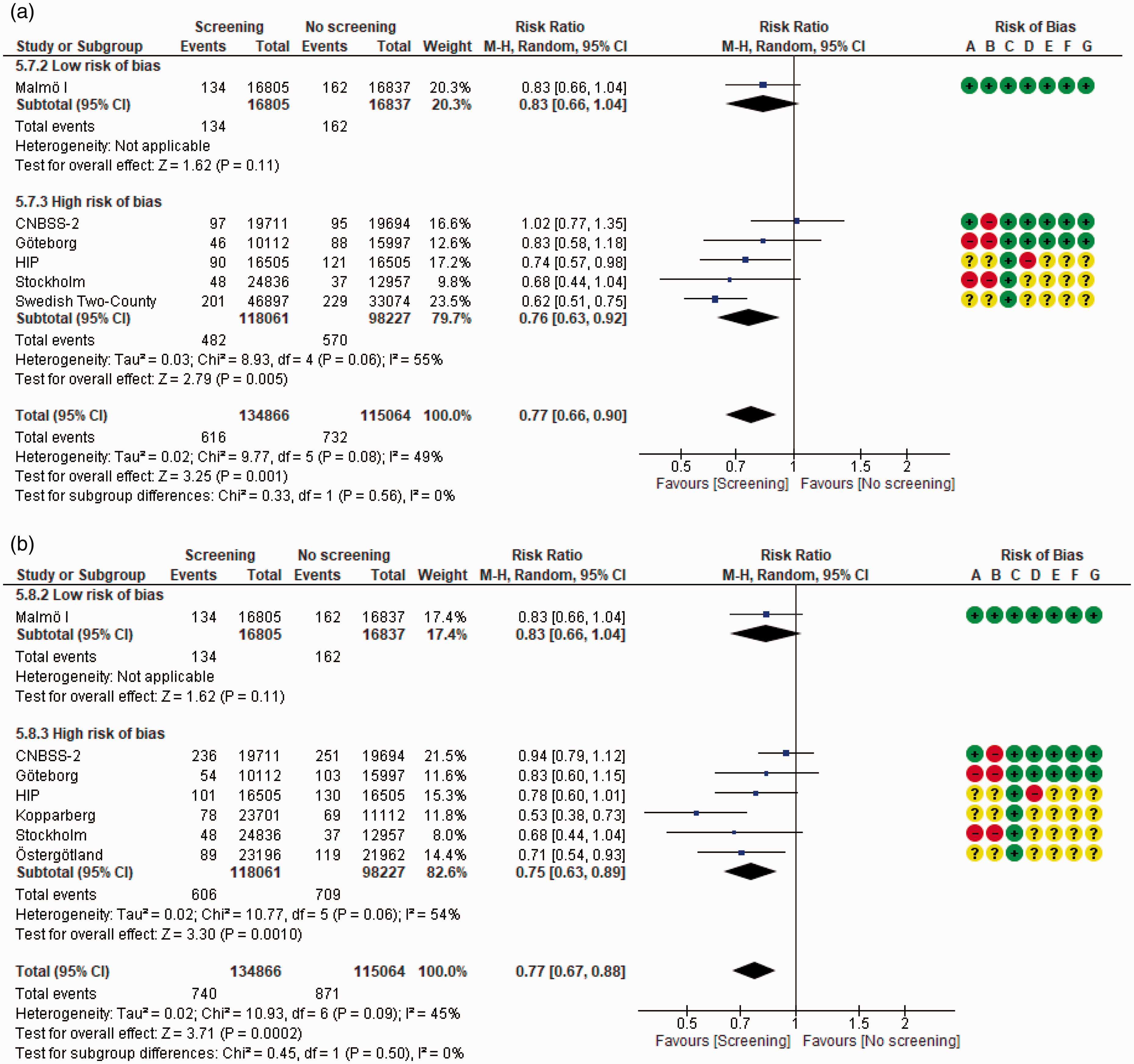
Effect on breast cancer mortality of mammography screening (women aged 50–69): (a) short case accrual, mean follow-up across studies 17.6 years; (b) longest case accrual, mean follow-up across studies 15.5 years. Risk of bias legend: (A) random sequence generation, (B) allocation concealment, (C) blinding of participants and personnel, (D) blinding of outcome assessment, (E) incomplete outcome data, (F) selective reporting and (G) other bias.
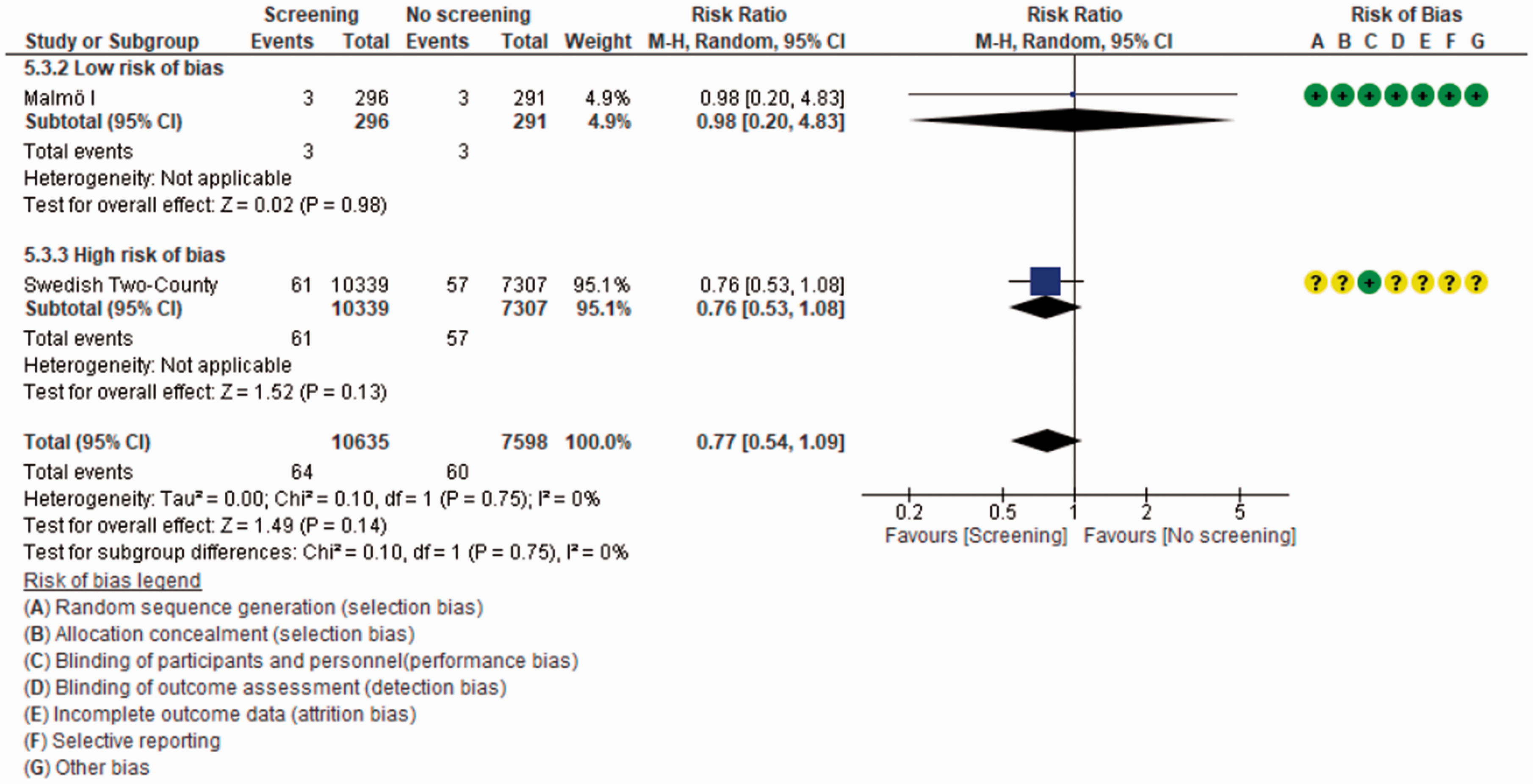
Effect on breast cancer mortality of mammography screening (women 70 years of age or older). Malmö I and Swedish two-county reported short case accrual estimate, follow-up across studies 9.5 years.
The risk difference in BC mortality for women aged 50–69 was 138 fewer deaths per 100,000 women invited to screening (95%CI 204 fewer to 60 fewer) using short accrual follow time, and 175 fewer deaths per 100,000 women invited to screening (95%CI 251 fewer to 91 fewer) using long accrual time (Table 2). Sensitivity analysis including only RCTs at low RoB yielded similar results.
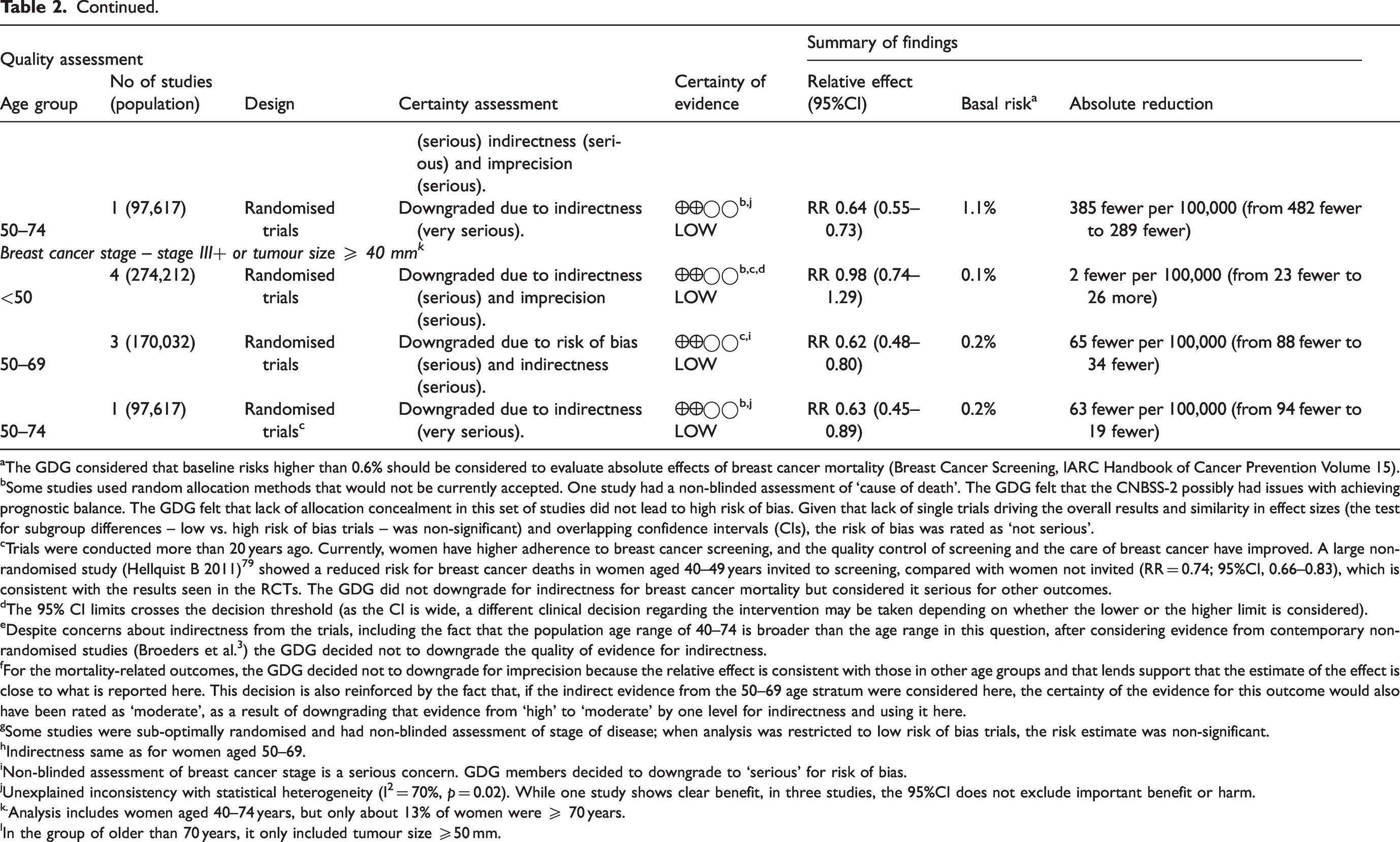
Summary of available evidence on desirable effects of breast cancer mammography screening by age groups.
aThe GDG considered that baseline risks higher than 0.6% should be considered to evaluate absolute effects of breast cancer mortality (Breast Cancer Screening, IARC Handbook of Cancer Prevention Volume 15).
bSome studies used random allocation methods that would not be currently accepted. One study had a non-blinded assessment of ‘cause of death’. The GDG felt that the CNBSS-2 possibly had issues with achieving prognostic balance. The GDG felt that lack of allocation concealment in this set of studies did not lead to high risk of bias. Given that lack of single trials driving the overall results and similarity in effect sizes (the test for subgroup differences – low vs. high risk of bias trials – was non-significant) and overlapping confidence intervals (CIs), the risk of bias was rated as ‘not serious’.
cTrials were conducted more than 20 years ago. Currently, women have higher adherence to breast cancer screening, and the quality control of screening and the care of breast cancer have improved. A large non-randomised study (Hellquist B 2011)79 showed a reduced risk for breast cancer deaths in women aged 40–49 years invited to screening, compared with women not invited (RR = 0.74; 95%CI, 0.66–0.83), which is consistent with the results seen in the RCTs. The GDG did not downgrade for indirectness for breast cancer mortality but considered it serious for other outcomes.
dThe 95% CI limits crosses the decision threshold (as the CI is wide, a different clinical decision regarding the intervention may be taken depending on whether the lower or the higher limit is considered).
eDespite concerns about indirectness from the trials, including the fact that the population age range of 40–74 is broader than the age range in this question, after considering evidence from contemporary non-randomised studies (Broeders et al. 3 ) the GDG decided not to downgrade the quality of evidence for indirectness.
fFor the mortality-related outcomes, the GDG decided not to downgrade for imprecision because the relative effect is consistent with those in other age groups and that lends support that the estimate of the effect is close to what is reported here. This decision is also reinforced by the fact that, if the indirect evidence from the 50–69 age stratum were considered here, the certainty of the evidence for this outcome would also have been rated as ‘moderate’, as a result of downgrading that evidence from ‘high’ to ‘moderate’ by one level for indirectness and using it here.
gSome studies were sub-optimally randomised and had non-blinded assessment of stage of disease; when analysis was restricted to low risk of bias trials, the risk estimate was non-significant.
hIndirectness same as for women aged 50–69.
iNon-blinded assessment of breast cancer stage is a serious concern. GDG members decided to downgrade to ‘serious’ for risk of bias.
jUnexplained inconsistency with statistical heterogeneity (I² = 70%, p = 0.02). While one study shows clear benefit, in three studies, the 95%CI does not exclude important benefit or harm.
k.Analysis includes women aged 40–74 years, but only about 13% of women were ≥ 70 years.
lIn the group of older than 70 years, it only included tumour size ≥50 mm.
Other cause mortality
In women under 50, mammography screening may make no difference to other-cause mortality, but the evidence is uncertain (RR 1.04; 95%CI 0.95 to 1.15; I2 = 62%) (very low certainty).18,28,42,44,50 Two trials were included in the 50–69 group27,44 and one trial in the 70–74 group; 44 in these age strata, mammography screening may also result in no difference (Table 2) (low certainty) (Supplemental material 3: Figures S5, S8 and S9).
Advanced BC
We defined advanced stage as either stage II or greater, tumour size ≥20 mm or ≥1 positive lymph node, which is consistent with stage IIA disease or higher. Additionally, we used a second definition of advanced disease as regional or metastatic or tumour size ≥ 40 mm, equivalent to stage III or higher.
Using the stage IIA or higher definition, in women under 50, mammography screening may reduce the risk of advanced disease, but the evidence is uncertain (RR 0.88; 95%CI 0.78 to 0.99; I2 = 0%) (very low certainty).23,24,28,34,43,45,50,51 In women aged 50–69, the effect size was similar (RR 0.80; 95%CI 0.64–1.00, I2 = 70%) (very low certainty).23,24,27,43,45,51 One trial including older women (aged 50–74) showed that mammography screening may reduce the risk of advanced disease (RR 0.64; 95%CI 0.55–0.73), 45 equivalent to 385 fewer cases (95%CI 482 fewer to 289 fewer) (Table 2) (low certainty) (Supplemental material 3: Figures S1 and S6).
Using the stage III or higher definition, in women under 50, screening may make little difference to the risk of advanced disease (RR 0.98; 95%CI: 0.74–1.29; I2 = 0%) (low certainty).23,24,28,34,45,50 In women aged between 50 and 69, mammography screening may reduce the risk of advanced disease (RR 0.62, 95%CI 0.48–0.80; I2 = 0%),23,24,27,45 which is equivalent to 65 fewer cases of advanced BC (low certainty). In women aged 50–74, mammography screening may reduce the risk of advanced disease (RR 0.63, 95%CI 0.45–0.89), 45 equivalent to 63 fewer cases (95%CI 94 fewer to 19 fewer) (Table 2) (low certainty) (Supplemental material 3: Figures S2 and S7).
Overdiagnosis
We identified three trials, the CNBSS-1, the CNBSS-2 and a subgroup of women aged 55–69 from the Malmo-I trial (women aged 45–54 received screening at the end of the study). In women aged 40–49, the estimates of overdiagnosis were 12.4% (95%CI 9.9–14.9) from a population perspective and 22.7% (95%CI: 18.4–27.0) from an individual perspective (moderate certainty).28,29 In women aged between 50 and 69, we estimated a pooled overdiagnosis of 10.1% (95%CI: 8.6–11.6; I2 = 0%) from a population perspective and 17.3% (95%CI: 14.7–20.0; I2 = 10%) from an individual perspective (Table 3) (moderate certainty).27,29,40
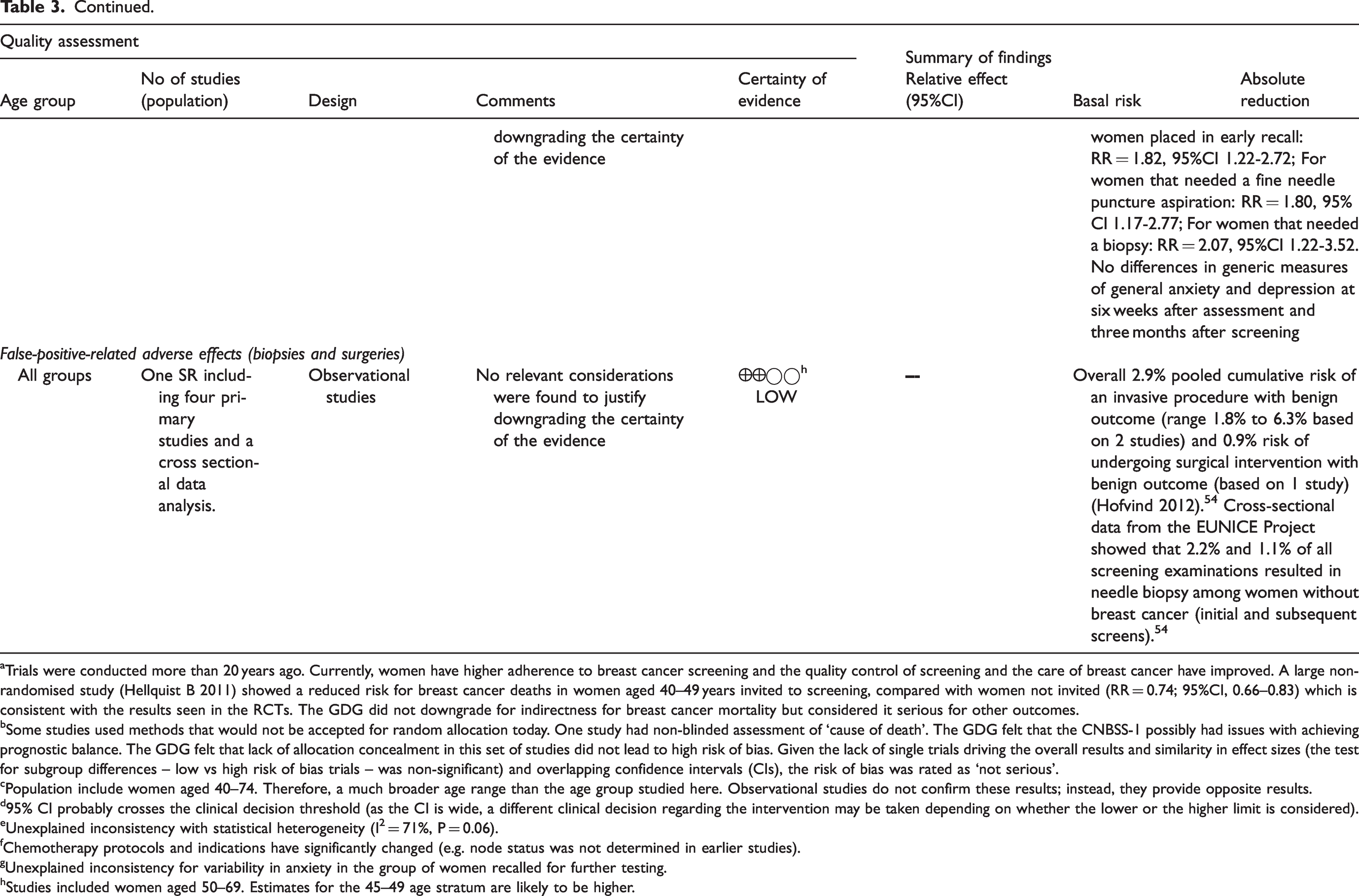
Summary of available evidence on undesirable effects of breast cancer mammography screening by age groups.
aTrials were conducted more than 20 years ago. Currently, women have higher adherence to breast cancer screening and the quality control of screening and the care of breast cancer have improved. A large non-randomised study (Hellquist B 2011) showed a reduced risk for breast cancer deaths in women aged 40–49 years invited to screening, compared with women not invited (RR = 0.74; 95%CI, 0.66–0.83) which is consistent with the results seen in the RCTs. The GDG did not downgrade for indirectness for breast cancer mortality but considered it serious for other outcomes.
bSome studies used methods that would not be accepted for random allocation today. One study had non-blinded assessment of ‘cause of death’. The GDG felt that the CNBSS-1 possibly had issues with achieving prognostic balance. The GDG felt that lack of allocation concealment in this set of studies did not lead to high risk of bias. Given the lack of single trials driving the overall results and similarity in effect sizes (the test for subgroup differences – low vs high risk of bias trials – was non-significant) and overlapping confidence intervals (CIs), the risk of bias was rated as ‘not serious’.
cPopulation include women aged 40–74. Therefore, a much broader age range than the age group studied here. Observational studies do not confirm these results; instead, they provide opposite results.
d95% CI probably crosses the clinical decision threshold (as the CI is wide, a different clinical decision regarding the intervention may be taken depending on whether the lower or the higher limit is considered).
eUnexplained inconsistency with statistical heterogeneity (I² = 71%, P = 0.06).
fChemotherapy protocols and indications have significantly changed (e.g. node status was not determined in earlier studies).
gUnexplained inconsistency for variability in anxiety in the group of women recalled for further testing.
hStudies included women aged 50–69. Estimates for the 45–49 age stratum are likely to be higher.
Rate of mastectomies
Across all age groups, women invited to screening may undergo more mastectomies (RR 1.20, 95%CI 1.11–1.30; I2 = 0%, 180 more in absolute terms) (low certainty) (Supplemental material 3: Figure S3).18,31,39,46
Provision of chemotherapy
Across all age groups, the evidence was uncertain with an RR of 0.86 (95%CI 0.53–1.40; I2 71%)18,39,46 (Table 3) (very low certainty) (Supplemental material 3: Figure S4).
Psychological effects
Uncertain evidence showed that mammographic screening may not produce anxiety in women given a clear result after a mammogram. 53 However, those requiring further investigations may experience significant anxiety, in the short and long term, depending on the extent of the additional exams (Table 3) (very low certainty). 53
False-positive-related psychological distress
One review, including 17 studies, suggested an increase in the scores of disease-specific BC measures of psychological distress with false-positive results, being largest for anxiety about BC (r = 0.22; 95%CI: 0.18–0.27) and smallest for fear (r = 0.08; 95%CI: 0.03–0.14) (low certainty). 55 In contrast, when using non-specific measures, the only suggested effect was a higher risk of generalised anxiety (r = 0.03; 95%CI: 0.00–0.07) (low certainty). 55
Another review included four studies evaluating psychological impact; 52 false-positive mammograms may be associated with negative psychological consequences when assessed using disease-specific measures (e.g. BC anxiety). Additionally, the risk of negative effects may be greater if a biopsy is required (RR 2.07; 95%CI 1.22–3.52) than if only further mammography is needed (RR 1.28; 95%CI 0.82–2.00) (low certainty). 52
False-positive-related procedures
One systematic review included four primary studies and an analysis of performance parameters from 20 screening programmes (low certainty). 54 One Norwegian study reported a cumulative risk of undergoing fine needle aspiration cytology, core needle biopsy (CNB) and having a surgical intervention with a benign outcome of 3.9%, 1.5% and 0.9%, respectively. 56 The largest study, from a Spanish screening programme, reported an estimated cumulative risk of 1.8% for undergoing an invasive procedure with a benign outcome. 57
RoB and certainty of the evidence
Our main concerns for BC mortality were: (1) the use of suboptimal random allocation methods, such as the date of birth to allocate women to each study arm in the Stockholm and Gothenburg trials;36,42 (2) in the CNBSS trials, participants underwent clinical breast examination before randomisation, and this information was available to the personnel in charge of allocation.27,28 However, no single trial drove the overall results in the subgroup analysis of low versus high RoB (Tables 2 and 3). This was corroborated in a post-hoc sensitivity analysis, where we explored the impact of excluding the CNBSS trials, which resulted in a non-meaningful change in the point estimates for the outcome breast cancer mortality (i.e. for women under 50, RR 0.84; 95%CI 0.73–0.98, for short case accrual) (Supplemental material 3: Figures S10 and S11).
For overdiagnosis, in the CNBSS trials, there was potential bias due to screening after the trial ended, as screening programmes were subsequently implemented under different province jurisdictions with an increased likelihood of attendance of women who had been screened during the trials.29,40 Also, the overdiagnosis results were simple cumulative incidence differences with no adjustment for lead time and, therefore, might be overestimated.
Other relevant limitations were related to indirectness, due to difference in quality control of screening and improvement of care over the last two decades, but given the consistency with more recent observational studies (see Discussion section), the GDG did not consider downgrading the evidence for this reason.
Discussion
Main findings
Our review shows that there is high certainty evidence that mammography screening reduces the risk of BC mortality in women between the ages of 50 and 69, with the number of deaths averted ranging from 138 fewer to 483 fewer per 100,000 women invited to screening, depending on the baseline risk assumed (from 0.6 to 2.1%). For other age groups, the evidence is not conclusive. Consistently, women invited to screening across all age groups showed a lower risk of advanced stages of BC.
There is moderate certainty that screening is associated with an increase in undesirable effects. Especially important was overdiagnosis, regardless of the calculation method, which was larger from an individual perspective in the younger age groups compared to older groups. 7 Mammography screening did not appear to produce significant negative psychological effects as long as the results were clearly communicated, while false-positive results, especially when further assessment is required, increased the number of invasive procedures and psychological distress.
Our results in the context of previous research
Consistent with our analysis, observational studies suggest a reduction of BC mortality after screening implementation. A systematic review of time trend studies estimated a BC mortality reduction from 1 to 9% per year and from 28 to 36% in studies comparing post- and pre-screening periods. 3 A pooled analysis of seven incidence-based mortality (IBM) studies from European countries showed a mortality reduction of 25% among invited women and 38% among those actually screened. 3 Another review which classified studies according to the quality of the methods used to estimate the expected mortality in absence of screening found an IBM risk reduction of 26% in women invited for screening from studies with robust approaches. 58
Our results suggest that screened women are diagnosed with less advanced disease. Observational evidence shows earlier BC staging at diagnosis in women who had received mammography screening. One Canadian registry-based study showed that screening attendees were more likely to have in-situ disease alone, and in those with invasive cancer, a lower proportion of grade III histology. 59 Furthermore, two studies using the SEER-Medicare database showed that extending mammography screening to elderly women decreased advanced stage at diagnosis.60,61
The interpretation of BC stage results from RCTs is precluded by stage migration bias due to the introduction of sentinel lymph node dissection 62 and by modifications in coding and classification practices. 63 Consequently, ecological studies have yielded conflicting results; for example, the incidence of BC stages II–IV has been reported to remain unchanged since the introduction of screening in the Netherlands. 64 To overcome these limitations, a systematic review suggested to use the primary tumour size as the most direct link to radiological detection; 65 their findings from observational studies suggested a reduction in BC advanced stages after the introduction of screening. 65
We observed an increased risk of mastectomies, which has not been consistently described in population studies. One Canadian study found that mastectomies were less frequently performed in screening attendees, 59 while women diagnosed in a New Zealand screening programme were more likely to undergo conservative surgery; 66 similar results were observed in women aged 40–49 from the US. 67 One Norwegian study reported that mammography screening was associated with an increase in mastectomy rates, which later declined, likely explained by changes in recommended surgical approaches. 68 It is noteworthy that a recent systematic review found that adherence to guideline recommendations on breast-conserving surgery is highly variable (35–95%). 69 Thus, the increase in mastectomies among RCTs may partly be due to lead time bias, the progress in BC care, or variation in clinical practice.
Our estimates on chemotherapy are limited by changes in clinical practices, but recent observational studies suggest similar results. One study using Italian population cancer registries, from 2009 to 2013, observed that the neo-adjuvant therapy indication was lower in provinces where a screening programme had been present for many years. 70 Another study that identified women aged 40–79 with incident BC from the British Columbia Cancer Registry (Canada) found that chemotherapy use was lower among regular screening participants after adjustment for age. 71
Our overdiagnosis estimates were in the range described in the literature for European screening programmes, which have been roughly estimated to range from 0 to 54% using unadjusted data and from 1 to 10% after adjustment for BC risk and lead time. 72 However, a proportion of the excess of incidence from the CNBSS trials occurred years after screening ceased in the intervention arm 24 and should not be considered as overdiagnosis. Overall, the certainty of the evidence of overdiagnosis was moderate, due to potential RoB, as women in the control group of the CNBSS trial might have received opportunistic or programmatic mammography screening at the end of the trial period.
There is no consensus about the method to estimate overdiagnosis. Most common approaches assess the difference in cancer incidence in the presence and absence of screening or make inferences about the lead time of BC. 73 One study observed that a long follow-up time is needed to account for lead time, as the excess of cumulative BC incidence will fall below 10% after a follow-up of 25 years in a simulated population. 74 Another study, applying a micro-simulation model to the Netherlands population, found that estimations made in earlier phases of the screening programme may overestimate overdiagnosis by a factor of 4, underlining the relevance of allowing an appropriate follow-up time to obtain reliable estimates. 75
There are discrepancies among previous systematic reviews on the assessment of RoB.4,76 In particular, a Cochrane review considered that only the CNBSS, Malmo and UK Age trials were at low RoB, showing a non-significant effect of mammography screening on BC mortality from those trials. 4 We considered the CNBSS trial as high RoB due to allocation of women by using open lists, and the inclusion of a clinical examination before randomisation which could have led to differential assignment;27,28 thus, only two RCTs were at low RoB with similar BC mortality effects when compared to the remaining studies. Consequently, in the 50–69 years strata, we did not downgrade our certainty for RoB due to the similarity in effect estimates across studies.
Some authors have proposed all-cause mortality as a better estimate of screening impact. This measure would be less prone to ascertainment bias of the cause of death, an issue described in the Swedish trials with higher all-cause mortality in the control group. However, authors of the Swedish trials4,77 reported no significant increased rate of death from other causes after appropriate adjustments for age distribution and lead time bias were implemented.18,47 Moreover, all-cause mortality would be an inefficient measure given the unfeasibly large sample size required to detect differences between groups. 47 We complemented our estimation of mortality impact with the results for other-cause mortality which suggested no difference between women invited or not to screening.
Balancing potential benefits and overdiagnosis, we estimate for 100,000 women invited to screening from age 50 to 69, at least 138 BC deaths would be avoided and 3240 BC would be diagnosed (2.7 per 1000 annual rate × 20 years × 0.6 mammography adherence) of which 550 (17%, individual perspective) could be overdiagnosed. Thus, for each BC death avoided, approximately four overdiagnosed cases will be managed. This estimate is in the range of previous systematic assessments of screening. 78 However, the potential bias in the overdiagnosis estimates means that this figure remains tentative.
Limitations and strengths
Our systematic review has some limitations, as no RCTs have sufficient statistical power to assess the benefit of screening on BC mortality according to age subgroups. Additionally, we included only English language articles; however, the risk of selection bias is probably small because we screened previous systematic reviews, and the GDG includes several international experts, making the possibility of missing studies unlikely. Although our original search was conducted up to April 2016, we conducted a new search in June 2018, and after looking at the results, the GDG decided not to update the systematic review.
Our review also has strengths: we used rigorous methods including the GRADE approach to rate the certainty of the evidence and included the longest follow-up data available from the RCTs and systematic reviews of observational studies. In contrast to previous systematic reviews, the consideration of contextual evidence allowed us to rate the certainty of evidence for BC mortality as high for women aged 50–69 and 70–74 and moderate for women aged 45–49. We also provided results stratified by age groups of interest for women, clinicians and policymakers.
Conclusions
Our findings have different implications depending on the stakeholder group. Guideline panellists (and policy makers) are more likely to formulate strong recommendations in women in the 50–69 age group than in other groups. In women under 50 or over 69, where the balance is less clear, conditional recommendations are more likely. Moreover, panels may specify further subgroups among women below 50, where baseline risk changes rapidly and recommendations could vary between the 40–44 and 45–49 age groups, as in the ECIBC guidelines 13 (https://ecibc.jrc.ec.europa.eu/recommendations/). Although informed decision-making should be recommended in all age groups, this will be especially important in these age groups where the balance is less clear.
A number of research priorities were identified with input from the GDG experts, which included: assessing the impact of different screening intervals; the identification of risk factors to stratify women who should start screening earlier (or at shorter examination intervals, such as women with dense breast tissue); better assessment of the magnitude of overdiagnosis with an emphasis on methods to estimate the actual impact across age groups; and the use of new technologies for screening (i.e. tomosynthesis).
Supplemental Material
sj-pdf-1-msc-10.1177_0969141321993866 - Supplemental material for Benefits and harms of breast cancer mammography screening for women at average risk of breast cancer: A systematic review for the European Commission Initiative on Breast Cancer
Supplemental material, sj-pdf-1-msc-10.1177_0969141321993866 for Benefits and harms of breast cancer mammography screening for women at average risk of breast cancer: A systematic review for the European Commission Initiative on Breast Cancer by Carlos Canelo-Aybar, Diogenes S Ferreira, Mónica Ballesteros, Margarita Posso, Nadia Montero, Ivan Solà, Zuleika Saz-Parkinson, Donata Lerda, Paolo G Rossi, Stephen W Duffy, Markus Follmann, Axel Gräwingholt and Pablo Alonso-Coello in Journal of Medical Screening
Footnotes
Availability of data and materials
All data sources used during this study are described in this published article and its additional information files. The datasets analysed are available from the corresponding author on reasonable request.
Acknowledgements
The authors would like to sincerely thank all members of the Guidelines Development Group of the European Commission Initiative on Breast Cancer for their participation in the discussions generated by this systematic review which led to the different recommendations they developed in the European Guidelines on Breast Cancer Screening and diagnosis (![]() ).
).
Authors’ contributions
Carlos Canelo-Aybar, Diogenes Seraphim Ferreira, Monica Ballesteros, Margarita Posso, Nadia Montero and Ivan Solá, Pablo Alonso-Coello were responsible for conducting the systematic review. Monica Ballesteros, Margarita Posso, Nadia Montero and Ivan Solá conducted the search and data extraction. Paolo Giorgi Rossi, Stephen Duffy, Markus Follmann, Zuleika Saz-Parkinson, and Axel Gräwingholt contributed to the definition of the research protocol and provided comments to the preliminary results of the systematic review. Carlos Canelo-Aybar and Pablo Alonso-Coello drafted the first version of the article. All authors contributed to the interpretation and reporting of the results and provided comments on subsequent versions of the article. All authors read and approved the final manuscript prior submission.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Zuleika Saz-Parkinson and Donata Lerda are current employees of the Joint Research Centre, European Commission. Carlos Canelo-Aybar, Diogenes Seraphim Ferreira, Monica Ballesteros, Margarita Posso, Nadia Montero, Ivan Solà and Pablo Alonso-Coello are employees of the Iberoamerican Cochrane Collaboration. Paolo Giorgi Rossi, Stephen Duffy, Markus Follmann, and Axel Gräwingholt are members of the ECIBC Guidelines Development Group. Diogenes Seraphim Ferreira was supported by the fellowship MTF 2015–02 from the European Respiratory Society during the conduct of the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The systematic review was carried out by Iberoamerican Cochrane Collaboration under the Framework contract 443094 for procurement of services between the European Commission’s Joint Research Centre and Asociación Colaboración Cochrane Iberoamericana.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
