Abstract
Objective
In mammography screening, interval cancers present a problem. The metric ‘screening sensitivity’ monitors both how well a programme detects cancers and avoids interval cancers. To our knowledge, the effect of breast cancer surrogate molecular subtypes on screening sensitivity has never been evaluated. We aimed to measure the 2-year screening sensitivity according to breast cancer subtypes.
Methods
We studied 734 women with an invasive breast cancer diagnosed between 2003 and 2007 after participating in one regional division of Quebec’s Mammography Screening Program. They represented 83% of all participating women with an invasive BC diagnosis in that region for that period. Tumours were categorized into ‘luminal A-like’, ‘luminal B-like’, ‘triple-negative’ and ‘HER2-positive’ subtypes. We used logistic regression and marginal standardization to estimate screening sensitivity, sensitivity ratios (SR) and sensitivity differences. We also assessed the mediating effect of grade.
Results
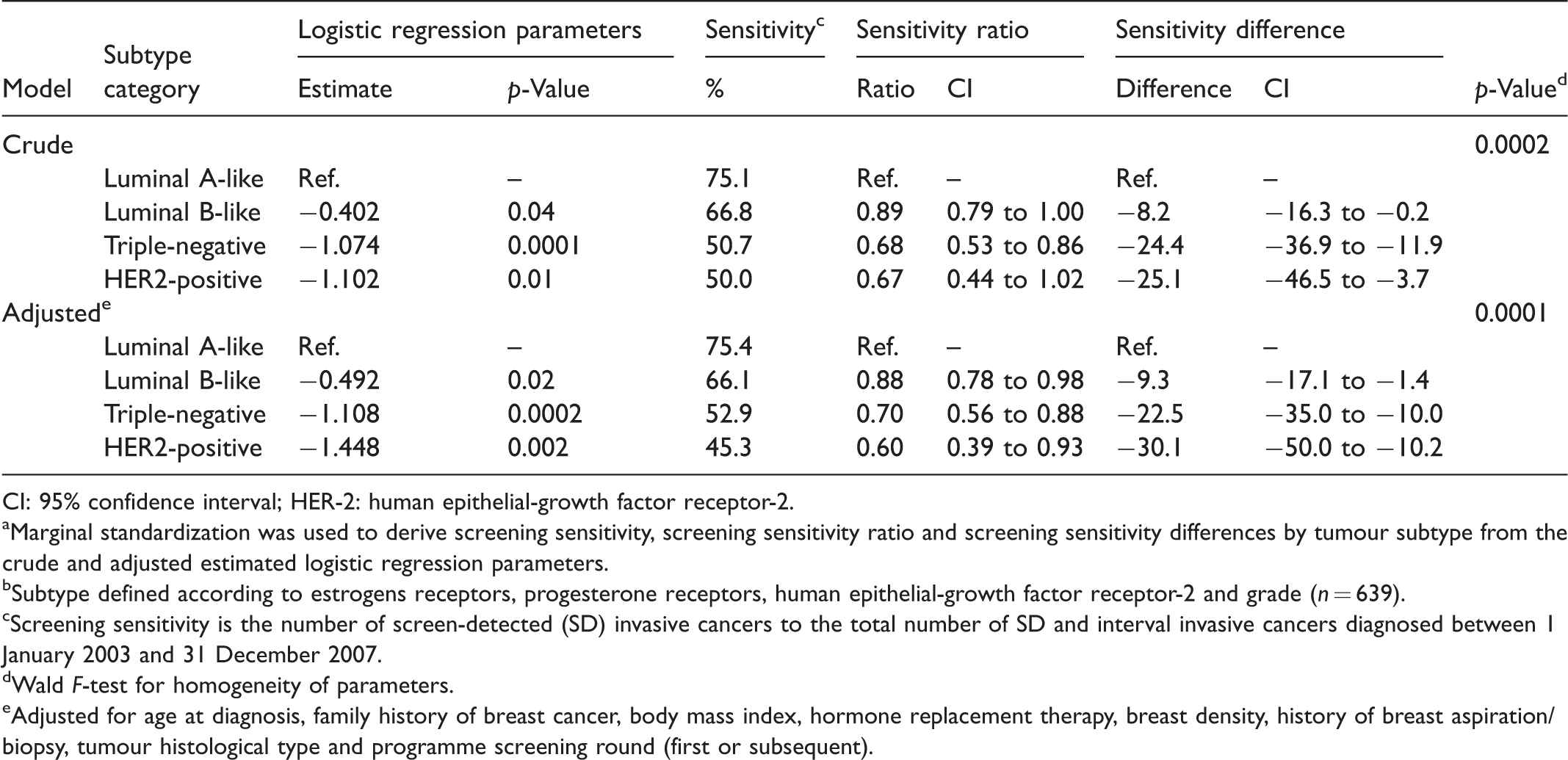
Adjusted 2-year screening sensitivity was 75.4% in luminal A-like, 66.1% in luminal B-like, 52.9% in triple-negative and 45.3% in HER2-positive, translating into sensitivity ratios of 0.88 (95% confidence interval [CI] = 0.78–0.98) for luminal B-like, 0.70 (CI = 0.56–0.88) for triple-negative and 0.60 (CI = 0.39–0.93) for HER2-positive, when compared with luminal A-like. Grade entirely mediated the subtype-sensitivity association for triple negative and mediated it partly for HER2-positive. Screening round (prevalent vs. incident) did not modify results.
Conclusion
There was substantial variation in screening sensitivity according to breast cancer subtypes. Aggressive phenotypes showed the lowest sensitivity, an effect that was mediated by grade. Tailoring screening according to women’s subtype risk factors might eventually lead to more efficient programs.
Among Canadian women, breast cancer accounts for one in four cancer diagnoses and one in eight cancer-related deaths, with 26,300 diagnoses and 5000 deaths in 2017. 1 The Canadian provinces have each adopted population-based mammography screening programmes, aimed to reduce morbidity and mortality by detecting breast cancer at an earlier stage, thus requiring less aggressive treatments and yielding better outcomes.2,3 These programmes also have shortcomings, including interval cancers, those cancers clinically diagnosed between a negative screen and the following scheduled screening. 4 Sensitivity, a standard mammography screening performance metric, can indicate whether a programme has successfully avoided interval cancers.4,5 Higher screening sensitivity increases the probability that the programme contributes to breast cancer mortality reduction, and decreases the risk of generating false reassurances. Screening sensitivity is affected by the characteristics of the programme (e.g. screening intervals, imaging technology, etc.), the target population (age, race, breast density, etc.), and the tumour (rate of disease progression, radiologic image, etc.).4–8
With the increased use of microarray-based gene expression profiling, traditional histopathology breast cancer classification has been reorganized into four molecular subtypes,9,10 each with a different incidence, prognosis, and treatment response.10,11 Treatment recommendations are now based on this classification.12,13 As a full genetic analysis of breast cancer is not easily available in clinical practice, the proposed surrogate definitions to differentiate the subtypes relies on the use of semiquantitative immunohistochemical (IHC) scoring of estrogen receptors, progesterone receptors and human epidermal growth factor receptor-2 (HER2) overexpression. These surrogate definitions classify breast cancers into luminal A-like, luminal B-like, HER2-positive and triple-negative. 12 Studies measuring the odds of interval cancer compared with screen-detected cancer according to the surrogate breast tumour subtypes all observed that triple-negatives were overrepresented in interval cancers.14–18 Only one study presented odds ratios separately for the luminal B-like, the triple-negative and the HER2-positive subtypes, in comparison with the less aggressive luminal A-like subtype, 17 and no study directly estimated the screening sensitivity according to tumour subtype.
We aimed to measure the association between breast tumour surrogate subtypes and mammography screening sensitivity within a regional division of the Quebec Breast Cancer Screening Program and to evaluate whether the subtype-sensitivity association was mainly mediated by tumour grade, a proxy for the proliferation rate of the tumour.
Methods
In the Quebec Breast Cancer Screening Program (Programme Québécois de dépistage du Cancer du sein, PQDCS) women aged 50–69 are invited for a biennial bilateral two-view mammogram at designated centres, and each mammogram is read by a radiologist (who must read at least 500 mammograms annually). Following a positive screen or following signs or symptoms after a negative screen, a medical assessment may occur either at an accredited or non-accredited reference centre, the former being women’s choice. The programme participation rate was between 51.7% and 59.2% during the study period.
The study took place in the Quebec City regional division of the PQDCS, which in 2017 carried out 39,444 of the 347,376 PQDCS screening episodes. The Centre des Maladies du Sein Deschênes-Fabia (CMSDF) is the only accredited reference centre for the Quebec City region. Figure 1 illustrates the study population selection process. Between 1 January 2003 and 31 December 2007, 1015 women with invasive breast cancers were diagnosed within 2 years of a screening mammogram. Of these 1015 women, 133 were excluded because, at time of screening, they were symptomatic or had undergone breast reduction/augmentation. A further 148 women were excluded because they were not assessed at the CMSDF accredited reference centre, which left 734 women in the study population. Screen-detected and interval cancers were identified using a validated algorithm that links the PQDCS database to the Quebec hospitals database and to the Quebec physician claims database. Every participant in the PQDCS provides informed consent allowing the use of their personal information for evaluation purposes. The CMSDF research ethics board waived study approval.

Flowchart for identifying the study population. PQDCS: Programme Québécois de dépistage du cancer du sein (Quebec breast cancer screening programme); CMSDF: Centre des maladies du sein Deschênes-Fabia (Quebec City regional accredited investigation centre).
The 2-year screening sensitivity was calculated as the number of screen-detected invasive cancers over the total number of screen-detected and interval invasive cancers diagnosed within 2 years of screening. 4 Screen-detected cancers were cases diagnosed within 6 months following a positive screen. Interval cancers were cases diagnosed either within 24 months of a negative screen or within 7–24 months following a positive screen. All laboratory tests were conducted at the accredited reference centre, and results were retrieved from the CMSDF database. Tumours were graded histopathologically using the Nottingham (Elston-Ellis) modification of the Scarff-Bloom-Richardson grading system. 19 Oestrogen and progesterone receptor status were considered positive if IHC assays stained at least 10% of cells. For HER2, a 3+ IHC assay score was deemed positive (overexpress) and a 0 or 1+ score was deemed negative (not overexpress). Following an equivocal 2+ score, gene amplification was performed and deemed positive if the ratio was greater than 2.2. Table 1 lists the subtype definition. We used grade and HER2 status to differentiate between luminal A-like and luminal B-like tumours. 12 Grade I–II was used as a proxy for Ki-67 less than 14% and grade III for Ki-67 of at least 14%.20,21 From the CMSDF database, we extracted age at diagnosis (50–54, 55–59, 60–64, 65–71 years), hormone replacement therapy (never/ever), body mass index (<20.0, 20.0–24.9, 25.0–29.9, 30.0–34.9, ≥35.0 kg/m2), histological type (ductal, others), first degree family history of breast cancer (none/any), mean tumour size, regional involvement, vascular invasion and cancer stage. From the PQDCS database, we extracted BI-RAD breast density (<25%, 25–49%, 50–75%, >75%), history of breast aspiration or biopsy (never/ever) and screening round (initial screening in the programme vs. any subsequent one).
Breast tumour surrogate molecular subtype definitions.
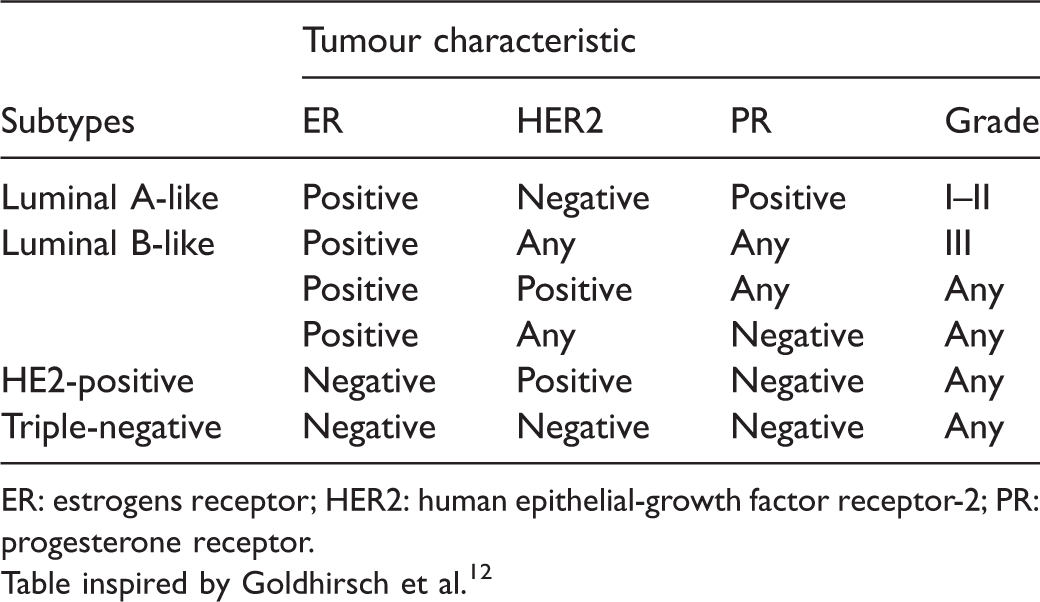
ER: estrogens receptor; HER2: human epithelial-growth factor receptor-2; PR: progesterone receptor.
Table inspired by Goldhirsch et al. 12
We first compared screen-detected and interval cancers according to women and tumour characteristics, and then estimated the association between tumour subtype (luminal A-like, luminal B-like, triple-negative or HER2-positive) and screening sensitivity using logistic regression. To facilitate result interpretation, we applied a marginal standardization, 22 which derives both average crude and average adjusted screening sensitivities from the logistic regression parameters, according to exposure level, for the whole study population. For each subtype category, using luminal A-like as the reference, we used the average screening sensitivities with the corresponding standard errors to calculate the crude and adjusted sensitivity ratios and the 95% confidence interval (CI). We also calculated sensitivity differences and the corresponding 95% CI. We adjusted the model by including age at diagnosis, hormone replacement therapy, body mass index, histological type, first-degree family history, breast density, history of breast aspiration or biopsy and screening round. We excluded from the analyses all subjects with a missing subtype value. When there were missing values for a confounding variable, we introduced a dummy category in the regression model, preventing further subject exclusion. Of the 734 women included in the study population, we were able to classify 639 (87.1%) by subtype. In the online Appendix, we present results where the missing values were handled by ‘multiple imputation’, with details on the multiple imputation method used.
In a secondary analysis, we explored whether the association observed between subtype and sensitivity was mediated by the co-variable grade, which is closely related to the proliferation rate of the tumour. 23 Assuming an association is causal, according to the ‘difference method’, when a presumed mediator is introduced in a fully adjusted statistical model and a meaningful drop is observed for the exposure–outcome association, the co-variable is likely to be a mediator (an intermediate step) on the causal pathway.24,25 Therefore, we compared the sensitivity, sensitivity ratios and sensitivity differences derived from the fully adjusted multivariate logistic regression model, with and without grade (i.e. the model’s presumed mediator). As grade is essential to differentiating luminal A-like from luminal B-like subtypes, in this secondary analysis, we merged the two luminal subtypes together, to avoid multicollinearity between the exposure and mediator variables. In this secondary analysis, the exposure variable, therefore, had only three categories, luminal, triple-negative and HER2-positive. Of the 734 women included in the study, we were able to classify 721 (98.2%) by this subtype definition combining both luminal A-like and B-like.
Finally, to verify whether screening rounds (initial vs. subsequent screens) modulate the subtype–sensitivity association, we fitted an interaction term between subtype and screening round in the multivariate models. A Wald F-test was used to verify the homogeneity of parameters. All analyses were performed with SUDAAN® release 11 and SAS® version 9.4 (SAS Institute, Cary NC). A two-sided p-value of less than 0.05 was considered statistically significant.
Results
Among the 734 women assessed at the accredited centre, 520 had a screen-detected cancer, and 214 had an interval cancer (Table 2). Women with a screen-detected cancer were less likely to have dense or very dense breasts, to have ever use hormone replacement therapy, or to have previous breast aspiration or biopsy. Their mean tumour size was smaller than interval cancers, and there was less regional involvement and vascular invasion. TNM stage was also more favourable.
Personal and tumour characteristics of the women included in analyses by mode of detection.
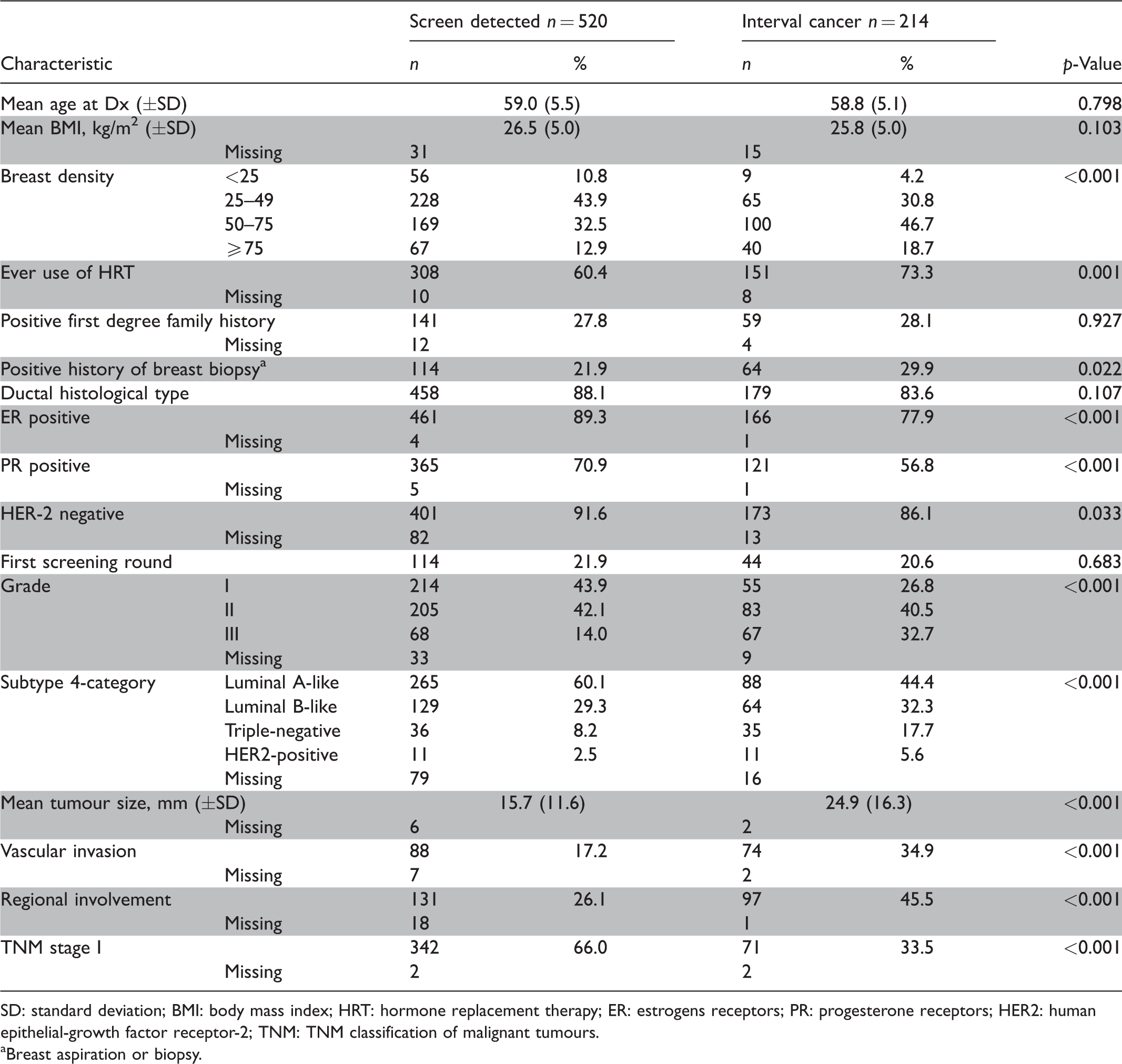
SD: standard deviation; BMI: body mass index; HRT: hormone replacement therapy; ER: estrogens receptors; PR: progesterone receptors; HER2: human epithelial-growth factor receptor-2; TNM: TNM classification of malignant tumours.
aBreast aspiration or biopsy.
In the primary analyses, we observed an adjusted 2-year sensitivity of 75.4% for the luminal A-like breast cancers (Table 3). In comparison with luminal A-like, the adjusted sensitivity was reduced by 9.3 points (95% CI = −17.1 to −1.4) for luminal B-like (sensitivity = 66.1%), by 22.5 points (95% CI = −35.0 to −10.0) for triple-negative (sensitivity = 52.9%), and by 30.1 points (95% CI = −50.0 to −10.2) for HER2-positive (sensitivity =45.3%). These correspond to adjusted sensitivity ratios of 0.88 (95% CI = 0.78 to 0.98) for luminal B-like, 0.70 (95% CI = 0.56 to 0.88) for triple-negative, and 0.60 (95% CI = 0.39 to 0.93) for HER2-positive. The Supplementary Table S1 online presents the logistic regression parameters and derived screening sensitivities from Table 3 after imputing missing subtype values instead of excluding subjects with a missing subtype value. Both approaches gave similar results.
CI: 95% confidence interval; HER-2: human epithelial-growth factor receptor-2.
aMarginal standardization was used to derive screening sensitivity, screening sensitivity ratio and screening sensitivity differences by tumour subtype from the crude and adjusted estimated logistic regression parameters.
bSubtype defined according to estrogens receptors, progesterone receptors, human epithelial-growth factor receptor-2 and grade (n = 639).
cScreening sensitivity is the number of screen-detected (SD) invasive cancers to the total number of SD and interval invasive cancers diagnosed between 1 January 2003 and 31 December 2007.
dWald F-test for homogeneity of parameters.
eAdjusted for age at diagnosis, family history of breast cancer, body mass index, hormone replacement therapy, breast density, history of breast aspiration/biopsy, tumour histological type and programme screening round (first or subsequent).
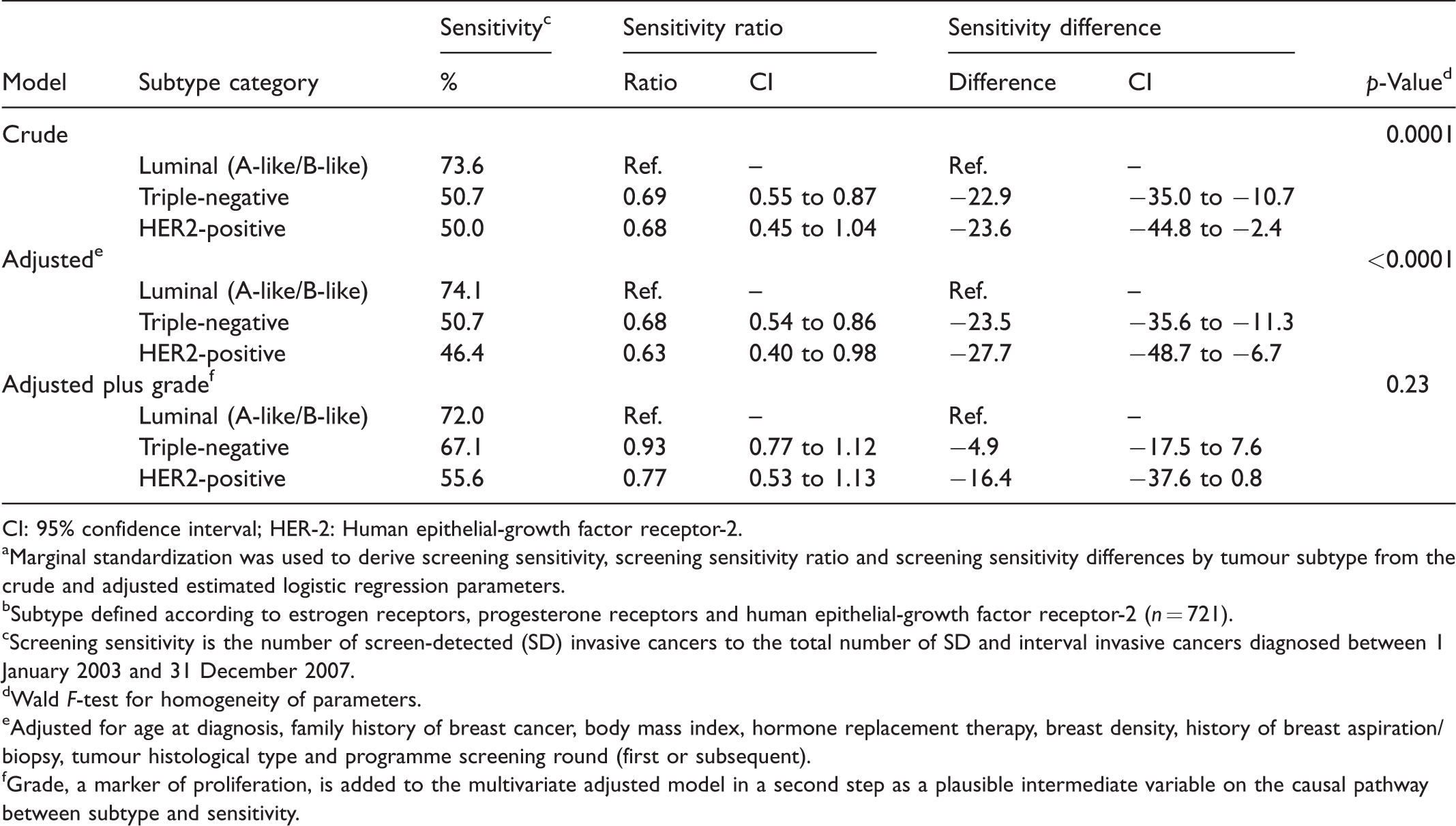
In the secondary analysis, before introducing grade to the fully adjusted multivariate model, the screening sensitivity for triple-negative tumours was 23.5 points lower (95% CI = −35.6 to −11.3) than that of luminal tumours. After adding grade to the model, it was only 4.9 points lower (95% CI = −17.5 to 7.6) (Table 4). In HER2-positive tumours, the screening sensitivity was 27.7 points lower (95% CI = −48.7 to −6.7) than that of luminal tumours before introducing grade to the model. After adding grade, it was only 16.4 points lower (95% CI = −37.6 to 0.8). When grade was added to the fully adjusted model, the absolute reductions in sensitivity were no longer statistically significant in triple-negative or in HER2-positive.
CI: 95% confidence interval; HER-2: Human epithelial-growth factor receptor-2.
aMarginal standardization was used to derive screening sensitivity, screening sensitivity ratio and screening sensitivity differences by tumour subtype from the crude and adjusted estimated logistic regression parameters.
bSubtype defined according to estrogen receptors, progesterone receptors and human epithelial-growth factor receptor-2 (n = 721).
cScreening sensitivity is the number of screen-detected (SD) invasive cancers to the total number of SD and interval invasive cancers diagnosed between 1 January 2003 and 31 December 2007.
dWald F-test for homogeneity of parameters.
eAdjusted for age at diagnosis, family history of breast cancer, body mass index, hormone replacement therapy, breast density, history of breast aspiration/biopsy, tumour histological type and programme screening round (first or subsequent).
fGrade, a marker of proliferation, is added to the multivariate adjusted model in a second step as a plausible intermediate variable on the causal pathway between subtype and sensitivity.
The Wald F-tests for the interaction between subtype and screening round (prevalent vs. incident screen) were not statistically significant (p-value = 0.45 for the multivariate model where luminal A and luminal B are distinct; p-value = 0.32 for the multivariate model where luminal A and B are combined).
Discussion
In the Quebec City regional division of the Quebec Breast Cancer Screening Program, the 2-year screening sensitivity for invasive cancers was 75.4% in luminal A-like, 66.1% in luminal B-like, 52.9% in triple-negative, and 45.3% in HER2-positive tumours. These results were not modified by screening round (prevalent vs. incident screen). For triple-negative tumours, no association remained when grade was added to the statistical model, suggesting that grade almost entirely mediates the subtype–sensitivity association. For HER2-positive tumours, the association was also greatly reduced and not statistically significant when grade was added to the model.
In studies comparing surrogate breast cancer subtypes of interval and screen-detected cancers, the odds ratios for interval presentation ranged from 2.3 to 4.7 for triple-negative tumours compared with the other subtypes.14–16,18 Every ratios were statistically significant. In the study classifying tumours according to a four-category subtype definition, using luminal A-like as the reference group, the odds ratios of interval presentation were 1.45 (95% CI = 0.68 to 3.07) and 1.30 (95% CI = 0.48 to 3.54) for two luminal B-like subgroups, 3.16 (95% CI = 0.81 to 12.26) for triple-negative, and 1.09 (95% CI = 0.26 to 4.55) for HER2-positive. 17 In our study, using luminal A-like as the reference category, the odds of interval presentation were 1.64 (95% CI = 1.08 to 2.45) for luminal B-like, 3.03 (95% CI = 1.68 to 5.42) for triple-negative and 4.25 (95% CI = 1.70 to 10.70) for HER2-positive.
To our knowledge, this is the first study to directly estimate the effect of IHC surrogate subtypes on the sensitivity of a mammography screening programme. Measuring the effect of subtypes on screening sensitivity through the comparison of screen-detected and interval cancers presupposes that such subtypes are stable over the course of pre-clinical and early clinical breast cancer. It has repeatedly been shown that in situ breast cancers display the full spectrum of molecular subtypes before any signs of invasiveness,26–30 pointing towards early determination of the cancer’s nature. The prognostic importance of subtypes is well established in early stage breast cancer,12,31 which also suggests that subtypes are stable characteristics in the early stage of the disease.
This is also the first study to explore the causal pathway between breast tumour subtypes and mammography screening sensitivity. The tumour’s radiological image, duration of pre-clinical phase, and growth environment (e.g. breast density) are the three main tumour-related components that may affect mammography sensitivity. We used grade, an indicator of the tumour’s proliferation rate, to explore whether the reduction in sensitivity for both the triple-negative and HER2-positive subtypes was driven by a short pre-clinical phase. Assuming the subtype–sensitivity association we observed is causal, our results suggest that a short pre-clinical phase entirely explains the poor sensitivity for triple-negative, and greatly explains the poor sensitivity for HER2-positive tumours. Given the importance of grade in differentiating luminal A-like from luminal B-like tumours, a shorter pre-clinical phase might also cause the reduction in sensitivity of luminal B-like tumours.
The results of this study suggest that current population-based mammography screening programmes fail to detect the most aggressive breast tumours, and that tailoring programmes to take into account the heterogeneity of the breast tumour pre-clinical phase would lead to more efficient breast screening. This would mean identifying women at a greater risk for aggressive breast cancer subtypes, and appropriately adjusting both their screening intervals and the associated screening imaging technology. The growing evidence that established breast cancer risk factors are differentially associated with each breast cancer subtype28,32,33 offers the hope that breast cancer subtype risk stratification will eventually be possible. This study also indicates that population’s characteristics affecting mammography screening sensitivity might be closely related to the population distribution of subtype risk factors. As research on breast cancer subtype risk factors evolves, integrating the target population’s subtype risk factors distribution to the programmes’ sensitivity indicators will become important.
This study has some limitations. First, in molecular cancer studies, missing biomarker values often arise due to impossible or inappropriate pathological assessment of small tumours, 34 and in our main analysis, we excluded 13% (95/734) of the study population because of missing subtype values. This proportion is relatively small, and results remained unchanged when multiple imputation was used instead of a the complete case analysis approach.35,36 Also, in the secondary analysis, where only 2% were excluded (13/734), results for triple-negative and for HER2-positive, in comparison with luminal tumours, were similar to those of main analysis in terms of magnitude and direction.
Second, we assumed that the 734 breast cancer cases assessed at CMSDF (83% of the 882 total cases in the programme's regional division.) adequately represented all cases diagnosed in the target population of the Quebec City regional division of Quebec breast cancer screening program. Characteristics of the CMSDF cases were similar to those of the excluded cases for all variables we considered as potential confounders (Supplemental Table S2 online), and overall sensitivity was similar between included and excluded cases (70.8% vs. 69.9%) (Supplemental Table S2 online). These estimated sensitivities closely resemble the 2003–2007 average programme sensitivity computed for the programme’s regional division (68.6%). Moreover, although the regional division represents 10% of the programme’s yearly mammography screening, there is only a 0.8 point difference between the regional (68.6%) and provincial (67.8%) 2003–2007 programme sensitivity. Altogether, this suggests that the CMSDF cases adequately represented the target population. Nevertheless, our study remains an institution-based study and should be replicated with population-based data, ideally including the whole programme target population instead of a regional division.
Third, the study population includes women screened between 2001 and 2007. The distribution of breast cancer’s subtypes may have since changed, paralleling demographic changes in subtype risk factors distribution. This should not affect the genuine relation between breast cancer subtypes and mammography screening sensitivity. During the study period, screen-film was the norm, whereas digital technology now dominates, but recent studies suggest that both technologies have similar screening sensitivity and generate screen-detected and interval cancers with comparable characteristics.37,38
Fourth, surrogate subtypes are based on tests that distinguish various immunoreactive cells. During the study period, the positivity threshold for hormone receptors was of at least 10% of immunoreactive cells, rather than the 1% now recommended. 39 This could have induced a degree of misclassification of tumours according to subtypes in our dataset, equally present in screen-detected and interval cases. This non-differential misclassification bias may have artificially attenuated the association measures. 40
Conclusion
The Quebec City regional division of the Quebec Breast Cancer Screening Program had a lower 2-year screening sensitivity for luminal B-like, triple-negative and HER2-positive breast cancers compared with luminal A-like cancers. This subtype–sensitivity association is mediated by tumour grade. These results demonstrate that the accuracy of mammography screening programmes varies considerably with tumour biology. Tailoring screening according to women’s subtype risk factors might eventually lead to more efficient breast cancer screening programmes.
Supplemental Material
Supplemental Material1 - Supplemental material for Breast cancer subtype and screening sensitivity in the Quebec Mammography Screening Program
Supplemental material, Supplemental Material1 for Breast cancer subtype and screening sensitivity in the Quebec Mammography Screening Program by Linda Perron, Sue-Ling Chang, Jean-Marc Daigle, Nathalie Vandal, Isabelle Theberge, Caroline Diorio, Julie Lemieux, Eric Pelletier and Jacques Brisson in Journal of Medical Screening
Supplemental Material
Supplemental Material2 - Supplemental material for Breast cancer subtype and screening sensitivity in the Quebec Mammography Screening Program
Supplemental material, Supplemental Material2 for Breast cancer subtype and screening sensitivity in the Quebec Mammography Screening Program by Linda Perron, Sue-Ling Chang, Jean-Marc Daigle, Nathalie Vandal, Isabelle Theberge, Caroline Diorio, Julie Lemieux, Eric Pelletier and Jacques Brisson in Journal of Medical Screening
Footnotes
Acknowledgements
We thank André Langlois (l’Institut national de santé publique du Québec) and Danielle Audet (Centre des maladies du sein Deschênes-Fabia) for their ongoing support throughout this work, and Marie-Hélène Guertin (l’Institut national de santé publique du Québec) for her helpful comments and suggestions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.