Abstract
Objective
To compare breast characteristics, compression parameters, and early performance measures (rates of recall, screen-detected and interval breast cancer, and histopathologic tumour characteristics) for mammographic screening at a stationary versus mobile screening unit.
Methods
Results from 92,408 mammographic screening examinations performed as part of BreastScreen Norway during 2008–2017 at either a stationary (n = 52,620) or mobile (n = 39,788) unit in Hordaland county were compared using descriptive statistics and generalized estimating equations. A generalized estimating equation for a binary outcome was used to estimate crude and adjusted odds ratios with 95% confidence intervals for the outcomes of interest. Adjusted generalized estimating equation models included age, breast volume, and density grade as covariates.
Results
Screening at the stationary unit was performed on smaller breasts with higher mammographic density, using lower compression force but higher pressure than at the mobile unit. Using the stationary screening unit as reference, for women screened at the mobile unit, the adjusted odds ratio was: for recall 0.94 (95% CI: 0.87--1.01), screen-detected breast cancer 0.92 (95% CI: 0.78--1.10), and interval breast cancer 1.17 (95% CI: 0.83–1.64).
Conclusions
The quality of care did not differ for women screened at the stationary versus the mobile unit, but there were differences between the women who attended the two units. Sociodemographic factors should be included in future analyses to fully understand the risk of breast cancer among women residing in urban versus rural areas.
Keywords
Introduction
BreastScreen Norway, administered by the Cancer Registry of Norway, offers biennial, population-based mammographic screening to women aged 50–691 at stationary and mobile screening units. In 2017, BreastScreen Norway's target population was 650,000 women, of whom approximately 17% were offered screening at a mobile unit. The attendance rate among women invited to mobile units was higher than that among women invited to stationary units (80% vs. 75%).
Early performance measures are quality indicators for breast cancer screening programs, and are usually presented for a screening program as a whole.2,3 Sociodemographic factors, including residential area, have been shown to affect both the incidence and risk of breast cancer, and could affect early performance measures.4–8 Because mobile units usually service rural areas with different sociodemographic characteristics from urban populations, it may be useful to stratify the evaluation of early performance measures. A higher rate of recall, screen-detected, and interval breast cancer has been observed among women screened in BreastScreen Norway at stationary compared with mobile units. 1 Characteristics of women residing in rural and urban areas could therefore have potential for stratifying early performance measures and for stratified mammographic screening. To our knowledge, no research in this area has been undertaken to date.
In mammographic imaging, the breast is compressed to improve image quality and to reduce radiation dose. 9 Breast compression parameters can be affected by breast characteristics, including breast volume, and mammographic density. 10 High mammographic density is an independent risk factor for breast cancer and affects early performance measures, as well as tumour histopathology.11,12 We are not aware of any research addressing whether there are systematic differences in breast characteristics and breast compression parameters between women screened at stationary versus mobile units.
About 17% of women attending BreastScreen Norway in Hordaland County were offered screening at a mobile unit in 2017. 1 Using data from digital mammographic screening examinations performed during 2008–2017, we investigated breast characteristics, breast compression parameters, and early performance measures for women screened at the stationary versus the mobile screening unit in Hordaland County.
Methods
The requirement to obtain written informed consent for this retrospective analysis of de-identified data from BreastScreen Norway was waived under the Cancer Registry Regulations. Institutional review board approval was not required. 13
Between 1 January 2008 and 31 December 2017, 199,260 screening examinations were performed among 63,655 women aged 50–71 residing in Hordaland County (Figure 1). Standard two-view digital mammography (cranio-caudal and medio-lateral oblique views) was performed by a team of two radiographers, either at a stationary unit in the city of Bergen, or at a mobile unit. The mobile unit was stationed at eight central locations during one screening round until 2013, when three of the locations were closed and the women were invited to the stationary unit. Mammography equipment from GE Healthcare was used at the stationary unit (GE Senographe DS™ from 2008–2014 and GE Senographe Essential™ from 2014–2017), and the mobile unit (GE Senographe Essential™). Images acquired at the stationary unit were stored directly in the Picture Archiving and Communication System at the breast centre. Images taken at the mobile unit were stored on an encrypted memory stick and taken to the breast centre, where they were transferred to the Picture Archiving and Communication System. All mammograms were read at GE workstations (Seno Advantage or Image Diagnost International). All radiographers and radiologists in this study were employed by the breast centre at Haukeland University Hospital and served both the stationary and the mobile units. The same pool of radiologists performed screen-reading and recall assessment on all women included in the study. All screening examinations were independently double read by two radiologists, who gave a score of 1–5 for each breast to indicate the level of mammographic suspicion for breast malignancy. 1 All examinations given a score of 2 or higher by one or both radiologists were discussed, and a consensus reached on whether to recall the woman for further assessment due to mammographic findings, hereafter referred to as recall.

Number of examinations available for the study, exclusions and final study population.
Screening examinations were used as the unit of analysis. Women could be included in the study population more than once if they attended several screening examinations during the study period. We excluded 14,689 examinations performed with digital breast tomosynthesis as part of the Tomosynthesis Trial in Bergen in 2016 and 2017 14 (Figure 1). To ensure the validity of breast compression parameters, we excluded examinations of women who attended both the mobile and the stationary unit (stationary n = 7494; mobile n = 11,516), as well as examinations by radiographers who performed <100 examinations during the study (stationary n = 33,473; mobile n = 162). We also excluded examinations where mammographic density and compression data from Volpara®Density™ were not available (stationary n = 15,001; mobile n = 5179), examinations that did not include exactly four standard mammographic images (left/right cranio-caudal view and left/right medio-lateral oblique view) (stationary n = 12,155; mobile n = 6919), and examinations where the women were recalled due to symptoms reported at screening, or technically unsatisfactory images (stationary n = 173; mobile n = 91). The final study population included 92,408 screening examinations performed among 44,702 women – 52,620 at the stationary and 39,788 at the mobile unit.
In this study, breast characteristics were defined as breast volume (cm3), mammographic density measured as volumetric breast density (VBD, %) and fibroglandular volume (cm3). VBD was classified into Volpara Density Grades (VDG) as follows: VDG1 (VBD <4.49%); VDG2 (VBD 4.5–7.49%); VDG3 (VBD 7.5–15.49%) and VDG4 (VBD ≥15.5%). 15 These categories are analogous to the BI-RADS 5th edition density categories a–d. 16 Breast compression parameters were defined as compression force (Newton [N]), compression pressure (kilopascal [kPa]), and compressed breast thickness (mm). Compression force was defined as the force applied by the radiographer during the examination and is measured in kg or N. Compression pressure was defined as the force divided by the area of the breast in contact with the compression paddle and is measured in kPa. Compressed breast thickness was measured in mm and defined as the thickness of the compressed breast.
Early performance measures included the rate of recall, screen-detected and interval breast cancer, and the positive predictive value of recalls and needle biopsies, as well as histopathologic tumour characteristics (tumour diameter, histologic grade, lymph node involvement, oestrogen and progesterone receptor status, and human epidermal growth factor receptor 2 status). The definition of screen-detected and interval breast cancer included ductal carcinoma in situ and invasive breast cancer. The recall rate was calculated as the proportion of recalls due to abnormal mammographic findings, biopsy rate as the proportion of needle biopsies performed at recall assessment, and rate of screen-detected breast cancer as the number of breast cancers diagnosed after recall, among the screening examinations. The rate of interval breast cancer was defined as the number of breast cancers diagnosed 0–24 months after a negative screening examination or 6–24 months after a false-positive screening examination, divided by the number of screening examinations. Positive predictive value of recalls was defined as the percentage of screen-detected breast cancer cases detected among recalls, and positive predictive value of needle biopsies as the percentage of screen-detected breast cancer detected among needle biopsies performed at recall assessments. Histopathologic tumour characteristics for invasive tumours included tumour diameter (≤10 mm, >10–≤20 mm and >20 mm), histologic grade (1, 2 and 3), lymph node involvement (positive/negative), and oestrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 status (positive/negative).
Information about breast characteristics and breast compression parameters was derived by an automated software for breast density assessment, Volpara®Density™ (version 15.1; Volpara Health Technologies Ltd, Wellington, NZ). 15 BreastScreen Norway supplied information about screening examinations, including recall, biopsies, cancer detection, and histopathologic tumour characteristics.
Descriptive statistics were used to compare distributions of age, breast characteristics, breast compression parameters, and early performance measures, including histopathologic tumour characteristics, stratified by screening unit. We presented means and standard deviation or median and interquartile range, depending on the distribution of the variable being described. We calculated 95% exact (Clopper–Pearson) confidence intervals (CIs) for the proportions associated with the histopathologic tumour characteristics.
Analyses of early performance measures and histopathologic tumour characteristics of screen-detected and interval breast cancer were presented as rates per 100 screening examinations. The independency assumption for standard regression models was violated because more than one screening examination could be included per woman, and because some variables changed over time. Generalized estimating equations (GEE) with a logit link function were therefore used to analyze differences in the two units.
Unadjused GEE was used to analyze differences in age, breast characteristics and compression parameters. Using a uni- and multivariable GEE for binary outcomes, we estimated the crude and adjusted odds ratios (OR) and 95% CIs for the outcome of interest, recall, screen-detected, and interval breast cancer when screening at a mobile versus a stationary unit (reference). Covariates in the adjusted models included five-year age groups (<55, 55–59, 60–64 and >64), quartiles of breast volume, and VDG (1–4).
We used STATA version 15 (Stata Corp, TX) for all statistical analyses. A p-value of <0.05 was considered statistically significant.
Results
The mean ages of women screened at the stationary unit and at the mobile unit were 59 and 60, respectively (p < 0.01, Table 1). The median breast volume was statistically lower for those screened at the stationary versus mobile unit (780 cm3 vs. 886 cm3), while the median volumetric breast density (5.4% vs. 4.5%) and fibroglandular volume (41.1 cm3 vs. 39.5 cm3) were higher at the stationary unit (p < 0.01 for all). The mean compression force and compressed breast thickness were statistically lower at the stationary unit than the mobile unit (115.5 N vs. 116.6 N and 57.1 mm vs. 60.0 mm, respectively), while the mean compression pressure was higher (12.0 kPa vs. 11.5 kPa) at the stationary unit (p < 0.01 for all).
Mean and standard deviation (SD), or median and interquartile range (IQR) for age, breast characteristics and compression parameters, by screening unit (stationary/mobile) for examinations performed in BreastScreen Norway (Hordaland County), 2008–2017.
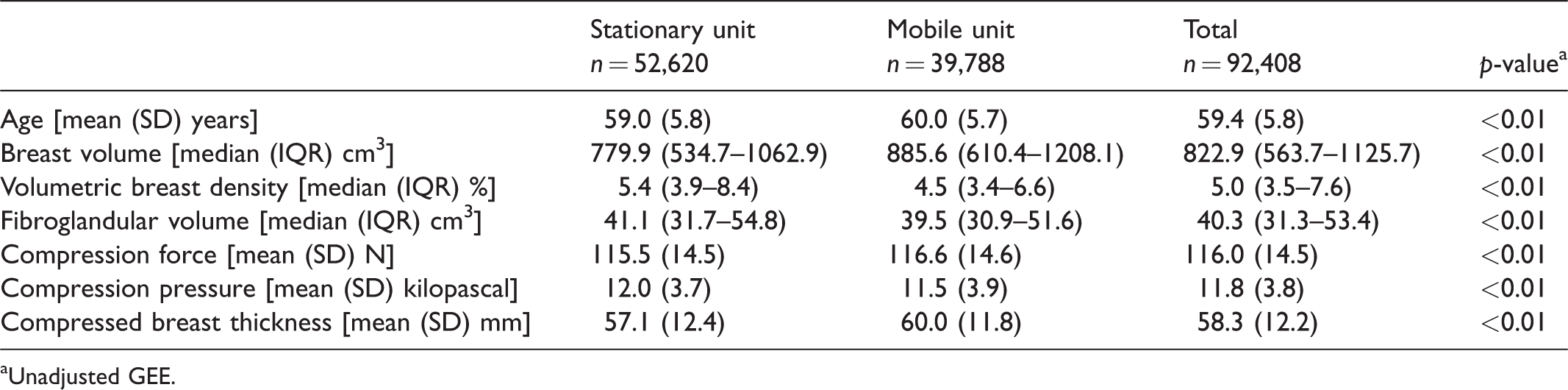
aUnadjusted GEE.
The unadjusted recall and biopsy rates were higher among women screened at the stationary versus the mobile unit (3.7% vs. 3.2%; and 1.8% vs. 1.5%, respectively), although the unadjusted rates of screen-detected and interval breast cancer did not differ statistically (Table 2). Based on the CI for the estimated proportions, we did not observe any statistical differences in histopathologic tumour characteristics for women diagnosed with screen-detected breast cancer after screening at the stationary versus the mobile unit (Table 3). Interval breast cancer diagnosed after screening at a stationary unit was more likely to be ≤10 mm (21.0% vs. 6.8%) and less likely to be grade 2 (23.4% vs. 42.6%) than interval breast cancer detected after screening at mobile units (Table 3). No other tumour histopathology differed between the two groups.
Frequencies and rates of early performance measures by screening unit (stationary/mobile) for examinations performed in BreastScreen Norway (Hordaland County), 2008–2017.
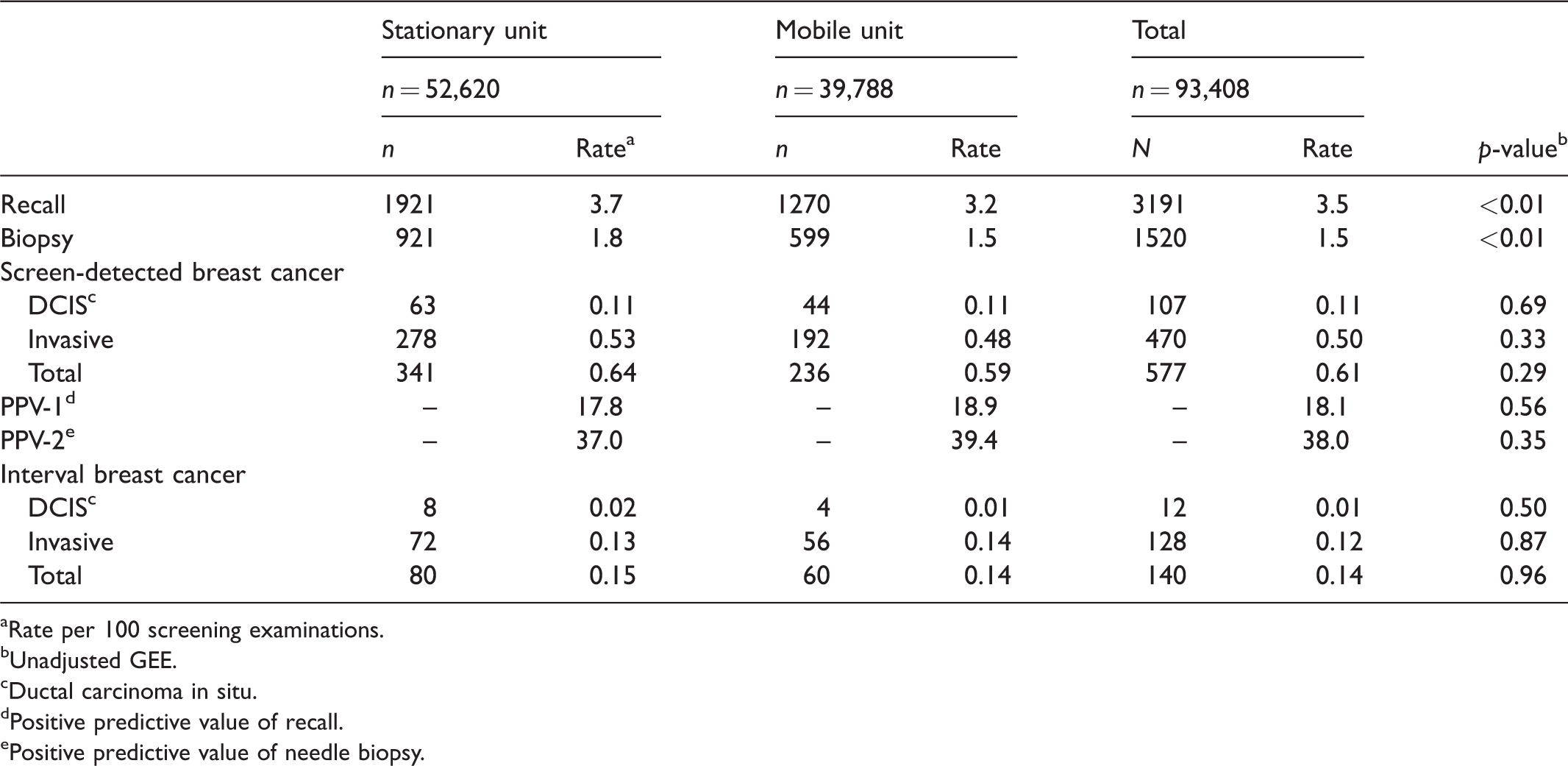
aRate per 100 screening examinations.
bUnadjusted GEE.
cDuctal carcinoma in situ.
dPositive predictive value of recall.
ePositive predictive value of needle biopsy.
Distribution (%; 95% confidence interval, CI) of histopathologic tumour characteristics of invasive screen-detected and interval breast cancer, by screening unit (stationary/mobile) for examinations performed in BreastScreen Norway (Hordaland County), 2008–2017.
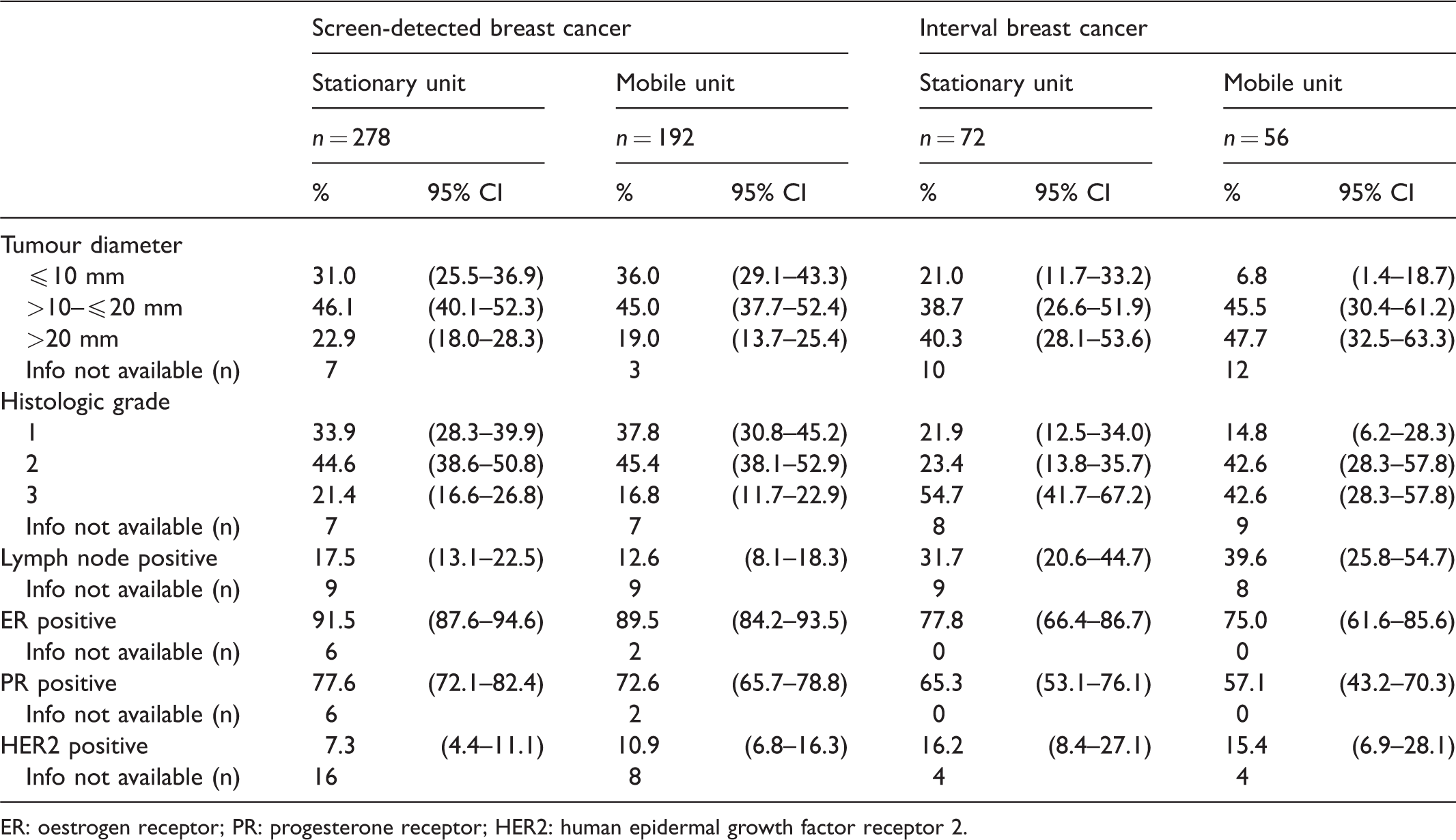
ER: oestrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2.
The adjusted odds ratio of recall, screen-detected breast cancer, and interval breast cancer did not differ between the two units (OR 0.94 (95% CI: 0.87--1.01); OR 0.92 (95% CI: 0.78--1.10); and OR 1.17, 95% CI: 0.83–1.64 respectively; p = 0.02; Table 4).
Crude and adjusted a odds ratios (OR) with 95% confidence interval (CI) of recall, screen-detected and interval breast cancer, by screening unit (stationary and mobile), in Hordaland County, 2008-2017.
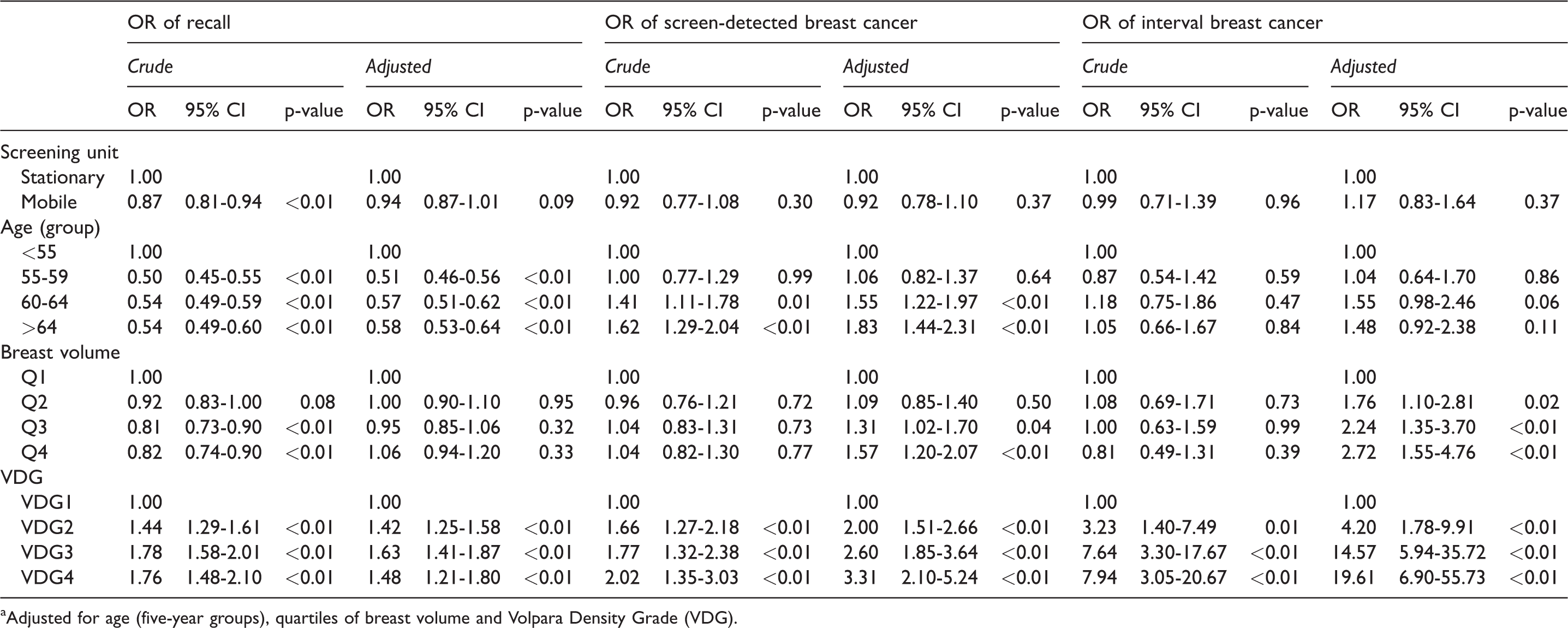
aAdjusted for age (five-year groups), quartiles of breast volume and Volpara Density Grade (VDG).
After adjusting for breast volume and VBD, the estimated compression force increased with age, while pressure decreased with age. Compression force was higher and the compression pressure lower for women of all ages screened at the stationary unit compared with the mobile unit (Figure 2(a) and (b)).
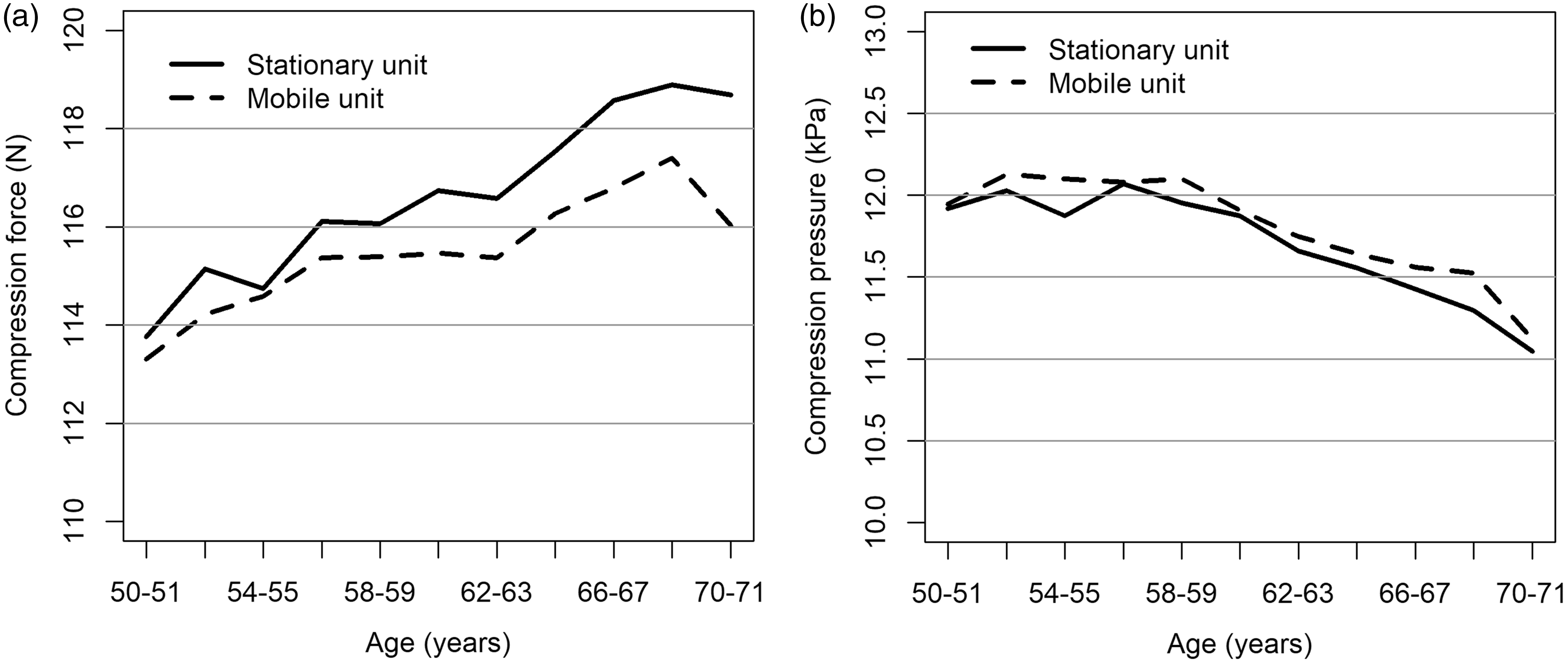
Compression force (a) and compression pressure (b) by screening unit (stationary/mobile) and two-year age groups, adjusted for breast volume and volumetric breast density, for screening examinations performed in BreastScreen Norway (Hordaland County), 2008–2017.
Discussion
This study is, to our knowledge, the first to compare breast characteristics, breast compression parameters, and early performance measures for screening at a stationary and mobile unit in a population-based breast cancer screening program. We observed that women screened at the stationary unit had smaller breasts, with higher mammographic density, than women screened at the mobile unit. Lower compression force and higher compression pressure were used at the stationary unit. The adjusted ORs for recall, screen-detected, and interval breast cancer did not differ between the two units. Histopathologic tumour characteristics did not differ between the two groups, except for less prognostically favourable tumour diameter and histologic grade for interval breast cancer among women screened at the mobile unit. Given that the mobile unit primarily serves the rural population, sociodemographic factors might be useful to consider as future stratification variables for early performance measures and personalized mammographic screening.
Studies have shown that women living in urban areas have a higher incidence of breast cancer than those in rural areas.7,17,18 Our study does not corroborate these findings, as we failed to observe a statistical difference in the rates of screen-detected interval breast cancer among women screened at a stationary (urban) or mobile (rural) unit. Women attending the stationary unit in our study had smaller and more mammographically dense breasts than women attending the mobile unit, which indicates that they have a different risk for breast cancer; however, small breast volume is also associated with low body mass index (decreased risk of breast cancer in post-menopausal women) and high mammographic density (increased risk of breast cancer).11,19 These two effects may counterbalance each other. Additionally, more accessible mammographic screening has been proposed as a possible explanation for higher breast cancer incidence in urban populations. 20 In our population-based screening program, all invited women receive an offer for screening with a fixed place and time for examination. The screening location is based on residential address, and the program aims to keep the one-way travel time to the screening unit below 1 hour. The criteria for the assigned location for examination might thus reflect sociodemographic differences among women invited to stationary versus mobile units in Norway. In 2019, the average travel time from a central point in the women’s residential locales is 25 min to the stationary unit and 16 min to the mobile unit in Hordaland. For those recalled for further assessment, travel time to the breast centre is 25 min and 2 h, respectively. Access to screening is similar for the women invited to the stationary or mobile units, which may also explain why we did not observe any statistical difference in cancer detection between these two groups. However, women screened at the mobile unit had interval breast cancer with a larger tumour diameter and higher histologic grade compared with those screened at the stationary unit. This might indicate a higher threshold to seek clinical mammography for women living in the rural area.
Several studies have shown that different lifestyle and distributions of sociodemographic factors among women living in urban versus rural areas are associated with breast cancer risk.7,21,22 A Norwegian study from 2005 reported a lower risk of breast cancer in women residing in rural compared with urban areas, even after adjusting for sociodemographic factors 4 ; however, this study included women aged 30–62 and the study period was before the startup of BreastScreen Norway. Our results, based on screened women, did not support these findings. BreastScreen Norway is administered centrally and ensures that all women have equal access to screening, follow-up, and treatment, independent of residential area and baseline risk factors. Our study showed that women living in urban and rural areas had similar risk for screen-detected and interval breast cancer, which we attribute to the implementation of the screening program. Other studies have reported more advanced breast cancer among those screened at mobile units, 17 which was partly observed for interval breast cancer in our study.
By offering screening at mobile units, BreastScreen Norway reaches women in their local area, which results in a higher attendance rate than for those invited to stationary units. 1 However, screening at a mobile unit presents some difficulties. In BreastScreen Norway, obtaining image storage and reading stations compatible with those at stationary locations has been challenging. The costs of procuring, maintaining, administering, and driving the unit to strategic locations based on the average journey for women in those areas must be considered in relation to the benefits. Locations for the mobile unit require access to electricity and washroom facilities, among other considerations. In addition, access to and the expenses associated with trained radiographers who are willing to travel and stay away from home for longer periods can also be a challenge.
Our study found that the compression force increased with age, while the pressure decreased. This could be explained by increased breast volume by age. 23 We also found that lower compression force and higher pressure were applied to women screened at the stationary unit, compared with the mobile unit. Women at the stationary unit had smaller breasts. This supports previous findings that breast characteristics influence breast compression parameters. 10 Continuous surveillance of early performance measures, stratified by screening unit (as a proxy for urban and rural areas) might, therefore, advance our knowledge about early performance measures in mammographic screening. This type of analysis could also improve knowledge about breast cancer risk factors, which is relevant in the progress and development of stratified screening for breast cancer. Stratified breast cancer screening based on breast cancer risk factors has been proposed to improve breast cancer screening effectiveness. 24 Mammographic density is one of the primary candidates to stratify screening protocols, but this study shows that residential areas might also be a useful candidate for stratified screening. However, stratified screening is a complex issue due to the vast number of factors that must be considered, including the practical consequences. 25
Our study was based on data from one county in Norway and had stringent exclusion criteria. For example, we only included examinations with four standard images, even though women with larger breasts can require additional exposures to image the entire breast when a standard-sized detector is used. The mammographic equipment used at the stationary unit from January 2008 to October 2014 only had a standard-sized image detector, while the equipment used at the mobile unit for the whole study period, and at the stationary unit from November 2014 to December 2017, offered the standard and a larger image detector. Women with larger breasts who obtained more than four images per examination were excluded, which would result in a lower average breast volume.
To ensure validity of the data, we only included screening examinations performed by radiographers with experience from both the stationary and mobile unit (more than 100 examinations per unit). Several radiographers had performed a high number of examinations at the stationary unit, but had never worked at the mobile unit, and so a larger proportion of examinations performed at the stationary versus the mobile unit was excluded, although this probably had a negligible effect on the outcomes of this study. In addition, we also excluded women who attended both the stationary and mobile units during the study period due to an administrative change in the invitation process.
Because radiologists are aware that women screened at the mobile unit experience longer travel times, they may increase their screening sensitivity to avoid undue inconvenience among women living in more rural areas. We observed a lower recall rate for women screened at the mobile unit, but this effect disappeared in adjusted analyses, suggesting that women receive comparable care independent of residential area.
Conclusions
This is the first study comparing breast characteristics, breast compression parameters, and early performance measures for women screened at a stationary versus a mobile unit in a population-based screening program for breast cancer. Women screened at the stationary unit had a lower mean breast volume and higher VBD than women screened at the mobile unit. Early performance measures were comparable for the two units, except for two parameters related to histopathologic characteristics of interval breast cancer. Despite this, we consider that the results indicate comparable quality of care for women screened at the two units. Our results could be relevant for personalized screening protocols in the future. Further research on this topic should include data from other counties, and examine sociodemographic factors in depth, to fully understand the effects of mammographic screening in women residing in urban versus rural areas.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Solveig Hofvind has permanent employment as a researcher at the Cancer Registry of Norway, independent of her job as the administrative leader of BreastScreen Norway.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
