Abstract
Background
Trials of cancer screening present results in terms of deaths prevented, but metastasis is also a key endpoint that screening seeks to prevent. We developed a framework for projecting overall (de novo and progressive) metastases prevented in a screening trial using prostate cancer screening as a case study.
Methods
Mechanistic simulation model in which screening shifts a fraction of cases that would be metastatic at diagnosis to being non-metastatic. This shift increases the incidence of non-overdiagnosed, organ-confined cases. We use estimates of the risk of metastatic progression for these cases to project how many progress to metastasis after diagnosis and tally the projected de novo and progressive metastatic cases with and without screening. We use data on stage shift from the European Randomized Study of Screening for Prostate Cancer (ERSPC) and data on the risk of metastatic progression from the Scandinavian Prostate Cancer Group-4 trial. We estimate the relative risk and absolute risk reductions in metastatic disease at diagnosis and compare these with reductions in overall metastases.
Results
Assuming no effect of screening beyond initial stage shift at diagnosis, the model projects a 43% reduction in metastasis at diagnosis but a 22% reduction in the cumulative probability of metastasis over 12 years in favor of screening. These results are consistent with the empirical findings from the ERSPC.
Conclusion
Any reduction in metastatic disease at diagnosis under screening is likely to be an overly optimistic predictor of the impact of screening on overall metastasis and disease-specific mortality.
Keywords
Introduction
A key rationale underlying cancer screening is that early detection of tumors while still localized will translate into a reduction in cancer mortality. In practice, however, the reduction in advanced stage incidence under screening does not always translate into a similar reduction in disease-specific deaths. In the European Randomized Study of Screening for Prostate Cancer (ERSPC), for example, the incidence of metastatic disease at diagnosis was reduced by 40%, but prostate cancer mortality was reduced by 20% at 12 years of follow-up.1–3
The expectation that early detection will reduce advanced-stage cancer, referred to as “stage shift,” is a key motivation for the development of novel screening tests.3,4 However, stage shift at diagnosis does not account for progressive cases that may develop after diagnosis. Any reduction in advanced-stage disease at diagnosis will translate into an increase in organ-confined diagnoses, thus increasing the pool of cases eligible to progress to metastasis. Progressive cases could attenuate any initial difference in the incidence of metastatic disease at diagnosis, allowing accumulating metastatic cases in a screened group to catch up with a non-screened group over time.
Most population-based screening trials do not compare the overall risk of metastasis in the screening and control arms during follow-up but focus instead on metastasis at diagnosis and disease-specific mortality. Empirical observation of the effect of screening on overall metastasis can be onerous and time-consuming, requiring similarly rigorous follow-up of localized cases on both trial arms for disease progression and recurrence. Being able to project the likely consequences of an observed stage shift at diagnosis for the overall difference in advanced-stage disease could help investigators to develop realistic projections of long-term screening impact.
We develop a modeling framework for projecting from the reduction in metastasis at diagnosis to the reduction in overall metastasis under screening. Our modeling framework clarifies the data and assumptions needed in general to project long-term outcomes of screening based on information about stage shift at diagnosis. We use the framework to project how lower incidence of metastasis at diagnosis in the ERSPC would translate into incidence of metastasis overall and compare it with the observed pattern of overall metastasis in the trial.
Methods
Overview
In a screened population, the observed increase in the number of cases that are localized at diagnosis reflects a combination of overdiagnosed cases and non-overdiagnosed cases. We assume that only non-overdiagnosed cases will progress to metastatic disease, so we require a way to partition the localized cases at diagnosis into overdiagnosed and non-overdiagnosed components. In our development, we use an established model of prostate cancer natural history, screening, and incidence to simulate the ERSPC trial and project the increase in localized incidence at diagnosis among participants in the screen arm.5–7 The natural history model permits us to quantify this increase and partition it into overdiagnosed and non-overdiagnosed cases. The model also produces a decline in metastatic disease at diagnosis under screening.
For non-overdiagnosed localized cases (i.e., cases that would be clinically detected in a patient’s lifetime in absence of screening), we project time to progression using data from an established treatment trial (Scandinavian Prostate Cancer Group-4 (SPCG-4) trial), 8 which provided detailed information on metastasis and disease-specific mortality among its (mostly clinically detected) localized participants.
In summary, the modeling framework includes three key components: (1) stage-specific (localized and metastatic) incidence of disease at diagnosis with and without screening; (2) partitioning of excess localized cases in the screen arm into overdiagnosed and non-overdiagnosed; and (3) distribution of time from diagnosis to metastasis of non-overdiagnosed, localized cases. Table 1 summarizes the simulation model data sources. We do not initially assume any effect of screening on the risk of progression; our primary model assumes that the effect of screening is only via a shift in the distribution of stage at diagnosis. We explore in sensitivity analyses how our projections might change if screening was to delay metastatic progression in addition to reducing metastatic incidence.
Components of the simulation model for projecting de novo and progressive metastasis.
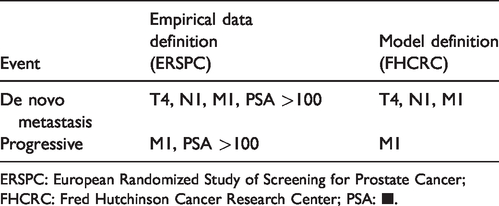
ERSPC: European Randomized Study of Screening for Prostate Cancer; FHCRC: Fred Hutchinson Cancer Research Center; PSA: ■.
ERSPC and SPCG-4 trial data
The ERSPC trial, initiated in 1993, is a large-scale randomized controlled screening trial for prostate cancer that compared Prostate-Specific Antigen (PSA) screening every 2–4 years to usual care between the ages of 55 and 69. We used data from N = 76,813 men (N = 36,270 screening; N = 40,543 control) from the four largest centers in the study (Netherlands (N = 34,833), Sweden (N = 11,852), Finland (Tampere location only) (N = 20,225), and Switzerland (N = 9903) with a median 12 years’ complete follow-up. The study protocol did not establish a common follow-up scheme for the ascertainment of metastasis; thus, patients were followed according to their center’s standard of care. In the ERSPC, metastasis at diagnosis was defined as M1 disease or PSA >100 ng/mL. For the purpose of this analysis and for greater consistency with the simulation model, metastasis at diagnosis also included N1 and T4 disease.
Metastasis (both at diagnosis and during follow-up) was recorded in the study database source documents from medical records reflecting clinical follow-up for metastasis at the discretion of the patients’ clinicians. 1 Patients with locally advanced disease were typically followed up every 3–6 months with a PSA test and clinical exam. Bone scans were performed in men with bone pain or other clinical suspicion of distant metastasis as well as in men with PSA ≥20 ng/mL and high Gleason score (GS) ≥7. 1 Progressive metastasis was defined as M1 or PSA > 100 ng/mL—a strong indicator of underlying metastatic disease even if not confirmed by pathology or imaging.
The SPCG-4 trial, initiated 19898 is a large-scale randomized controlled treatment trial comparing radical prostatectomy to watchful waiting for localized prostate cancer cases (<75 years, life expectancy >10 years, moderately high differentiation according to the World Health Organization definition, clinical stage ≤T2, PSA <50 ng/mL and negative bone scan). The average age of the participants was 65 years. We used data from N = 695 men (N = 347 intervention; N = 348 control) in the SPCG-4 trial with 29 years’ complete follow-up for metastasis after date of diagnosis. Metastasis was defined as a positive bone scan. As part of the study protocol, all men in both trial arms were actively followed every six months for the first two years, and annually thereafter, with PSA tests and clinical exams. Bone scans were obtained annually until 2003; thereafter, they were performed biennially unless the patient had a PSA increase or showed other clinical signs of progression. Men in both groups received hormonal therapy if metastases were confirmed, mainly orchiectomy or gonadotropin-releasing hormone analogs as lifelong therapy. 9 The SPCG-4 trial took place at a time when most men in Scandinavia were not PSA-screened (the proportion of PSA-detected T1c tumors was 36/695 = 5.2%), 9 and collected metastasis and death as outcomes. As a result, it provides high-quality information about the timing of progressive metastasis in men who were clinically diagnosed.
Model of screening and stage-specific incidence
To project the incidence of de novo metastatic disease, we used the Fred Hutchinson Cancer Research Center (FHCRC) microsimulation model of prostate cancer natural history, as described in detail previously.6,7 The model links an individual man’s PSA growth to the onset and progression of a preclinical prostate tumor so that, on average, an individual with a faster PSA growth rate has a shorter time to clinical diagnosis and a higher probability of metastatic stage at diagnosis. Tumor grade is assigned at disease onset and does not change over time. Under screening, a fraction of preclinical prostate tumors that would have been diagnosed at a metastatic stage in the absence of screening are detected at a localized stage. For each individual, the model generates a date and stage of clinical diagnosis and similarly a date and stage of screen-diagnosis given the screening protocol. This allows for the identification of cases shifted earlier by screening and generates a lead time for each screen-detected case. For non-overdiagnosed cases, post-screen-diagnosis events (e.g., disease progression and survival) are generated beginning at the end of the lead time in accordance with the SPCG-4 trial, our source for progression events, which constituted a mostly clinically diagnosed population.
The FHCRC model was originally calibrated to U.S. population trends in disease incidence by age and stage. 6 For the ERSPC simulation, select natural history parameters of the FHCRC model were adjusted to approximate observed incidence in the trial by age, stage, grade, and study year, while accounting for age distributions at randomization, attendance rates, and biopsy patterns in each center. 5 We used the model to simulate outcomes for individuals in the participating Netherlands, Sweden, Finland, and Switzerland centers, all of which recorded metastasis after diagnosis and were represented in the published analysis of the trial. 1 The model replicated biopsy compliance in the trial that depended on age and PSA in each center. Biopsy sensitivity was assumed to be 80%. 10 All analyses were conducted on data collected up to 12 years following randomization. Metastasis at diagnosis was defined in the model as M1, N1, or T4 disease and progressive metastasis as M1, reflecting the different technologies used for identification of metastatic disease at diagnosis (rectal exam, surgery) and post-diagnosis (PSA tests, imaging).
Mechanism of screening benefit
When patients with prostate tumors that would have been diagnosed at a metastatic stage in the absence of screening are screened when their cancer is still localized, the stage-shift mechanism 11 dictates that their post-diagnosis progression should be based on their localized disease status (Figure 1). The base model assumes no additional benefit of screening beyond the stage shift.
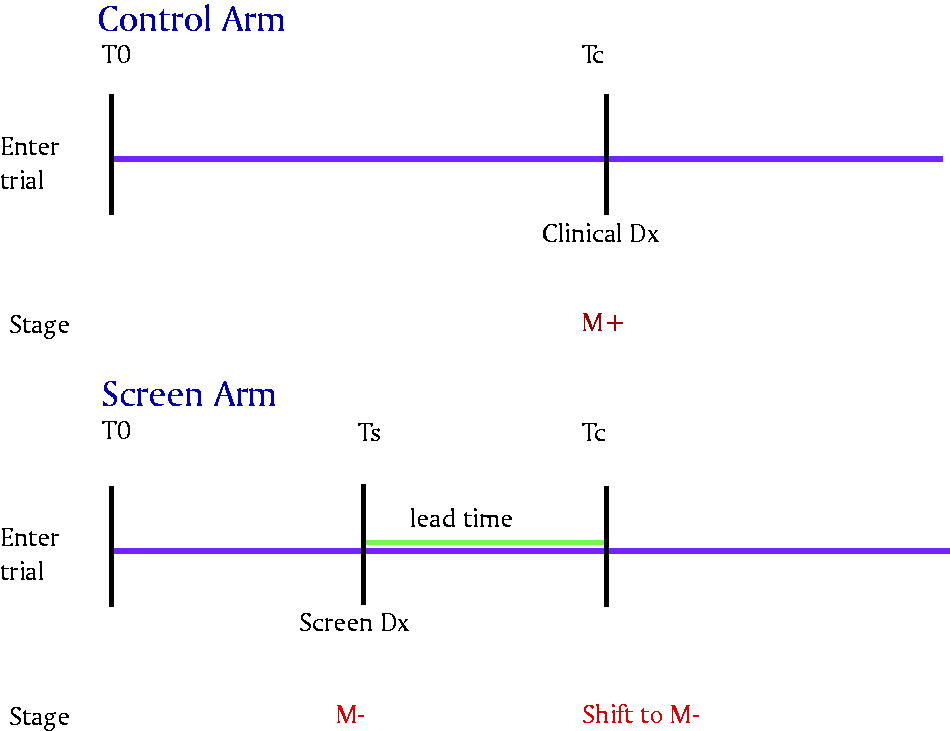
Stage shift mechanism. The same man diagnosed with metastatic (M+) disease in absence of screening at the point of clinical diagnosis (Tc) may be detected with localized disease (M−) disease under screening. Under the stage shift mechanism, he retains M− disease post lead time and at Tc.
Model of metastatic progression
Metastasis after diagnosis was projected from the point of clinical diagnosis (post lead time in the screen arm). The risk of metastasis after diagnosis was based on the SPCG-4 trial that compared radical prostatectomy versus watchful waiting for localized prostate cancer. 8 We fit a Weibull regression model to time to metastasis as a function of age, grade (GS <7 versus ≥7), and treatment arm and used this model to simulate times of metastatic progression after diagnosis (Supplementary Appendix, Methods A). The simulation assumes that the fraction in each arm receiving curative treatment was the same as in the ERSPC (72.5% of men in the control arm received either radical prostatectomy or radiation, as did 70.5% of men in the control arm). 12 We model the risk of metastasis in the absence of curative treatment using the SPCG-4 control arm and the benefit of radiation on metastatic progression as equivalent to the benefit of surgery.
Analysis of outcome measures
The goal of our analyses was to compare projected screening benefit in terms of metastasis at diagnosis and overall metastasis. We quantified screening benefit via three different measures: (1) hazard ratio (HR) comparing the time from entry to the trial until metastasis in the screen versus the control group, (2) relative risk (RR) or metastasis rate ratio measured at 12 years in the screen versus the control group, and (3) absolute risk (AR) difference at 12 years (control group minus screen group). All metrics considered other-cause death and end of study as censoring events (Supplementary Appendix, Methods B).
Sensitivity analyses
We considered three types of sensitivity analyses pertaining to the benefit of screening, the benefit of treatment, and the risk of metastasis, as described in Supplementary Appendix, Sensitivity Analyses. In brief, the sensitivity analyses allowed us to consider (1) an additional benefit of screening on progressive metastasis beyond the stage shift (applying only to screen arm), (2) an improved benefit of treatment for localized disease (e.g., corresponding to adjuvant or early salvage treatment) applying to both arms, and (3) a different risk of metastasis than was observed in the SPCG-4 population (applying to both arms).
Results
Metastasis at diagnosis in the simulation
By the end of the 12-year study, the screen arm accumulated fewer de novo metastatic cases at diagnosis than the control arm, reflecting the stage shift effect of screening (Figure 2). Notably, the cumulative incidence of de novo metastasis in the screen and control arms in the simulated trial replicated the ERSPC trial fairly closely (Figure 3).
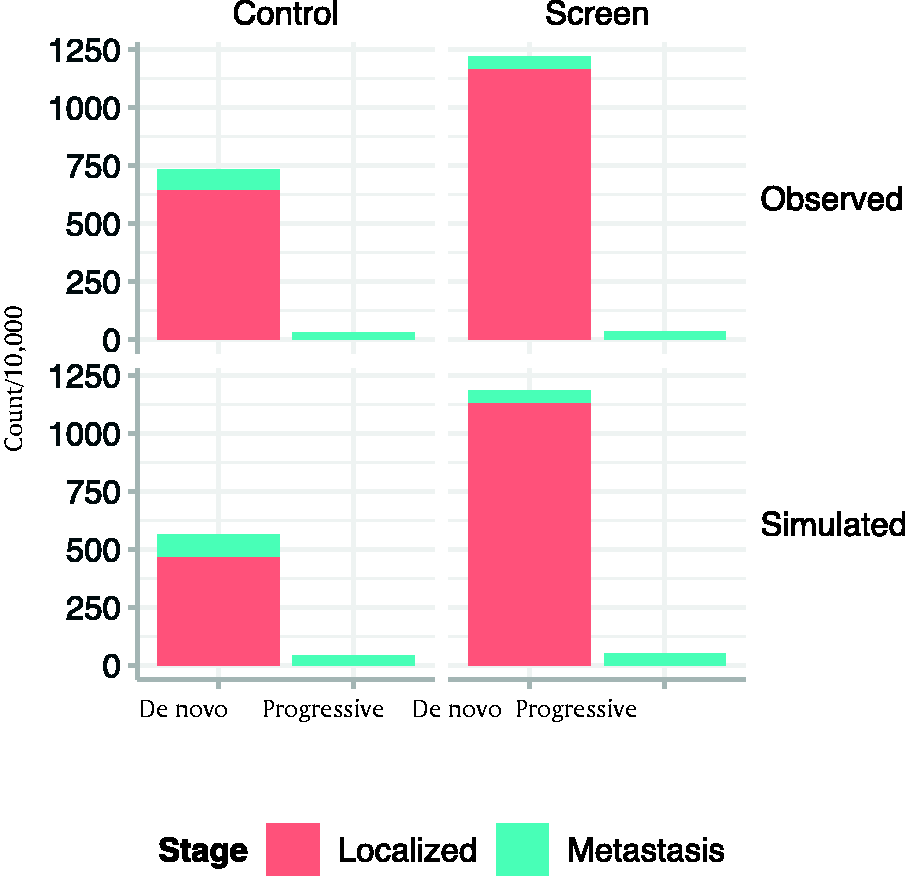
Model-projected and observed cases of localized disease, de novo metastasis, and progressive metastasis at 12 years follow-up in screen and control arms.
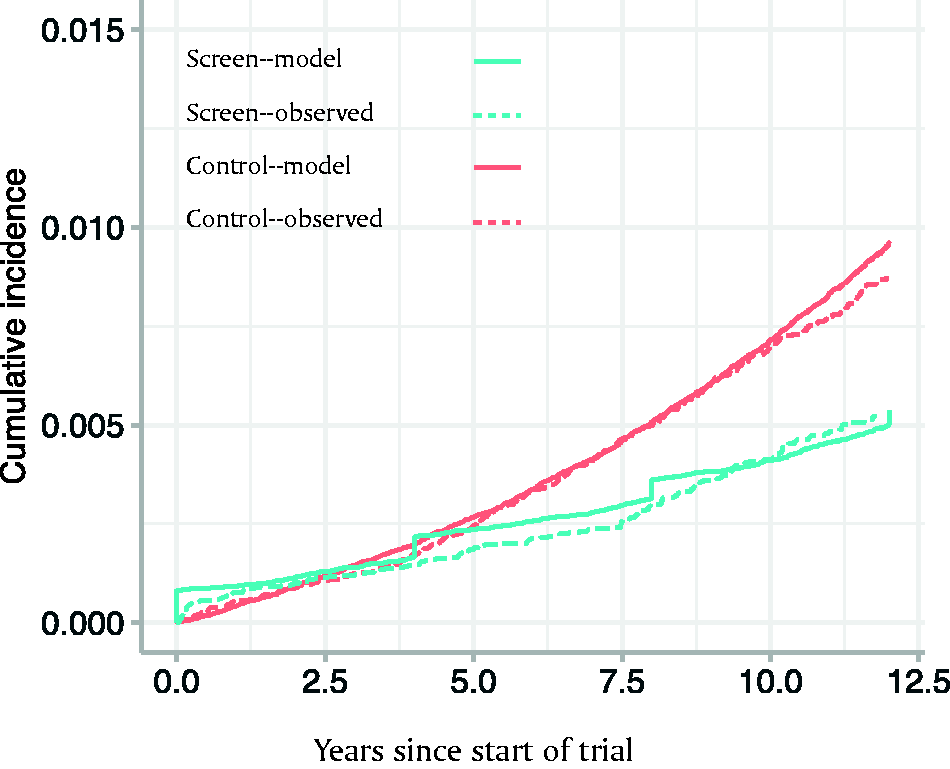
Comparison of cumulative incidence of de novo metastasis in observed ERSPC and simulated trial. Jumps in the simulated screen arm are due to simplified scheduling of the predominantly quadrennial PSA screening in the model.
Modeled effect of screening on metastasis at diagnosis
Screening had a substantial relative and absolute benefit for reducing de novo metastasis in the simulated trial. By the end of the simulated trial, the RR of de novo metastasis in the screen versus control arm was 0.57, the HR was 0.60, and the AR reduction (ARR) was 39.6/10,000 men (Table 2).
Simulated and observed trial events after 12-year trial.
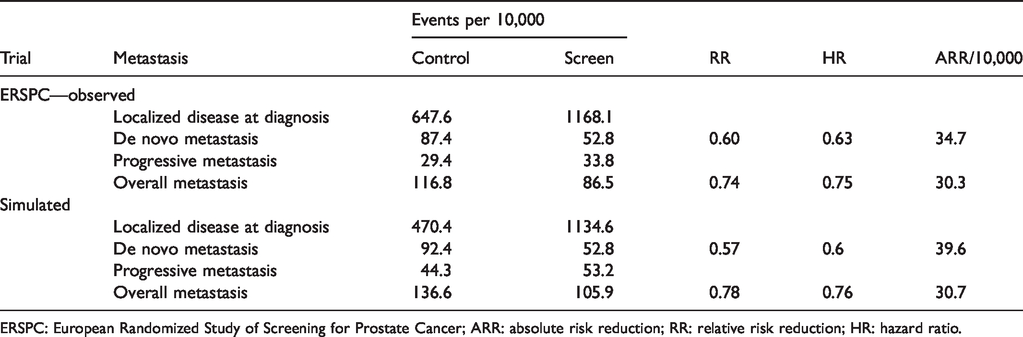
ERSPC: European Randomized Study of Screening for Prostate Cancer; ARR: absolute risk reduction; RR: relative risk reduction; HR: hazard ratio.
Progression to metastasis in the SPCG-4 trial
Figure 4 shows the results of the Weibull regression model fit to the SPCG-4 trial and used to project progressive metastasis in the simulated trial. The SPCG-4 trial found radical prostatectomy to have a HR of 0.53 on risk of metastasis after diagnosis, in favor of surgery (Supplementary Appendix, Methods A). 8 Across the age ranges included, the Weibull regression estimates an approximately 9%–10% and 16%–19% cumulative probability of metastasis 10 years after clinical diagnosis in men with GS < 7 in the treated arm and untreated arms, respectively. GS ≥7 was associated with a 2.8 (95% confidence interval [2.1, 3.7]) times higher hazard and a correspondingly higher cumulative probability of metastasis (Figure 4).
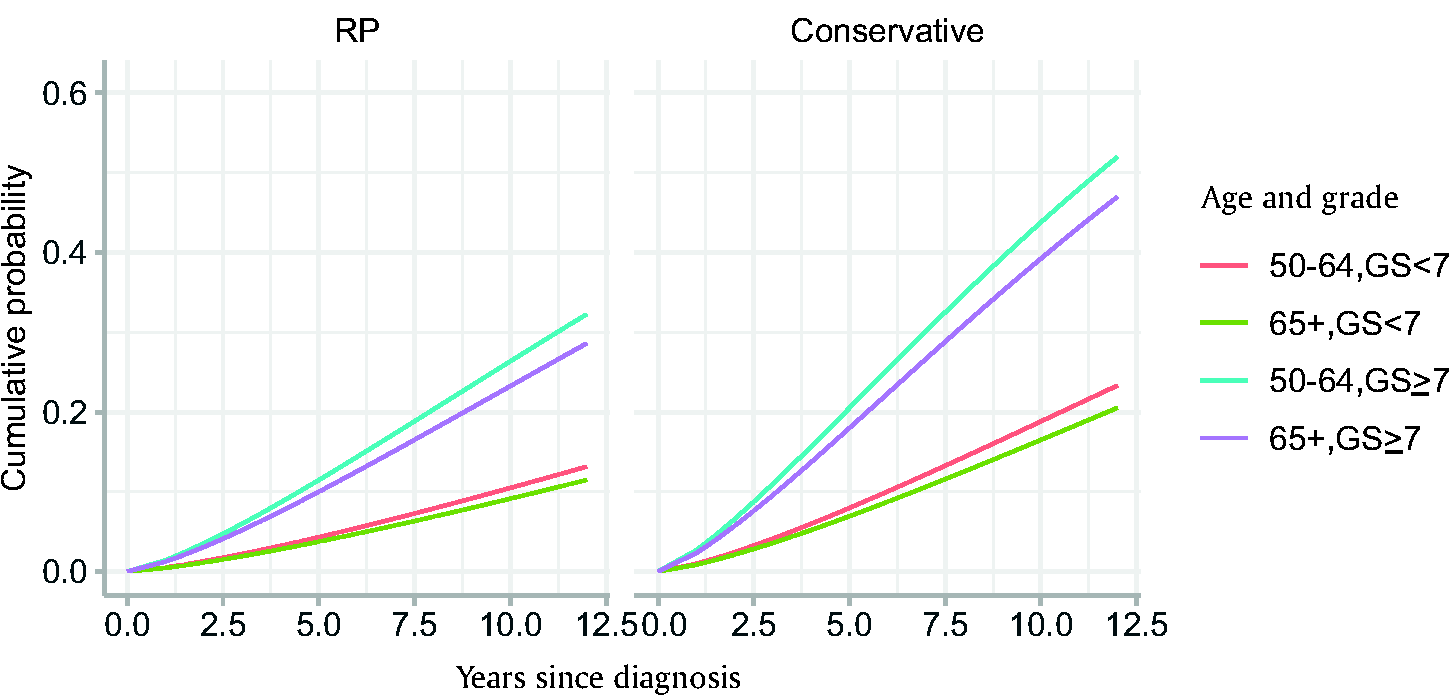
Cumulative distribution functions used to project metastasis after diagnosis based on Weibull models fit to the Scandinavian Prostate Cancer Group-4 trial.
Modeled effect of screening on progressive and overall metastasis
Figure 5 shows stacked plots of the cumulative incidence of developing progressive and de novo metastasis in the simulated trial; the total curve represents the cumulative probability of developing either type of metastasis. Compared with the effect of screening on de novo metastasis, the accumulation of progressive metastases in the screen and control arms leads to an attenuation in absolute and relative benefit for metastatic disease overall. The RR at 12 years for metastasis overall in screen versus control arms is 0.78 in the simulated trial, and the HR is 0.76. The ARR at 12 years is 30.7/10,000 men (Table 2).
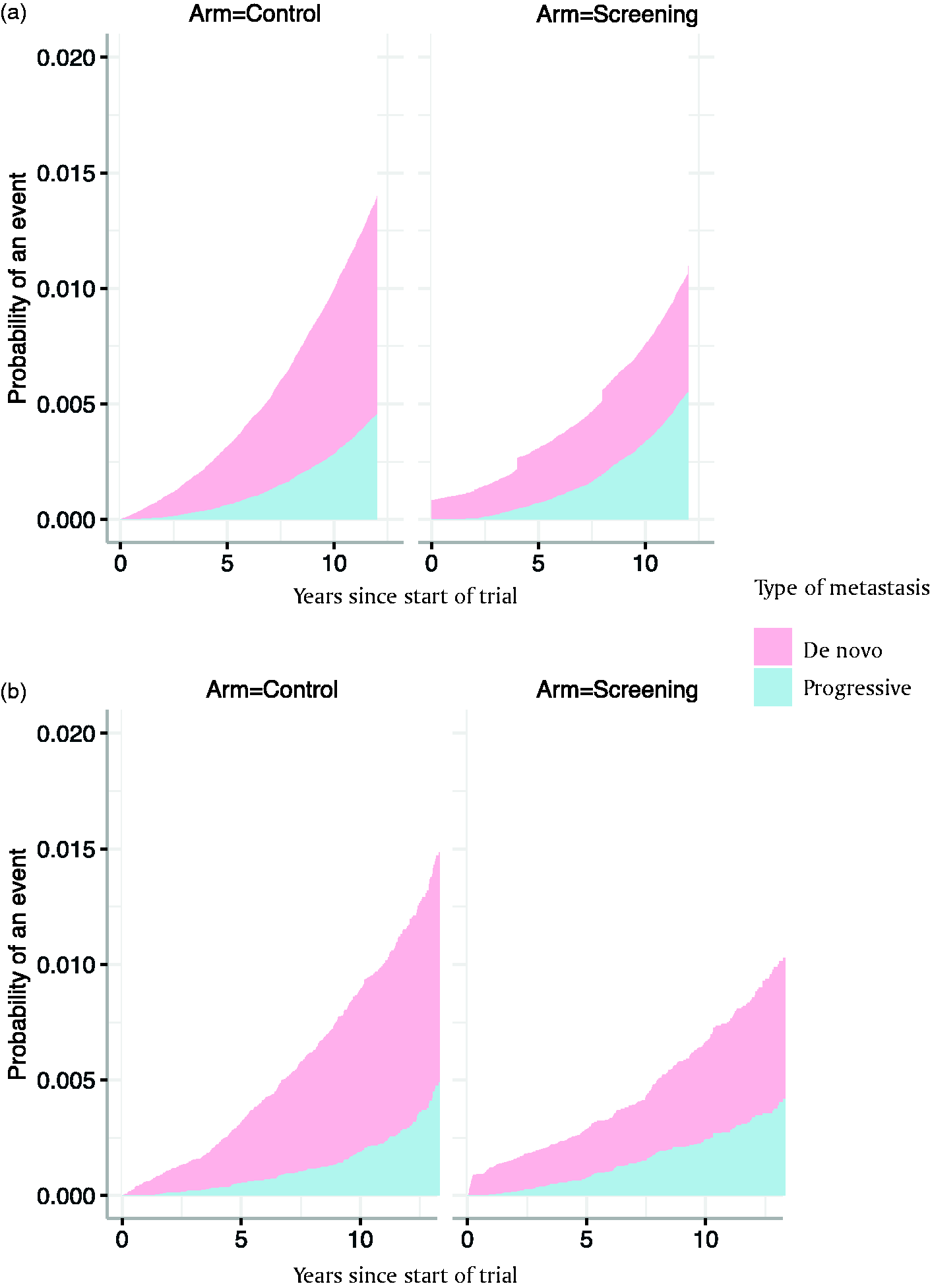
Cumulative incidence of de novo and progressive metastasis in (a). The simulated trial and (b). Observed ERSPC trial.
Comparison of simulated trial with ERSPC metastasis at diagnosis and overall metastasis
As in the simulated trial, the empirical ERSPC demonstrated the stage-shift benefit of screening on de novo metastasis (Figure 2). The simulated trial was broadly in agreement with the empirical ERSPC as far as an attenuation of the absolute and relative benefit of screening on metastasis overall relative to de novo metastasis. At diagnosis, the RR of de novo metastasis in screen versus control arms was 0.60; for overall metastasis, it was 0.74. The ARR was 34.7/10,000 for de novo metastasis and 30.3/10,000 for metastasis overall (Table 2).
There were also some differences between the simulated trial and what was observed in the ERSPC. In particular, the simulated trial projected fewer localized cases at diagnosis and more progressive metastasis cases than the empirical ERSPC (Table 2).
Sensitivity analyses
Under the assumption of additional benefit of screening (enhanced reduction of progression to metastasis in the screening arm only), the RR for metastasis overall associated with screening became more favorable. However, treatment for screened men with localized disease would need to reduce the risk of metastasis dramatically—by an additional 80%—in order for the RR for overall metastasis RR to match the RR for metastasis at diagnosis
Discussion
In this study, we present a novel modeling framework to project the expected benefit of cancer screening on the cumulative incidence of metastasis. Rather than focusing only on the reduction in distant stage disease at diagnosis (“stage shift”), this measure combines de novo and progressive cases and is likely to be a more accurate proxy for mortality reduction than the commonly used stage shift at diagnosis.
Our framework provides a mechanistic understanding of how screening might impact disease stage at and after diagnosis. Screening reduces the number of cases with advanced stage disease at diagnosis in the screening group relative to the control group, but this leads to a larger pool of non-overdiagnosed localized cases eligible to progress. The model reproduces the ERSPC result of a greater accumulation of progressive metastases in the screen arm relative to the control arm. This accumulation translates into an attenuation of both the relative and absolute benefit of screening for overall metastasis compared to metastasis at diagnosis.
While we use the ERSPC as an exemplar trial for our case study, we acknowledge that the existing simulation model is not a perfect replication of reality. Our definition of metastasis at diagnosis was chosen to enhance compatibility between the model-projected incidence of metastasis at diagnosis and that observed in the ERSPC. After diagnosis, we counted ERSPC cases with PSA > 100 ng/mL as metastatic since they were likely clinically metastatic even if they lacked imaging or other clinical records. Still, the model-projected risk of progressive metastasis was higher than was observed in the ERSPC. This could potentially be attributable to intensive surveillance for metastasis in the SPCG-4 trial, which was our source for post-diagnosis progression, but it could also simply be due to the fact that the ERSPC and SPCG-4 populations were not contemporaneous and could have had other differences.
There are several potential current and future applications of our framework. Distant metastasis is an important endpoint of screening trials alongside cancer mortality because it has a tremendous impact on individuals’ quality of life. 13 Similarly, metastasis is a strong surrogate outcome for survival in clinical trials of new therapeutics, due to the long natural history of prostate cancer from diagnosis to progression and death. 14 Our framework offers a short cut to project the effectiveness of novel tools for cancer screening in terms of distant metastasis.
A limitation of our modeling framework is that it is based on assumptions that may not fully reflect the benefit of screening or the risk of metastasis after localized diagnosis. We addressed several of these assumptions in our sensitivity analyses. In the first sensitivity analysis, we examined the possibility that screening may confer an additional benefit beyond just the stage shift on the risk of progressive metastasis. 15 In the second sensitivity analysis, we reduced the risk of progressive metastasis in treated men on both arms to accommodate adjuvant or early salvage treatments for PSA recurrence that might not have been present in the SPCG-4 cohort. In the third sensitivity analysis, we addressed the possibility that risk of metastasis after diagnosis may be very different for today’s cancer patients as compared to the SPCG-4 trial. Our conclusions were generally robust across all the sensitivity analyses.
In conclusion, metastasis is a key endpoint that impacts both quality of life and cancer mortality. We developed a novel framework that mechanistically explains how screening might impact the cumulative risk of cancer metastasis. Using this framework, we show that, in general, expectations of screening benefit that are based on stage shift at diagnosis should be tempered when considering the endpoint of disease mortality.
Supplemental Material
sj-pdf-1-msc-10.1177_0969141321989738 - Supplemental material for Impact of cancer screening on metastasis: A prostate cancer case study
Supplemental material, sj-pdf-1-msc-10.1177_0969141321989738 for Impact of cancer screening on metastasis: A prostate cancer case study by Jane Lange, Sebastiaan Remmers, Roman Gulati, Anna Bill-Axelson, Jan-Erik Johansson, Maciej Kwiatkowski, Anssi Auvinen, Jonas Hugosson, Jim C Hu, Monique J Roobol, Sigrid V Carlsson and Ruth Etzioni in Journal of Medical Screening
Footnotes
Authors contributions
Study concept and design: RE; acquisition of data: AB-A., J-EJ, SR, MR, MK, AA, JH, and SVC; analysis and interpretation of data: all authors; drafting of the manuscript: JL and SVC; critical revision of the manuscript for important intellectual content and final approval of the submitted version: all authors; statistical analysis: JL, RG, and SR; and obtaining funding: all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JL, SVC, RG, and RE were supported by a grant from the National Cancer Institute as part of the Cancer Intervention and Surveillance Modelling Network (U01-CA199338-02). RG was also supported by the National Cancer Institute (R50-CA221836).SVC’s work on this paper was supported in part by a Cancer Center Support Grant from the National Cancer Institute made to Memorial Sloan Kettering Cancer Center (P30-CA008748), a Specialized Program Of Research Excellence (SPORE) grant from the National Cancer Institute to Dr. H. Scher (P50-CA092629), and a career development award from the National Cancer Institute (K22-CA234400). JCH’s work was supported by The Frederick J. and Theresa Dow Wallace Fund of the New York Community Trust. RE’s work is partially supported by the Rosalie and Harold Rea Brown Endowment. The funding bodies had no role in the preparation, review, or approval of the manuscript or the decision to submit the manuscript for publication. The funding of the ERSPC trial was obtained from national cancer research funding agencies, European funding in the form of Framework programs, private sponsors, and a grant from the former Beckman/Hybritech company. The maintenance of the ERSPC central database is sponsored by the European Association of Urology.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
