Abstract
Objective
To compile current usage of serum-based prenatal screening for Down syndrome in the United States and compare it with results from a similar 2011/2012 survey.
Setting
The College of American Pathologists maternal screening proficiency testing survey includes a supplemental question on the first of three yearly distributions.
Methods
Information regarding tests offered and the monthly number of pregnancies tested for US-based laboratories were reviewed. Results were stratified by size of laboratory, tests offered, and pregnancies tested. Findings were compared to an earlier survey.
Results
Fifty-six laboratories reported they will have screened 1,131,336 pregnancies in 2020. Of these, 36% are screened by stand-alone first trimester testing, 48% by stand-alone second trimester testing, and 16% using tests that integrate results from both trimesters. Eighty percent of all serum screens were provided by the five laboratories that performed the most screens (at least 50,000). These five performed similar proportions of first or second trimester screens (42.2% and 41.8%, respectively). Compared to eight years earlier, there are now 54% fewer laboratories. Pregnancies screened using the first trimester, second trimester, and integrated protocols were lower by 27%, 69%, and 72%, respectively. The serum screening activity in the US showed a 62% decrease from 2012 levels. During 2012–2020, the number of cell-free DNA tests increased from negligible to 1,492,332.
Conclusions
Maternal serum screening for common aneuploidies has changed significantly in eight years with fewer laboratories, a shift toward larger laboratories and a 2.5-fold reduction in pregnancies tested, likely due to the introduction of cell-free DNA screening.
Keywords
Introduction
Advances in knowledge of biochemical and physical markers associated with common autosomal aneuploidies have greatly changed the landscape of screening tests available to pregnant women. Prior to the mid-1980s, the only available prenatal screening test for Down syndrome was the maternal age at delivery. Beginning in the late 1980s, the well-described association with maternal age was combined with maternal serum alpha-fetoprotein (AFP) measurements that were already being performed for open neural tube defect (NTD) screening.1,2 In the early 1990s, additional second trimester markers were added. 3 First trimester serum markers were identified and used4,5 and an ultrasound marker was also included.5–7 These combinations of biochemical markers in both first and second trimesters as well as ultrasound-based physical measurements such as nuchal translucency (NT) have improved screening, opened options earlier in pregnancy for low risk populations 8 and have been well accepted. Maternal plasma testing using next-generation sequencing of cell-free DNA (cfDNA) from the feto-placental unit present in maternal circulation has entered the clinical screening arena; initially only available to high risk pregnancies.9–11 As experience with cfDNA has increased, this option is now being offered to a wider number of pregnant women, including pregnancies at lower risk for aneuploidy.12,13 For open NTD, the approach has slowly drifted from using maternal serum AFP measurements in second trimester to the wider use of ultrasound for screening anatomical abnormalities.14,15 Maternal serum AFP screening for open NTD should, however, continue to be offered to all pregnancies when an earlier diagnosis is desired or when ultrasound is not readily available.
The College of American Pathologists (CAP) Maternal Screening subgroup of the Biochemical and Molecular Genetics Committee reviewed external proficiency testing surveys and exercises, compiled the utilization data from the first survey of 2020, and compared those results to utilization data from the 2012 survey. 16 The aim is to document potential changes in the numbers of active laboratories, their testing volumes, as well as the types of testing offered (e.g., first trimester, second trimester).
Materials and methods
Data were derived from the yearly CAP maternal screening proficiency testing programs (the Fetal Protein or FP surveys) for laboratories offering serum-based screening for Down syndrome (https://documents.cap.org/documents/2020-survey-catalog.pdf) in the first and second trimesters of pregnancy. The FP series of surveys provide five simulated maternal serum challenges in three mailings per year and includes challenges for open NTD in the second trimester. Participants reported the specific screening test as well as the average monthly volume of each test performed as part of the first (A) mailing each year. These data were collected from all participating laboratories, but analysis was restricted to those located in the US for the purposes of the present study. These raw data were extracted several months after the deadline for submission to ensure late results were included. Data were reviewed for consistency and those laboratories with a potential discrepancy were contacted by CAP staff and issues clarified. Monthly volumes in April 2020 were converted to a yearly rate for 2020. A small percentage of samples analyzed in laboratories located in the US were collected from women living outside of the country, but it was not possible to account for these. A similar non-US workload was also likely present in the 2012 report. 17 Workload was also collected from US laboratories from the CAP Non Invasive Prenatal Testing (NIPT) Exercise for cfDNA screening tests during its first (A) distributions, both in 2019 and 2020. Average monthly volumes were converted to a yearly rate for 2019 and 2020. At its inception in 2018, the NIPT program was designated an exercise, in order to provide options for laboratories as alternate proficiency assessments. Responses from these surveys and exercises form the basis of the current report on the proportion of women in the United States receiving serum screening for Down syndrome. These data were then compared to similar analyses of US laboratories from 2012. 17 The changes in each type of screening test and numbers of pregnancies screened were computed.
Results
A total of 56 laboratories located in the US reported the average monthly types of Down syndrome testing performed and numbers of screens performed in 2020. Table 1 summarizes the types of tests and the annualized workload. Second trimester testing was the most commonly offered, with 55 of the 56 laboratories screening 543,648 women. The most common second trimester test, accounting for more than 99% of all second trimester testing, was the Quadruple test (usually maternal age in combination with AFP; uE3: unconjugated estriol; hCG: human chorionic gonadotropin; and DIA: dimeric inhibin-A measurements). The first trimester test (usually maternal age in combination with NT, pregnancy-associated plasma protein A (PAPP-A), and hCG or the free beta subunit of hCG), was used to screen 411,948 women. Various forms of the integrated test (combining results from both the first and second trimester tests) were used to screen another 175,740 women. Overall, this documents that at least 1.13 million women in the US received some form of serum screening for Down syndrome in 2020.
Types and annual numbers of Down syndrome serum-based screening tests performed in US laboratories in 2020.
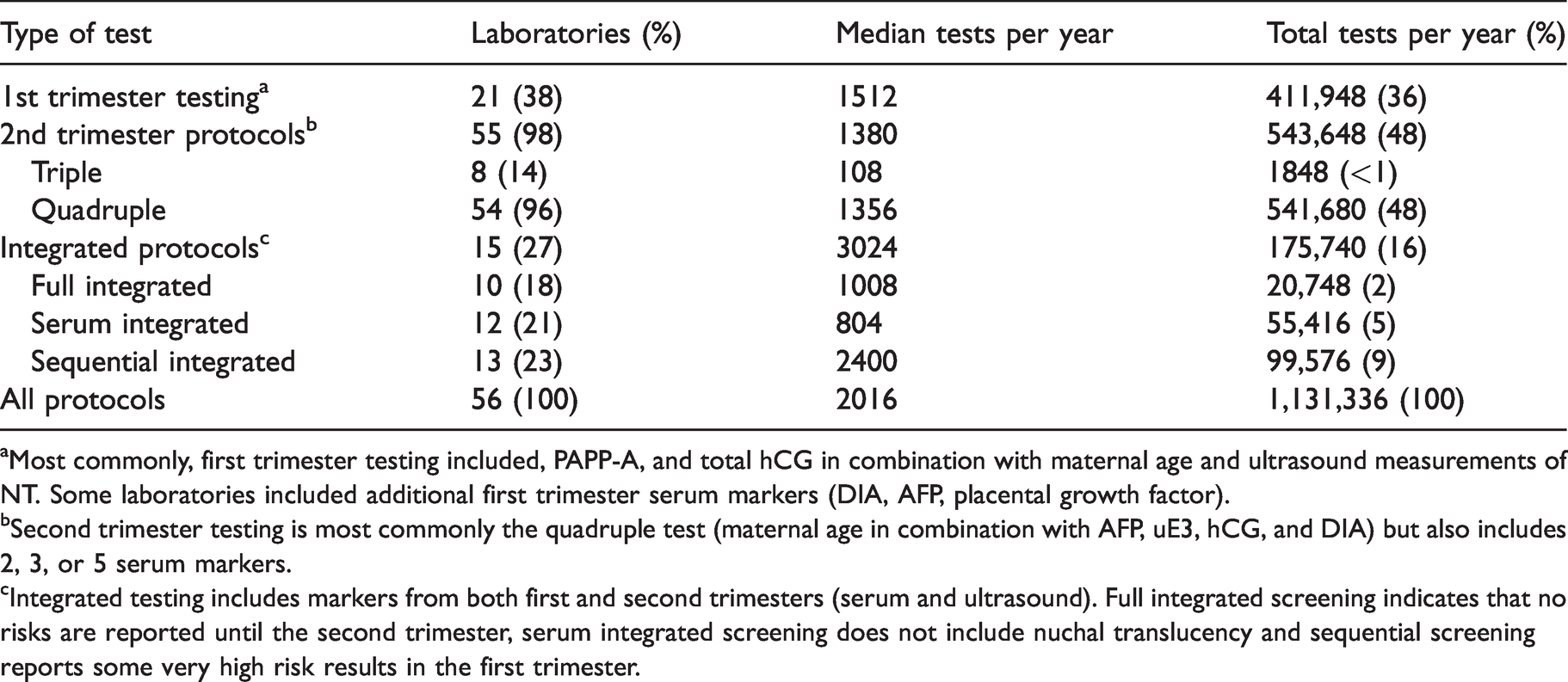
aMost commonly, first trimester testing included, PAPP-A, and total hCG in combination with maternal age and ultrasound measurements of NT. Some laboratories included additional first trimester serum markers (DIA, AFP, placental growth factor).
bSecond trimester testing is most commonly the quadruple test (maternal age in combination with AFP, uE3, hCG, and DIA) but also includes 2, 3, or 5 serum markers.
cIntegrated testing includes markers from both first and second trimesters (serum and ultrasound). Full integrated screening indicates that no risks are reported until the second trimester, serum integrated screening does not include nuchal translucency and sequential screening reports some very high risk results in the first trimester.
Table 2 provides an illustration of the distribution of sample testing in US laboratories stratified by their yearly screening workload. Low-volume laboratories (those screening fewer than 1000 samples per year) represented 30% of all laboratories, but screened only 0.9% of the pregnancies reported. Most of the testing performed by these laboratories was in the 2nd trimester. In the US, there is a positive association between the yearly number of tests performed and the number of screening protocols offered. For any laboratory reporting less than 49,999 pregnancies per year, the most common protocol selected was still the 2nd trimester. This was not the situation with the five laboratories reporting at least 50,000 pregnancies per year. In these laboratories, testing was most commonly performed in the first trimester versus second trimester (383,592 vs. 379,032 respectively). Although they represented only 9% of all screening laboratories, they reported 80.2% of all samples tested.
Numbers and types of Down syndrome serum-based screening tests, stratified by the numbers of pregnancies screened in 2020.
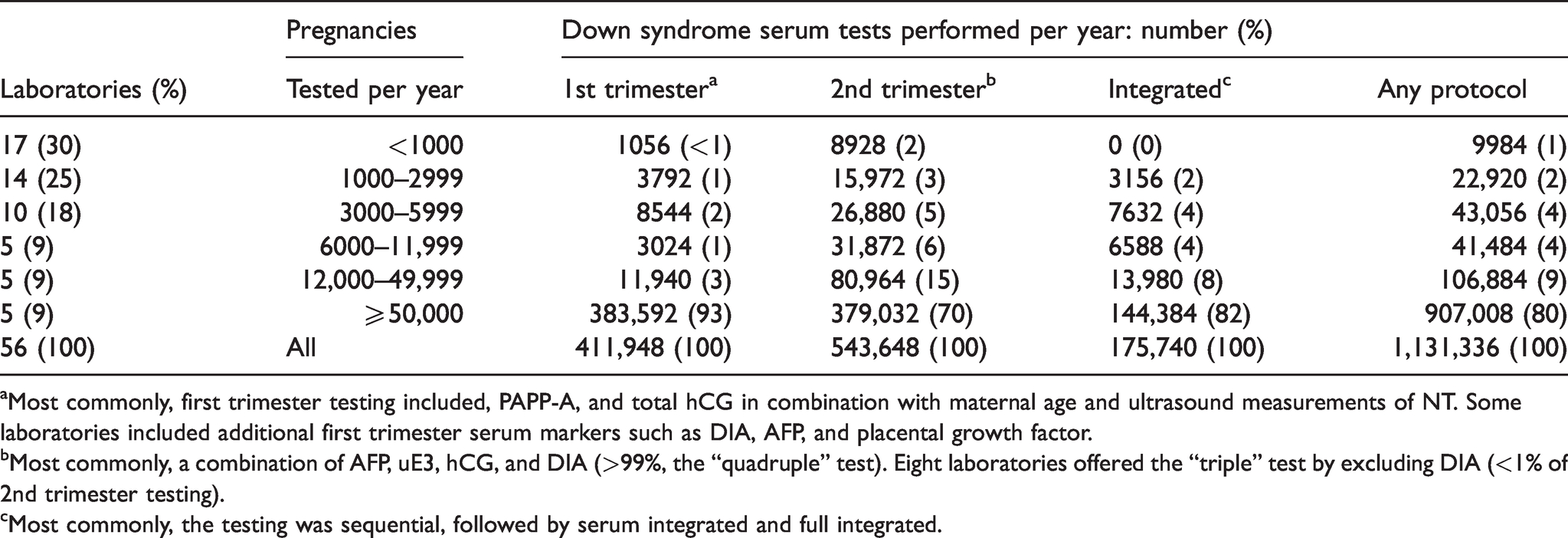
aMost commonly, first trimester testing included, PAPP-A, and total hCG in combination with maternal age and ultrasound measurements of NT. Some laboratories included additional first trimester serum markers such as DIA, AFP, and placental growth factor.
bMost commonly, a combination of AFP, uE3, hCG, and DIA (>99%, the “quadruple” test). Eight laboratories offered the “triple” test by excluding DIA (<1% of 2nd trimester testing).
cMost commonly, the testing was sequential, followed by serum integrated and full integrated.
Table 3 provides data on the types of screening protocols stratified by the yearly workload. Among the 46 laboratories in the two lowest stratification categories (up to 11,999 pregnancies tested per year), 30 (65%) offered only 2nd trimester protocols. For the 10 laboratories in the two highest stratification categories (over 12,000 pregnancies tested per year), only one (10%) offered only 2nd trimester protocols. Six of these 10 offered all of the screening protocols.
Types of Down syndrome serum-based screening tests offered by each laboratory, stratified by their total annual number of pregnancies screened in 2020.
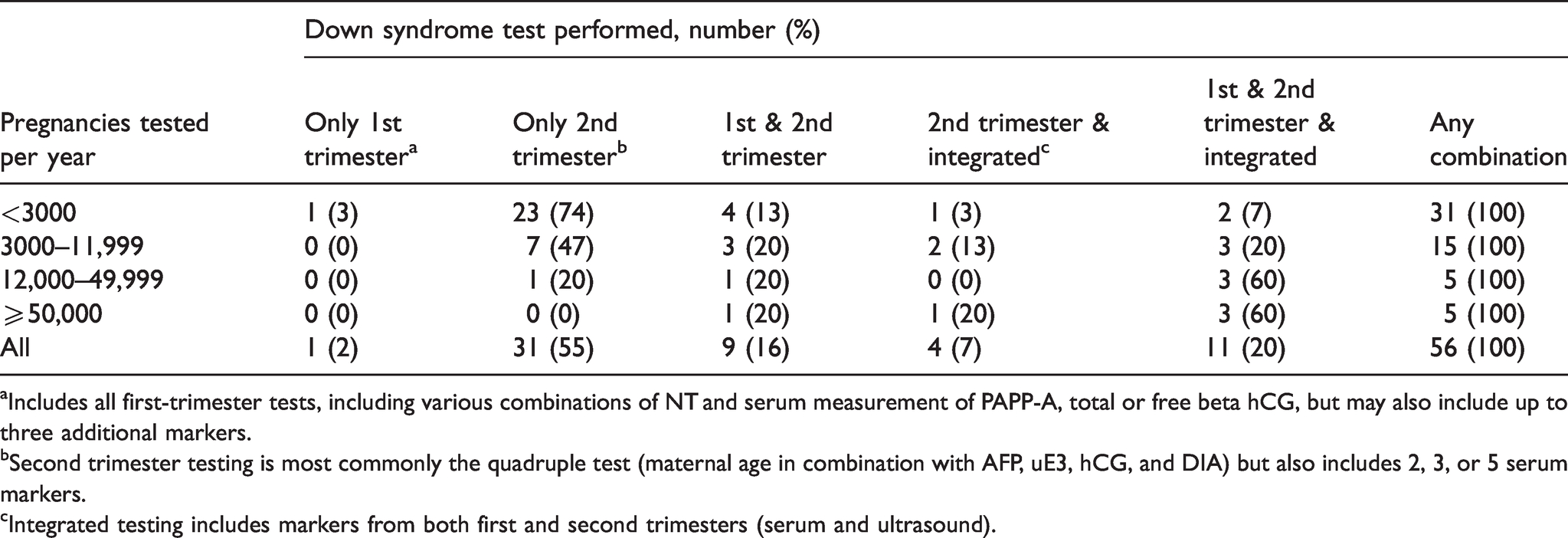
aIncludes all first-trimester tests, including various combinations of NT and serum measurement of PAPP-A, total or free beta hCG, but may also include up to three additional markers.
bSecond trimester testing is most commonly the quadruple test (maternal age in combination with AFP, uE3, hCG, and DIA) but also includes 2, 3, or 5 serum markers.
cIntegrated testing includes markers from both first and second trimesters (serum and ultrasound).
A comparison of workload between 2012 and 2020 showed clear differences in ordering trends (Table 4). Overall, second trimester tests decreased by 69%, from 1.77 million to 0.54 million. First trimester screening decreased by 27%, from 0.57 to 0.41 million. Integrated testing decreased by 72%, from 0.63 to 0.18 million. The total number of screening tests decreased by 62% over the past eight years, from 2.96 to 1.13 million. Finally, the number of US laboratories providing serum screening decreased from 123 to 56; a diminution of 54%.
Comparison of Down syndrome screening testing using serum markers in 2020 and in 2012.a
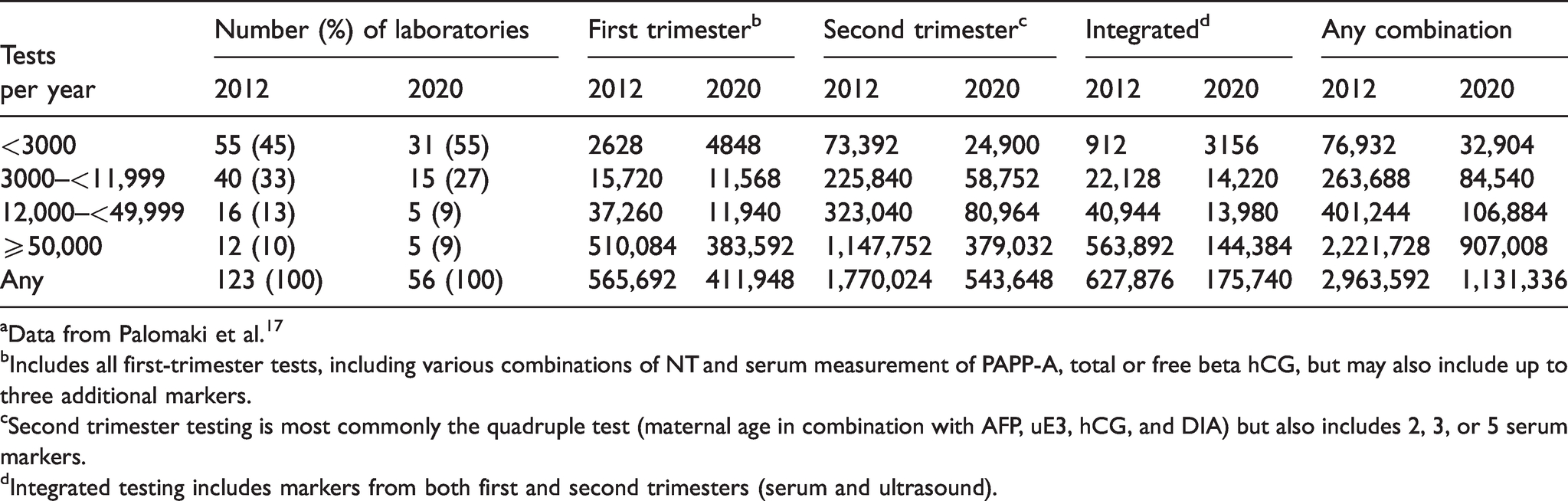
aData from Palomaki et al. 17
bIncludes all first-trimester tests, including various combinations of NT and serum measurement of PAPP-A, total or free beta hCG, but may also include up to three additional markers.
cSecond trimester testing is most commonly the quadruple test (maternal age in combination with AFP, uE3, hCG, and DIA) but also includes 2, 3, or 5 serum markers.
dIntegrated testing includes markers from both first and second trimesters (serum and ultrasound).
Information was also available for the use of the second trimester AFP measurements alone to screen for open NTD. In the 2020 survey, 45 of the 56 laboratories (80%) provided open NTD screening as a stand-alone test to 241,680 pregnancies; a median of 1380 pregnancies tested per laboratory. By comparison, in 2012,85 laboratories were providing measurement of AFP for open NTD screening, to 235,992 pregnancies, with a median of 720 pregnancies tested per laboratory.
Table 5 provides limited information on cfDNA testing in the US. The methodologies are divided into two main approaches. Targeted sequencing is utilized by 57% of the laboratories testing less than 50,000 pregnancies per year, whereas the whole genome approach is utilized by 67% of the laboratories performing at least 50,000 pregnancies per year. Overall, 1,492,332 cfDNA tests were expected to be performed in 2020 by the 13 participating US laboratories. Based on information collected in the 2019 NIPT exercise, an estimated 9% of all samples received were likely collected from outside of the US (134,000). Furthermore, 44% of tests were likely performed in pregnancies considered to be at high risk (e.g., serum screen positive, advanced maternal age, abnormal ultrasound). When applying the 2019 rates for out-of-country testing to the 2020 volumes with tests sent from outside of the US removed, the total would be reduced to 1.36 million. Of these, an estimated 600,000 would have been at high risk while the remaining 760,000 would represent a more general pregnancy population.
Methodologies and annual numbers of cfDNA screening tests performed in US laboratories in 2020.
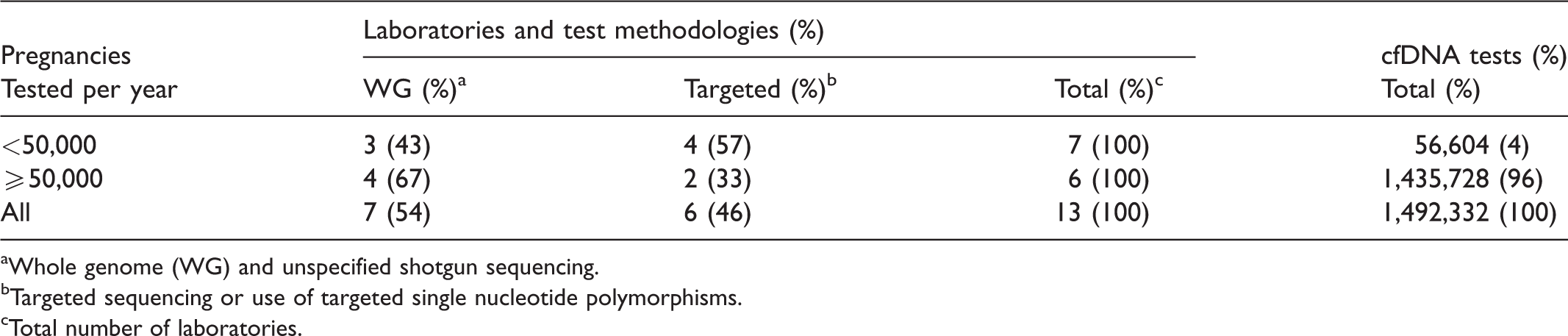
aWhole genome (WG) and unspecified shotgun sequencing.
bTargeted sequencing or use of targeted single nucleotide polymorphisms.
cTotal number of laboratories.
Discussion and conclusion
In the US, the frequency at which pregnant women select their screening option has changed over the past eight years based upon the CAP maternal screening surveys. These results documented that the proportion of women choosing any serum screening protocol for Down syndrome has dropped by more than half (62%). This decline can be explained by technology changes and the strengths and weaknesses of the 2020 survey compared to that in 2012.
In 2012, serum screening for Down syndrome was generally associated with about a 5% screen positive rate. 18 Those pregnant women who received a screen positive result were then offered diagnostic testing. The diagnostic procedures that were available in 2012 were amniocentesis (available after 15 weeks gestation) and chorionic villus sampling (available between 9 and 13 weeks gestation). Both procedures are associated with a small rate of fetal loss. 19
Testing using cfDNA as a second tier screening test has been available in the US since late 2011. Initially, cfDNA was often elected by women after they received a screen positive serum test, who were of advanced maternal age (i.e., 35 years or older), had an abnormal ultrasound or had other “high risk” characteristics. Excluding advanced maternal age, up to 15% of the population were likely to be having secondary screening via cfDNA testing. In the 2020 NIPT exercise, it was not possible to distinguish whether indication for testing was as a primary or secondary screen. In the 2019 CAP NIPT exercise, laboratories reported that approximately 44% of all US samples tested were in the “high risk” categories. In 2020, the American College of Obstetricians and Gynecologists recommended that cfDNA be offered to all pregnant women as a primary test along with other options. 13 However, secondary screening will likely continue to be a major indication. Such a recommendation is warranted as the detection rate for Down syndrome and other common trisomies is high (96 to 99%) so few affected pregnancies will be missed. As important, the false positive rate is low (<0.5%), so that more than 99% of women undergoing cfDNA testing can be provided with reassurance that their pregnancy is unlikely to be affected and avoid invasive testing. The use of cfDNA as a primary test will also have an impact on the number of invasive procedures and on traditional cytogenetic karyotyping. Its use as a primary test also impacts the ability to estimate the number of women having Down syndrome screening, as it would not be appropriate to add the serum and cfDNA screening statistics together.
A limitation to the retrospective use of data provided as part of FP surveys and NIPT exercises may be incomplete reporting by laboratories. Some laboratories enrolled in CAP surveys do not respond to the optional queries associated with each survey. In the 2012 survey, 17 multiple methods were employed to help ensure as complete an ascertainment as possible. Some laboratories elect to not participate in the CAP FP surveys, but rather choose other sources for their maternal serum screening proficiency testing. In the 2012 report, laboratories that enrolled in other external quality assessment schemes were also identified and were offered the opportunity to provide information. Also, two years of responses to the supplemental questions (2011 and 2012) were available and compared responses by individual laboratory. This allowed an additional 11 laboratories to be included that provided responses to the queries in 2011, but did not respond to those queries in 2012, even though they still participated. These methods were not used in the 2020 analysis which may provide some under-reporting. Even with these limitations, the proportion of each screening option provides a reasonable representation of the current serum-based screening for Down syndrome in the US.
A review of the US birth numbers indicates 3,952,841 livebirths in 2012 and 3,745,540 in 2019; a 5% reduction. 20 In 2012, there were 2.9 million pregnancies screened by serum markers (73%) and only a small number of pregnancies were screened by cfDNA as this methodology was offered by one laboratory in late 2011 and was restricted to high risk pregnancies. The estimation from the current data indicates 1.1 million pregnancies screened by serum markers (30%) and up to 1.3 million pregnancies screening by cfDNA (35%). These data most likely are due to a technological replacement rather than a service reduction.
The shift of prenatal care by health care providers to earlier in the pregnancy is observed through the continued use of serum markers in the first trimester (a lesser reduction of 27% between 2012 and 2020) compared to a higher reduction of both second trimester and integrated testing in 2020. Another contributing factor may be testing for open NTD in the second trimester. This would require a second sample where cfDNA screening was chosen as a primary screen. Another reason for the continued ordering of serum screening is the use of other serum markers to identify pregnancy outcomes other than common autosomal trisomies, such as preeclampsia.21,22 Lastly, and perhaps most importantly, insurance coverage is well-established for serum-based screening. This is not yet the case for cfDNA-based screening in the US. A state-wide offering of cfDNA screening at no cost to patients or their insurance company found increases in uptake at the five largest group practices ranging from 8% to 40%. 23 This suggests that if economic barriers were reduced, a higher proportion of the population would likely choose cfDNA as a screening tool for the risk assessment for Down syndrome and other common autosomal trisomies.
These data allow for some speculation and reflection. There are many smaller volume laboratories whose primary workload is linked with only second trimester screening for Down syndrome. Laboratory management for those institutions might consider discussions with their local health care providers and disseminate the benefits of earlier screening to their pregnant population. Requests for second trimester AFP only for risk for open NTD continue to be ordered at a steady state. Some patients may select to decline screening for aneuploidy but consent to screening for open NTD. Others choose first trimester Down syndrome testing using serum and ultrasound markers or cfDNA and opt for second trimester AFP to gain information prior to a targeted ultrasound that is usually performed at around 20 weeks. In some situations, due to a model of decentralized health care systems, second trimester AFP is requested in order to avoid delays in diagnosing pregnancies with open NTD that could happen with delays in booking appointments for ultrasound. Laboratory management should review its requirement for offering second trimester AFP, based on local needs. Although not common in the US, many screening programs in Europe 24 are maintaining first trimester serum based screening with ultrasound markers and using a lower risk cut-off level (e.g. 1 in 500) such that the detection rate and false positive rate are both higher. The cfDNA testing is then offered to women with screen positive results. This can be done using a contingent 25 or reflex model 26 while maintaining the benefits of routine first trimester scanning with performance approaching stand-alone cDNA screening at a lesser cost.
Prenatal screening for Down syndrome and other common autosomal trisomies using serum markers is clearly becoming less common in the US, but was still the primary Down syndrome screen for over one million pregnancies in 2020. Such screening is continuing to move from smaller local programs offering a limited screening menu to larger national screening laboratories providing a wider choice. The introduction of cfDNA screening is becoming an important component in prenatal screening and may become the predominant screening test in the US in the near future.
Footnotes
Acknowledgements
We thank Pamela M Provax, MT(ASCP) the CAP staff person for providing assistance in coordinating the group and their activities.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NL, PW, ERA, RGB, and GEP are/were members of the College of American Pathologists/American College of Medical Genetics and Genomics Biochemical and Molecular Genetics and Genomics Committee. NL, PW, ERA, RG, TL, and GEP have no conflicting stock ownership, patents, or licensing arrangement and have not been paid for publicizing a study. GEP is an Associate Editor for the Journal of Medical Screening but did not participate in any decisions regarding manuscript review or acceptance.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical declaration
Study design is based on voluntary responses by participants in a College of American Pathologists external educational survey to report the specific serum-based screening test as well as the average monthly volume of each test performed as part of the first (A) survey mailing each year. Neither patient consent nor IRB approval was required as this analysis did not include individual patient level data.
