Abstract
Objectives
Unconjugated estriol (uE3) is used as a marker for fetal aneuploidy in maternal serum screening tests. The goal of this study was to examine the validity of a new immunoassay for uE3 that uses a monoclonal antibody (m-uE3) rather than the more commonly used polyclonal antibody (p-uE3).
Setting
Assays were performed in the Special Chemistry laboratory at Women and Infants Hospital of Rhode Island.
Methods
Residual fresh (n = 100) and frozen (n = 533) second trimester serum samples from routine clinical care were tested using p-uE3 and/or m-uE3 assays. Assay results were compared between methods using Bland–Altman plots. A median equation was developed for m-uE3 results. Down syndrome risks were compared between the two assays in a case–control sample set (21 cases each matched with five controls for the completed week of gestation, duration of freezer storage and race).
Results
Log-transformed serum uE3 levels were highly correlated between the assays (r = 0.93, p < 0.001), with the m-uE3 assay levels yielding, on average, 23% higher (standard deviation of differences in log uE3 concentrations = 0.07) results. Assay and gestation-based median equations were calculated and used to convert m-uE3 concentrations to multiples of the median (MoM). The m-uE3 MoM values fit a log Gaussian distribution well with a log standard deviation of 0.11. Down syndrome risk results were not significantly different between assays.
Conclusions
The m-uE3 assay, with results expressed in MoMs, is suitable for screening and as a monoclonal-based assay offers the advantage of a predictable and indefinite supply of antibody to perform the assay.
Introduction
Prenatal serum screening is performed worldwide to assess the risk of fetal aneuploidy, such as trisomy 21 (Down syndrome). Testing can be offered in the first or second trimester, or both, using various combinations of serum and ultrasound markers.1–3 In the first trimester, between 10 and 13 weeks, maternal serum levels of human beta (free or total) chorionic gonadotropin (hCG) and pregnancy-associated plasma protein A (PAPP-A), combined with a nuchal translucency ultrasound measurement, provide for 85% detection of fetal Down syndrome with a 5% screen-positive rate. Similar screening performance can be achieved in the second trimester, between 15 and 20 weeks, using a combination of four serum markers (Quad test); alpha-fetoprotein (AFP), unconjugated estriol (uE3), hCG, and inhibin A. These tests also provide pregnancy-specific risks for trisomy 18 and, in some programs, trisomy 13 as well. Integrated screening, 3 a combination of first and second trimester markers, provides the best serum screening performance, with up to 95% detection at a 5% positive rate.
New and improved methods for screening have evolved using cell-free DNA in maternal plasma. Although this testing format provides excellent performance, geographical and financial barriers to access remain. 4 Continued provision of maternal serum screening tests remains necessary at this time.
uE3 is a useful component in serum screening, reducing the false-positive rate by about a quarter at 80% and 90% detection rates for the second trimester test (from 3.6% to 2.7%) 5 and Integrated test (from 0.9% to 0.7%), respectively. 2 Levels of uE3 are described as an indicator of fetal well-being. 6 For example, very low levels of uE3 are associated with a rare genetic condition involving cholesterol deficiency, Smith–Lemli–Opitz syndrome, that leads to severe intellectual handicap in affected children. 7 Other fetal problems, such as poor growth, X-linked ichthyosis, and structural anomalies, have also been linked to a low maternal serum uE3 level. 8
Several manufacturers provide reagents for the measurement of uE3 in serum on automated laboratory platforms. Hitherto, polyclonal antisera have been used for uE3 measurement in serum, such as in Siemens Diagnostics (Malvern, PA, USA), Beckman Coulter (Chaska, MN, USA), and PerkinElmer (Turku, Finland) assay methods. However, a polyclonal antiserum is a limited and finite resource. The antiserum is only available as long as the animal that produces it is alive, and the generation of an equivalent antiserum in another animal is uncertain. By contrast, monoclonal antibodies avoid this problem by immortalizing the clone of cells that produce the selected antibody in the laboratory. The goal of this study was to examine the validity of a new immunoassay for unconjugated estriol that uses a monoclonal antibody (m-uE3).
Methods
Samples
Residual second trimester maternal serum samples received in the laboratory at Women and Infants Hospital (<48 h after collection) were used for method comparison. Samples were run fresh on both assays. To determine appropriate medians9,10 for the m-uE3 assay, approximately 100 samples from unaffected pregnancies for each completed week of gestation, from 15 to 19 weeks (n = 533, Table 1), were retrieved from −20°C freezer storage. None of these samples had been previously thawed, and all had been refrigerated at 4°C for less than 1 week before being transferred to freezer storage at −20°C. The gestational age (decimal weeks) and maternal weight (pounds) associated with each sample were recorded. 11 Unconjugated estriol levels in serum are reported to be stable for up to five freeze-thaw cycles. 12
Median gestational age and m-uE3 values observed for second trimester serum samples from unaffected pregnancies.

Data fit well to log linear regression (p < 0.001) as shown by similarity of observed and expected median levels at each week and as depicted in Figure 3.
A separate case–control set from 21 pregnancies with Down syndrome, each associated with five matched controls, was also retrieved from −20°C freezer storage. Matching criteria were completed week of gestation, sample collection date within 6 months, and race (Black or Non-black). Original prenatal screening results for the case–control samples were recorded for comparison of Down syndrome risks with those derived from results that incorporated the m-uE3 assay. Ten of the cases had originally been tested per physician request using the second trimester Quad test and 11 had been tested using Integrated screening. The risk was calculated using the Alpha software (Logical Medical Systems Ltd, London, UK). The project was approved by the Institutional Review Board at Women and Infants Hospital.
Immunoassay
Testing was performed on the Beckman Coulter automated chemiluminescent immunoassay platform (DxI) using the current p-uE3 and/or the new m-uE3 that uses a rabbit anti-estriol antibody developed by Beckman Coulter. The interassay coefficients of variation for m-uE3 in this study were 14% at 3.4 ng/mL and 7% at 7.4 ng/ml. Case–control set samples had also been tested on the DxI at the time of receipt for AFP, p-uE3, hCG, inhibin A, and PAPP-A when a first trimester sample was submitted for Integrated screening.
Statistical analyses
Linear regression and Bland–Altman analyses 13 were used to compare results between assay methods in fresh samples. Using the larger dataset of approximately 100 stored samples at each week of gestation, a log-linear regression of m-uE3 concentrations against gestational age was performed and the resulting equation was used to convert results in ng/mL to gestation-specific multiples of the median (MoM). Those data were then assessed using a probability plot analysis (with suitable truncation limits) for goodness of fit to a log Gaussian distribution and to estimate the logarithm standard deviation (SD) for unaffected pregnancies. The SD was compared with those from published literature. Log-linear regression of MoM values against maternal weight was performed to generate a weight adjustment equation. Using the case–control set, the median m-uE3 MoM in cases was compared with that in controls, and the trisomy 21 detection and false-positive rates were compared using p-uE3 and m-uE3 assay results.
Results
m-uE3 and p-uE3 results were highly correlated (Figure 1, r = 0.93, p < 0.001); however, the m-uE3 levels were on average 23% higher, with a SD of 0.0743 (log[m-uE3]-log[p-uE3]) (Figure 2). After excluding one low outlier, the slope of the relationship between the mean and the difference was not statistically significant (n = 99, Slope 0.0603, 95% CI −0.019 to 0.1394; p = 0.134), indicating a constant proportional difference across uE3 concentrations.
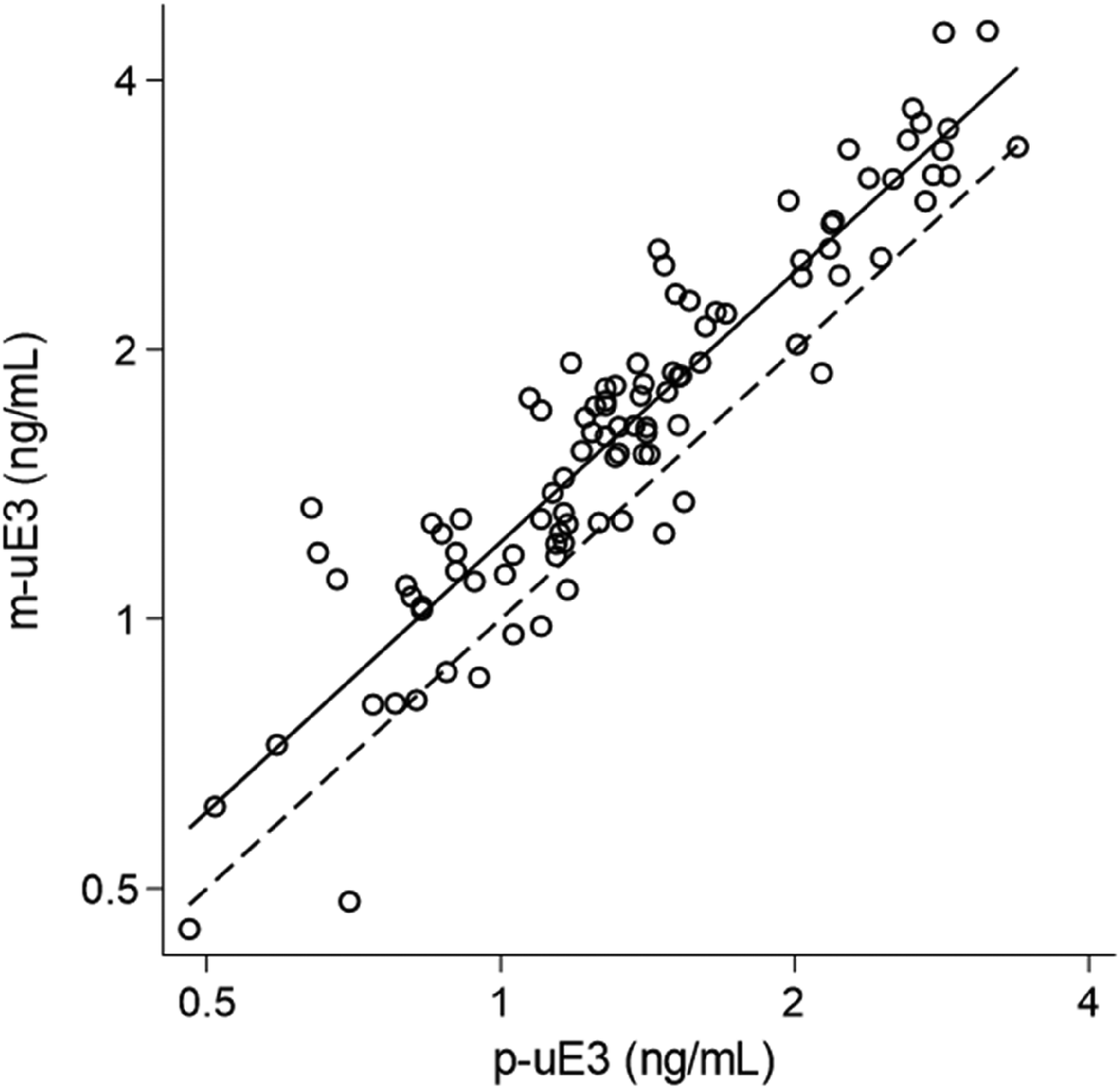
Linear regression analysis of m-uE3 and p-uE3 assay results for 100 fresh second trimester serum samples.
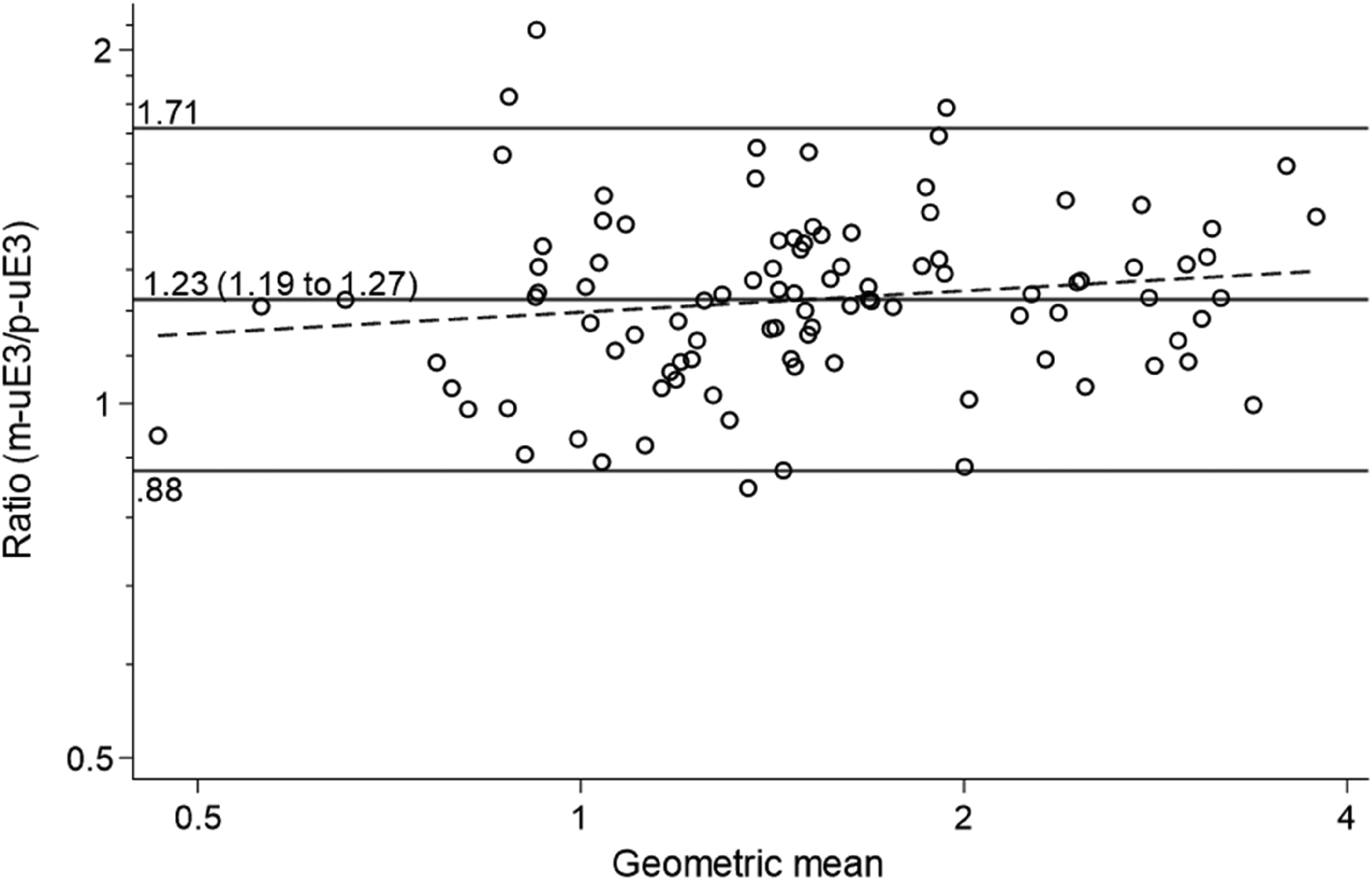
Bland–Altman analysis showing the relationship of the m-uE3 and p-uE3 assays on the DxI instrument. Values are, on average, 23% higher with the new monoclonal assay. One low outlier value was excluded.
The m-uE3 concentrations increased with increasing gestational age in log-linear manner defined as Log10(uE3) = −1.57155 + 0.0148231×GA (Slope p-value < 0.001, Figure 3). The number of samples tested at each gestational week, along with median gestational age and observed and expected m-uE3 median values, are shown in Table 1. Using the derived median equation, m-uE3 values were converted to gestational age-specific MoM. The m-uE3 MoM values fit a log Gaussian distribution well (Figure 4). The slope of the line, between the 10th and 90th centiles, provides an estimate of the log SD and was 0.1114. As expected, there was an inverse relationship between m-uE3 MoM values and maternal weight (Table 2).
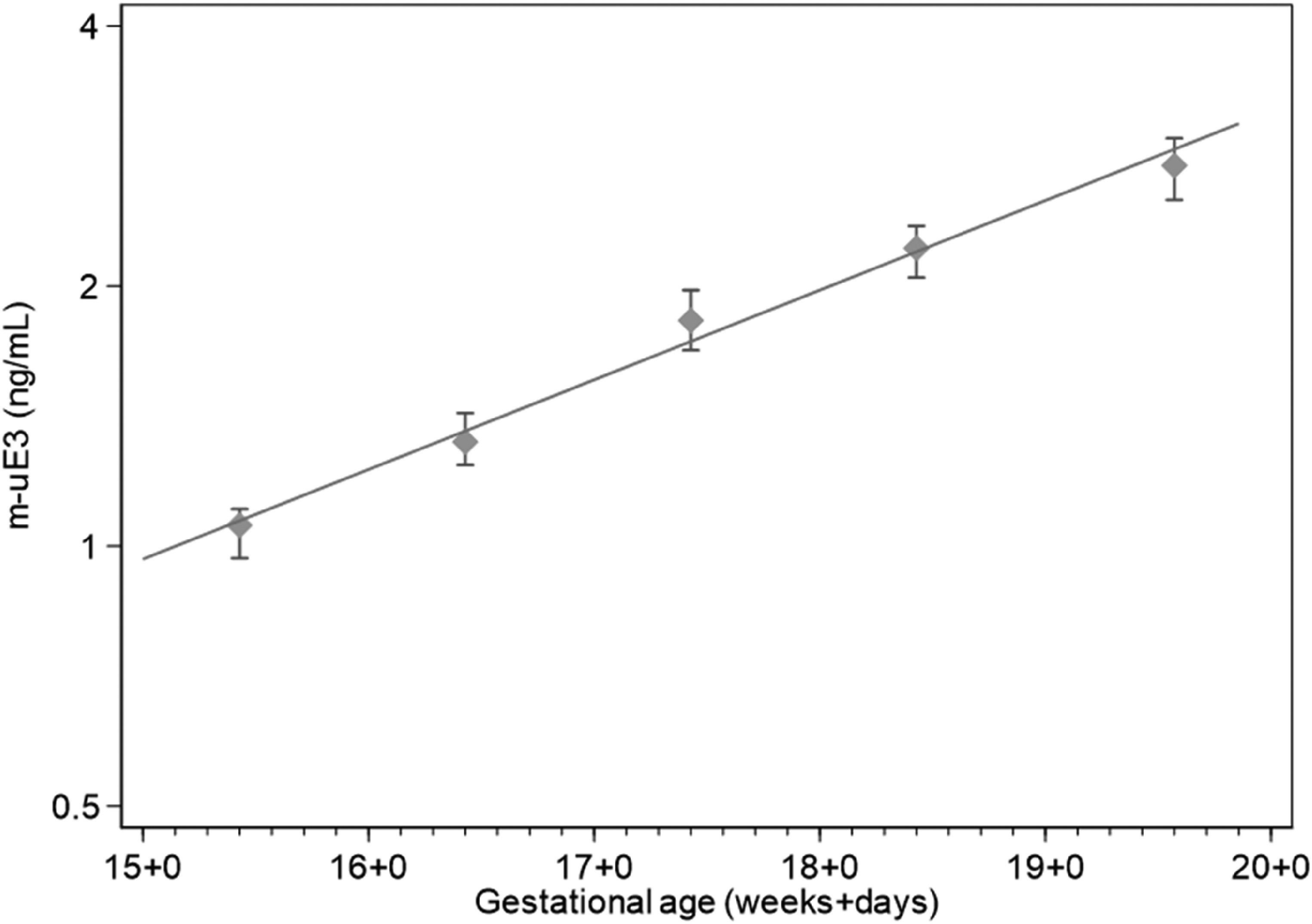
Regression analysis (log-linear) of m-uE3 assay results and gestational age. Median and 95% confidence interval shown at each week of gestation, but regression is based on all data points.
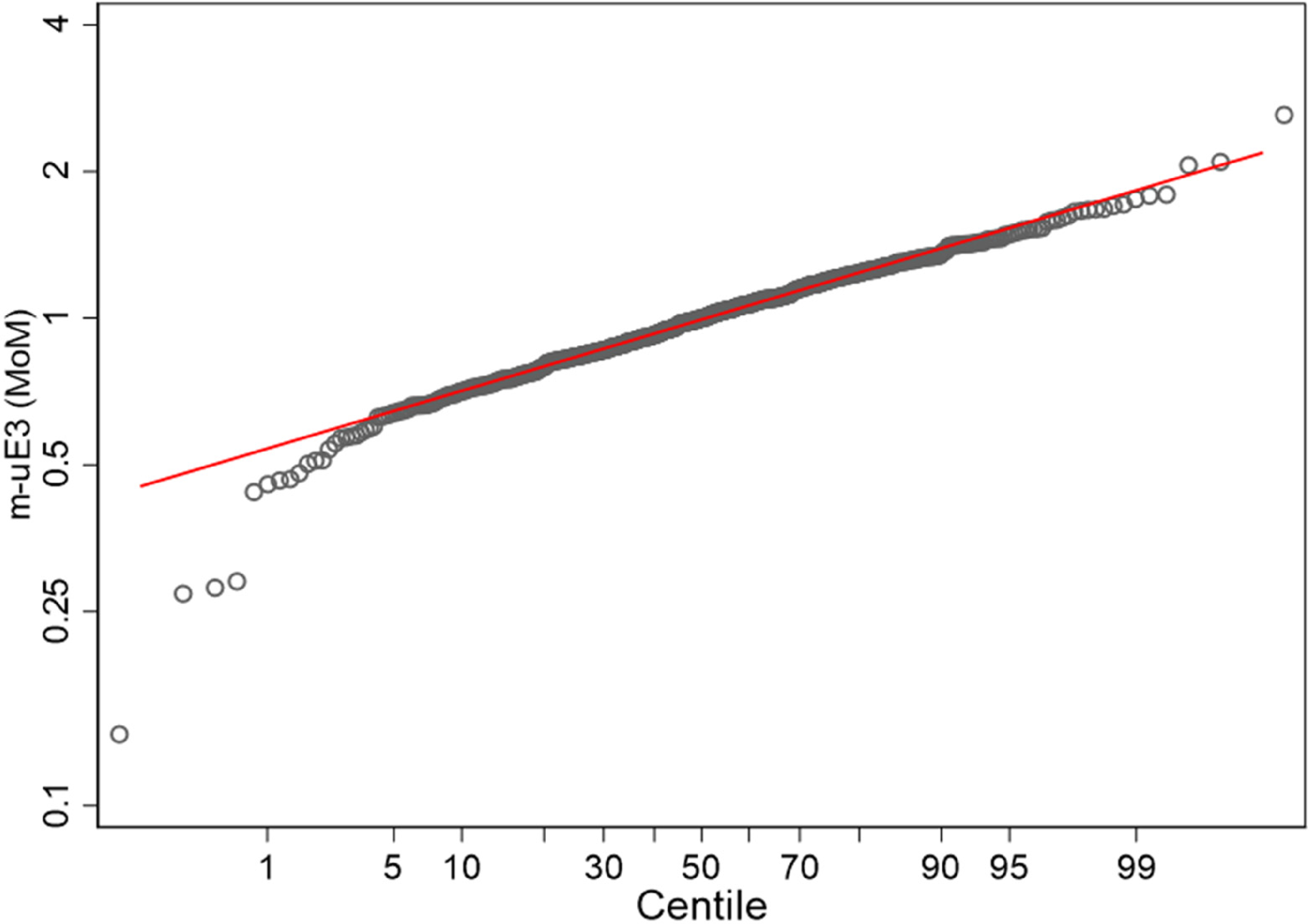
Probability plot showing fit to a Gaussian distribution of m-uE3m MoM values using data calculated from the log-linear median equation.
Analysis of observed median m-uE3 MoM values and maternal weight.
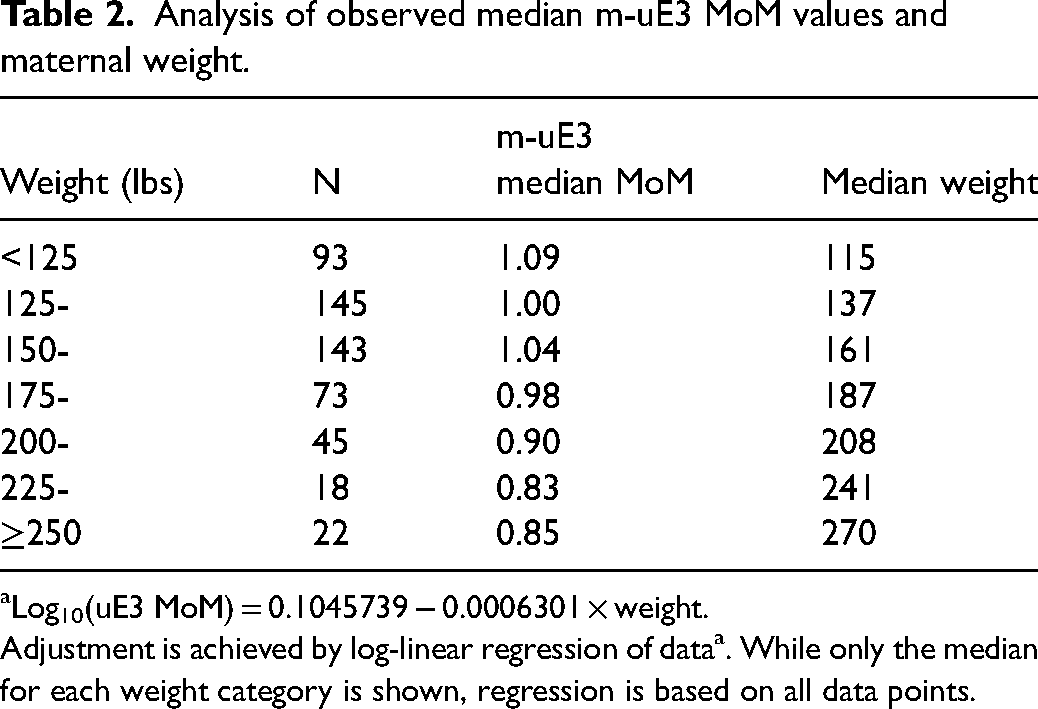
Log10(uE3 MoM) = 0.1045739 − 0.0006301 × weight.
Adjustment is achieved by log-linear regression of dataa. While only the median for each weight category is shown, regression is based on all data points.
The median uE3 MoM in Down syndrome cases was the same, and the Down syndrome detection and false-positive rates were not significantly different for the m-uE3 and p-uE3 assays, regardless of whether the Quad or Integrated screening test was performed (Table 3).
Median uE3 MoM in Down syndrome (DS) pregnancies and detection and false-positive rates, sorted by type of screening test, for m-uE3 and p-uE3.

Wilcoxon signed rank test.
McNemar's test. Quad test cutoff second trimester risk of 1 in 270, Integrated test 1 in 110.
Discussion
The monoclonal antibody used on the DxI platform (Beckman Coulter Inc.) provides acceptable performance for use in Down syndrome screening. In this study, levels obtained in second trimester serum samples were consistently higher with the m-uE3 assay (23%) than with the current p-uE3 assay. The m-uE3 assay was calibrated by nominal mass assignment to a certified reference material, Cerilliant CRM Estriol (Round Rock, TX, USA), whereas the p-uE3 assay was calibrated to a GC-MS standard. Regardless, this difference in absolute levels between methods can be considered by the use of an assay-specific median equation to generate MoM values. This is not an unusual burden as laboratory-specific medians are required routinely in prenatal screening. Data provided in the present manuscript should only serve as a guide to other laboratories interested in adopting the monoclonal assay.
Analyses of m-uE3 MoM values were consistent with expectations. The log SD for unaffected pregnancies was at the lower end of the range previously reported using other uE3 assays (0.1208–0.1476,2,5 without weight correction). The median MoM in 21 Down syndrome cases was similar between assays and showed the expected reduction in levels (0.7 MoM). Furthermore, serum screening performance was similar for both Quad and Integrated test protocols regardless of the assay used; detection rates were identical and screen-positive rates for Down syndrome did not differ significantly between assays and met published test-specific estimates.
To our knowledge, this is the first automated monoclonal uE3 assay commercially available and it offers the advantage of an indefinite and stable supply of antibody, unlike polyclonal antisera that rely on live animals for production. The m-uE3 assay performs as expected in second trimester prenatal maternal serum screening.
Footnotes
Declaration of conflicting interests
NJW is a director of Logical Medical Systems. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded in part by Beckman Coulter Inc through a grant paid to Women and Infants Hospital.
