Abstract
Objectives
Various lifestyle and occupational factors have been associated with an increased risk of breast cancer, but there is limited research investigating the relationship between these factors and participation in breast cancer screening. This study explores the associations between lifestyle and occupational factors and participation in breast mammography screening among women living in Western Australia.
Methods
This study involved 1705 women aged 40 and older who participated as controls in the Breast Cancer Environment and Employment Study conducted in Western Australia. Self-reported questionnaire data were collected on participation in mammography screening, demographic factors, and lifestyle and occupational variables (smoking, physical activity, alcohol consumption, body mass index, use of contraceptive pill and hormone replacement therapy, breastfeeding, occupation, and participation in shift work). Multivariate modified Poisson regression was used to identify variables associated with ever participation in breast mammography screening.
Results
Just over 88% of women reported having ever had a mammogram. Likelihood of having ever had a mammogram was higher among women who had ever used hormone replacement therapy (adjusted prevalence ratio (aPR) = 1.05, 95% CI 1.02–1.07). Women who worked in clerical occupations (aPR = 1.06, 95% CI 1.01–1.11) or home duties (aPR = 1.05, 95% CI 1.00–1.11) were also more likely to report having ever had a mammogram compared with those in professional or technical occupations.
Conclusions
Participation in mammography screening was found to differ by lifestyle and occupational factors. These results have important implications for public health strategies on improving screening participation.
Introduction
Breast cancer is the most commonly diagnosed cancer and the second leading cause of cancer death in Australian women, with an estimated 17,586 new cases diagnosed and 3087 deaths in 2017. 1 Australia has a high breast cancer survival rate, with 90% of those diagnosed in 2009–2013 still alive five years after diagnosis. The survival rate is even higher (98%) when breast cancer is diagnosed at an early stage, 2 reinforcing the importance of early detection through cancer screening. Mammography is the most common mode of breast cancer screening, 3 and is the only means of population-based screening that has been shown to reduce breast cancer mortality. 4 A modelling analysis using data from the population-based BreastScreen Australia programme demonstrated that participation was associated with a 21–28% reduction in mortality. 5
BreastScreen invites all women aged 50–74 to free two-yearly mammograms. 4 Those aged 40–49 and 75 and older are also eligible for free mammograms, but are not actively invited. In 2015–2016, 54.8% of the target population (1,772,540 women) received a screening mammogram, a slight increase from the 53.7% reported for 2013–2014. BreastScreen does not report ‘ever’ screening rates, but 2014–2015 data from a nationally representative sample of Australian households showed that 77% of women aged 50 and older reported having ‘ever’ been screened for breast cancer. 6
Participation in BreastScreen has been found to differ by age (highest in those aged 65–69) and area of residence (highest in those living in outer regional areas), but not across socioeconomic groups. 4 Other recent Australian studies have found various demographic and health factors to be associated with participation in breast cancer screening. Married women, those living in outer regional or remote areas, and those with private health insurance have higher rates of participation in mammographic screening.7,8 Women with a family history of breast cancer and those who have ever used hormone replacement therapy (HRT) also have higher rates of participation.8,9
It has been hypothesized that participation in breast cancer screening may be associated with lifestyle factors such as alcohol use and physical activity. It has been reported that those participating in healthy lifestyle behaviours are also more likely to participate in preventive health behaviours, including cancer screening. 10 Whilst many lifestyle factors have been found to be associated with breast cancer risk, fewer studies have investigated their association with screening participation. 9 A Swedish study found that current smokers, those who had not consumed any alcohol in the past year, and those participating in low levels of physical activity were less likely to participate in breast mammography screening. 11 An Australian study found that overweight or obese women and those who participated in adequate physical activity were more likely to have ever had a mammogram, 9 and that women with a higher number of unhealthy lifestyle risk factors (e.g. alcohol consumption, obesity, physical inactivity, and HRT use) were more likely to have participated in breast mammography screening.
Limited evidence also suggests that participation may be associated with occupational factors, including shiftwork 12 and employment status. 8 A United States study found that women working alternative shifts were less likely to have had a mammogram in the last two years than those working regular daytime shifts, 12 while an Australian study found that those who were employed were more likely to have had a mammogram in the past two years. 8 A recent study of Australian nurses and midwives also found that shift workers and those working full-time were less likely to participate in breast screening compared with those working regular office hours and part-time or casual hours, respectively. 13 This may be related to practical considerations and such women being less able to attend cancer screening appointments than those working regular or shorter hours. 12
Our study aims to further investigate the lifestyle and occupational factors associated with participation in breast mammography screening among women living in Western Australia.
Methods
We used data from the Breast Cancer Environment and Employment Study (BCEES), a case-control study conducted in Western Australia in 2009–2011. Full details of the methodology have been provided elsewhere. 14 The study was approved by the Human Research Ethics Committees of the Western Australian Department of Health (project number 2009/28) and the University of Western Australia (project number RA/4/1/2331). Data were collected by a self-administered postal questionnaire. All participants provided written informed consent. The BCEES included 1205 breast cancer cases (response rate 57.8%) and 1789 frequency age-matched controls (response rate 41.1%) who were randomly selected from the Western Australian electoral roll. We analysed data from control women aged 40 and older (n = 1705), in line with current BreastScreen screening recommendations.
Participation in breast mammography screening was ascertained by the question: ‘Have you ever had a mammogram (breast x-ray)?’. Demographic data collected included age, highest level of educational attainment, country of birth (Australia or other), main language spoken in the home (English or other), and area of residence (metropolitan or non-metropolitan). Socioeconomic status was derived from participants’ residential postcodes and coded according to the Socio-Economic Indexes for Areas Index of Relative Socioeconomic Advantage and Disadvantage 2011. 15 We also assessed the health variables: family history of breast cancer (yes/no), menopausal status (pre- or post-menopausal), and number of children.
The lifestyle variables assessed comprised smoking status, physical activity, alcohol consumption, body mass index, contraceptive pill and HRT use, and breastfeeding duration. These factors were assessed as they are part of the World Cancer Research Fund cancer prevention recommendations for reducing risk of cancer, including breast cancer, and/or they have been associated with risk of breast cancer.16–18 Smoking status was classified as current, former, or never, with current and former smokers combined into a single group for analysis. Physical activity was assessed using a modified version of the Chasan-Taber Physical Activity Questionnaire, which assesses recreational, household, and transport-related physical activity.19,20 All activities reported were assigned a metabolic equivalent (MET) value, derived from the Compendium of Physical Activities, 21 and MET-hours/week calculated by multiplying the MET-value by its frequency and duration. We then averaged MET-hours per week conducted over the lifetime, and classified into quartiles for analysis. Alcohol consumption was assessed as the usual number of alcoholic drinks consumed per week (categorized into <1, 2–3, 4–6, or 7+ drinks). Body mass index (BMI) was based on self-reported current weight and height and trichotomized into not overweight (<25 kg/m2), overweight (25 < 30 kg/m2), or obese (≥30 kg/m2). Participants were also asked whether they had ever used the combined oral contraceptive pill (yes/no) and HRT (yes/no). Breastfeeding was assessed only for those women who reporting having given birth to at least one child. Participants were asked how many months in total they breastfed their children, and duration was then categorized into ≤6 months, 7–12 months, 13–24 months, and >24 months for analysis.
A full occupational history was collected for each participant, including job title for each job held for more than six months, and time spent in each job. Occupation was coded according to the International Standard Classification of Occupations-1968 (ISCO-68). 22 We used data on the longest job held over the lifetime as well as the most recent job held. Where the participant held more than one job for the same amount of time, we used the most recent of those jobs in the longest job held analysis. Women were also asked whether their job involved “night work, shift work, or work at unusual hours”. Those who answered yes to this question were then asked a series of questions including “Did you ever work between the hours of midnight and 5 am?”; the answer to this question was used to define exposure to graveyard shift (yes/no).
We used modified Poisson regression with robust sandwich variance estimation 23 to estimate the prevalence ratio (PR) and 95% confidence intervals (CI) of participating in breast mammography screening by lifestyle and occupational variables. Analyses controlled for those sociodemographic and health variables which were significantly associated with both screening participation and lifestyle or occupational variables, where relevant. We used backward stepwise elimination with a cut-off of p < 0.10 to arrive at the final model. All statistical tests were two-sided with significance established at α=0.05. All analyses were conducted in Stata 14 (College Station, TX).
We created a series of healthy lifestyle indices (HLI) based on the seven lifestyle variables investigated. We used a binary score (0/1) for each factor whereby a score of 1 indicated healthier behaviour (i.e. not a current or former smoker; those reporting ≥59 MET-hours per week; alcohol consumption of ≤1 drink per week; BMI <25 kg/m2; never use of oral contraceptive pill; never use of HRT; breastfeeding duration of >6 months). We then summed the binary score for each of the factors. For the overall HLI, scores ranged from 0 (least healthy) to 7 (most healthy). As a small number of individuals were practicing less than three or more than six healthy behaviours, we combined the scores into five categories (0–2, 3, 4, 5, and 6–7 factors). A test for trend was conducted by entering the original HLI score into the model as a continuous variable.
We also conducted sensitivity analyses by excluding various factors from the HLI. First, we excluded smoking, as this factor has not been found to be associated with breast cancer risk. Scores ranged from 0 to 6, with scores combined into five categories for analysis (0–1, 2, 3, 4, 5–6). Next, we excluded use of oral contraceptive pill, HRT, and breastfeeding duration, and created a separate reproductive behaviour index comprising these factors. Scores for the reproductive index ranged from 0 to 3, while scores for the revised HLI (smoking, physical activity, alcohol consumption, BMI) ranged from 0 to 4.
Results
A total of 1494 women aged 40 and older (88.1%) reported having ever had a mammogram (Table 1). Older women and those with a family history of breast cancer were more likely to report having ever had a mammogram, while those who were pre-menopausal were less likely to have had a mammogram. Likelihood of having had a mammogram increased with number of children; however, this trend was no longer significant after controlling for other demographic factors.
Ever breast mammogram screening, by sociodemographic characteristics.
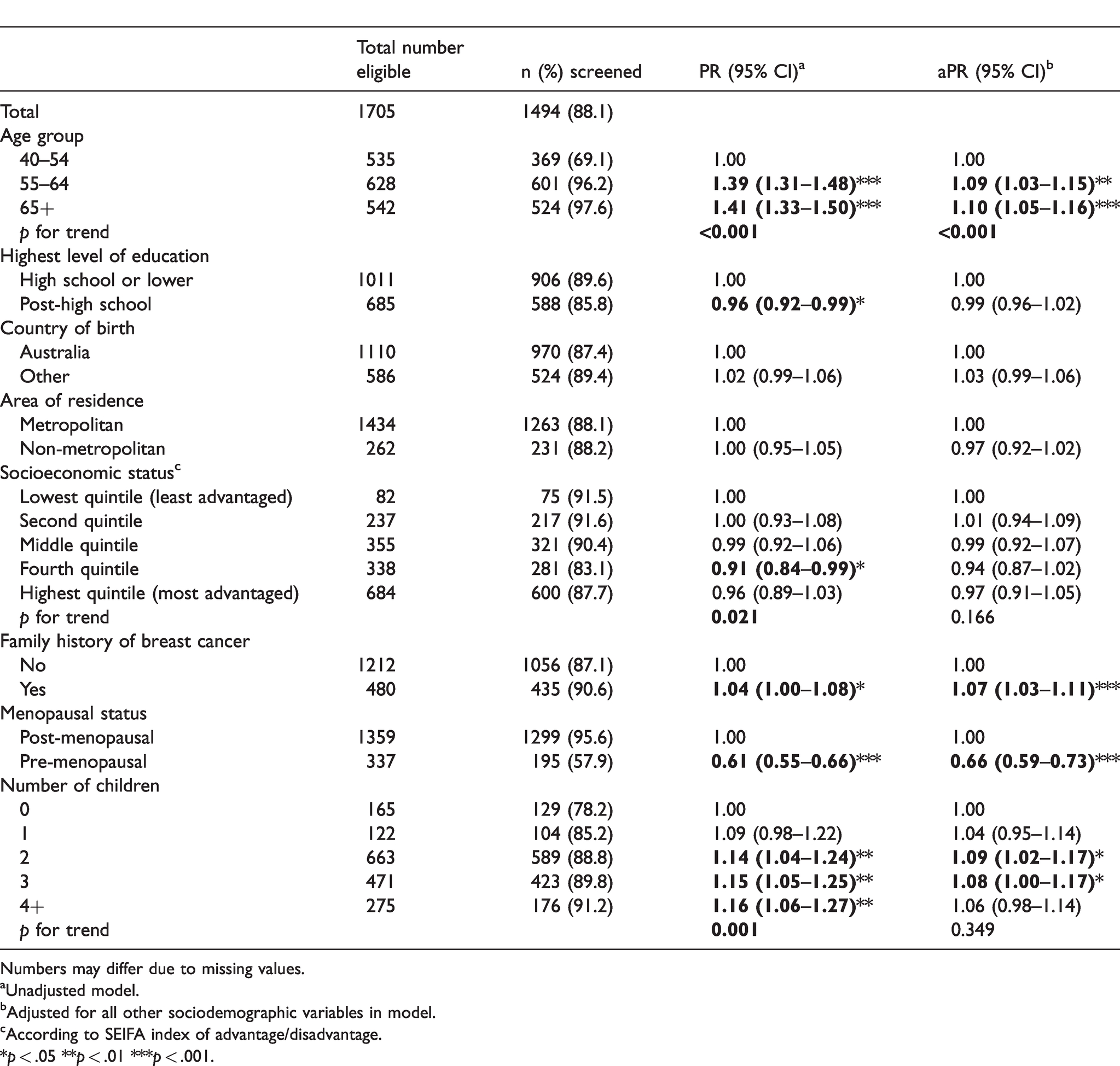
Numbers may differ due to missing values.
aUnadjusted model.
bAdjusted for all other sociodemographic variables in model.
cAccording to SEIFA index of advantage/disadvantage.
*p < .05 **p < .01 ***p < .001.
When considering lifestyle factors, women who were more physically active and those who had ever used HRT were more likely to have ever had a mammogram (Table 2). Likelihood of having ever had a mammogram did not differ by smoking status, alcohol consumption, BMI, contraceptive pill use, or breastfeeding duration. When lifestyle factors were considered together in an overall HLI, there was no significant association with having ever had a mammogram (Table 3). When smoking was excluded from the HLI, those practicing a higher number of healthy behaviours were less likely to have ever had a mammogram, but this association attenuated when controlling for demographic variables. When considering lifestyle behaviours only (smoking, physical activity, alcohol consumption, BMI), there was a significant trend whereby those practicing a higher number of healthy behaviours were more likely to have ever had a mammogram, but again, this association attenuated when controlling for demographic variables. When considering reproductive behaviours only (ever use of oral contraceptive, HRT, breastfeeding duration), there was a significant trend whereby those practicing a higher number of healthy behaviours were less likely to have ever had a mammogram. This association remained after controlling for demographic variables.
Ever breast mammogram screening, by lifestyle characteristics.
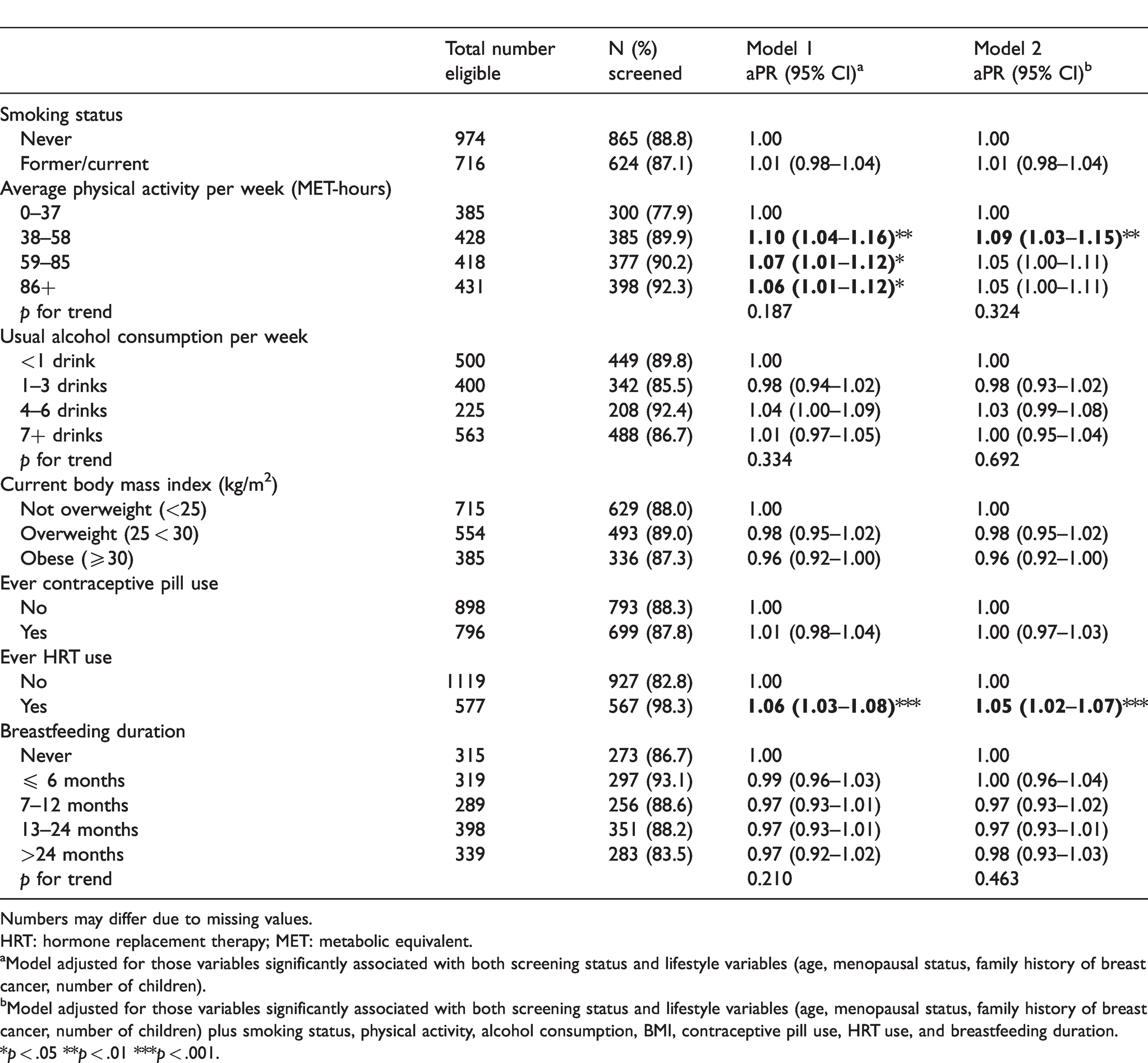
Numbers may differ due to missing values.
HRT: hormone replacement therapy; MET: metabolic equivalent.
aModel adjusted for those variables significantly associated with both screening status and lifestyle variables (age, menopausal status, family history of breast cancer, number of children).
bModel adjusted for those variables significantly associated with both screening status and lifestyle variables (age, menopausal status, family history of breast cancer, number of children) plus smoking status, physical activity, alcohol consumption, BMI, contraceptive pill use, HRT use, and breastfeeding duration.
*p < .05 **p < .01 ***p < .001.
Ever breast mammogram screening, by healthy lifestyle indices.
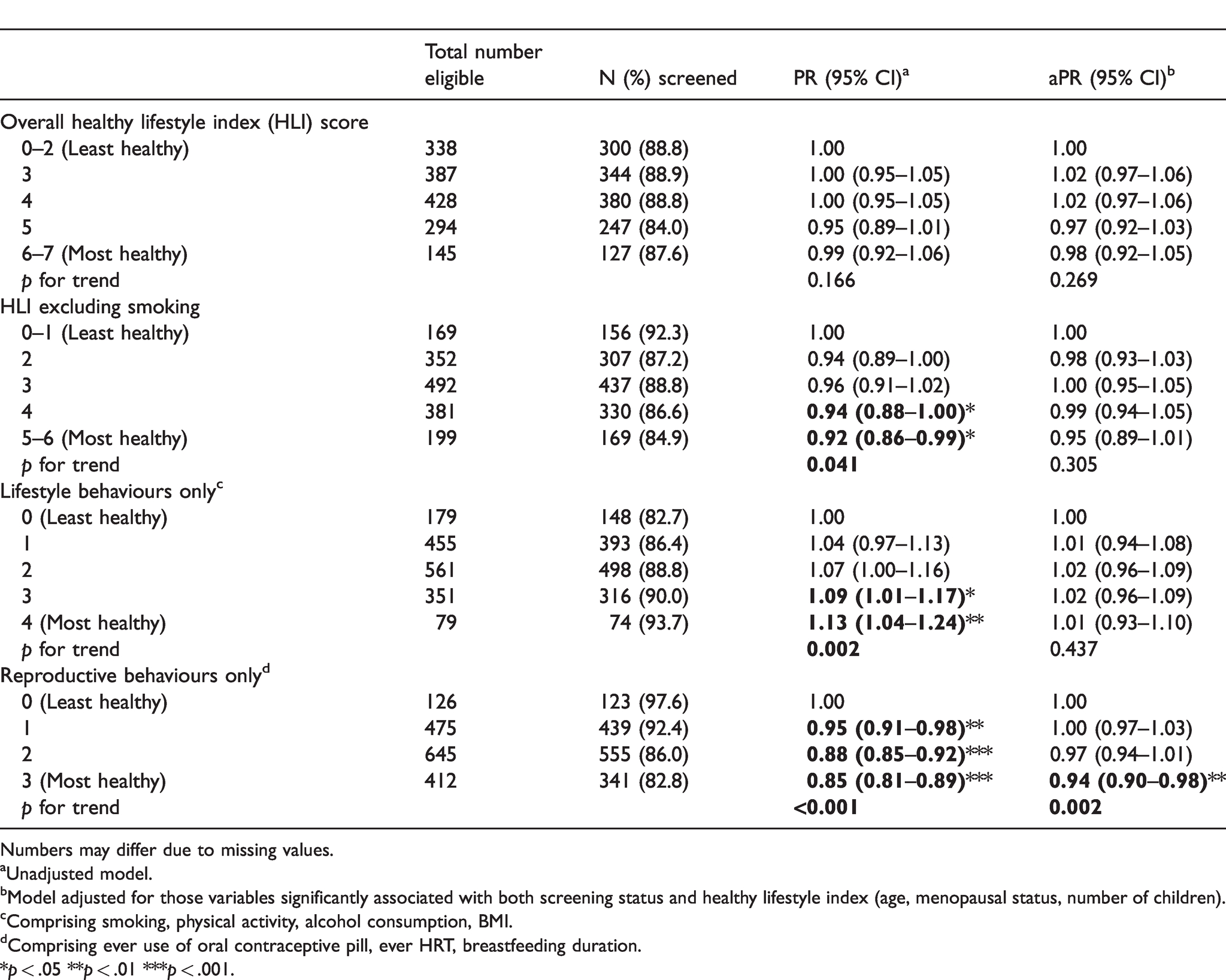
Numbers may differ due to missing values.
aUnadjusted model.
bModel adjusted for those variables significantly associated with both screening status and healthy lifestyle index (age, menopausal status, number of children).
cComprising smoking, physical activity, alcohol consumption, BMI.
dComprising ever use of oral contraceptive pill, ever HRT, breastfeeding duration.
*p < .05 **p < .01 ***p < .001.
The likelihood of having ever had a mammogram was higher in women who reported working in clerical occupations or home duties compared with those in professional or technical occupations (Table 4). There was no difference in likelihood of having ever had a mammogram by shift work status or duration.
Ever breast mammogram screening, by occupational characteristics.
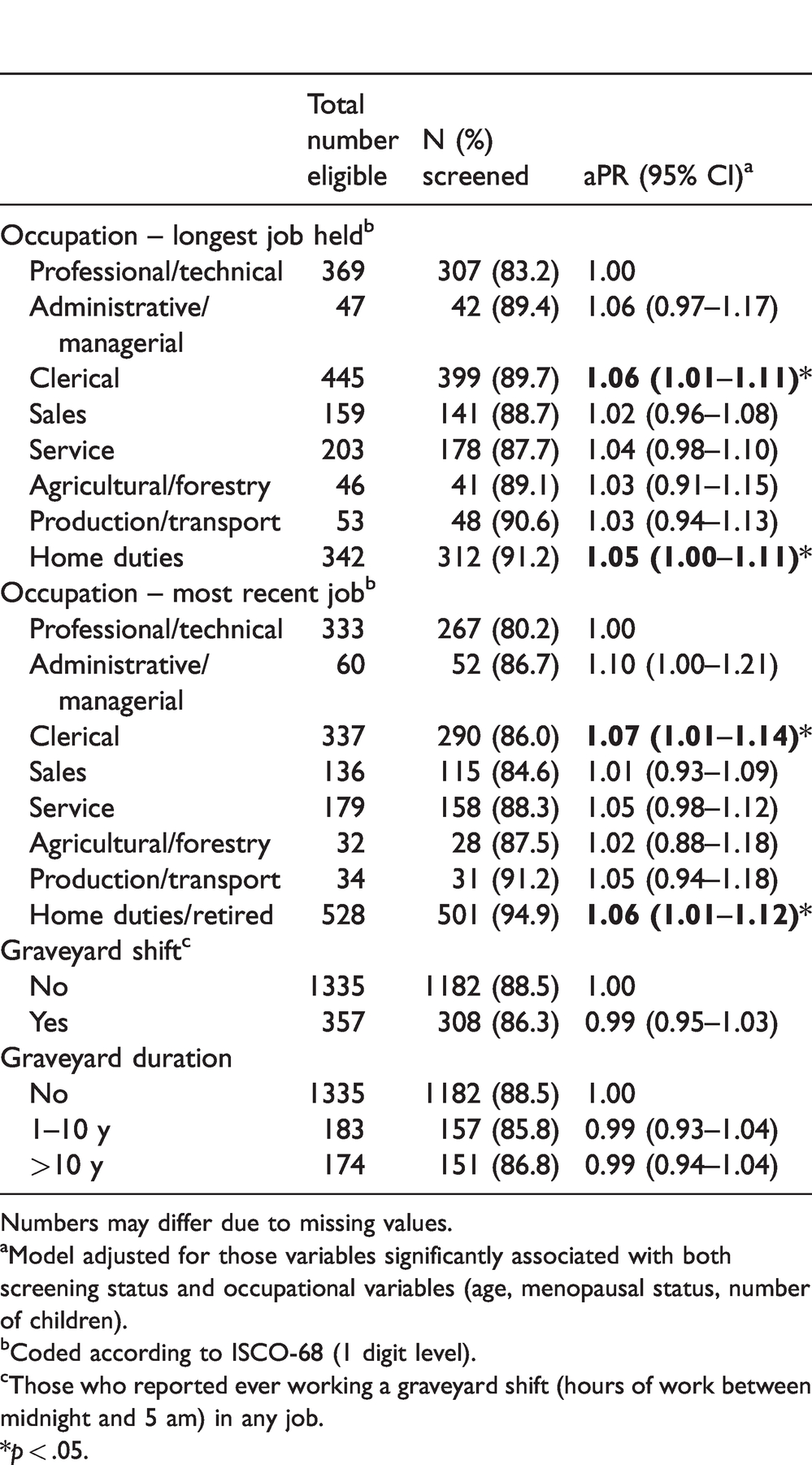
Numbers may differ due to missing values.
aModel adjusted for those variables significantly associated with both screening status and occupational variables (age, menopausal status, number of children).
bCoded according to ISCO-68 (1 digit level).
cThose who reported ever working a graveyard shift (hours of work between midnight and 5 am) in any job.
*p < .05.
Discussion
This study found that 88% of women aged 40 and older had ever had a breast mammogram. Participation was higher in older women and in those with a family history of breast cancer, in line with findings reported in previous Australian studies.7–9 The 88% participation rate is in line with nationally representative data from the National Health Survey conducted in 2014–2015, which found that 77% of women aged over 50 reported having ‘ever’ been screened for breast cancer. 6 Our estimate also lies within the range of studies carried out in other States of Australia (e.g. 65% in South Australia 9 ), and other developed countries (92% in Sweden 24 and 60.5% in Spain 25 ). Discrepancies in participation rates reported between studies are probably due to the different inclusion and exclusion criteria of the studies, sample sizes, type of screening program, and mammogram screening recommendations.
We found that women who had ever used HRT were more likely to have ever had a mammogram, in line with other Australian studies. A previous nationwide study found that women using HRT were 1.4 times more likely to have had a mammogram in the past three years, 7 while a survey of South Australian women found that those who had ever used HRT were 3.7 times more likely to have ever had a mammogram. 9 The authors of the South Australian study noted that the association may result from increased contact with medical practitioners, who may then recommend screening to women using HRT due to the associated increased risk of breast cancer. In support of this, past research has found an association between frequency of general practitioner visits and breast screening participation, 7 and recommendations from medical practitioners have been found to predict screening behaviour. 26 We also found that those practicing a higher number of healthy reproductive behaviours (never use of oral contraceptive and HRT, and breastfeeding duration of six months or greater) were less likely to have ever had a mammogram. It is likely that this finding is strongly influenced by the association between HRT use and likelihood of having had a mammogram.
We did not find a relationship between other known breast cancer risk factors, including alcohol consumption and overweight/obesity, and participation in breast screening. This is in contrast to past research, which has found that women who did not consume alcohol were less likely to participate, 11 and that those who were overweight or obese were more likely to have ever had a mammogram. 9 We did find some limited evidence that women practicing more physical activity were more likely to have ever had a mammogram, in line with past research. 11
We found limited evidence that participation in breast mammography screening was associated with occupation. Those working in clerical occupations or home duties were more likely to have ever had a mammogram than those in professional or technical occupations. Past Australian research has not investigated differences in screening participation by occupation, although studies in the United States have found higher rates of mammography screening participation among workers in the construction, health care, and services industries, as well as white collar occupations.27,28 Previous studies have also found that those working in shifts were less likely to participate in breast mammography screening,12,13 but we did not find any difference in screening participation by shift work status. This is line with a study conducted in Korea, which found that regular day time and alternative shift workers did not differ in their participation in breast cancer screening. 29 This suggests that more research is needed to clarify the relationship between shift work and screening participation.
This study has several strengths including the large sample size of women aged 40 and older living in Western Australia, and the comprehensiveness of the data collection, allowing for adjustment for a number of important variables associated with lifestyle and occupational factors and mammography screening. The cross-sectional design, however, limited causal and directional conclusions. Another limitation was that mammography screening was based solely on self-report, and women may have over-estimated their screening behaviour. This may be particularly so in the older age groups, where we found a very high proportion of women (97.6%) reporting having ever screened. A meta-analysis of the accuracy of self-reported mammography use relative to medical records found a high sensitivity (94.9%) and specificity (61.8%) of self-report, 30 and our overall participation rate roughly reflects that reported in the National Health Survey data, a nationally representative sample of Australian households. 6
It may also be that those having a mammogram for purposes other than screening, including diagnostic purposes, have been included in our estimate of screening participation, as our question did not specifically exclude non-screening mammograms. Data from the United States indicate that approximately 7% of mammograms in women without prevalent breast cancer are conducted for non-screening purposes. 31 Given that women typically have multiple mammograms over their lifetime, and that our measure was of ‘ever’ having had a screening mammogram, it is likely that even those reporting a diagnostic mammogram may have also had a screening mammogram in the past. Thus, the magnitude of this potential overestimation is likely to be minimal.
In addition, body height and weight were not objectively assessed. Previous studies have shown that weight is often under-reported, especially among overweight and obese women, leading to an underestimation of body mass index. 32 Another limitation is that physical activity was collected through self-reported questionnaire, which may have led to an under- or over-estimation of physical activity, as a recent meta-analysis reported that self-report measures of physical activity differed in their accuracy and led to both under- and over-estimation depending on the measure. 33 Finally, the possibility of selection bias cannot be ruled out, particularly given the low response rate (41.1%) of controls in the BCEES. Women participating in this study may have been more likely to be invested in breast cancer and breast health, and arguably may have been more likely to have had a mammogram.
Conclusion
We found that participation in breast mammography screening was lower among those participating in less physical activity, and in those who had not used HRT. These findings have important implications for public health strategies on how to improve screening participation, providing information about the groups who are least likely to screen. Identifying factors associated with breast mammography screening participation can assist health professionals, and particularly general practitioners, to identify those who are less likely to participate, and consequently to encourage them to undergo screening. These findings will also enable those at high risk of not being screened to be targeted for additional reminders, including, for example, by post, telephone, or email.
Footnotes
Acknowledgements
We thank the many women who participated in this study, as well as the members of the BCEES team (Lin Fritschi, Allyson Thomson, Ann D’Orsogna, Terry Slevin, Jennifer Girschik, Pierra Rogers, Terry Boyle, Jane Heyworth, Deborah Glass, Christobel Saunders, Troy Sadkowsky).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Breast Cancer Environment and Employment Study was funded by a National Health and Medical Research Council Australia (NHMRC) Project Grant #572530 and by a grant from the Cancer Council Western Australia.
