Abstract
Objective
To compare interval cancer rates from the Irish breast screening programme, BreastCheck, for the period 2000–2007 with those from other European countries.
Methods
Data from BreastCheck was linked to National Cancer Registry breast cancer registrations, to calculate numbers of women screened, screen-detected cancers, and interval cancers, by year of screening, in the first and second years after screening, and by initial or subsequent screen. Estimated underlying cancer incidence from the period 1996–1999 inclusive was used to calculate proportionate incidence. We calculated the interval cancer ratio as an alternative measure of the burden of interval cancers.
Results
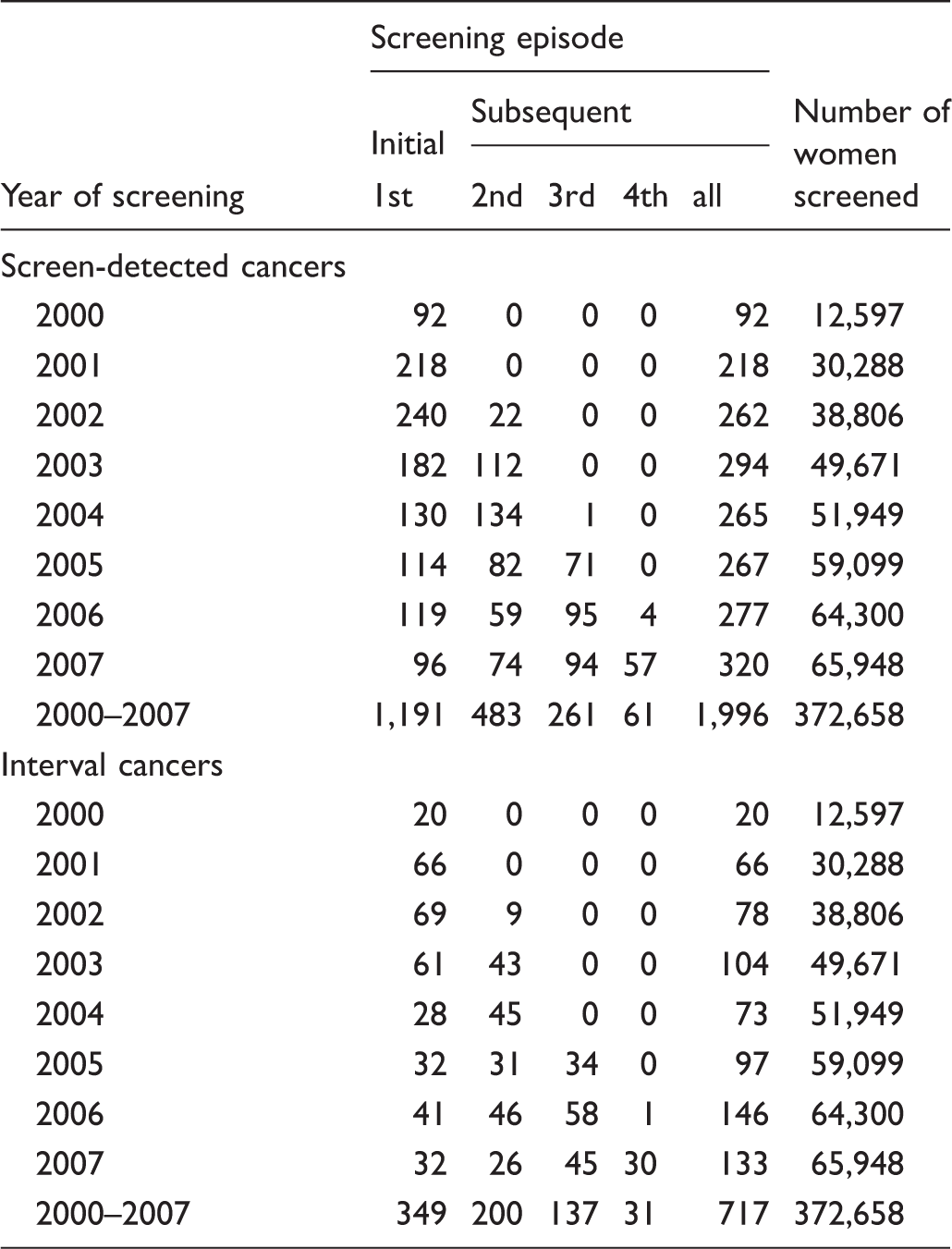
There were 372,658 screening records for 178,147 women in the period 2000–2007. The overall interval rate was 9.6 per 10,000 screens. In the first year after screening, the interval cancer rate was 5.8 per 10,000 screens and this increased to 13.4 in the second year after screening. The screen detection rate for the period was 53.6 per 10,000 screened for all screens combined. Initial screens produced a higher detection rate at 66.9 per 10,000 screened compared with subsequent screens with a screen-detected rate of 41.4 per 10,000 screens.
Conclusion
Interval breast cancer rates for the first years of the programme are within acceptable limits and are comparable with those in other European programmes. Nationwide roll-out together with the adoption of digital mammography may have an impact on interval cancer rates in future years.
Introduction
Mammographic screening programmes, introduced in many countries with the aim of reducing breast cancer mortality,1–3 must balance the costs and benefits of screening. While these are still the subject of debate,4,5 the consensus is that well-organized and systematically conducted breast screening, with rigorous internal and external quality control, is effective at the population level.6,7 Population-based screening programmes usually have no internal mechanism for monitoring the incidence of breast cancer in the interval between screens, which is generally done by following up screening attendees through population-based cancer registries. 7 Interval cancer rates can provide evidence on the sensitivity of the screening test, and on appropriate screening intervals, and may provide further information on the natural history of the disease. 8 If interval cancer rates are high, further investigation may be required into why this is happening and to ascertain the nature of the interval cancer. Most programmes with at least one full round of screening have reported on interval cancer rates. 9
Following a pilot programme in the Dublin region, 10 the Irish breast cancer screening programme, BreastCheck, commenced in 2000 in the east of the country, covering about 50% of the eligible population, and was extended to the rest of the country beginning in 2007. 11 A full round of screening of the entire national eligible population was achieved in 2013. Women aged 50–64 are invited for free screening every two years. Two-view mammography and double reading of mammograms were used from the outset of the programme, and digital mammography was adopted in 2007.12,13
We aimed to evaluate the incidence of interval cancers in the early years of the Irish programme and to compare this with recent data from population-based screening programmes in other European countries.
Methods
Ireland has a mixed public-private health system; screening mammography is available through the public national BreastCheck programme. Some opportunistic screening is performed in private hospitals and clinics, but we do not have access to this data. A linked dataset suggests that there were 765 in-situ cancer diagnoses within two years of a screening (with no further invasive breast cancer in the period 2000–2009) of which 176 (23%) were interval cancers. This paper describes invasive cancers detected in women attending the organized programme, BreastCheck.
Data was provided by BreastCheck on all women screened from 2000–2007. On registration, each woman was asked to tick a box by BreastCheck to consent to her data being shared with the National Cancer Registry. After the exclusion of a small number of duplicate observations and records relating to women outside the screening age group, there were 372,658 screening records for 178,147 women. Using probabilistic matching methods, these records were linked to breast cancer registrations, both invasive (ICD10 C50) and
For each woman the follow-up time from screening to the end of 2009, or age 66, or date of breast cancer diagnosis, whichever occurred first, was calculated to give woman years at risk (wyar) overall, for initial (prevalent) and subsequent (incident) screen and by year of screening. If a woman had more than one tumour, only the earliest invasive tumour was retained in the dataset. If two invasive tumours were diagnosed at the same time, the one with worse prognosis was retained. Cancers of unknown type and those coded as Paget’s disease and phyllodes tumour were excluded from the dataset. Each successfully linked cancer was classified as screen-detected or interval (diagnosed within two years of a negative screen). It was not possible to tell if 527 cancers diagnosed more than two years after a screen were due to a missed screening episode or delay by either the programme or the woman, so these were excluded from the analysis. These women were considered lapsed attenders 15 and considered a separate group from those with screen detected or interval cancers. Numbers and rates are given here for invasive breast cancer only.
Screen detected (interval) cancer rates per 10,000 screens are calculated by dividing the number of screen detected (interval) cancers by the number of women screened and multiplying by 10,000. Several methods exist to describe the incidence rate of interval cancers: as a rate for a specified period (the number of interval cancers divided by the number of wyar for that period); as a proportion of the expected rate for that period (proportionate incidence);
7
or a proportion of total cancers in the screened population (number of interval cancers divided by the total number of screen-detected and interval cancers for that period). This last method, the interval cancer ratio has been argued by Andersen
We used Joinpoint, 17 a piecewise log-linear model with limited constraints on the positions of the joinpoints, to examine whether there were trends in screen-detected or interval cancer rates over the time period and to estimate any annual percent changes in the rates. Finally, we abstracted relevant information from papers identified on PubMed to report interval cancer rates from other population-based breast cancer screening programmes in Europe.
Results
Number of screen-detected and interval cancers by year and screening episode, 2000–2007.
Numbers and rates of screen-detected and interval cancers, overall and for initial and subsequent round, by age at screening.
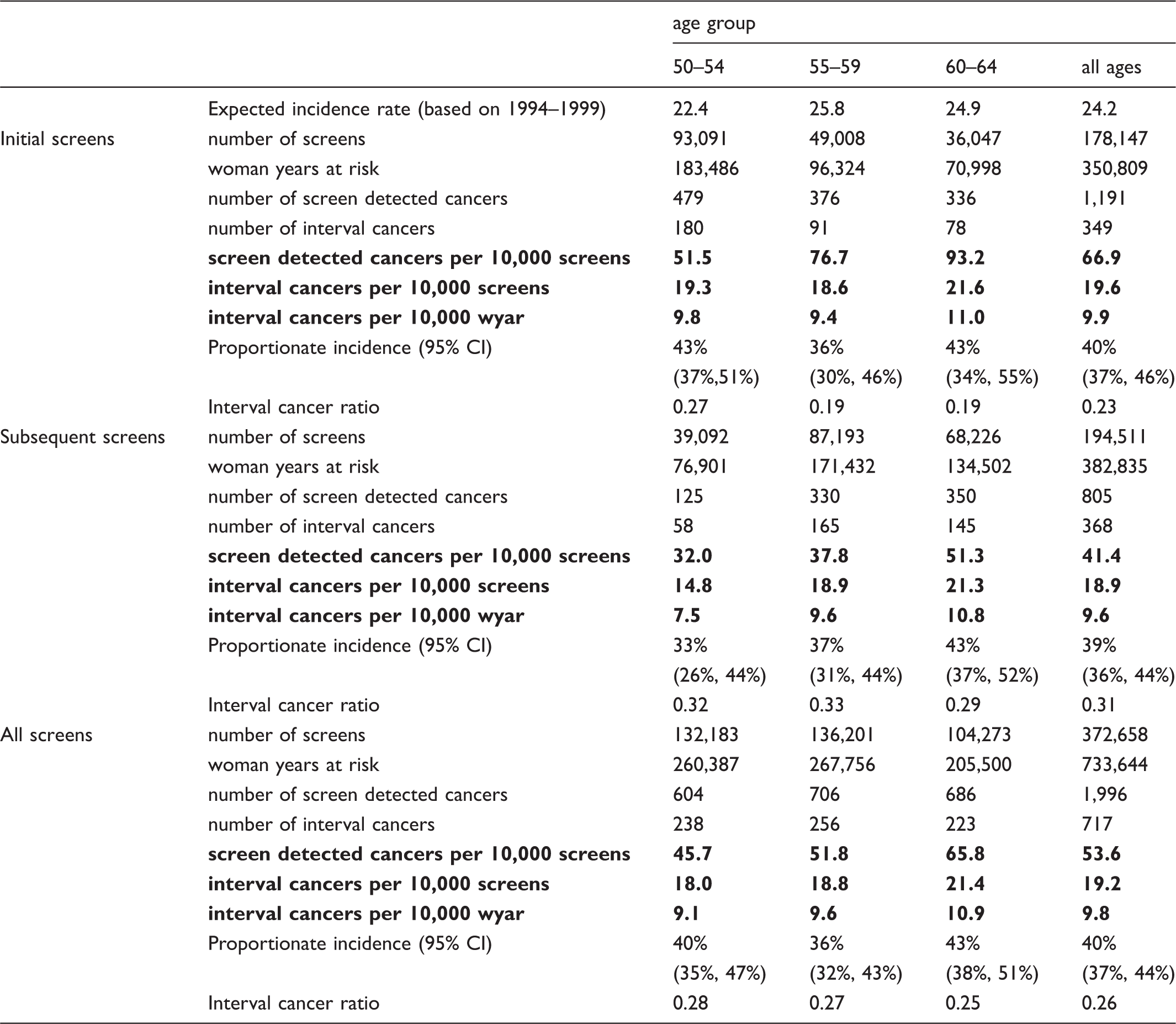
There were 717 invasive interval cancers diagnosed in women aged 50–64 at screening and aged 50–66 at diagnosis (table 2). The interval cancer rate was 19.6 per 10,000 screens for initial screens and 18.9 per 10,000 screens for subsequent screens (table 2). Proportional incidence of interval cancers and interval cancer ratio are presented for three age-groups and overall in table 2.
Woman years at risk, numbers and rates of interval cancers, by time since last negative screen and screening year.
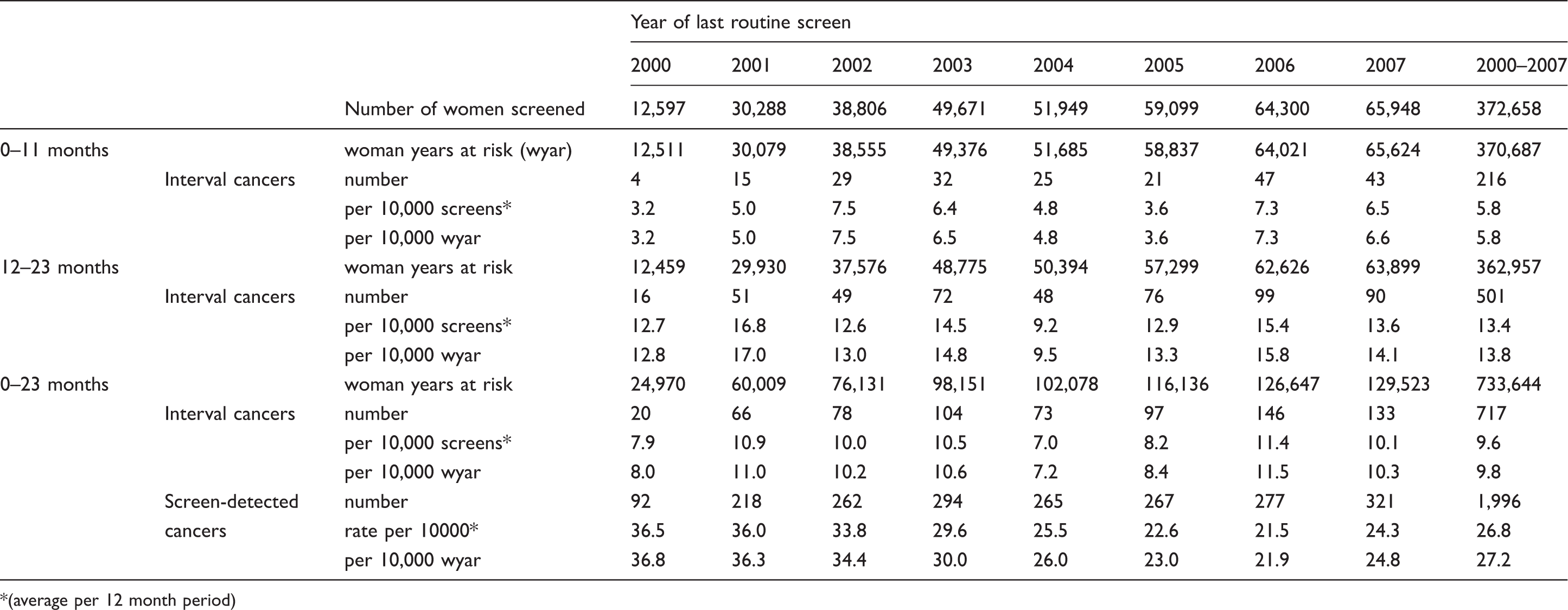
(average per 12 month period)
Comparison of interval cancer rates and proportionate incidence in Ireland and other European countries, ordered by proportionate incidence (0–23 months).
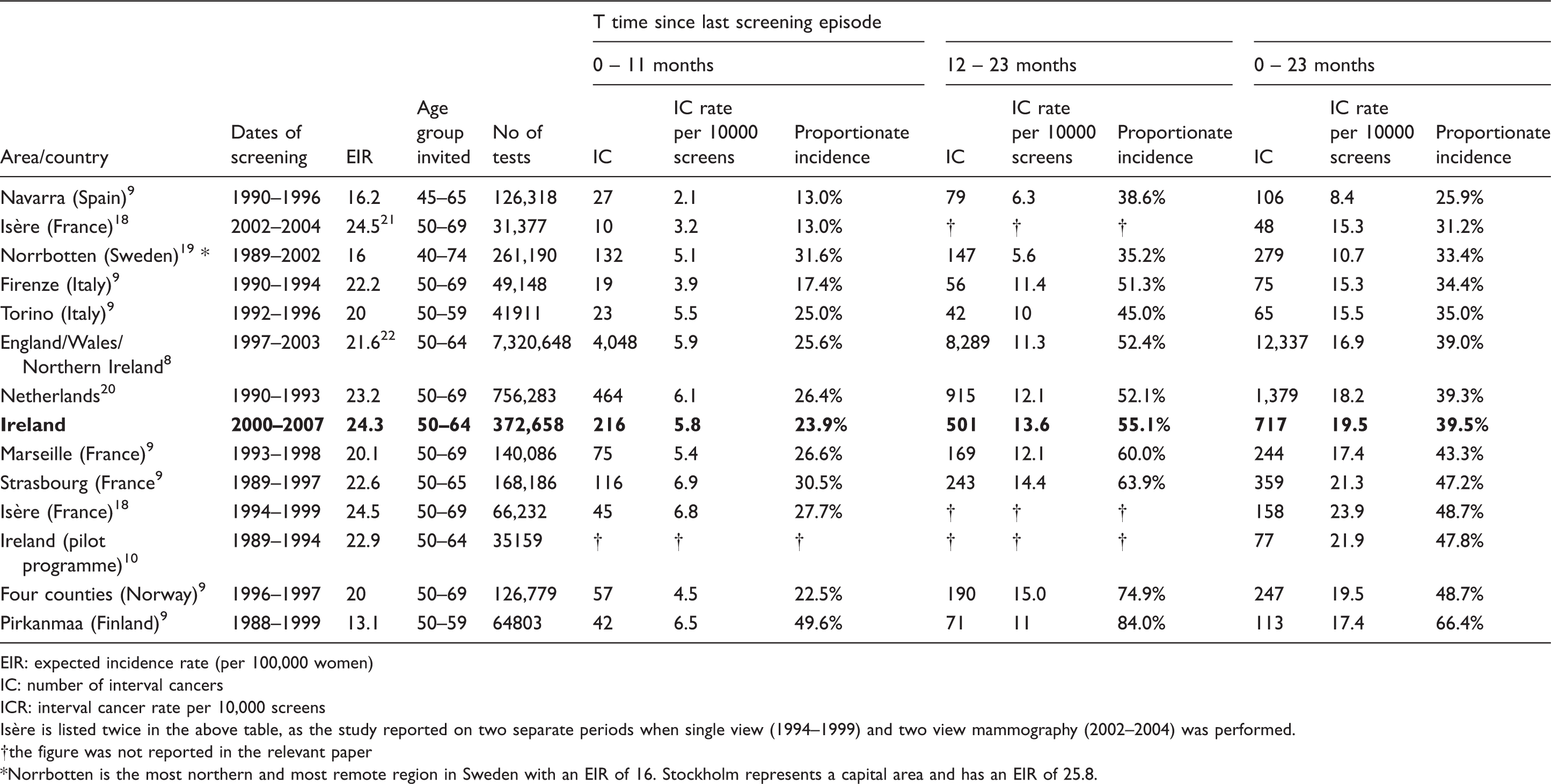
EIR: expected incidence rate (per 100,000 women)
IC: number of interval cancers
ICR: interval cancer rate per 10,000 screens
Isère is listed twice in the above table, as the study reported on two separate periods when single view (1994–1999) and two view mammography (2002–2004) was performed.
the figure was not reported in the relevant paper
Norrbotten is the most northern and most remote region in Sweden with an EIR of 16. Stockholm represents a capital area and has an EIR of 25.8.
Comparison of interval cancer ratios in Ireland and other European countries, ordered by overall interval cancer ratio.
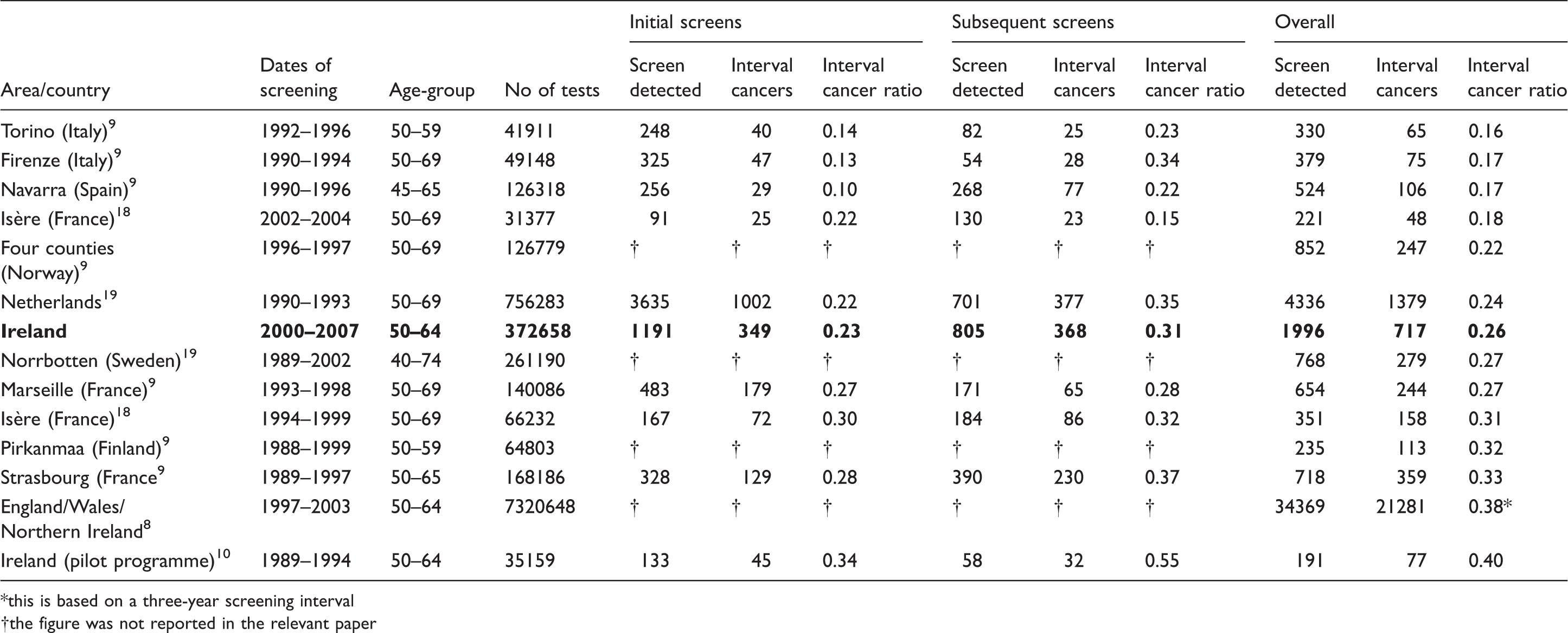
this is based on a three-year screening interval
the figure was not reported in the relevant paper
Discussion
A strength of this study is the high level of completeness of the National Cancer Registry data in Ireland, in which information on cancer diagnoses for women who reside in one part of the country but are treated for cancer in another part (common in rural populations) is captured. With 372,658 women participating in the programme in the period 2000–2007, there were sufficient numbers to compare incident and subsequent screens in addition to the number of early (0–11 months) and late (12–23 months) interval cancers. The main limitation is the difficulty in calculating the background incidence, which makes inter-country comparison problematic.
BreastCheck sets a minimum standard of no more than 7.5 interval cancers per 10,000 women screened in the first year, and 12.5 per 10,000 in the second year, following a screening episode. 23 The “achievable” standard was set as 5.0 per 10,000 women screened in the first year, and 7.5 per 10,000 in the second year. For 2000–2007, the programme comfortably met the minimum standard. The rate was slightly above the “achievable” standard in the first year after screening, however, in the second year after screening episode, the rate was 13.4 per 10,000 - above the minimum standard of 12.5.
The interval cancer rate in Ireland is in the mid-range compared with rates published for a number of European screening programmes, although there is little information on interval cancer rates covering recent periods (table 4 and18–20,24), and some of the published European data precede the routine use of double view mammography, which is standard in the Irish programme.
Interval cancers include those potentially detectable at the time of screening, and true negatives (including occult cancers). The incidence of true interval cancers depends on the background incidence of cancer and on the proportion of rapidly growing tumours in this background incidence. A high background incidence of breast cancer will lead to a higher interval cancer rate. The estimated background incidence in Ireland (based on incidence during 1995–1999) was relatively high, second only to one of the other European programmes listed, and may have been an under-estimate of the true background incidence during 2000–2007 if the upward trend in incidence rates observed prior to 2001 24 was maintained. However, the incidence of interval cancers relative to the expected rate (proportionate) was comparable with the other regions and countries described here, as was the interval cancer ratio. The calculation of background incidence outside a trial situation is difficult. In Ireland, due to the pilot programme, 10 the entire population was not screening naïve and thus background rates may have been slightly lower than presented. However, the upwards trend in incidence rates in the period 1994–1997 was not carried forward, so these may be underestimated. The interval cancer ratio is a useful alternative measure for regions that have mature screening programmes, unstable incidence rates in the period prior to the programme, or where there is no population based cancer registry to supply such information.
Detection through mammography carried out privately, between BreastCheck rounds, may have inflated the number of cancers classified as intervals. We have indirect evidence of private mammography via the number of in-situ interval cancers found in this group.
Interval rate estimation depends on complete case ascertainment and successful case linkage to the screening register. The published registry data cited here was from areas covered by population-based cancer registries, linked to screening registers. Incomplete linkage of registered cancers to the screening register will result in under-estimation of interval cancer rates, while incomplete linkage to screen-detected cancers will lead to overestimation. In Ireland, almost all screen-detected breast cancers reported to the Registry by BreastCheck were successfully linked to registrations, using probabilistic matching software and identifiers including name, address, date of birth, and hospital medical record number. Only two cancers detected by BreastCheck could not be linked to a National Cancer Registry registration.
In breast cancer screening programmes choices must be made between high sensitivity, which will reduce the number of interval cancers but increase false positive rates, or high specificity, which will reduce recall rates at the expense of increasing the number of interval cancers. While avoiding interval cancers must be a priority, false positive results cause distress to the women involved and may reduce future participation rates; 25 for a programme to be effective, participants need to be retained and attend for multiple screens. Digital mammography, which may have better performance characteristics than conventional mammography,26,27 was introduced by the Irish programme in 2007, and this could reduce interval cancer rates.
Conclusion
The interval cancer rates over the first eight years of BreastCheck were centred in the range reported from other population-based screening programmes in Europe. Although the programme achieved the minimum standard it had set itself in the first 12 months after screening, it did not attain the “achievable” standard, especially in the second year after screening. It is important for all population-based screening programmes to undergo periodic external evaluation, including estimation of interval cancer rates. The challenges inherent in maintaining quality during nationwide programme roll-out mean that assessing interval cancer rates will remain important for the Irish programme in coming years.
Footnotes
Acknowledgements
We thank staff at BreastCheck for providing data and for their assistance during this project, and the Tumour Registration Officers of the National Cancer Registry.
The Authors have no conflicting interests to declare.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
