Abstract
Objective
To evaluate performance of the first round of HPV-based screening in Tuscany region and compare it with the prior round of Pap-based screening
Setting
Tuscany region of Italy, where HPV-based cervical cancer screening started in 2013, with a strong level of centralization screening tests at the Regional Laboratory for Cancer Prevention (ISPRO).
Methods
The transition from Pap- to HPV-based screening was initiated for older women and at 3 out of 12 Tuscany Local Health Units (LHUs). Data from the Florence and Grosseto LHUs (about 300,000 women) were analysed and performance screening indicators estimated.
Results
HPV-based indicators recorded good performance, with increased compliance vs. the Pap-based programme. We registered a substantial decrease in waiting times from sampling to test reporting, probably related to the centralization strategy. Since the screening protocol was the same and conducted at a single laboratory, we could hypothesize that the difference in HPV positivity (6.8% in Florence vs. 8.4% in Grosseto) was due to a real difference in HPV prevalence among women of the two LHUs. The transition to HPV-based screening led to a significant increase both in colposcopy referral rate (4.3% vs. 1.2%) and CIN2+ detection rate (8.3‰ vs. 3.4‰).
Conclusions
HPV-based is more effective in detecting high-grade precancerous and cancerous lesions than Pap-based screening and is characterized by an “anticipatory effect” in the detection of CIN2+ lesions. The transition from Pap-based to HPV-based screening programme should include increased resources dedicated to colposcopy services. Centralization in a laboratory with long experience in this field promotes efficiency of the screening process.
Introduction
According to several international agencies, there is clear scientific evidence that HPV-based cervical cancer screening, performed with clinically validated DNA tests for oncogenic HPV types and with an appropriate protocol that includes cytological triage, is more effective than Pap-based screening in preventing invasive cervical cancer.1–6 Primary HPV screening also allows screening intervals to be extended safely, because of its enhanced sensitivity 7 , and is more cost-effective than Pap-based screening.8–10
The introduction of a test with higher sensibility (HPV test) required well-structured protocols in order to guarantee acceptable levels of specificity. According to the European Guidelines, 11 only clinically validated HPV tests that detect high-risk HPV types (HR-HPV) should be used for screening purposes. 12 For HPV-positive women it is suggested to perform a Pap test as “reflex test” (cytology triage), in order to increase the specificity of the new algorithm with HPV as primary test. Only HR-HPV-positive women showing abnormal cytology undergo colposcopy examination, while those with normal cytology repeat the HPV test after one year (one-year recall).
In Italy, cervical cancer screening with primary HPV test is gradually replacing primary screening with Pap test. The National Health Plan 2014–2018 required all regions to introduce HPV-based screening in women over 30–35 and up to 64 years old by 2019. In 2013, Tuscany was the first region in Italy to introduce HPV as primary screening test for women aged 34–64 years.
The transition from Pap test to HPV test in a screening programme is a very complex task, and needs to be supported by well-defined protocols and guidelines.11,13,14 Organized population screening programmes should be regularly monitored to evaluate effectiveness and to identify potential harms, in order to ensure a high level of quality.15,16 To define every phase of the transition process, the Tuscany Regional Government, in 2012, set up a timetable for all the required steps in two official documents (DGRT 1049/2012 and 1235/2012). Furthermore, it was decided to centralize testing in the Regional Laboratory for Cancer Prevention within the Institute for cancer prevention, research and oncological network (ISPRO, Florence).
An information campaign about the new screening programme was launched to ensure high levels of compliance among women eligible for screening. A monitoring system of quality assurance for each phase of the programme was implemented.17,18 Since 2016, all Local Health Units (LHUs) in Tuscany have been gradually included in the new programme so that, by 2020, it was fully implemented in the whole region. The sole laboratory has a catchment area each year of approximately 850,000 women aged 34–64 years.
The aims of this study were: i) to evaluate the performance of the first round of HPV-based screening implemented in two LHUs in Tuscany (Florence and Grosseto) in 2013–2017, and ii) to compare results of the HPV-test programme with those recorded in the previous 2010–2012 Pap-test programme.
Methods
Switch from Pap to HPV-based screening: The “tuscany model” of organization
The organized cytology based screening programme in the Tuscany region started in the 1980s and, with the last update, consisted of an active invitation to women aged 25–64 years to undergo a Pap test every 3 years. The transition from Pap-based to HPV-based screening started in 2013 in three LHUs (Grosseto, Florence and Viareggio) and for older women (age range 55–64 years), in order to avoid a sudden increase in colposcopy service workloads, while younger women (aged 45–54 and 34–44 years) were invited, respectively, in the second and third year (Table S1). The fourth and fifth years were dedicated to re-calling all non-respondent women and inviting newly eligible resident women, in order to increase compliance to the screening programme.
As a consequence of this transition, there was a marked decrease in cytology workload and a progressive increase in number of HPV tests. Women were invited to the first screening round with HPV test after 3 years from the last normal Pap test, in order to avoid an immediate imbalance in the workloads of health professionals.
Cervical cancer screening information materials were updated and sent by regular mail, together with the letter of invitation, in order to introduce the eligible women to the new screening campaign and point out the new inter-screening interval of 5 years. In each area, a widespread network of dedicated sampling centres was organized and the new informational material was exhibited in their waiting rooms.
Complete integration of information systems between the central laboratory and the screening centres allowed full traceability of all phases, from the invitation letter to the production of the laboratory report. The first HPV-based screening round was completed, for the three LHUs, in 2017 and the one-year recall in 2018.
The cervical cancer screening protocol
The current HPV screening protocol is shown in Figure 1. Eligible women received a letter of invitation with an appointment date (that could be changed by phone or e-mail) for sampling, to be performed locally by a midwife or gynaecologist who first explained the new procedure. All cervical samples were sent to the regional laboratory at ISPRO and the Pap reflex test (cytology triage) was prepared and read for all HPV-positive women, who were immediately referred for a colposcopy if the HPV test was positive and cytology triage showed any grade of abnormality (ASC-US+) or was inadequate. Women who were HPV positive but with normal cytology triage were actively re-invited after one year to repeat the HPV test Women with persistent HPV infection at one-year recall were referred for a colposcopy, while HPV-negative women (cleared infection) were invited to the next screening round after 4 years (5 years from the baseline screening test). HPV-negative and HPV-positive/triage-negative women were informed of the results by a letter sent by regular mail. Women with HPV+/Pap test triage ≥ ASC-US or inadequate were invited by phone to attend for a colposcopy in their local reference screening centre.
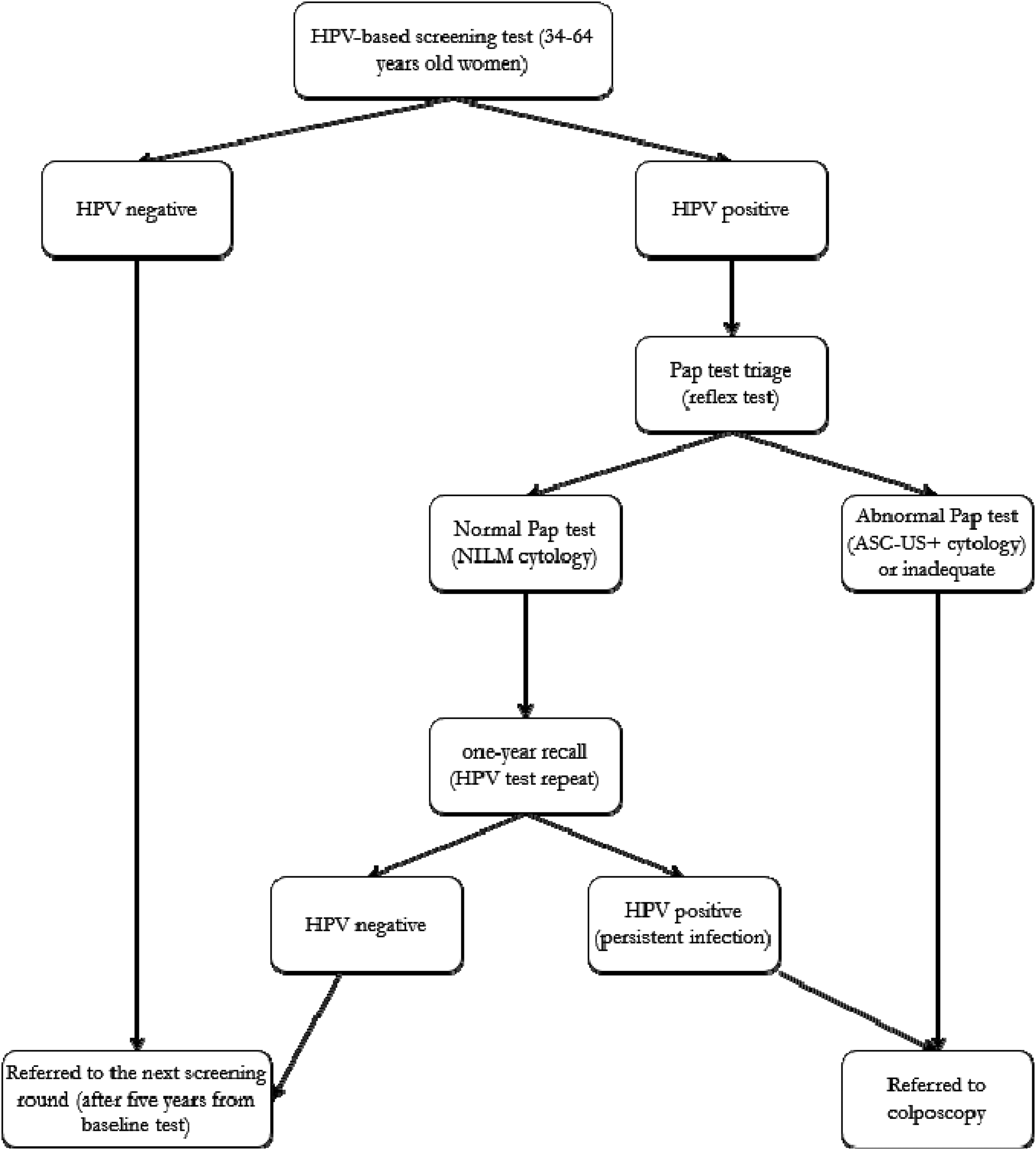
HPV primary screening protocol in tuscany region. NILM: Negative for intraepithelial lesion malignancy; ASC-US: Atypical squamous cells of undetermined significance.
Sample collection
Several training courses for midwives and gynaecologists were organized for the whole region. In a first phase, double sampling was performed: cells were first collected by spatula and cytobrush for conventional Pap-test preparation and, subsequently, by the cervical sampler brush specific for HPV testing (STM; Specimen Transport Medium, Qiagen, Hilden, Germany). Only the conventional slides from HPV-positive women were stained using Papanicolaou procedures and read by cytologists. In a second phase (Grosseto from June 2014 and Florence from 2016), cervical samples were collected in ThinPrep vials containing PreservCyt solution (Hologic Corp., Marlborough, MA, USA), which allows to execute both liquid-based cytology and HPV test
Laboratory procedures and quality control
All screening tests (HPV and Pap) were performed in the ISPRO Regional Cancer Prevention Laboratory. From January 2013 to May 2016, HPV-DNA testing was performed using the Hybrid Capture® 2 (HC2) HR-HPV test (Qiagen). Subsequently, the cobas® 4800 HPV test (Roche Diagnostics, Pleasanton, CA, USA) was used. Both tests are clinically validated and have been approved for screening purposes also by the Italian Group for Cervical Cancer Screening (GISCi) and National Observatory of Screening (ONS). 19
HC2 HR-HPV test
The HC2 test is able to identify 12 high-risk HPV types (16,18,31,33,35,39,45,51,52,56,58 and 59) and HPV68, which is classified by IARC 20 as probably carcinogenic (group 2A). The test was performed according to the manufacturer's instructions for samples collected in STM or Thin Prep solution, in the latter case using the QIAsymphony instrument which performs an automated DNA extraction. Samples with a relative light unit/positive control (RLU/PC) ratio ≥ 1 were considered positive.
Cobas 4800 HPV test
cobas 4800 is a real-time PCR-based assay able to detect the 13 HPV types identified by HC2 with the addition of HPV66, which is classified as possible oncogenic type by IARC (group 2B), and has as internal control gene the Beta globin (BG). According to the method's algorithm, a test was considered: “HPV Positive” if Ct value for HPV16 was ≤ 40.5 and/or Ct value for HPV18 and/or the 12 “other” genotypes was ≤ 40.0; “HPV Negative” if HPV DNA was not detected and the internal control had a Ct value ≤ 40; “Invalid” if HPV was not detected and the internal control had a Ct value > 40; or “Failed” if errors occurred in clot detection or sample processing. Invalid and Failed samples were repeated two additional times and, if the results were confirmed, the woman was invited to repeat the cervical sampling.
Cytology triage
The conventional Pap test slides were prepared manually by midwives or gynaecologists, fixed by spray, and then placed in a slide container for transport to the laboratory. Liquid-based cytology (LBC) slides were prepared (only for HPV-positive samples) with an automated platform (ThinPrep 5000 with Autoloader, Hologic). Papanicolaou staining was done using an automated platform (Leica Multistainer, Leica Biosystems Nussloch GmbH).
Cytology results (primary and triage) were reported according to the Bethesda System for reporting cervical cytology (TBS 2001 until 2015, then TBS 2014 was used). All cytologists were aware to reduce as much as possible the ASC-US category according to the indications of the GISCi. 21 Cytologists were not blinded to the HPV screening test results.
Quality control
The Regional Cancer Prevention Laboratory performed daily internal quality controls both for HPV test and Pap test, according to GISCi recommendations,21,22 and took part in a national external quality assurance programme specific for HPV Screening and Pap Test Triage (VEQ HR-HPV screening and VEQ Pap screening, Regional Reference centre of Quality assessment of Careggi Hospital). Moreover, for HPV testing, the laboratory took part also in the UK-NEQAS, QCMD and Dicocare external quality assurance programmes (EQA). Results of EQA programmes were always within the expected range.
Clinical management
Colposcopy was performed locally in each LHU, according to GISCi national guidelines and to the regional protocol.23,24 Colposcopy-directed biopsies were read by pathologists of Azienda Ospedaliera Universitaria Careggi (AOU Careggi, Florence) and Grosseto Hospital, respectively. Histological results were classified as negative, cervical intraepithelial neoplasia grades 1–3 (CIN1, CIN2, CIN3), in situ adenocarcinoma (AIS) or invasive adenocarcinoma, or squamous cell carcinoma according to WHO classification.
Women with a diagnosis of CIN or more severe lesions (CIN2+) were referred for treatment. Women with a negative colposcopy and/or biopsy result of ≤ CIN1 were referred for a specific follow-up protocol depending on the cytology triage result, as described in the GISCI guidelines. 23
Data retrieval and statistical analysis
Aggregated data from the Grosseto and Florence screening programmes were retrieved from the annual standardized surveys that the ONS administered to managers of each Italian LHU screening programme, for the monitoring and evaluation of screening programmes. Quality controls of survey data were carried out at the regional level. Surveys from 2013 to 2017 were used for collecting data from the HPV-based screening (women aged 34–64); surveys from 2010 to 2012 for the Pap-based screening (women aged 25–64). Since annual surveys recorded aggregated data by five-year age group (from 25–29, 30–34 to 60–64 years for Pap-based screening; from 34 alone, 35–39 to 60–64 for the HPV-based screening), only aggregated data of women aged from 30–34 to 60–64 years were selected from the Pap-based screening surveys for comparison with the HPV-based screening.
The following performance indicators both at baseline and at the one-year recall in women who were HPV+/Pap-, stratified by age group, 17 were estimated: compliance to invitation; positivity at HPV test; distribution of Pap results by diagnostic category; referral rate to colposcopy; positive predictive value (PPV) for CIN2+ at colposcopy (proportion of colposcopies with histologically confirmed CIN2+); detection rate (DR) for CIN2+ (proportion of women with histologically confirmed CIN2+). Moreover, the proportion of Pap tests reported within 30 days in the last year of the Pap programme (2012) and of HPV tests reported within 30 days in 2016 (the first year with available waiting times for the HPV programme), in addition to the proportion of women undergoing colposcopy within 8 weeks of a positive baseline test result, were calculated. In the case of HPV positivity, waiting time within 8 weeks of a positive result included the time needed for cytological triage.
Results of the 2013–2017 HPV programmes in the Florence and Grosseto LHUs were compared with those of the previous 2010–2012 Pap test programmes by calculating ratios of the main indicators, with 95% confidence intervals (CI). Similar ratios were calculated to study differences between the two HPV programmes.
Results
Participation
From January 2013 to December 2017, 218,483 and 57,321 women aged 34–64 years in Florence and Grosseto LHUs, respectively, were invited to the first screening round with HPV test (Table 1, Figures 2a and b). The overall participation was 56.7% and 53.2% for Florence and Grosseto, respectively. Compliance gradually increased from 54.0% among women aged 34 years in Florence (39.0% in Grosseto) to 62.0% among women aged 40–44 in Florence (56% in Grosseto) and then gradually decreased to 50.0% among women aged 60–64 in both areas (Figure 3).
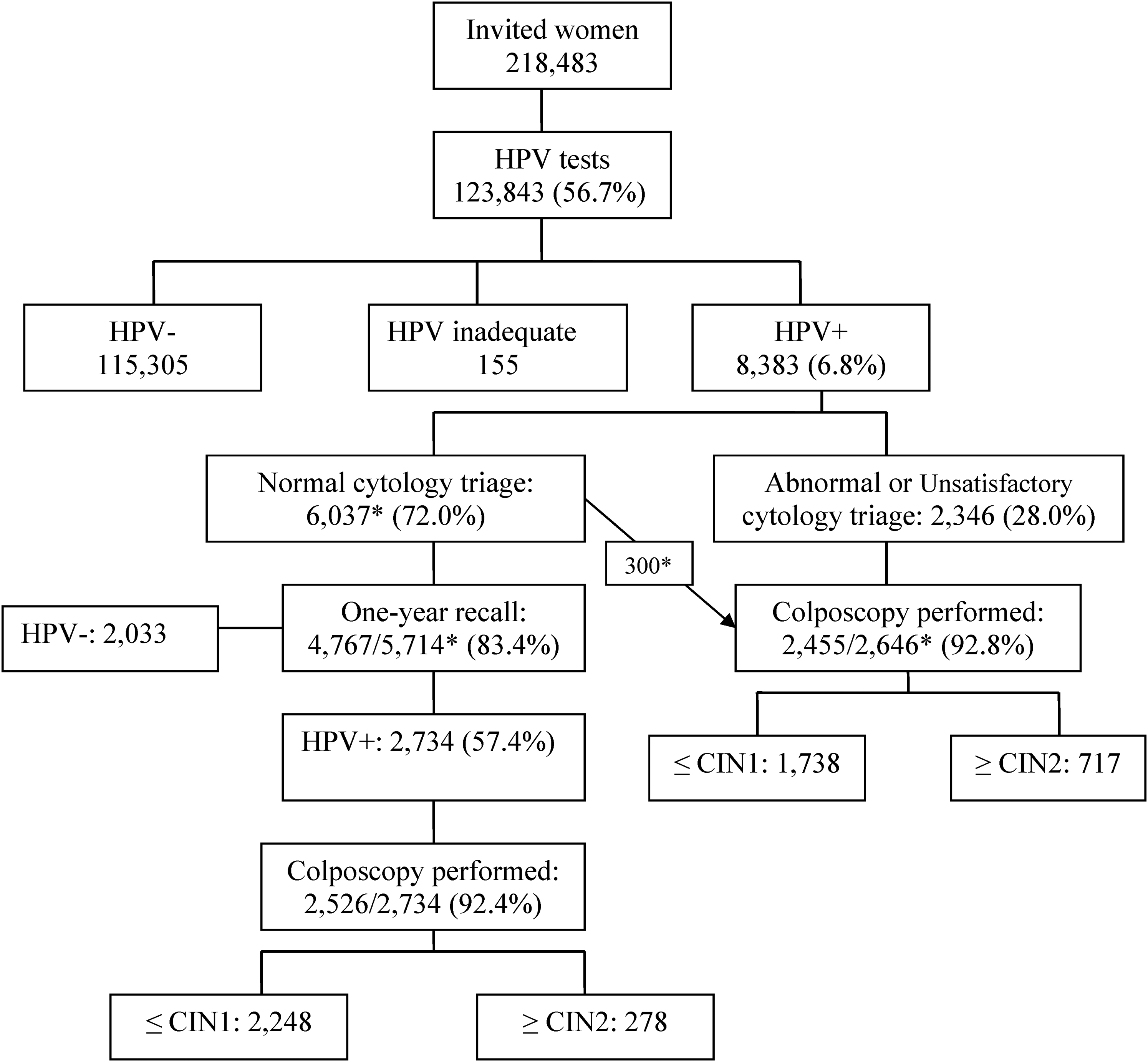
Flow chart and main data of the HPV test-based screening programme, Florence local health unit, 2013–2017. * Among 6037 women HPV+/cytology- at baseline, 300 were recruited to the active arm of the NTCC2 trial and underwent colposcopy, according to the study protocol. Among one-year recalls, 23 women were excluded as in active follow-up.
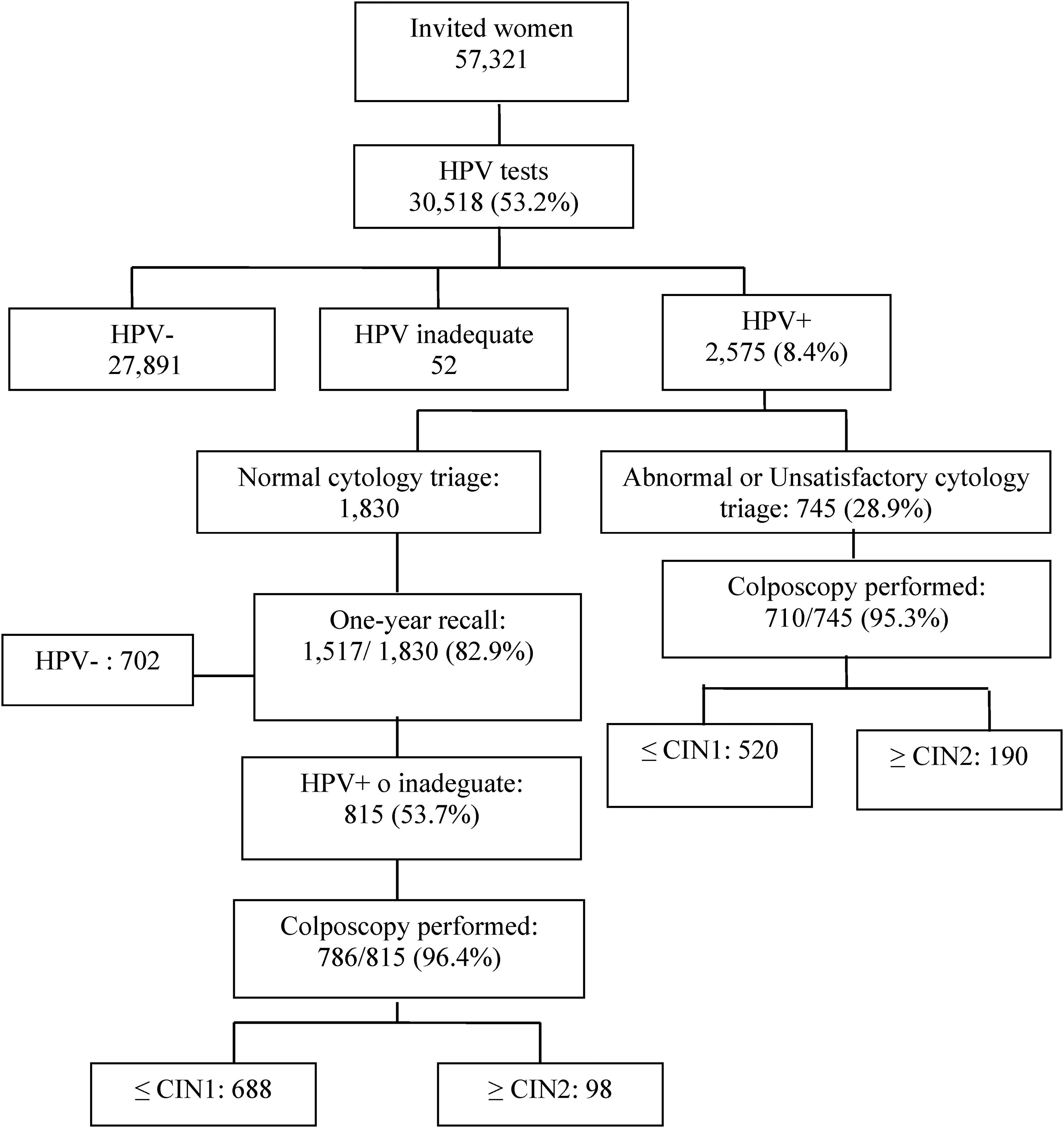
Flow chart and main data of the HPV test-based screening programme, grosseto local health unit, 2013–2017.
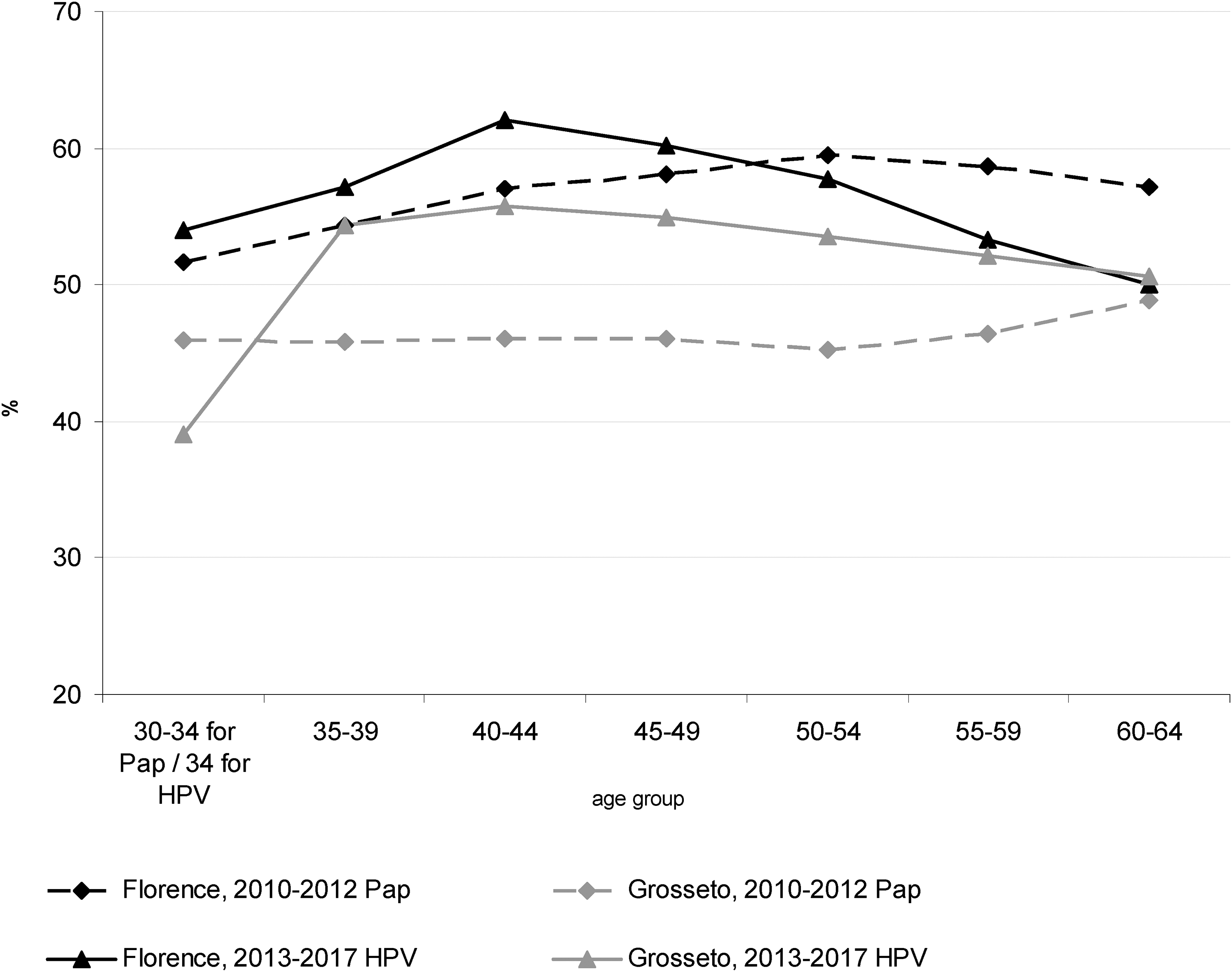
Participation in the 2013-2017 first round of HPV-based cervical screening and in the 2010-2012 Pap-based screening by age group, florence and grosseto.
Results of the HPV test-based screening programmes at baseline (2013–2017) and at the one-year recall among women HPV+/cytology- at baseline.
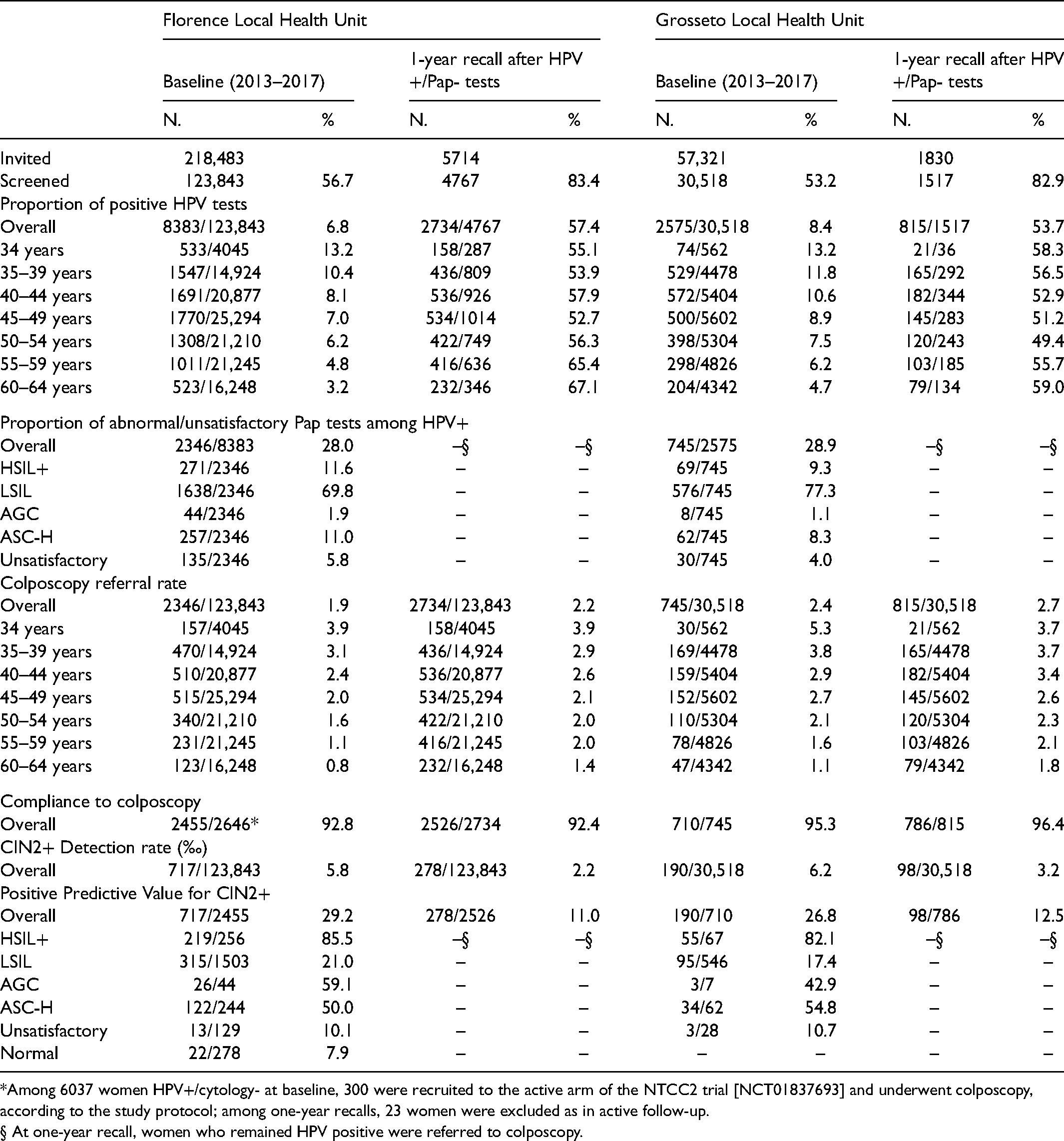
*Among 6037 women HPV+/cytology- at baseline, 300 were recruited to the active arm of the NTCC2 trial [NCT01837693] and underwent colposcopy, according to the study protocol; among one-year recalls, 23 women were excluded as in active follow-up.
§ At one-year recall, women who remained HPV positive were referred to colposcopy.
HPV test results at baseline
At baseline, the overall HPV positivity rate was 6.8% in Florence and 8.4% in Grosseto (Table 1; Figures 2a and b) and was strictly related to age: it decreased gradually from 13.2% among women aged 34 years in both areas to 3.2% in Florence and 4.7% in Grosseto among women aged 60–64 years (Figure S1).
Cytology triage
Among HPV+ women, 2346 women (28.0%) in Florence and 745 (28.9%) in Grosseto had an abnormal or unsatisfactory cytology triage result and were therefore referred for colposcopy. Most cytology examinations were low-grade squamous intra-epithelial lesions (LSIL; 69.8% in Florence, 77.3% in Grosseto), followed by high-grade squamous intraepithelial lesions or more severe (HSIL+: 11.6% in Florence; 9.3% in Grosseto), atypical squamous cells, high-grade not excluded (ASC-H: 11.0% in Florence; 8.3% in Grosseto) and atypical glandular cells (AGC: 1.9% in Florence; 1.1% in Grosseto; Table 1). Unsatisfactory cytology examinations (proportion of inadequate Pap test results) were very few in both programmes: 1.6% in Florence and 1.2% in Grosseto (Figure S2).
Colposcopy and histology results at baseline
Among screened women, 1.9% in Florence and 2.4% in Grosseto were referred immediately to colposcopy at baseline. Colposcopy referral rate at baseline decreased by age: from 3.9% in Florence and 5.3% in Grosseto among 34-year-old women to 0.8% in Florence and 1.1% in Grosseto among those aged 60–64 (Figure S3). Compliance to colposcopy was high in both programmes: 92.8% in Florence and 95.3% in Grosseto (Table 1). CIN2+ DR at baseline was 5.8‰ in Florence and 6.2‰ in Grosseto. The Positive Predictive Value (PPV) for CIN2+ lesions at baseline was 29.2% in Florence, from 85.5% for HSIL+ cytology examinations to 10.1% for unsatisfactory Pap tests. Similar results were recorded in Grosseto: the PPV for CIN2+ was 26.8%, from 82.1% for HSIL+ to 10.7% for unsatisfactory Pap tests.
Screening indicators of HPV-based screening programme at one-year recall
Attendance for the one-year recall protocol was 83.4% in Florence and 82.9% in Grosseto and the persistence of HPV infection was, respectively, 57.4% and 53.7%. The HPV positivity was higher among older women (aged ≥ 55 years; Table 1, Figure S1). The colposcopy referral rate at one-year recall was 2.2% in Florence and 2.7% in Grosseto, and decreased by age: from 3.9% and 3.7% among 34-year-old women to 1.4% and 1.8% among those aged 60–64 (Figure S3). Compliance to colposcopy was high in both programmes (92.4% in Florence; 96.4% in Grosseto). At one-year recall, CIN2+ DR was 2.2‰ in Florence and 3.2‰ in Grosseto. The PPV for CIN2+ in the HPV-persistent women was 11.0% in Florence and 12.5% in Grosseto.
Overall screening indicators
Cumulative colposcopy referral rates (baseline+ one-year recall) for Florence and Grosseto were, respectively, 4.1% and 5.1%. Overall DR for CIN2+ was 8.0‰ in Florence and 9.4‰ in Grosseto, and it decreased with age: from 18.3‰ in Florence and 32.0‰ in Grosseto among women aged 34 years to 3.6‰ in Florence and 4.6‰ in Grosseto among those aged 60–64 (Figure S4). The overall PPV for CIN2+ was 20.0% in Florence and 19.3% in Grosseto (Table 2). Considering both programmes, 38 invasive carcinomas (13 invasive adenocarcinomas, 12 full invasive squamous carcinomas, 13 microinvasive squamous carcinomas), 5 AIS, 676 CIN3 and 564 CIN2 were diagnosed at baseline and at one-year recall.
Comparison of Pap tests and HPV tests in organized screening programmes in florence and grosseto local health units, and comparison between florence and grosseto HPV screening programmes, 2013–2017.
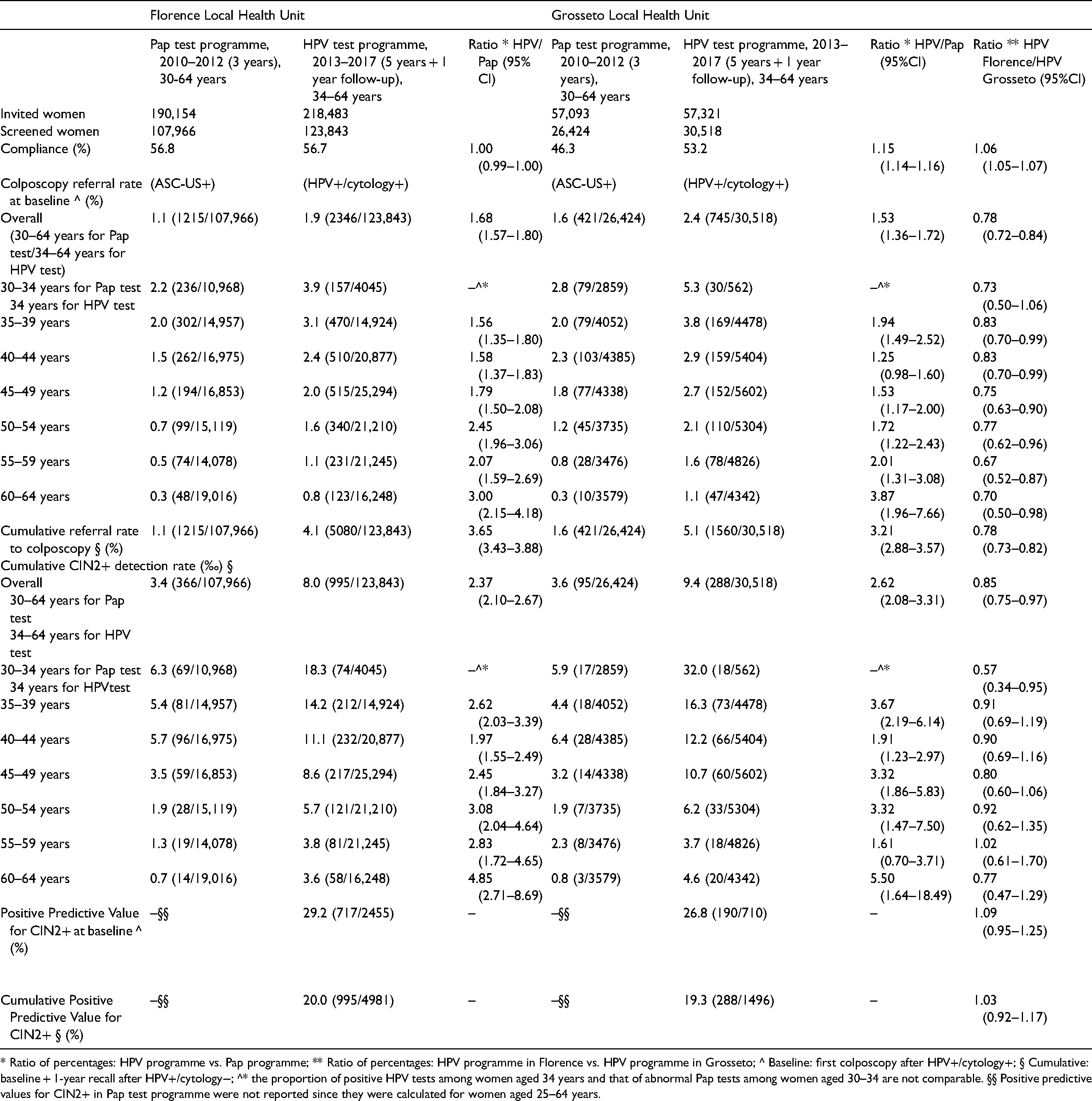
* Ratio of percentages: HPV programme vs. Pap programme; ** Ratio of percentages: HPV programme in Florence vs. HPV programme in Grosseto; ^ Baseline: first colposcopy after HPV+/cytology+; § Cumulative: baseline + 1-year recall after HPV+/cytology−; ^* the proportion of positive HPV tests among women aged 34 years and that of abnormal Pap tests among women aged 30–34 are not comparable. §§ Positive predictive values for CIN2+ in Pap test programme were not reported since they were calculated for women aged 25–64 years.
Comparison between main indicators of the first round of the HPV-based programme with the last round of the Pap-based programme (2010–2012)
Compliance in Grosseto significantly increased from 46.3% in the Pap-based to 53.2% in the HPV-based programme, whereas in Florence a compliance of about 57% in both programmes was recorded (Table 2; Figure 3). Colposcopy referral rates at baseline increased by more than 50% in both areas, especially in women aged ≥ 50 years old, with a doubling or tripling of referral rates at baseline. Moreover, cumulative referral rates (i.e. at baseline and at one-year recall) more than tripled in both areas (Florence: 1.1% in Pap-based vs. 4.1% in HPV-based programme; ratio: 3.65; 95%CI: 3.43–3.88; Grosseto: 1.6% in Pap-based vs. 5.1% in HPV-based programme; ratio: 3.21; 95%CI: 2.88–3.57). Finally, cumulative DR for CIN2+ more than doubled after the transition from Pap- to HPV-based programme (respectively, from 3.4‰ to 8.0‰ in Florence and from 3.6‰ to 9.4‰ in Grosseto), with important results registered especially in older women (Table 2). Considering both the Florence and Grosseto LHU programmes (Figure S4), the DR for CIN2 increased from 0.78‰ in Pap-based to 3.65‰ in the HPV-based programme (ratio: 4.68; 95%CI: 3.80–5.76); the DR for CIN3 increased from 2.40‰ to 4.41‰ (ratio: 1.84; 95%CI: 1.61–2.10) and the DR for AIS and invasive adenocarcinoma increased from 0.05‰ to 0.08‰ (ratio: 1.89; 95%CI: 0.72–4.96), whereas the DR for squamous carcinoma remained approximately at the same level (Pap-test: 0.21‰; HPV: 0.16‰; Table S2).
Waiting time for HPV- and Pap-based screening
The proportion of Pap tests reported within 30 days (time between sampling and the reporting date of laboratory tests) was, in 2012, around 35% in both LHUs. This improved to 100% for HPV tests in the new programme, in 2016. The proportion of women undergoing colposcopy within 8 weeks of a positive screening result from the baseline test changed from 81% in Florence and 64% in Grosseto with the Pap-based programme to 55% in Florence and 94% in Grosseto with the HPV-based programme.
Discussion
In Tuscany, the transition from a Pap-based to a HPV-based screening programme was well-planned according to regional laws that specified, as a crucial point, the centralization of screening tests in a single laboratory where there was great experience in this field. Furthermore, the result of ongoing implementation of the HPV-based programme from 2013, and the model of invitation by age range (starting with older women) and by LHU, was sustainable in terms of workload, especially for second-level healthcare professionals. The data obtained after the end of the first round of screening have been used i) to compare the performances of the two considered HPV-based programmes (Florence LHU vs. Grosseto LHU); and ii) to compare the new HPV-based programme with the previous Pap-based round.
Similarities between the two HPV-based programmes
Several HPV screening indicators of Florence and Grosseto programmes reported very similar results: compliance at baseline and at one-year recall, compliance to colposcopy at baseline and at one-year recall, cytological triage results, PPV at baseline and at one-year recall. CIN2+ DR at baseline was comparable for all age ranges, except for women aged 34 years for whom it was recorded as significantly higher in Grosseto than in Florence (respectively 32.0‰ and 18.3‰; ratio: 0.57; 95%CI: 0.34–0.95), probably due to the low proportion of participating women aged 34 years in Grosseto compared to Florence (1.8% vs. 3.3%).
Similarities between the two HPV-based programmes could be explained with three main reasons: 1) an organized screening programme was already active in these two LHUs, for several years; 2) the transition from Pap test to HPV test was well planned and homogeneously organized in the Tuscany region, applying findings of European randomized controlled trials (RCTs); 3) the laboratory tests (HPV test and cytological triage) were performed at a single central laboratory, with a high grade of specialization and experience in cervical cancer screening. These aspects allowed us to analyze HPV screening indicators starting from a similar organization in all Tuscan LHUs and to interpret any different performance indicators in terms of differences in population features and/or in effectiveness of the local second-level services (colposcopy and histology).
Differences between the two HPV-based programmes
HPV positivity data, colposcopy referral rate and CIN2+ DR are comparable to those recorded in other Italian regions25,26 and those registered in the Italian HPV trial (NTCC: New Technologies in Cervical Cancer). 27 A lower HPV positivity rate at baseline was recorded in the Florence LHU compared to the Grosseto LHU (respectively, 6.8% and 8.4%; ratio: 0.80), especially in women aged ≥ 40 years old (ratios ranged from 0.67 to 0.77). The lower colposcopy referral rate at baseline recorded in Florence compared to Grosseto (respectively, 1.9% and 2.4%; ratio: 0.78), and the lower cumulative referral rate (i.e. at baseline and at one-year recall) in Florence (4.1%) versus Grosseto (5.1%; ratio: 0.78) were exclusively due to a lower HPV positivity rate at baseline in Florence. The lower cumulative DR of CIN2+ registered in Florence (Florence 8.0‰ vs. Grosseto 9.4‰; ratio: 0.83) was due, in part, to the different CIN2+ DR at baseline in women aged 34 years and, largely, to the different DR at one-year recall (respectively, 2.2‰ in Florence and 3.2‰ in Grosseto; ratio: 0.70). Thus, these differences are strongly related to the difference in HPV positivity rate registered at baseline between the two LHUs. Since the screening protocol was the same, there was a single laboratory and a previous Pap-based screening programme was active in both the LHUs (women aged 25–64), we could hypothesize that the difference in HPV positivity at baseline was due to a real difference in HPV prevalence that probably reflects different behaviours between women who live in two different geographical areas, as hypothesized by other colleagues. 28
Pap-based and HPV-based programme comparison
HPV-based screening was well accepted by women; in fact there was a substantial increase in compliance, compared to the previous Pap-based round, especially for the Grosseto LHU (from 46.3% to 53.2%; ratio: 1.15; 95%CI: 1.14–1.16). As demonstrated by the follow-up of the European RCTs (6), our data showed that HPV-based screening is clinically superior and more effective in detecting high-grade precancerous (CIN2+) and cancerous lesions than Pap-based screening. This crucial aspect is confirmed by the two or three-fold increase in overall DR for CIN2+ reported after the transition from Pap- to HPV-based screening in both the Florence and Grosseto LHUs (respectively, from 3.4‰ to 8‰ and from 3.6‰ to 9.4‰). This improved performance in CIN2+ DR was registered for all age ranges, especially in older women (see the ratio “HPV/Pap” in Table 2).
The difference in CIN2+ DR between traditional Pap test and HPV+/triage Pap test registered at baseline (respectively, 3.4‰ vs. 5.8‰ for Florence and 3.6‰ vs. 6.2‰ for Grosseto) was due to better performance of the Pap test as triage test in comparison to its use as primary test. This is probably attributable to the selection of a HPV-positive population, characterized by a greater risk of developing cervical cancer. Furthermore, this increase in CIN2+ DR, as hypothesized by other colleagues in experimental phase, 29 could also be caused by a lower false negative rate that characterizes the cytology triage, due to the greater attention of cytologists in reading Pap test slides from HPV-positive women, and to the use of this knowledge for interpreting cell abnormalities that would otherwise be considered as irrelevant.
The better performance of the HPV-based screening programme resulted in, as a disadvantage, a significant increase in colposcopy referral rate, as registered in other screening programmes in Europe.30,31 Furthermore, increased diagnosis of CIN2+ with high potential for spontaneous regression, especially in younger women, could lead, as reported by other colleagues, 32 to an increased risk of obstetric complications.
It should be stressed also that adenocarcinoma DR increased about two fold (Table S2) with respect to the previous Pap-based screening. Thus, HPV-based screening seems to be more efficient even in detecting adenocarcinomas, although this type of cancer is less preventable than squamous cancer for three principal reasons: 1) shorter pre-clinical phase, 2) more difficult cell sampling, 3) a small proportion of adenocarcinomas (especially the non-usual type) are not HPV-related.
HPV versus Pap-based screening performance indicators: Waiting times
Waiting times as performance indicators changed radically after the transition from Pap-based to HPV-based screening. As expected, we registered a substantial decrease in waiting time, from sampling to test, reported in both LHUs. This was probably strictly related to the high automation of the screening test procedure and also to the centralization strategy, which allowed the use of a high-throughput molecular instrument to assure shorter times for HPV test processing. Furthermore, the centralization strategy and the reorganization of screening services guaranteed a greater professional expertise assuring higher efficiency, to overcome local problems that caused a lengthening of waiting times, such as the fragmentation of Pap-test reading activity in each of the 12 LHUs, some of which had staff shortages.
On the other hand, the waiting times for colposcopy increased in Florence. This was probably due to the increase in referral rate that characterized the HPV-based programme in the first round of screening. In Grosseto, however, the proportion of women undergoing colposcopy within 8 weeks significantly increased, since the expansion of colposcopy services implemented for the introduction of HPV-based screening was sufficient to cope with the increased demand, given the lower number of colposcopies in Grosseto compared to Florence (1560 vs. 5080).
Pap-based programme comparison and possible consequences on the first round of HPV-based screening
Despite the higher proportion of abnormal Pap tests (ASC-US+, corresponding to the colposcopy referral rate) registered in the Grosseto LHU (1.6% vs. 1.1% in Florence LHU; ratio: 0.71; 95%CI: 0.63–0.79), the CIN2+ DR was substantially comparable (respectively 3.4‰ in Florence and 3.6‰ in Grosseto). In the Pap-based programme (2010–2012) the compliance to colposcopy was, respectively, 91.5% for Florence and 83.2% for Grosseto (data not shown). After the transition to HPV-based screening, in the Grosseto LHU there was a substantial increase in compliance to colposcopy (95.3%; IC 95% 93.8–96.8 vs. 79.6–86.8), probably due to a reorganization process that involved this LHU in 2013. So, the similar CIN2+ DRs of Florence and Grosseto, against the difference in number of ASC-US+ lesions, could be attributable to the lower compliance to colposcopy registered Grosseto with the Pap-based programme. Moreover, compliance to the Pap-based programme was significantly lower in Grosseto (56.8% in Florence vs. 46.3% in Grosseto; ratio: 1.23; 95%CI: 1.21–1.24).
These aspects could further explain the difference in HPV positivity and in the CIN2+ DR registered for the two HPV-based programmes at baseline. In fact, in addition to the possible real difference in HPV positivity between women in Grosseto and Florence, it can be said that the increase in compliance in Grosseto from the last round of the Pap-based programme to the first round of the HPV-based programme could have determined that several women who had never performed a Pap test, or did not attend a previous colposcopic examination, had their first screening episode in the first HPV-based round.
Strengths and limitations
This paper has some limitations. First, the study was not based on longitudinal data for participants; instead, it was based on aggregated data retrieved from annual surveys conducted to estimate performance indicators for the monitoring and evaluation of screening programmes in Italian LHUs. Second, we conducted a retrospective comparison of Pap-based screening with HPV-based screening organized during a subsequent time period. Third, the comparison between these screening programmes was not conducted considering exactly the same age groups (Pap-based from 30 to 64 years; HPV-based from 34 to 64 years).
This study has also several strengths. The centralization of laboratory tests and the regional screening and follow-up protocols applied in both the Florence and Grosseto LHUs allowed us to correctly interpret and compare several screening indicators. Moreover, the implementation of a laboratory quality control system allowed monitoring of the whole process of laboratory testing. Finally, the standardized data collection through annual surveys allowed us to retrieve data on 288,751 women attending cervical cancer screening programmes in the two LHUs, from 2011 to 2017.
Conclusion
The transition from Pap-based to HPV-based screening in the Tuscany region, planned in 2012 (taking as crucial starting point the results of European RCTs) and implemented from 2013, before the development of the European guidelines and with decisive regional support, was characterized by the centralization of screening tests at a single laboratory with long experience in this specific field. This feature improved the efficiency and quality of the screening process leading to significantly improved screening performance indicators.
Our results confirmed findings from the European RCTs and other studies in Europe, i.e. that HPV-based screening is more effective in detecting high-grade precancerous and cancerous lesions, with an anticipatory effect on the detection of cervical lesions that with the Pap-based screening would probably have been detected in subsequent screening rounds.
Referral to colposcopy increased significantly in the first round, with an inevitable increase in the number of women involved in follow-up after negative colposcopy for CIN2+ lesions and after treatment of identified lesions. Thus, the decision to move from Pap-based to HPV-based screening should include increased programme resources dedicated to colposcopy services.
It is now clearer that the residual risk of developing a CIN2+ lesion after a negative test in the first HPV-based screening round should be very low. 33 Therefore, the management of women with a positive test in a successive HPV-based round who were negative in the previous round could probably be different. In fact, this population is characterized by a lower risk of developing a CIN2+ lesion because they are women with a recent HPV infection.
Within the next few years, the first cohort of women vaccinated during adolescence will be invited for screening. The widespread coverage of both cervical HPV screening and HPV vaccination has the potential to avert a huge number of cervical cancer cases.34,35 Thus, it is essential to start thinking how the current screening protocols could be modified. The introduction of new triage methods or biomarkers that can support cytology 36 could optimize the follow-up protocols and cost-effectiveness ratio, and reduce the risk of unnecessary colposcopy and treatments.
Supplemental Material
sj-docx-1-msc-10.1177_09691413211067922 - Supplemental material for Implementation of a centralized HPV-based cervical cancer screening programme in Tuscany: First round results and comparison with the foregoing Pap-based screening programme
Supplemental material, sj-docx-1-msc-10.1177_09691413211067922 for Implementation of a centralized HPV-based cervical cancer screening programme in Tuscany: First round results and comparison with the foregoing Pap-based screening programme by Francesca Carozzi, Elena Burroni, Massimo Confortini, Giampaolo Pompeo, Simonetta Bisanzi, Filippo Cellai, Irene Paganini, Paola Mantellini, Anna Iossa, Lisa Lelli, Carmelina Di Pierro, Marzia Matucci, Stefania Cannistrà, Roberta Rosati, Alessandra Mongia, Carmen Beatriz Visioli, Giuseppe Gorini, Cristina Sani and the Tuscany HPV-based Screening Working Group in Journal of Medical Screening
Footnotes
Acknowledgements
All the authors would like to thank all the health professionals that participate to the cervical cancer screening process of Tuscany region, especially the health professionals who work everyday for the Institute for cancer research, prevention and oncological network (ISPRO).
Authors’ Contributions
Carozzi F., Burroni E., Pompeo G., Cellai F. and Mongia A. developed the concept and design of this study. Carozzi F., Burroni E., Pompeo G., Cellai F. and Mongia A. wrote the first draft of the Manuscript. Carozzi F., Cellai F. and Pompeo G. wrote the “Background” and the “Methods” paragraphs of the Manuscript. Clinical data collection, analysis and curation were performed by Gorini G. and Visioli C.B.. Carozzi F. and Gorini G. wrote the “Results” paragraph of the Manuscript. Pompeo G. wrote the “Conclusion” and “Discussion” paragraphs of the Manuscript. All the authors contributed actively to the review and the finalisation of all the paragraphs of the Manuscript and approved the submitted version.
Ethics Statement
Not applicable.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
