Abstract
Objective
To assess the secondary outcome of overall mortality in the randomized Prostate, Lung, Colorectal, and Ovarian cancer screening trial.
Methods
In the Prostate, Lung, Colorectal, and Ovarian trial, subjects were randomized to usual care or intervention. In the intervention arm, men and women received annual chest radiographs and two sigmoidoscopy exams. Men also received annual prostate-specific antigen tests and digital rectal exams, and women also received annual CA125 tests and trans-vaginal ultrasounds. Poisson regression and Cox proportional hazards models were used to assess differences across trial arms in overall mortality and overall mortality excluding deaths from trial cancers (OMEX). Due to slight age imbalances in later trial years, age-adjusted rate ratios and hazard ratios were computed.
Results
There were 76,678 men and 78,209 women randomized, with median follow-up of 17 years. In men there was a significant reduction in both overall mortality (age-adjusted rate ratio = 0.966; 95% CI: 0.943–0.989; p = 0.004) and OMEX (age-adjusted rate ratio = 0.970, 95% CI: 0.946–0.995; p = 0.02) in the intervention versus usual care arm. In women, no reduction was seen in either overall mortality (age-adjusted rate ratio = 1.002) or OMEX (age-adjusted rate ratio = 1.006). In both sexes combined, there was a significant reduction in overall mortality (age-adjusted rate ratio = 0.980; 95% CI: 0.963–0.999; p = 0.036) but not OMEX (age-adjusted rate ratio = 0.985; 95% CI: 0.965–1.004; p = 0.13). Results were similar using age-adjusted hazard ratios.
Conclusion
In the Prostate, Lung, Colorectal, and Ovarian trial, there was a small but significant reduction in overall mortality in men, and in both sexes combined, and a small but significant reduction in overall mortality excluding trial cancer deaths in men.
Introduction
In cancer screening trials the primary endpoint is generally mortality from the cancer of interest,1–3 rather than overall mortality, because such deaths typically represent a very small proportion of all deaths. Under the assumption that screening has little or no effect on deaths from causes other than the cancer of interest, the power of any single trial to find a significant reduction in all-cause mortality is very low. Accordingly, almost all individual cancer screening trials have failed to find a statistically significant reduction in overall mortality (OM), even if they show a statistically significant reduction in cancer-specific mortality. This has led to questions on whether such screening actually saves lives, or simply replaces one type of death with another within the same timeframe. It has also been argued that the lack of an OM reduction could be indicative of mis-attribution bias, where an apparent reduction in cancer-specific mortality is caused by preferentially classifying screening arm deaths from the cancer of interest, or from complications of diagnosis or treatment of that cancer, as due to other causes or vice versa (preferentially classifying control arm non-cancer deaths as due to the cancer of interest). 4 In contrast, an observed significant reduction in OM could not be caused by mis-attribution bias. To assuage such concerns, it is of interest to demonstrate a significant reduction in OM in cancer screening trials, either in individual trials or in meta-analyses of trials.
Uniquely, the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial evaluated screening modalities for multiple cancer types.5,6 Deaths directly related to the trial interventions would be expected to comprise a larger proportion of total deaths than in a single cancer screening trial; however, deaths from all the trial cancers combined (three for each gender) still comprise only around 10–15% of all trial deaths, a relatively low proportion.
Because PLCO was an ongoing trial, previous analyses of OM for each specific cancer being assessed excluded deaths from the other trial cancers.7–10 A recent overview paper of the PLCO trial reported, for the first time, comprehensive all-cause mortality, including deaths from all trial cancers. Based on data from 11 years of median follow-up, there was no difference in the all-cause mortality rate across trial arms. 6 Here we report a more detailed investigation of comprehensive OM based on extended follow-up findings with median follow-up of about 17 years. An important component of OM is mortality from the four trial cancers. For the same period as OM is currently being evaluated (median 17 years), mortality from prostate and colorectal cancer has recently been reported.11,12 We here summarize those findings. Mortality from lung and ovarian cancer has previously been reported only for shorter follow-up periods (median 11 and 14 years, respectively).9,10 Here we report for the first time lung and ovarian cancer mortality for the current follow-up period. It is also of interest to examine all deaths, excluding those from the trial cancers. Ancillary findings from the screening exams, and their downstream sequelae, may have an effect on other causes of death. In addition, the act of participating in trial screenings could alter health behaviours.
Methods
The details of the PLCO trial design and methods have been previously described.5,6 Briefly, the PLCO trial enrolled men and women aged 55–74 between 1993 and 2001 with no history of prostate, lung, colorectal, or ovarian cancer, and not currently undergoing cancer treatment. Participants were recruited at 10 institutions across the United States, and randomized to an intervention or usual-care arm. Institutional review boards at each institution approved the trial, and all participants provided written consent. For men, the screening exams offered were annual prostate-specific antigen (PSA) exams for six years, annual digital rectal exams (DREs) for four years, annual chest radiographs (CXRs) for four years (three years for never smokers), and flexible sigmoidoscopy (FSG) at year 0 and year 5 (year 3 for those randomized prior to April 1995). Women received FSG and CXR at the same schedule as men, and additionally received annual CA125 blood tests for six years, and annual trans-vaginal ultrasound for four years. Diagnostic follow-up of positive screens was carried out by participants’ health care providers. It was not managed by the trial.
From 1993 to 2010, deaths were ascertained primarily through mailed Annual Study Update questionnaires and subsequently obtained death certificates, supplemented by linkages to the National Death Index (NDI). Beginning in 2011, PLCO switched to a centralized follow-up process that utilized linkages to the NDI to assess mortality. A portion of subjects declined to be re-consented (‘refusers’) and opted out of extended follow-up. Non-refusing subjects (about 85% of those alive in 2011) were followed for mortality until the end of 2015. Refusers were followed for mortality until their date of refusal (generally in 2011).
Deaths through 2009 that were potentially related to a trial cancer underwent death review in a blinded (to trial arm) fashion, to determine cause of death. For non-reviewed deaths in that period and subsequent deaths, the underlying cause of death from the death certificate or NDI was used as the cause of death.
Due to the modest difference (see ‘Results’ section) in refusal rates across arms, and the shorter follow-up times of refusers, there was a slight imbalance in age between arms over the later follow-up years of the trial (post-2011). Therefore, we employed Poisson regression and Cox proportional hazards regression models that controlled for age. For Poisson regression, we used indicator variables for five-year age groups of all person-years (PYs) of follow-up to compute the rate ratio (RR) across arms. For the Cox regression, we used time-varying covariates for five-year age groups to compute the hazard ratio (HR) across arms. Additionally, we utilized a Cox model where age was the underlying time variable instead of years on study. All of the age-adjusted models gave similar point estimates (RR or HR) and p-values. We display the results of the age-adjusted Poisson and Cox regressions. Differences in model findings with the adjusted versus unadjusted models are shown in the online Appendix. Kaplan–Meier curves were utilized to plot the cumulative probability of death by trial arm.
Because men and women underwent different screening regimens, we performed the primary analysis separately for each group. A secondary analysis was performed for both sexes combined. We examined both OM as well as OM excluding deaths from the trial cancers (OMEX).
As stated above, prostate and colorectal cancer specific mortality results for this follow-up period were recently published, with unadjusted (for age) RRs reported. These RRs are essentially the same as those computed here using the age-adjusted models. For lung and ovarian cancer deaths, results are reported here using the age-adjusted models.
For context, we compared mortality in the PLCO control arm with that of the general United States population. Specifically, we computed standardized mortality ratios (SMRs) of observed versus expected (times 100) death rates for all deaths and trial cancer deaths. Expected rates were computed using calendar year, age, and sex-specific United States population death rates applied to corresponding PYs of observation in PLCO. 13
Results
Men
Table 1 displays the demographics and smoking history of the 38,340 and 38,338 men randomized to the intervention and usual care arms, respectively. The median (25th/75th) follow-up for mortality was 16.9 (13.9/19.2) and 16.7 (13.4/19.0) years in the intervention and usual care arms, respectively. Among those alive at the time of transition to centralized follow-up (end of 2011), 11.2% of intervention arm and 15.2% of usual care arm men refused further follow-up. Refusers in each arm were slightly older at the time of transition than non-refusers (median age 77 and 76 for intervention and usual care arm refusers, respectively, versus median age 75 in each arm for non-refusers).
Baseline characteristics of PLCO Trial participants.
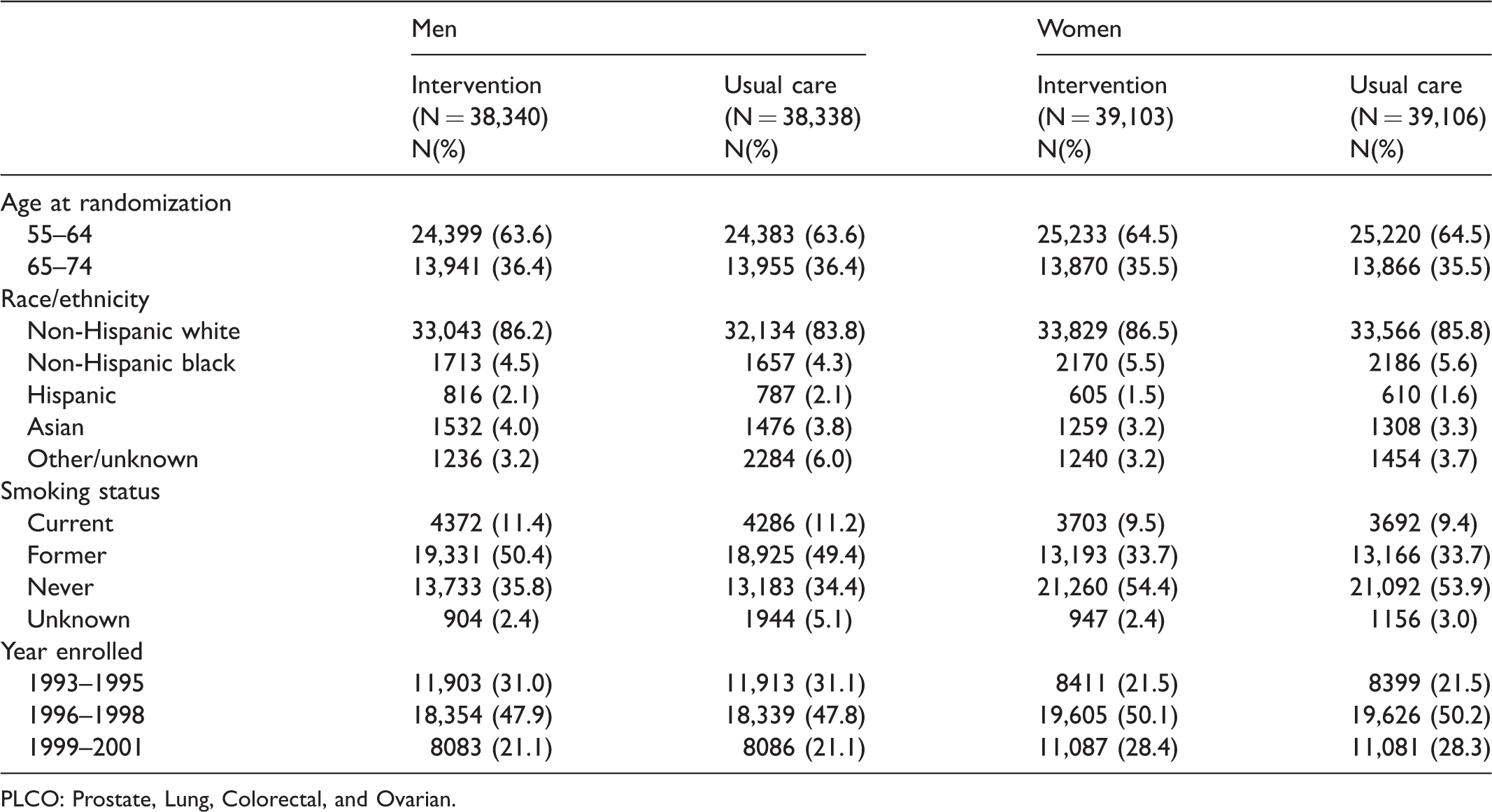
PLCO: Prostate, Lung, Colorectal, and Ovarian.
There were 13,609 and 13,842 deaths in the intervention and usual care arms, respectively, giving rates (per 10,000 PYs) of 226.3 and 233.0 (Table 2). The RR for the age-adjusted Poisson regression models for OM was 0.966 (95% CI: 0.943–0.989; p = 0.004); the Cox regression model (age-adjusted) gave similar results, HR = 0.966 (95% CI: 0.944–0.989; p = 0.004). For OMEX the age-adjusted RR was 0.970 (0.946–0.995; p = 0.02), with a similar estimate for the HR (Table 2). Figure 1(a) shows the cumulative probability of death by arm for OM and OMEX. A slight separation of the curves begins to emerge around year 12.
Overall and cause-specific mortality by trial arm for men, women, and both sexes combined.
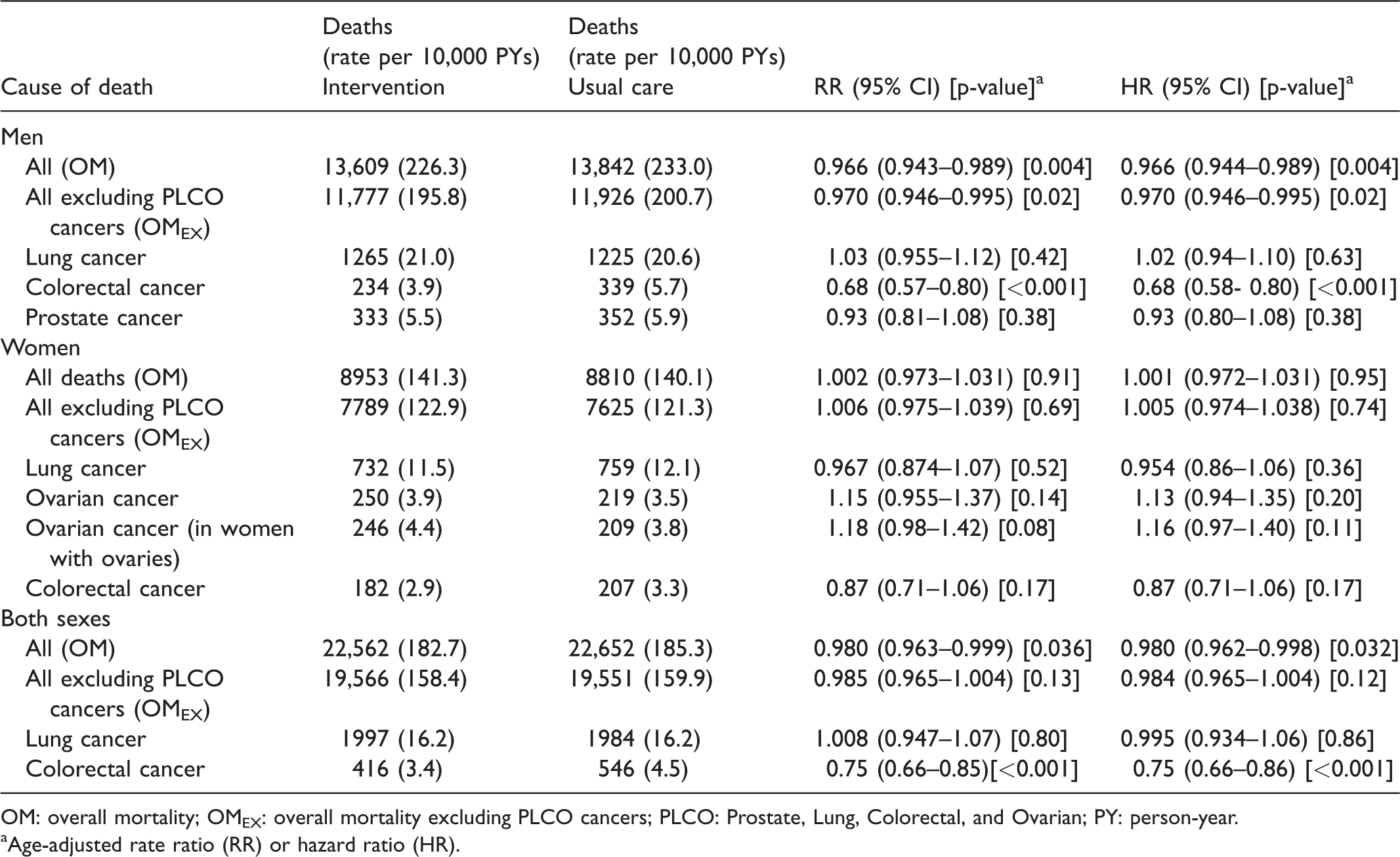
OM: overall mortality; OMEX: overall mortality excluding PLCO cancers; PLCO: Prostate, Lung, Colorectal, and Ovarian; PY: person-year.
Age-adjusted rate ratio (RR) or hazard ratio (HR).
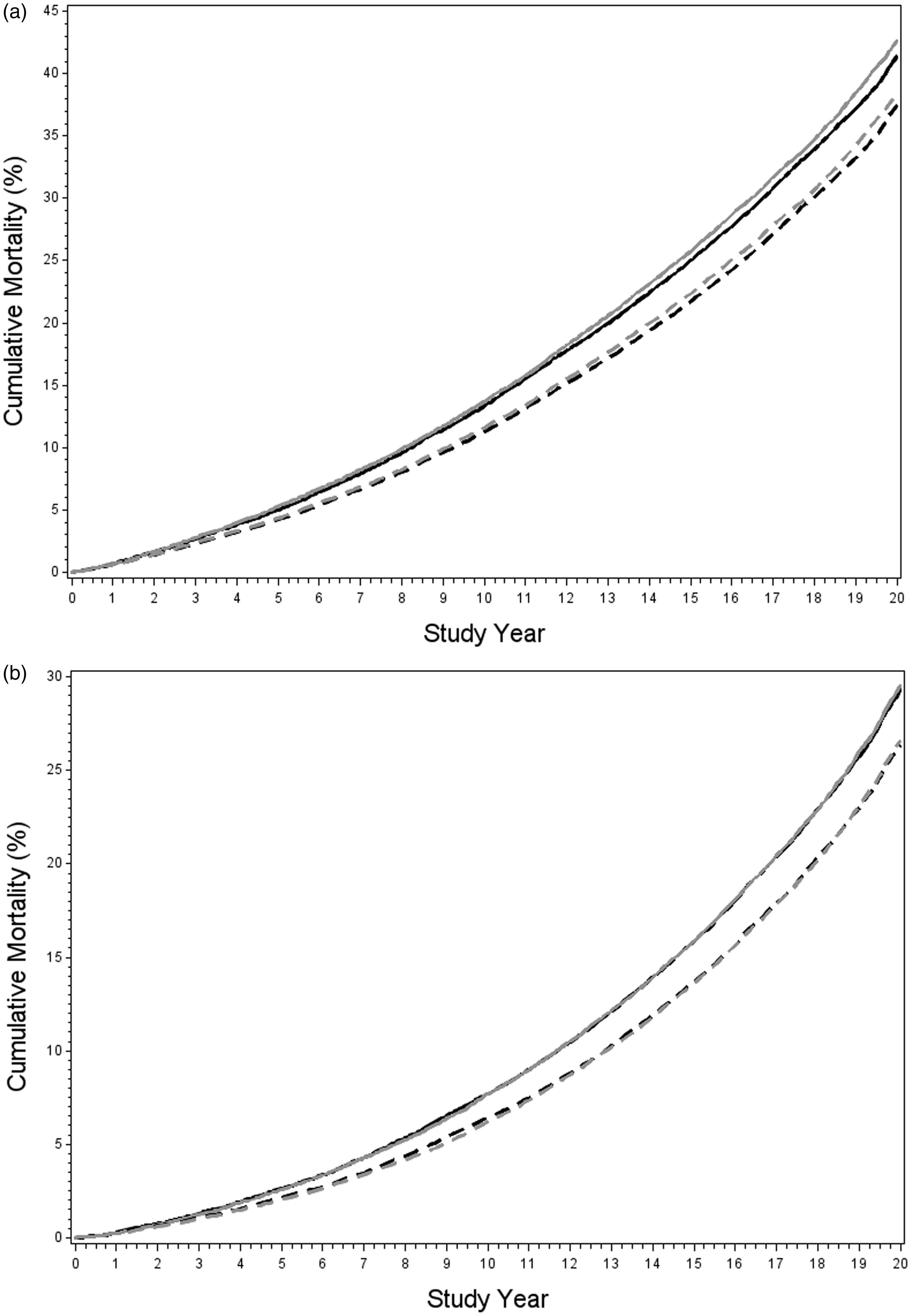
(a) Cumulative OM and OM excluding deaths from PLCO cancers (OMEX) in men by trial arm. Grey is usual care arm, black is intervention arm; solid lines are OM, dashed lines are OMEX and (b) cumulative OM and OM excluding deaths from PLCO cancers (OMEX) in women by trial arm. Grey is usual care arm, black is intervention arm; solid lines are OM, dashed lines are OMEX.
Table 2 also shows mortality from trial cancers. Deaths from lung cancer totalled 1265 in the intervention arm and 1225 in the usual care arm, giving rates (per 10,000 PYs) of 21.0 and 20.6, respectively. The RR from the adjusted Poisson regression was 1.03 (95% CI: 0.955–1.12). The HR was similar. RRs for prostate and colorectal cancer were 0.93 (95% CI: 0.81–1.08) and 0.68 (95% CI: 0.57–0.80), respectively. Causes of death by trial arm are displayed in Table 3. Cancer and cardiovascular diseases each comprised a little less than one-third of all deaths in each arm.
Causes of death by trial arm.
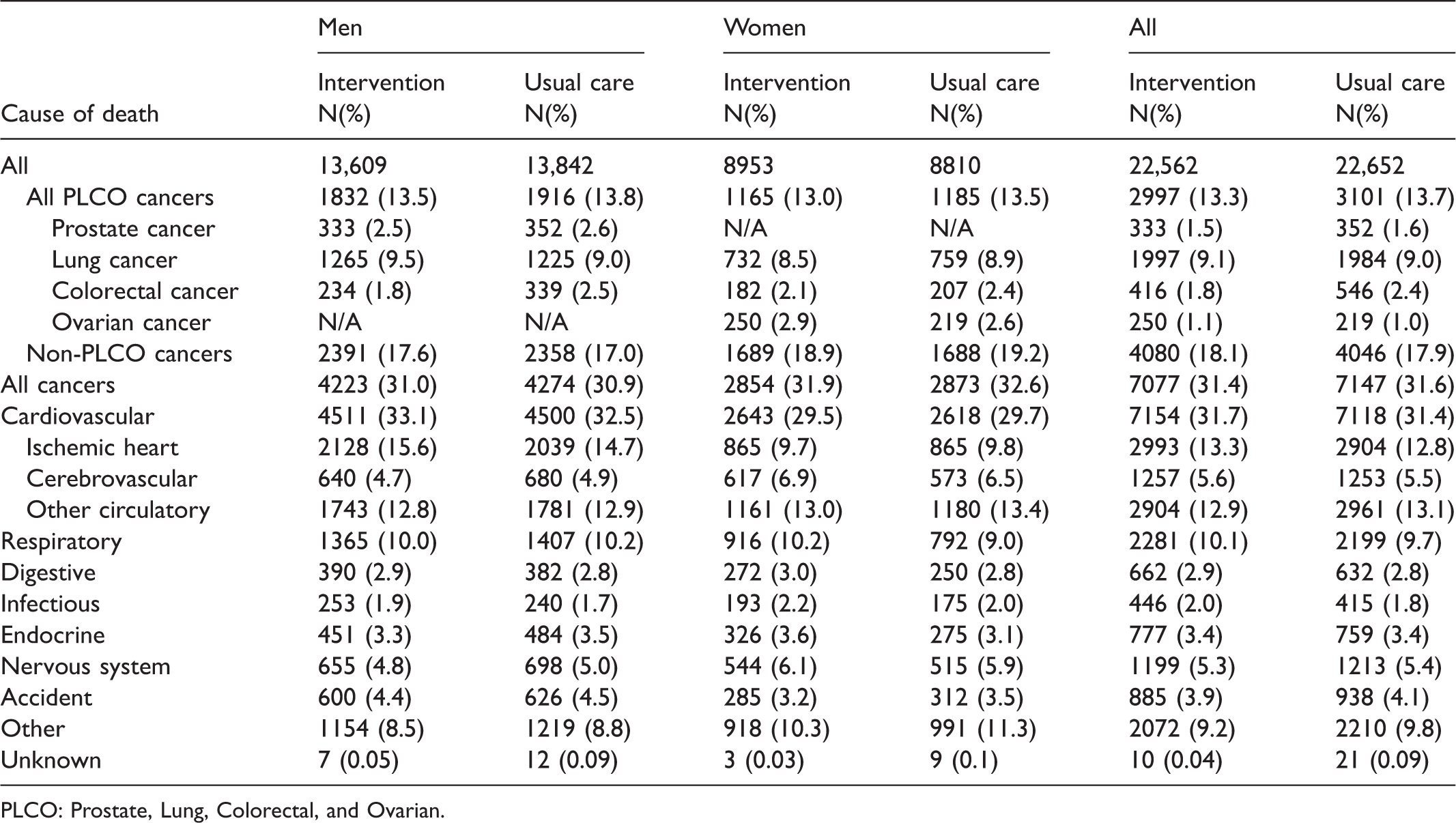
PLCO: Prostate, Lung, Colorectal, and Ovarian.
Women
The demographics and medical history of the 39,103 and 39,106 women randomized to the intervention and usual care arms, respectively, are shown in Table 1. The median (25th/75th) follow-up for mortality was 16.9 (14.9/18.8) and 16.8 (14.6/18.7) years in the intervention and usual care arms, respectively. Among those alive at the time of transition to centralized follow-up (end of 2011), 14.9% of intervention arm and 19.0% of usual care arm women refused further follow-up. Refusers in each arm were slightly older at the time of transition than non-refusers (median age 77 and 76 for intervention and usual care arm refusers, respectively, versus median age 75 in each arm for non-refusers).
There were 8953 and 8810 deaths in the intervention and usual care arms, respectively, giving rates (per 10,000 PYs) of 141.3 and 140.1 (Table 2). The RR for OM in the age-adjusted model was 1.002 (0.973–1.031). The age-adjusted HR was similar. The RR for OMEX was 1.006 (0.975–1.039). The cumulative probability of death for OM and OMEX by arm is displayed in Figure 1(b). The curves are virtually superimposable throughout the entire follow-up period.
Among all women there were 250 and 219 ovarian cancer deaths in the intervention and usual care arms, respectively, giving rates (per 10,000 PYs) of 3.9 and 3.5 and a RR from the adjusted Poisson regression of 1.15 (95% CI: 0.96–1.4). The HR was similar (Table 2). Restricting the analysis to women with at least one ovary at baseline (N = 34,251 and 34,300 for intervention and usual care, respectively), there were 246 (rate = 4.4) and 209 (rate = 3.8) ovarian cancer deaths in the intervention arm usual care arms, respectively; RR = 1.18 (95% CI: 0.98–1.4). Deaths from lung cancer among all women totalled 732 in the intervention versus 759 in the usual care arm (adjusted RR = 0.97; 95% CI: 0.87–1.07). The RR for colorectal cancer was 0.87 (95% CI: 0.71–1.06). Table 3 shows causes of death by trial arm. As with men, cancer and cardiovascular diseases each comprised slightly less than a third of all deaths in each arm.
Both sexes
OM rates (per 10,000 PYs) were 182.7 in the intervention arm and 185.3 in the usual care arm, giving an adjusted RR for OM of 0.980 (95% CI: 0.963–0.999; p = 0.036) (Table 2). For OMEX, the adjusted RR was 0.985 (0.965–1.004; p = 0.13). For both OM and OMEX, there were borderline statistically significant interactions of sex by trial arm. Interaction p-values were 0.06 for OM and 0.07 for OMEX (p-values were the same for both Poisson and Cox adjusted regression models). Lung cancer death rates were 16.2 in each arm. The adjusted RR was 1.01 (95% CI: 0.95–1.07). The RR for colorectal cancer was 0.75 (95% CI: 0.66–0.85).
SMRs
SMRs for OM over the entire follow-up period were 67% (95% CI: 66–68%) in men and 59% (95% CI: 58–60%) in women. SMRs for OMEX were similar. SMRs increased by time period in both sexes. For 0–5, 5–10, 10–15, and 15–21 years from randomization, SMRs (for OM) were 49, 61, 72, and 82%, respectively, in men, and 38, 53, 63, and 73%, respectively, in women. For trial cancers, SMRs for men were 61% (95% CI: 58–64%) for lung, 63% (95% CI: 56–70%) for colorectal, and 57% (95% CI: 51–63%) for prostate, while for women SMRs were 62% (95% CI: 57–65%) for lung, 55% (95% CI: 47–63%) for colorectal, and 98% (95% CI: 85–111%) for ovarian.
Discussion
In this analysis of OM in the intervention versus usual care arm of the PLCO trial after extended follow-up, a small but statistically significant reduction was observed in men, but no reduction was observed in women. In men, a significant reduction was also observed for OM excluding deaths from trial cancers (OMEX). For both sexes combined, a small but significant reduction was observed for OM, but not for OMEX.
In the PLCO trial, only one of the three screening regimens evaluated in men, sigmoidoscopy for colorectal cancer, found a significant reduction in cancer-specific mortality. The reduction in OM of 3.4% (i.e. RR = 0.966) was mostly due to deaths excluding those from the trial cancers, with the reduction in OMEX of 3.0% (RR = 0.970). It is not clear why there was a reduction in OMEX in men, and why there was no hint of a reduction in OMEX or OM in women. In women, none of the three trial cancer-specific mortality RRs (ovarian, lung, colorectal) reached statistical significance, and the observed reduction across arms in colorectal cancer mortality, which was significant for both sexes combined, was of a lesser magnitude in women (RR = 0.87) than in men (RR = 0.68). Women had slightly lower compliance with colorectal cancer screening than men (81% versus 86%), 7 but this difference is too small to explain the difference in RRs. Also, the differential RRs by sex for colorectal cancer mortality do not explain the difference across sexes in the RR for OMEX.
In addition to compliance, screening in the control arm (denoted as contamination) could also have affected the OM difference across arms. Contamination was relatively high for colorectal cancer screening, with 25.8 and 46.5% having received sigmoidoscopy screening and any endoscopic screening (sigmoidoscopy or colonoscopy), respectively, during the screening phase of PLCO. 7 This contamination could have increased the RR for colorectal cancer deaths towards the null, and thus also increased the RR for OM towards the null. Endoscopic contamination was similar among men and women (slightly lower in women), so this would not explain the difference in RRs by sex. There was no hint of a mortality reduction with lung or ovarian cancer screening, so contamination with respect to those cancers is not likely to have affected the RR for OM. Contamination rates were generally low anyway, around 3–5% for the ovarian screening modalities and 10% for CXR.10,14 For prostate cancer, contamination with PSA screening was relatively high. Annual contamination rates were 40–50%, and 74% of control arm men received at least one PSA screen during the trial’s screening phase. 15 It is possible that a modest true reduction in prostate cancer mortality was diluted due to this contamination, which also would have increased the RR for OM towards the null in men. Use of the PLCO screening tests prior to randomization was high for some tests, around 50% for PSA, DRE, and CXRs, but was equal across arms, so would not have been likely to have affected the RR for OM. 7
RRs for OM reported for the original trial follow-up period (median follow-up around 11 years and maximum follow-up 13 years) 6 were similar to those observed here for the extended follow-up period (0.968 versus 0.966 in men and 0.983 versus 0.980 for both sexes combined). However, the findings for the original period were not statistically significant.
In general, for cancer screening trials, and specifically in PLCO, there are several possible explanations for a reduction in intervention versus control arm mortality excluding deaths from the trial cancer(s). These include incidental findings observed on the screening exams or diagnostic follow-up that result in life-saving interventions, health behavioural changes due to being in the intervention arm and receiving screenings, and deaths actually from a trial cancer being wrongly attributed to another cause, differentially by arm. However, for these factors to be operating here, they would have to be differential by sex. In addition, there is random chance, which cannot be completely ruled out here, with the observed p-value of 0.02 in men.
As men and women received a different battery of screening exams, there could be differences in these factors by sex. Even with the same screening tests, there could still be a gender difference based on different distributions of causes of death in men versus women, and/or differences in health behaviours. A prior analysis of PLCO CXR screening found substantial proportions of subjects with non-lung cancer related abnormalities, including pulmonary fibrosis, pleural fibrosis, and COPD. 16 Men were significantly more likely than women to have these conditions reported. The category with the largest differential across genders and arms was respiratory disease. In men there were 42 (3%) fewer respiratory deaths in the intervention versus usual care arm, whereas in women there were 124 (16%) more such deaths in the intervention arm. However, it is not clear to what extent, if any, this is related to reported ancillary findings on screening CXRs. The PLCO population overall was significantly healthier than the average population, but SMRs were similar for men (67%) and women (59%).
The National Lung Screening Trial was one of the only individual screening trials to show a reduction in all-cause mortality. 17 Because the trial population was selected as at high risk for lung cancer, and lung cancer has the highest mortality in the United States for any cancer, the proportion of all deaths that were from the cancer of interest was very high (24.1%). In comparison, in PLCO, deaths from all trial cancers combined comprised only 13.5% of all deaths. A meta-analysis of four sigmoidoscopy screening trials, including PLCO after a median 11 years of follow-up, showed a significant decrease in all-cause mortality (RR = 0.97; 95% CI: 0.96–0.99). 18 In contrast, a meta-analysis of mammography trials did not show a significant reduction in all-cause mortality, with a combined RR estimate of 0.99 (95% CI: 0.97–1.002) over nine screening trials. 19
For OM and OMEX, the percentage risk reduction in the intervention arm was small. Therefore, even small imbalances in age, if not adjusted for, could cause appreciable changes in the risk reduction estimate, and in turn affect the p-value and confidence intervals. As shown in the online Appendix, due to the small age differences, the models that did not control for age (unadjusted Poisson and Cox regressions) gave different findings from the models that did control for age, and in fact gave different findings from each other. In contrast, the two models that controlled for age gave very similar results.
Though the percentage reductions for OM and OMEX were small, whether these represent true decreases in mortality and were not due to chance is important. First, OM is one of the few examples of a cancer screening trial showing a significant decrease in OM. Second, because the event (OM or OMEX) is relatively frequent, the impact could be substantial in absolute terms. For example, the absolute difference across arms in colorectal cancer deaths in men, using the adjusted RR and control arm rate, was 1.8 per 10,000 PYs. In comparison, the absolute difference across arms in OMEX (all deaths excluding PLCO cancers) in men was 6.0 per 10,000 PYs.
For lung cancer, this is the first update of mortality since the reporting from the original trial period. That original analysis showed an RR of 0.99 (95% CI: 0.876–1.22). 10 With extended follow-up, the RR remained essentially the same (RR = 1.01). A prior update for ovarian cancer mortality, through a median of almost 15 years of follow-up, showed an RR for women with ovaries of 1.06 (95% CI: 0.87–1.30). 9 The current update, with about two additional years of follow-up, has increased the RR modestly to 1.18 (0.98–1.4).
Conclusion
In the PLCO multiphasic cancer screening trial, there was a small but statistically significant reduction in OM in both sexes combined after a median 17 years of follow-up. In men, there was a small but significant reduction in OM, and in OM excluding deaths from trial cancers. These findings strengthen the argument that cancer screening can actually save lives and does not just replace cancer deaths with those from other causes.
Supplemental Material
Supplemental material for Overall mortality in men and women in the randomized Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial
Supplemental Material for Overall mortality in men and women in the randomized Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial by Paul F Pinsky, Eric A Miller, Claire S Zhu and Philip C Prorok in Journal of Medical Screening
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The PLCO trial was funded by the National Cancer Institute, ClinicalTrials.gov number NCT00002540.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
