Abstract
Objective
To analyze the age- and trial-specific effects of the breast cancer screening trials with mammography in Malmö, Stockholm, and Göteborg.
Methods
The original trial files were linked to the Swedish Cancer and Cause of Death Registers to obtain date of breast cancer diagnosis and date and cause of death. Relative risks and 95% confidence intervals were calculated using the evaluation model (only breast cancers diagnosed between date of randomization and date when the first screening round of the control group was completed were included in the analysis).
Results
Women aged 40–70 at randomization in the Malmö I and II, Stockholm, and Göteborg trials were followed-up for an average of 30, 22, 25, and 24 years, respectively. The overview of all trials resulted in a significant decrease of 15% in breast cancer mortality. The variation by consecutive 10-year age group at randomization was small—from 21% in the age group 40–49 to 11% in the age group 50–59. After adjustment for age, there was a significant reduction in breast cancer mortality in the Göteborg trial (26%), and a non-significant reduction in the Malmö I and II and Stockholm trials (12%, 15%, and 5.8%, respectively).
Conclusions
The overview showed a 15% significant relative reduction in breast cancer mortality due to invitation to mammography screening. Heterogeneity in age, trial time, attendance rates, and length of screening intervals may have contributed to the variation in effect between the trials.
Introduction
Mammography service-screening programs for breast cancer aim to decrease the breast cancer mortality. The scientific basis for these programs was the randomized breast cancer screening trials. Of the eight randomized controlled trials (RCTs) performed worldwide, four have been conducted in Sweden. The Swedish trials performed in Malmö, Kopparberg and Östergötland (two-county or WE), Stockholm, and Göteborg had similar designs: all were population based and compared invitation to breast screening with mammography alone versus no invitation. This design differed from the Health Insurance Plan (HIP) of Greater New York, the Edinburgh trial, and the Canadian National Breast Screening Study (CNBSS), which all evaluated mammography combined with breast self-examination and/or clinical breast examination, and the HIP and the CNBSS trials were not population based. The UK Age Trial that started in 1991 invited women aged 40–41 to annual screening until age 50, to evaluate the effect of inviting women below age 50 to screening.
To improve the precision in the estimates in the Swedish RCTs and to facilitate analysis by age at randomization, the Swedish Cancer Society initiated an overview (meta-analysis using individual patient data) of the Swedish trials in the late 1980s. The first results of the overview with a follow-up through 1989 were published in 1993, 1 and an update through 1993 focusing on the age group 40–49 was published in 1997. 2 The latest update through 1996 did not include the Kopparberg part of the WE trial. 3 Both the first and the third overview showed that mammography screening reduced breast cancer mortality by about 20%. At the latest follow-up, through 1996, the median follow-up time varied from 9.1 years in part II of the Malmö trial to 19.2 years in part I of Malmö trials. We therefore aimed to extend the follow-up through 2007 and to analyze the age-specific and trial-specific effects on breast cancer mortality in the trials in Malmö, Stockholm, and Göteborg.
Methods
Overview of the randomized controlled trials in Malmö (MMST I and II), Stockholm, and Göteborg.
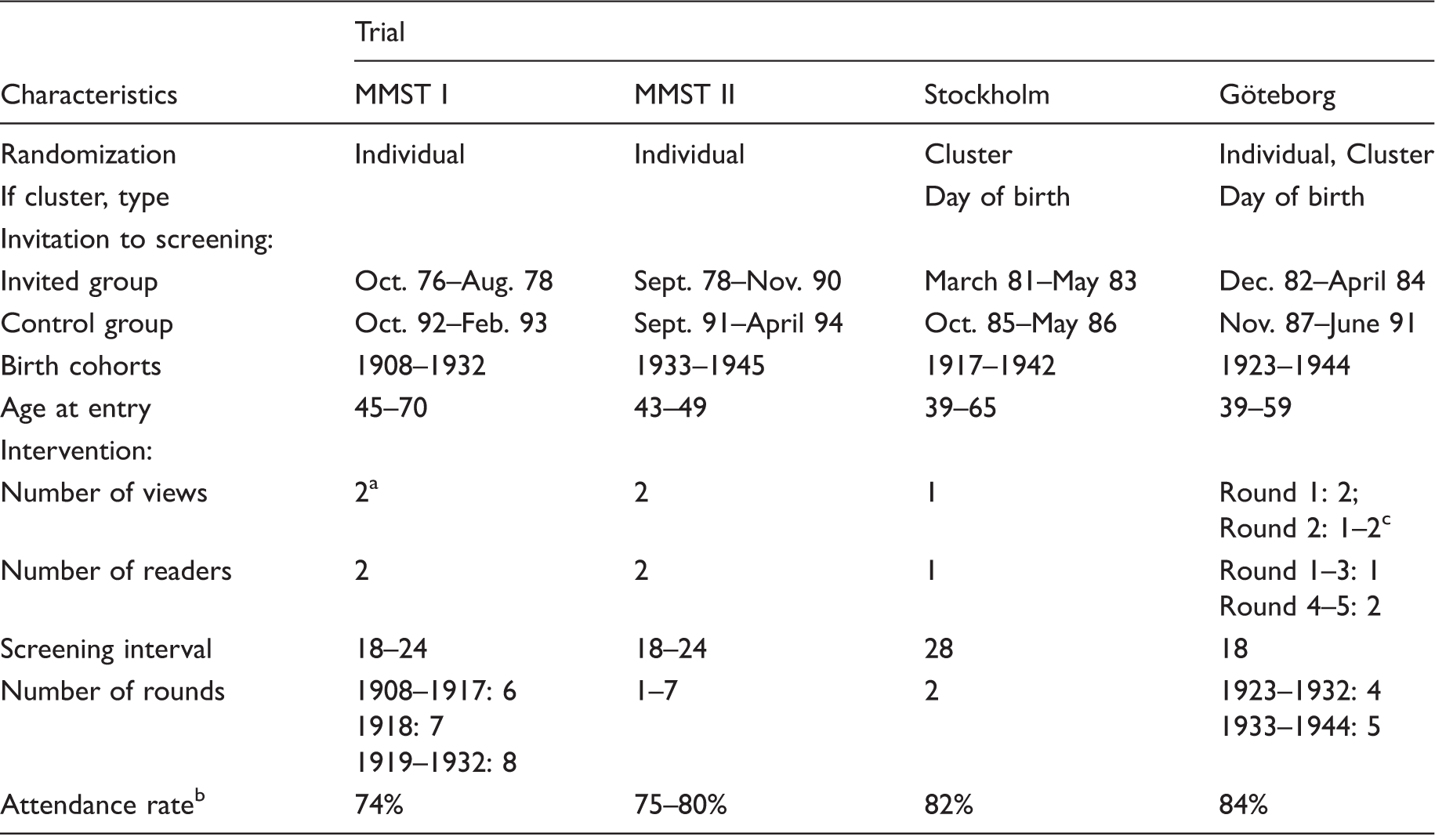
MMST: The Malmö Mammographic Screening Trial.
From round three single or two views according to parenchymal pattern.
First round.
Depending on the density of the breast.
The Malmö Mammographic Screening Trial (MMST)
This trial included women in the city of Malmö from October 19764,5 and was conducted in two parts. In MMST I, women born between 1908 and 1932 were randomized individually stratified by year of birth. Women were invited to screen-film mammography alone, in the first two rounds with two views (craniocaudal and oblique), and in subsequent rounds with either two views or the oblique view alone, depending on the parenchymal pattern. A single oblique view was used for women whose breasts were mainly fatty on mammography, and two views for women with dense breasts. Women born 1908–1917, 1918, and 1919–1932 were invited to 6, 7, and 8 screening rounds, respectively. Women in the control group, born 1908–1922, were never invited to screening, while women born 1923–1932 were first invited to screening in 1992–1993 (Table 1). In age groups where screening continued after the end of the trial time (aged under 70), screening was performed in the invited group only up until age 70 for women born 1918–1922 and in women born 1923–1932 until 1989. The service screening program in Malmö started in 1990, and both the invited and the control group of women born 1923–1932 was screened up until age 70 in the following years.
After MMST I closed in August 1978, women reaching age 45 were continuously randomized to the Malmö trial part II, using the same protocol as in MMST I. MMST II comprised all women born from 1933 to 1945 living in Malmö between 1978 and 1990. These women were randomly allocated to receive an invitation to screening. The plan was to invite these women when they turned 45, beginning in 1978. Due to limited resources, the plan could not be strictly adhered to, which means that, some years, no women could be invited, while other years, two or even three birth-year cohorts were randomized and invited to examination. The median age at entry was 45 (range 43–48). The last birth-year cohort, women born in 1945, was invited in 1990. Women born 1933–1936, 1937, 1938, 1939–1943, and 1944 were invited to 7, 6, 3, 2, and 1 screening round, respectively. After that, women in the invited and control groups were transferred to the service-screening program that started in 1990. The first screening round of the control group took place between 1991 and 1994.
The Stockholm trial
About 60,000 women in the southern Stockholm born between 1917 and 1942 (aged 39–65) were randomized by day of birth, to receive an invitation to mammography alone with an oblique single-view mammography, or no intervention.6,7 About 40,000 women were allocated to the invited group and 20,000 to the control group. Screening started in March 1981. The screening interval was 28 months between the first and the second round and 24 months between the second and the third rounds in 1985–1986, when the control group was invited to screening (Table 1).
The Göteborg trial
Between December 1982 and April 1984, all women born between 1923 and 1944 (aged 38–59) who lived in the city of Göteborg were randomized.8,9 To make it possible to reinvite women every 18 months, the ratio of women randomized to the invited group versus the control group was 1:1.2 in women aged 39–49 and 1 to 1.6 in women aged 50–59. Women born from 1923–1932 were invited to four screening rounds between December 1981 and April 1988, and women born from 1933–1944 were invited to five screening rounds between September 1983 and August 1990. Women in the control group, born from 1923 to 1932 were invited to their first screening round between September 1987 and April 1988, and women in the control group born from 1933 to 1944 were invited to their first screening round between February and April 1990 (Table 1). Two-view mammography was used unless the observations at the previous screening round indicated that single-view mammography would be adequate, depending on the density of the breast. Mammograms were single read at the first three screening rounds. In the fourth round, double reading was successively introduced and completely established by the end of the fifth round.
Diagnostic work-up
In all screening trials, mammography was coded 1–5, 1 and 2 indicating normal or benign mammography, 3 a suspected finding probably benign but needs further examination, and codes 4 and 5 a suspect or clear malignant lesion. All women with suspicious findings on the mammogram (code 3–5) were recalled for supplementary mammography. If the suspicious lesion persisted after complete mammography, the woman was referred to the trial surgeons for a complete triple examination, including clinical examination, fine needle aspiration cytology, and/or core biopsy performed by palpation, stereotactic technique, or guided ultrasound. If cytology or core biopsy were not conclusive, a surgical excision was performed.
Randomization methods
The randomization procedure has been described in detail elsewhere. 3 In summary, individual randomization was used in the Malmö trials and in the second part of the Göteborg trial (women born between 1936 and 1944). During the first part of the Göteborg trial (women born between 1923 and 1935 and randomized between 21 December 1982 and 3 November 1983), day of birth was used for randomization, with varying days for each year cohort. The Stockholm trial used randomization by day of birth. Between March 1981 and April 1982, women born on days 1–20 in 1917–1941 were invited to screening, women born on days 1–10 in the invited group and women born on days 11–20 in the control group, and between May 1982 and May 1983, women born on days 21–30 in 1918–1942 were included in the invited group, and women born on days 11–20 in 1942 were included in the control group. 6
Inclusion and exclusion criteria
Number of women randomized by trial, study group, and 5-year age group. Only women aged 40–74 were included in the overview.
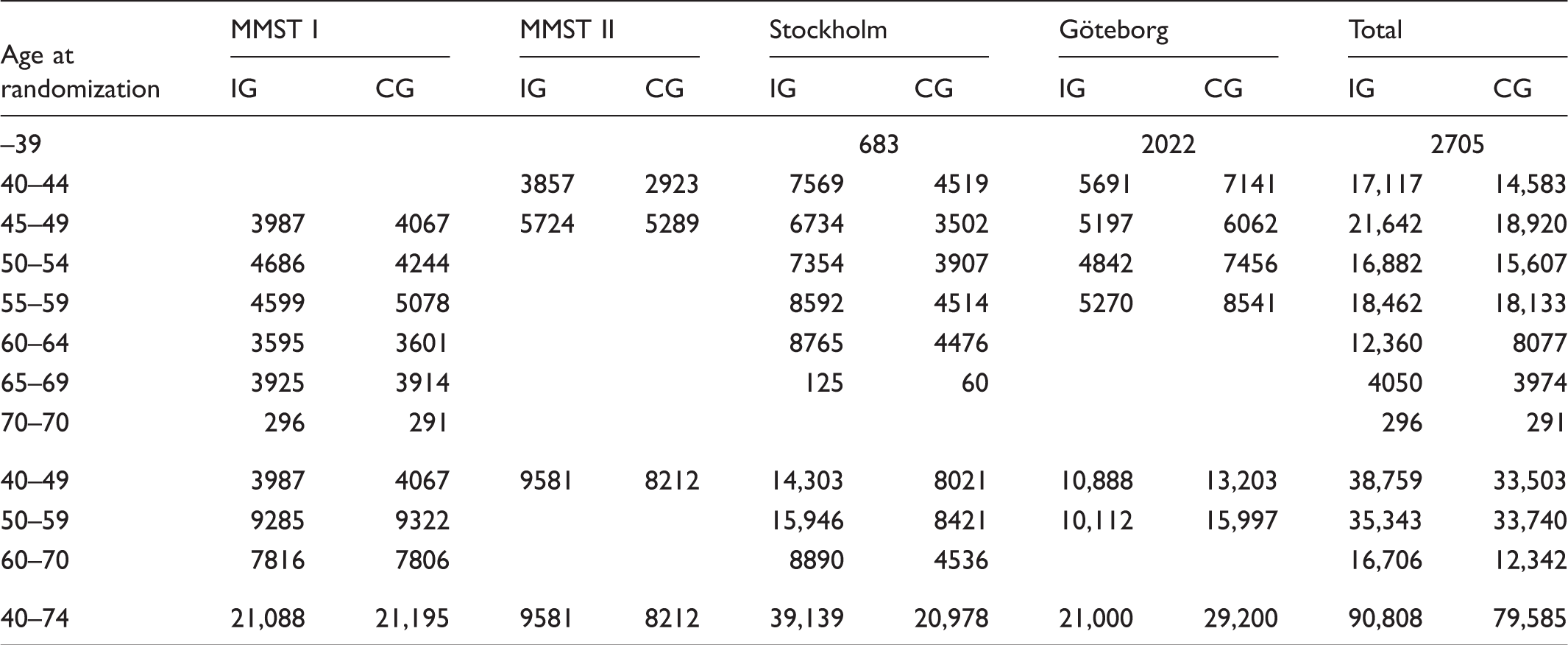
IG: invited group; CG: control group; MMST: The Malmö Mammographic Screening Trial.
End points
The primary end point in this overview was breast cancer as underlying cause of death according to the Swedish Cause of Death Registry. Data on deaths from other causes were also retrieved from the same register. Our previous comparison of breast cancer as the underlying cause of death and breast cancer present at death as determined by an independent end-point committee in a “blind” review based on available clinical records and autopsy protocols with the officially recorded underlying cause of death 10 revealed a close concordance. We have also earlier shown that using excess mortality (i.e., deaths among breast cancer cases reported to the Cancer register minus the expected number of deaths), an outcome measure that is independent of cause of death determination, gave the same results. 11 As the estimated benefit associated with invitation to breast cancer screening was almost identical, irrespective of end-point measure and source of cause of death determination used, this overview used only the officially recorded breast cancer as underlying cause of death.
Data retrieval
The original trial files, selected from the official population register, which served as basis for the randomization, were obtained from the principal investigator of each participating trial. All individuals were identified through their unique personal identification number. For each woman, information on date of randomization and allocation group was added. Records were linked with the Swedish Cancer Register and the Swedish Cause of Death Register at the National Board of Health and Welfare, to obtain date for breast cancer diagnosis and pathological-anatomical diagnosis (PAD), and date and underlying cause of death. The end date for the computerized follow-up was 31 December 2007. In contrast to earlier follow-ups, only cases reported to the Cancer Register with breast cancer and a PAD = 096 were included. Breast cancer deaths reported to the Cause of Death register but not reported to the Cancer Register were not included in the analysis, but the numbers are reported separately.
Statistical methods
To minimize possible “dilution” of the effect of screening in the invited group from screening in the control group, we developed an evaluation model and a follow-up model (described in detail previously 3 ) to analyze outcome. Briefly, the follow-up model includes as an event all women with breast cancer diagnosed after date of randomization, who died with breast cancer as the underlying cause of death before date of follow-up. The evaluation model is similar, but has a restriction on diagnosis, so that only breast cancers diagnosed during the trial time (i.e., between date of randomization and completion of first screening round of the control group) are included in the analysis. With respect to the extended follow-up time after closing the trials, only the evaluation method was used in this update.
In MMST I, women in the control group born 1908–1922 were never invited to screening. Women born 1908–1917 were invited to their 6th and last screen between January 1986 and June 1987, women born 1918 were invited to their 7th and last screen between April and May 1988, and women born 1919–1922 were invited to their 8th and last screen between April and December 1988. Women born 1908 received their last invitation to screening between 6 June and 30 September 1986; thus, the end date for inclusion as a breast cancer case for this birth cohort was set as 30 October 1988, and so on. In contrast to earlier follow-ups of the MMST I, end date was set based on when women in the invited group were no longer invited to screening, i.e., one month after the last invited women in the birth cohort. Women in the control group born 1923–1932 were invited to screening after the 8th and last screening round of the invited group. In MMST II, the Stockholm and Göteborg trial women in the control group were invited to screening after the last screening round of the invited group.
Data were analyzed using SPSS. Relative risks (RR) and 95% confidence intervals were calculated. The breast cancer mortality by time since randomization was calculated for year x as number of breast cancer deaths year x divided by number of women-years year x. The cumulative risk was then obtained by summarizing the yearly ratios. To obtain a total cumulative risk, a weighted sum of the trial-specific cumulative risks was calculated, using the number of women randomized in each trial as weights. The same weights were used for the invited and the control group. The software R was used to estimate the total efficacy of each trial adjusted for age, the age-specific efficacy of all trials adjusted for trial, and the overall efficacy of the three trials adjusted for age and trial applying a multiplicative Poisson regression model, including the covariates group (invited/control), trial and age group, and women-years at offset.
Number of women needed to invite to screening during trial time to prevent a breast cancer death during follow-up was estimated as the number of women invited to screening divided by the number of breast cancer deaths prevented; the number of women needed to screen was calculated as number of women invited to screening divided by the number of breast cancer deaths prevented multiplied by the screening attendance rate.
Results
Median follow-up time by trial and 5-year age group.
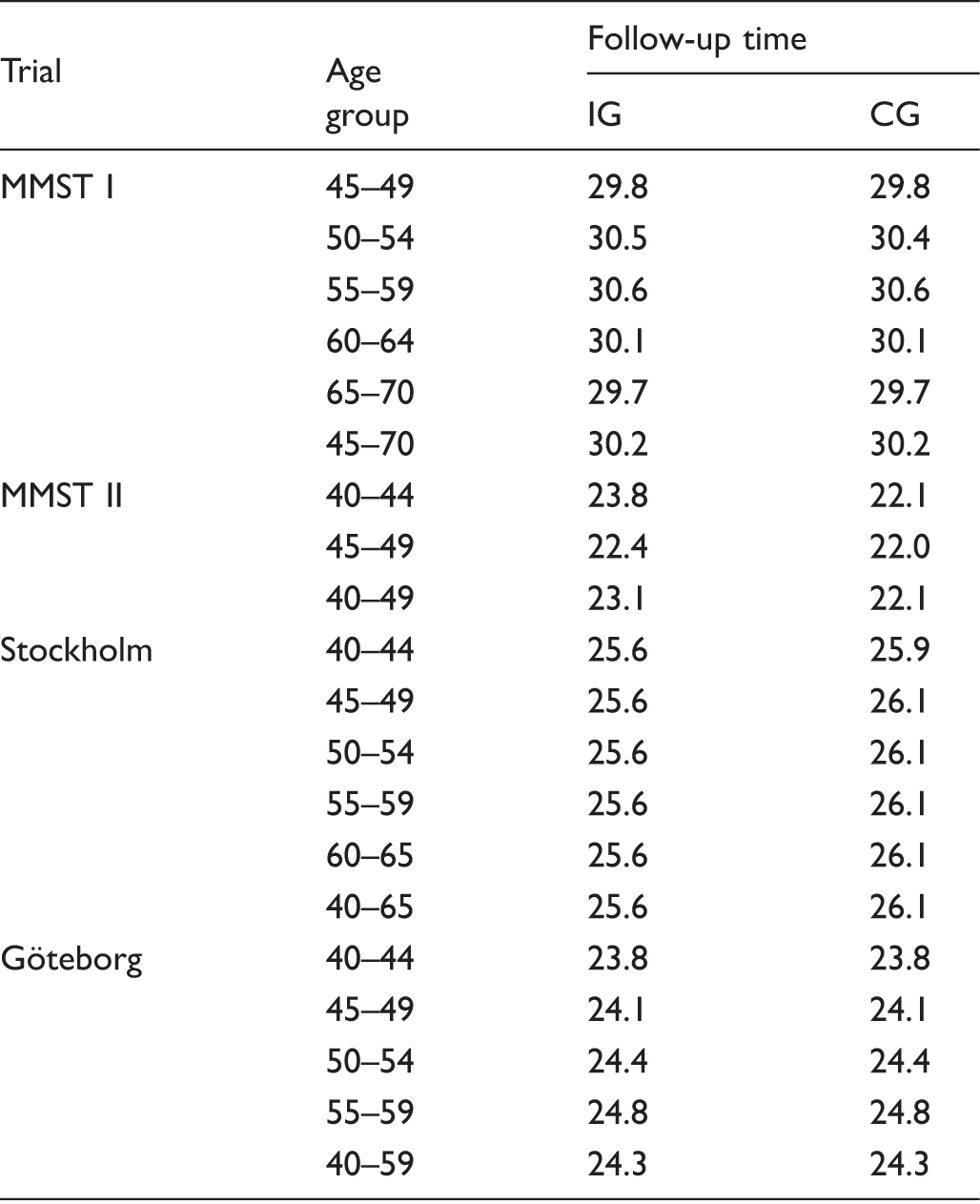
IG: invited group; CG: control group; MMST: The Malmö Mammographic Screening Trial.
Number of 1000 women-years, number of breast cancer deaths, relative risk, and 95% confidence intervals by trial and age at randomizaton.
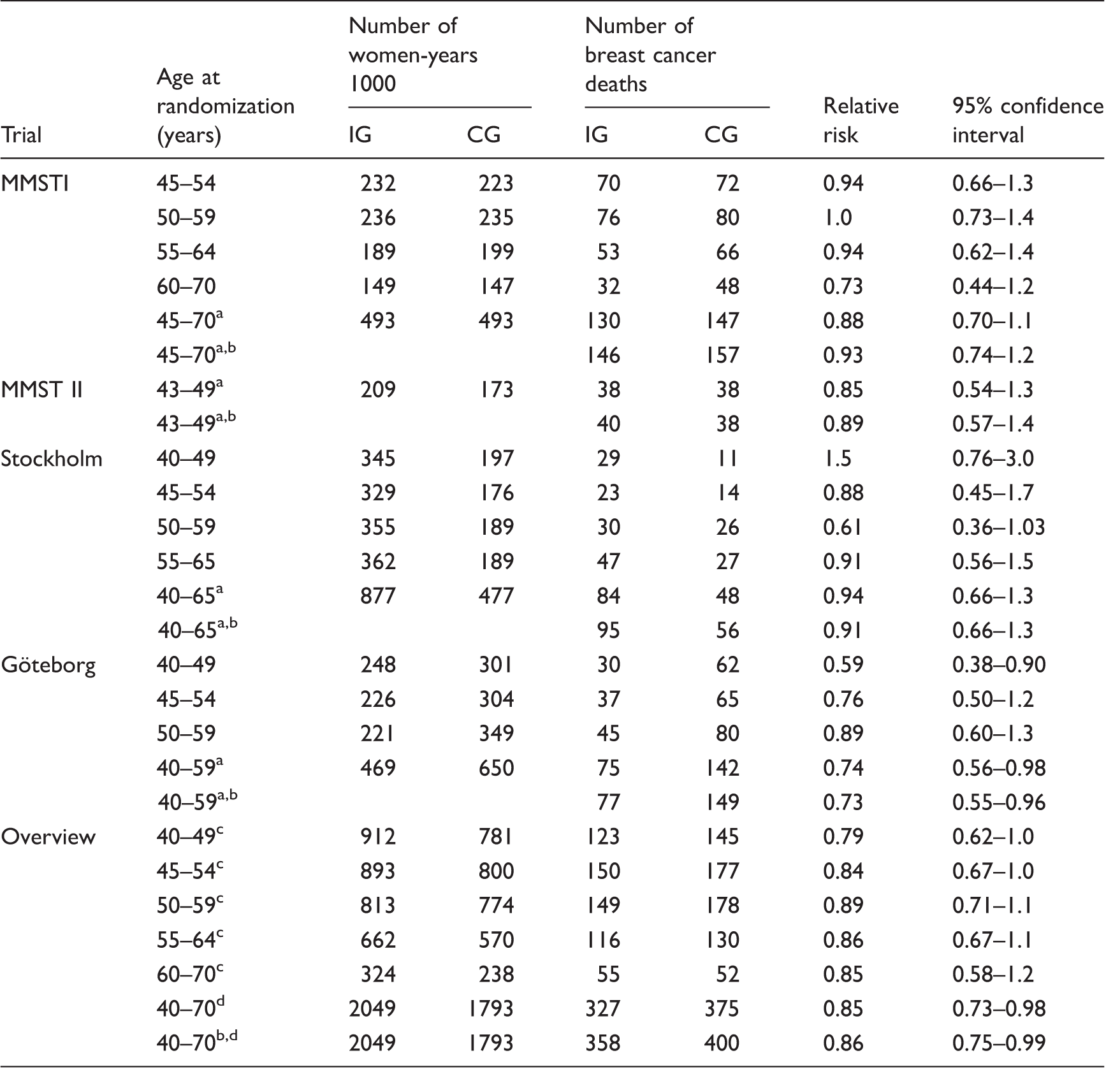
MMST: The Malmö Mammographic Screening Trial.
Adjusted for age.
Including breast cancer deaths not reported to the cancer registry.
Adjusted for trial.
Adjusted for age and trial.
The average trial time (from date of invitation to date when screening of the corresponding control group was finalized) varied: 4.3 years in Stockholm, 6.8 years in Göteborg, 7.8 years in MMSTII, and 12.4 years in MMST I. The non-significant reduction in breast cancer mortality in the MMST I and II and Stockholm trials, after adjustment for age, was 12%, 15%, and 5.8%, respectively. The only trial with a significant reduction in breast cancer mortality was the Göteborg trial (RR = 0.74; 95% CI: 0.56–0.98).
Including breast cancer deaths not reported to the Cancer Registry added 31 and 25 cases to the invited and control groups, respectively, so inclusion of these cases did not affect the results (trial and age-adjusted RR = 0.86 (95% CI: 0.75–0.99)).
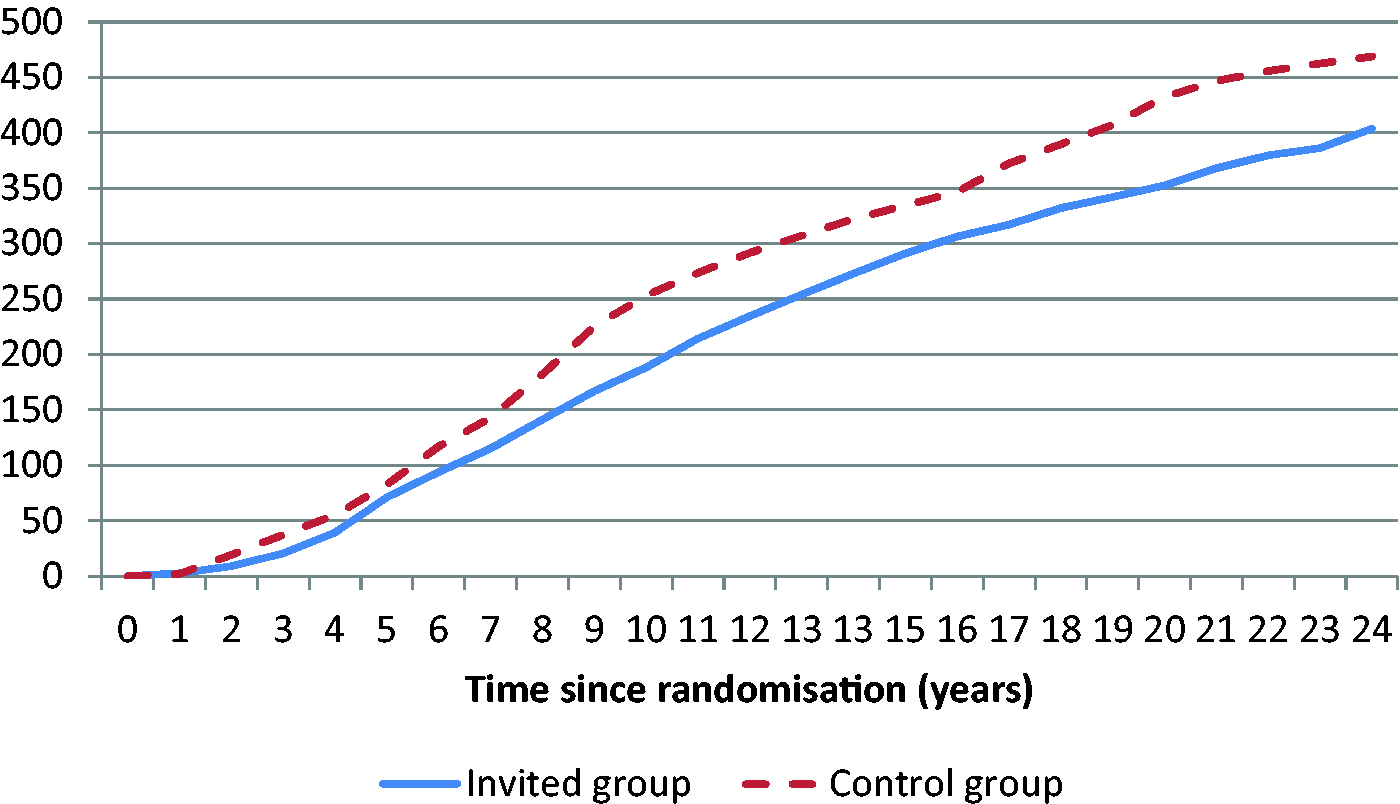
Cumulative breast cancer mortality per 100,000 women by trial is presented in Figure 1, and for the overview of the trials in Figure 2. In the Göteborg trial, the curves diverged markedly during the first 10 years after randomization, after which they diverged slightly. In the Stockholm trial, the curves also diverged during the first 10 years, after which the gap between the curves diminished, and after 20 years it had almost disappeared. In MMST I, the curves diverged slightly after 8 years and more evidently after 16 years, after which the difference was maintained. In MMST II, the curves started to diverge three years after randomization.
Cumulative breast cancer mortality/100,000 women by trial (IG: solid line, CG: dashed line). Weighted cumulative breast cancer mortality/100,000 women.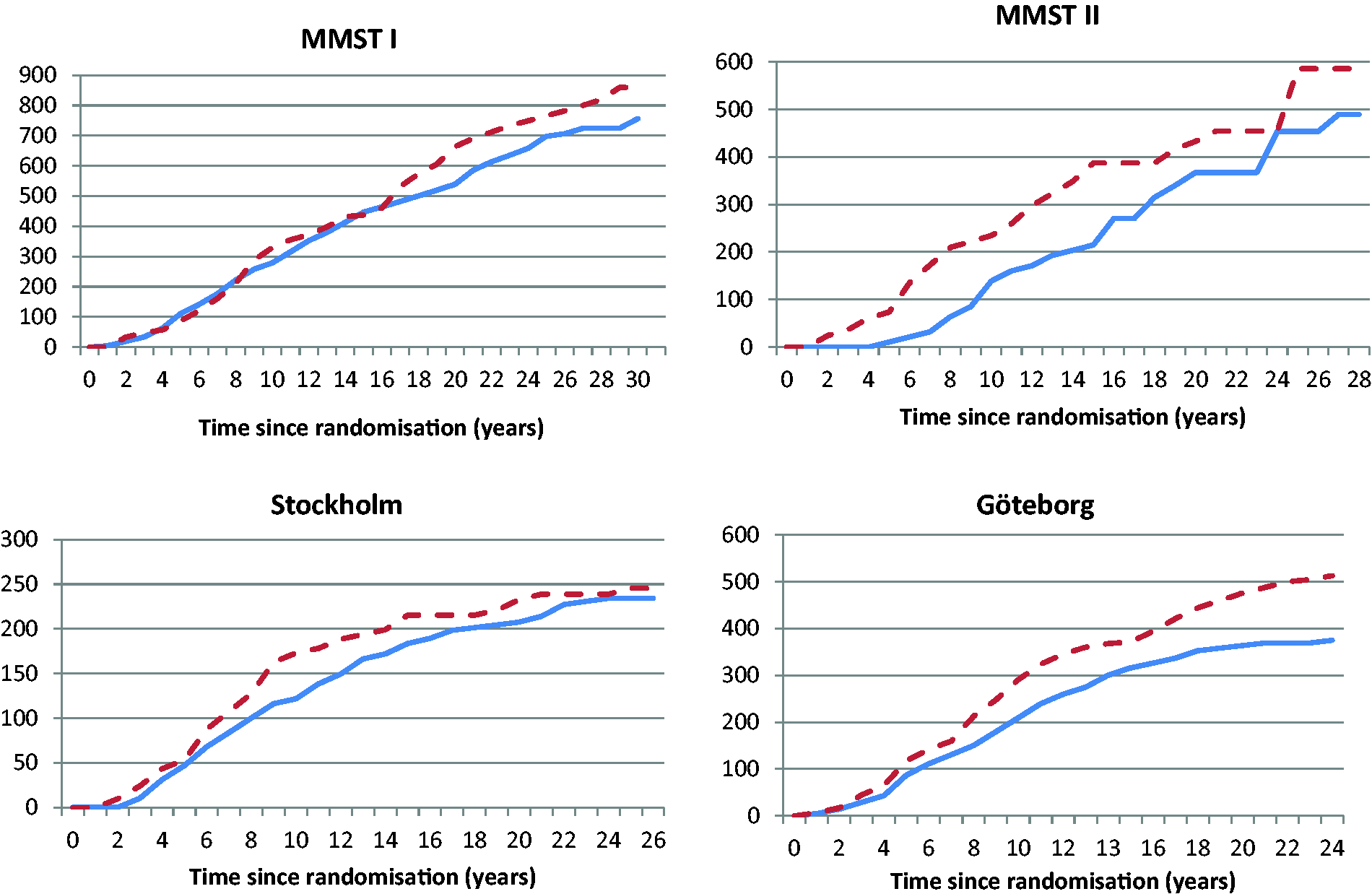
Number of breast cancer deaths, expected number of deaths and deaths prevented (Expected–Observed) in the invited group and number needed to invite (NNI) to screening (invited/prevented) and number needed to screen (NNS) (screened/prevented).
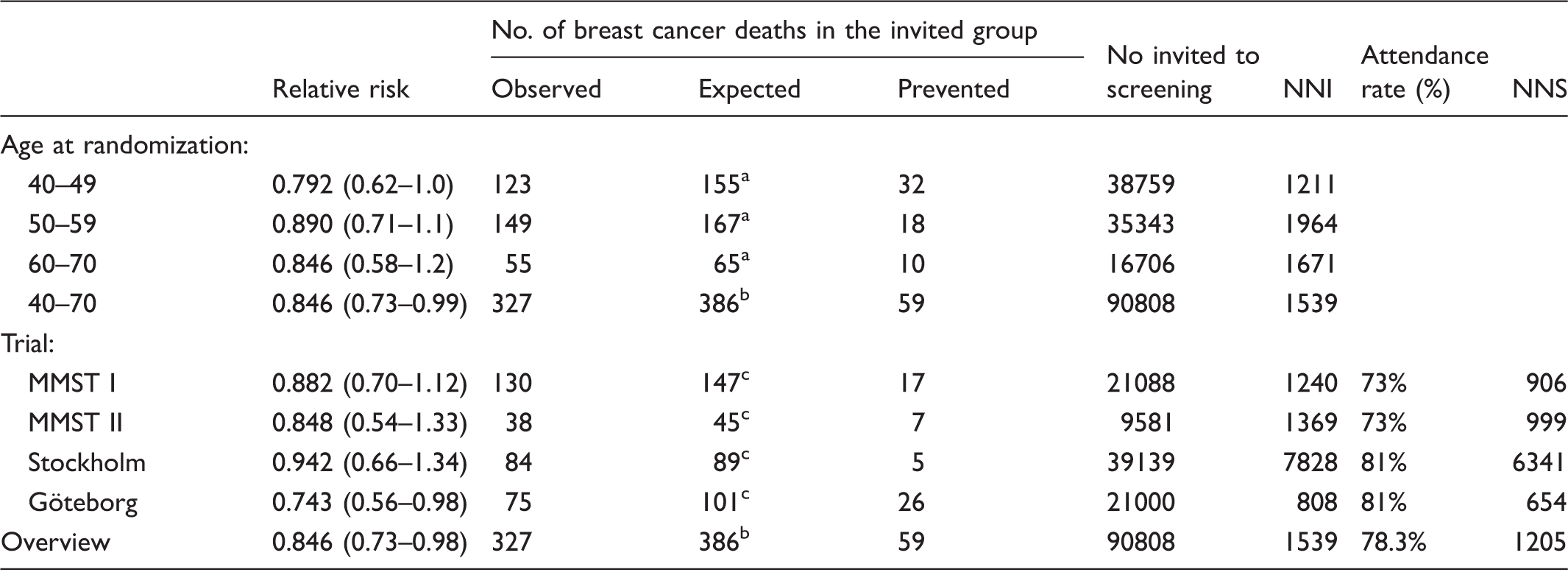
MMST: The Malmö Mammographic Screening Trial.
Adjusted for trial.
Adjusted for age and trial.
Adjusted for age.
Discussion
The Malmö, Stockholm, and Göteborg mammography screening trials have been followed up for 22–30 years, taking into account only breast cancers diagnosed between randomization and completion of the first screening round of the control group, and the efficacy of the intervention remains significant at 15% relative reduction of the breast cancer mortality. The non-significant effect in the age groups 40–49, 50–59, and 60–70 at invitation was 21%, 11%, and 15%, respectively. The youngest trial (Göteborg) showed the highest relative effect (26%), and the lowest effect was found in the Stockholm trial (5.8%). These differences might be due to differences in length of screening intervals, trial time, and program sensitivity. The cumulative breast cancer mortality per 100,000 women by time since randomization varied by trial. In MMST I, the effect during the first 15 years after randomization was marginal, after which it increased. In the Stockholm trial, the effect peaked 10 years after randomization, after which it decreased. In the Göteborg trial, the effect increased by time since randomization.
The relative effect in this follow-up was lower than earlier follow-ups, but the study base has varied over time. The first overview 1 also contained the two-county (WE) trial, and the second overview 3 contained the E-part (Östergötland) of the WE trial. In the 30-year follow-up of the WE trial, the morality curves diverged throughout the whole follow-up period. 12 In our study, the curves diverged markedly during the first 10–12 years, after which the difference between the invited group and the control group was maintained. Breast cancer today is a more “chronic” disease, with longer survival, but women still die from breast cancer 15–20 years after diagnosis. Today women live longer due to better medical treatment, with an increased risk of developing a contralateral breast cancer or a second primary breast cancer. The effect is still impressive, considering the short intervention period with only two screening rounds in the Stockholm trial and only 4–5 screening rounds in the Göteborg trial. According to the EU guidelines, women aged 50–69 should be invited to screening every second year (i.e., 10 screening rounds).
Comparison with other RCTs
Similar long-term follow-up of the RCTs on mammography screening has so far only been presented by the two-county trial. 12 The longest follow-up of the HIP, Edinburgh, CNBSS I, and CNBSS II trials is 10–14, 14, 11–16, and 25 years, respectively.13–17 With a trial time of approximately 7 years and 29 years of follow-up, in the WE trial, the relative reduction in breast cancer mortality was 31%. This is slightly higher than in the Malmö, Stockholm, and Göteborg trials and was achieved in spite of a longer screening interval (women aged 40–49 were invited every 24 months, and women aged 50–74 every 33 months). The curves for the cumulative breast cancer mortality per 100,000 women in the WE trial diverged during the whole follow-up period, as in the Göteborg trial. Number needed to undergo screening for seven years to save one life was as low as 414, using the local end-point committee, and 519 using Statistics Sweden, which is considerably lower than in our overview, and can only partly be due to a higher attendance rate in the Kopparberg trial.
Strengths and limitations
In the first overview of all four Swedish trials, an independent external end-point committee assessed the cause of death for all incident breast cancer cases using the cause of death certificate, autopsy protocol, histopathology report, and medical records. 10 The statistical analysis using breast cancer as the underlying cause of death or breast cancer prevalent at death according to the end-point committee, and breast cancer as the underlying or underlying/contributory cause of death according to the cause of death register, resulted in almost identical estimates. 1 Thus, in this follow-up, we used only breast cancer as the underlying cause of death according to the high-quality national cause of death register.
In addition, a comparison between the two-county trial using the WE trial evaluation protocol and the overview evaluation protocol showed that the differences were negligible. 18
Duffy and Smith 19 estimated the effect in screening trials when follow-up for mortality continues beyond the screening phase and found that “all designs incurred a bias against screening.” The approach with the smallest bias was the approach that was applied in this study (i.e., include a single screen of the control group conducted at the end of the screening phase and include only deaths from breast cancers diagnosed during the screening phase in both arms up to completion of the single control screen).
A limitation of our study is that there were limited data on the amount of contamination. The trials were performed in the three largest cities in Sweden with access to mammography screening. It was estimated, based on small surveys, that in Malmö, Stockholm, and Göteborg, 24%, 20%, and 20% of women, respectively, had had a mammogram before their first invitation to the screening program.
Another limitation is that the Swedish randomized trials were performed a long time ago, and important improvements in technical and in treatment strategies have since taken place. This means that the results may not be fully representative and applicable in a modern screening setting. Nevertheless, robust data from randomized trials represent the strongest evidence, especially with long follow-up.
Conclusions
At 22–30 years follow-up of the MMST I and II, Stockholm, and Göteborg trials, the overview showed a significant 15% relative reduction in breast cancer mortality due to invitation to mammography screening. Except for chance, heterogeneity in age, trial time, attendance rates, and length of screening intervals may have contributed to a variation in effect between trials. The variation in absolute effect by trial is most likely to have been due to length of trial time.
Footnotes
Acknowledgement
We thank Associate Professor Ingvar Andersson (MD, PhD) for valuable contributions to the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Swedish Cancer Society.
