Abstract
Objective
Biliary atresia, a rare newborn liver disease, is the most common cause of liver-related death in children and the main indication for paediatric liver transplantation. Early detection and surgical intervention with a Kasai portoenterostomy offers the best chance for long-term patient survival. We conducted a cost-effectiveness analysis to compare no universal screening with screening using either a home-based infant stool colour card with passive card distribution strategy, or conjugated bilirubin testing.
Methods
A Markov model was developed, with structure, costs, and probabilities informed by the literature and clinical expert opinion, to simulate a newborn cohort over a 10-year time horizon. Health benefits were expressed as life-years gained. This analysis was conducted from the perspective of the Canadian publicly funded health care system (all costs in Canadian dollars). Both deterministic and probabilistic analyses were conducted.
Results
Screening using a home-based colour card with passive card distribution was a cost-effective option. For a population of 392,902 annual births in Canada, this strategy cost approximately $192,000 more than no universal screening but led to eight life-years gained (incremental cost-effectiveness ratio (ICER) = $24,065 per life-year gained). Screening using conjugated bilirubin testing versus the colour card cost $2,369,199 more and led to five more life-years gained (ICER= $473,840 per life year gained), and so was not cost-effective.
Conclusions
A home-based screening program using infant stool colour cards with a passive distribution strategy could be highly cost-effective when administered at a low unit cost and with a reasonable screening performance.
Keywords
Introduction
Biliary atresia (BA), a rare paediatric liver disease and the most common cause of liver death and transplantation in children,1,2 presents in the first three weeks of life and is accompanied by jaundice and pale (acholic) stools. Incidence in Canada is estimated at 5.25/100,000 births. 2 Left untreated, BA typically leads to liver failure induced mortality by age two. 3 Kasai portoenterostomy (KP) is a surgical procedure that can restore bile flow and prevent the need for early liver transplantation in children, if performed in the first two months of life. Infant age at the time of the KP is the most important prognostic factor for a successful outcome; the older the infant at the time of surgery, the less likely it is that they will survive long term with their native liver and without the need for liver transplantation. 3 The most favourable outcomes are achieved if the KP is performed before 30 days of age, and the poorest outcomes are associated with KP surgery at more than 90 days. 3
Japan and Taiwan have introduced home-based universal screening programs using an infant stool colour card (ISCC) to detect BA early and thus accelerate the KP within the optimal time-window.4–8 An important element of these programs is that the ISCC is reviewed directly with parents at the routine infant one month vaccine clinic appointment, if the parents have not flagged a concern earlier. The ISCC screening program has performed well in studies, with a sensitivity and specificity of 76.5% and 99.9%, respectively. 4 In addition, a retrospective study conducted at British Columbia Women’s Hospital using the ISCC found a sensitivity and specificity of 83% and 99.9%, respectively. 9 In this study, parents were asked to perform the screening for the first month and contact the screening centre directly with any concerns, as a check-up with a physician does not routinely occur until two months. Another newly developed screening option is a conjugated bilirubin test, for which a United States study found a sensitivity and specificity of 100% and 98%, respectively. 10
Two studies have examined the cost-effectiveness of screening for BA using an ISCC. The first, conducted in Canada, found that over a time horizon of 10 years, a passive ISCC screening strategy was cost-effective compared with no screening (incremental cost-effectiveness ratio (ICER) $22,000 CAN per life year gained). 11 A second study, conducted in the United States over a 20-year time horizon, found that screening with an ISCC was a dominant strategy compared with no screening (i.e. less costly and more effective). 12 Neither study, however, assessed the cost-effectiveness of conjugated bilirubin testing.
In Canada, the United States, and most of Europe, there is currently no universal screening program for BA. This study aims to assess the cost-effectiveness of screening for BA using a universal, home-based BA ISCC program, or screening with conjugated bilirubin, compared with no screening.
Methods
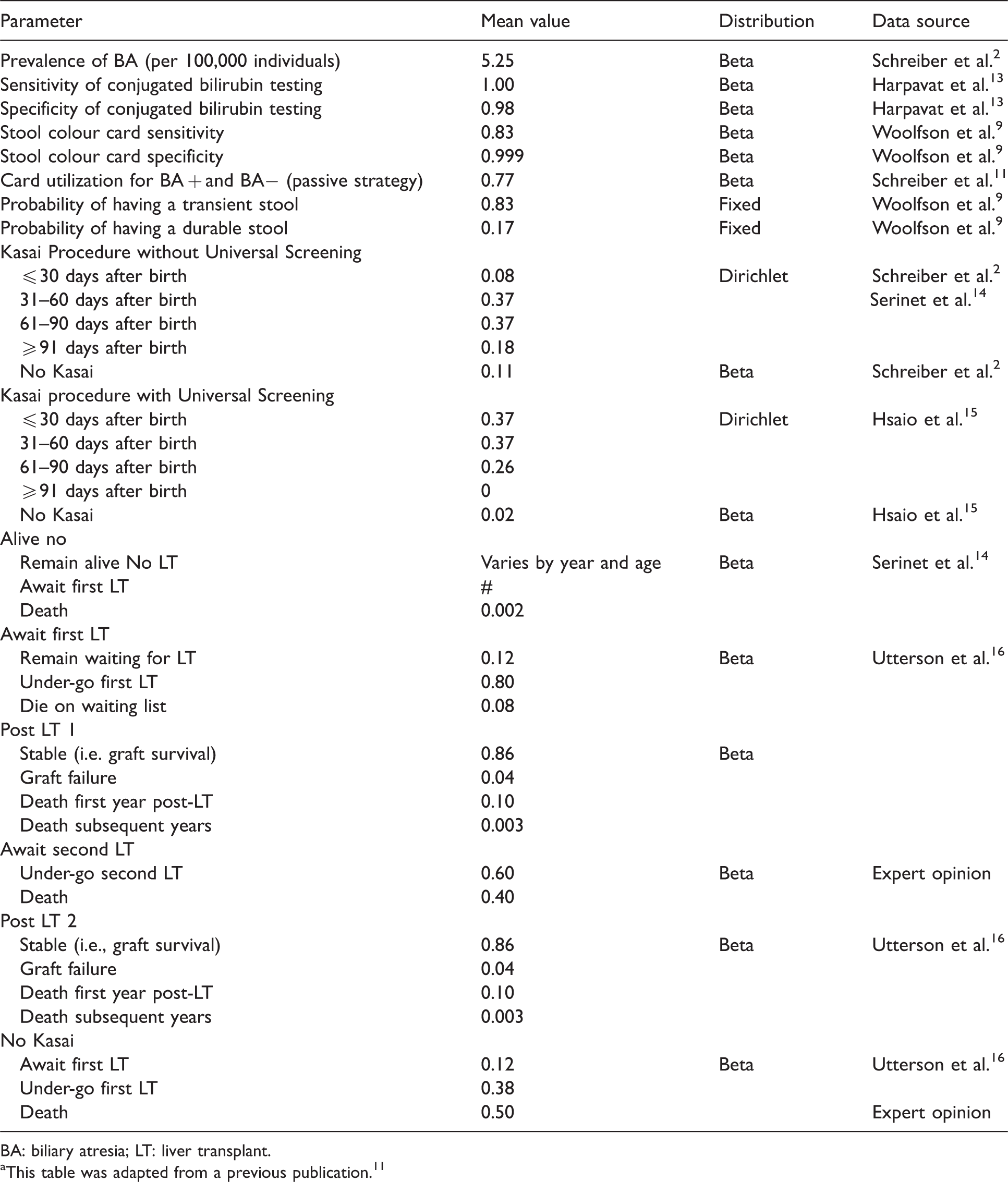
A combined decision tree and Markov model with yearly cycle lengths was developed using TreeAge DATA Pro. 11 The model simulated a cohort of newborns (born over a 12-month period) and followed the cohort over a 10-year time horizon (chosen to avoid extrapolation into adulthood, as a paediatric population was being modelled). The model structure was informed by existing literature and discussions with clinical experts. To model the different screening strategies, we first distinguished between true BA positive and true BA negative cases. Parents’ use of the colour card (utilization rate) was considered. Patients who received a KP are modelled by age at KP (≤30 days, 31–60 days, 61–90, and ≥91 days after birth). The Markov model (Figure 1) consisted of six states: alive no liver transplant, awaiting first liver transplant, post first liver transplant, waiting for a second liver transplant, post second liver transplant, and death. After a patient with BA receives a KP, they move to the “alive no liver transplant” state. At each yearly cycle, the patient has a probability of remaining in this state, awaiting a liver transplant, or death. The probability of moving to the liver transplant state is based on the age at Kasai. 2 BA patients who did not receive a KP move immediately to the “awaiting first liver transplant” state. The Markov process simulated infants’ move from one health state to another, the movements determined by the transition probabilities.
Markov model structure.
We used data from the literature or clinical expert opinion to assign costs and probabilities. Health benefits were expressed as life-years gained rather than quality-adjusted life-years, as utility values were not readily available for this patient population. The incremental cost-effectiveness ratio was calculated by dividing the difference in cost by the difference in health benefit between the screening strategies.
Two universal screening strategies were compared with no universal screening. The first was screening all newborn infants with conjugated bilirubin testing in hospital prior to maternity discharge. No prospective large screening study using conjugated bilirubin testing has been reported. A retrospective study found the sensitivity and specificity of testing to be 100% and 98%, respectively.10,13 The second strategy was home-based screening with the ISCC using a passive card distribution strategy. In this strategy, the ISCC is distributed at the hospital on maternity discharge. The family is instructed to monitor their infant’s stool colour through the first 30 days of life and to directly contact the screening centre by phone (toll free), email, or fax if an abnormal stool colour is detected. The card utilization rate was estimated at 77% (60–94%), and based on data from a Canadian home-based ISCC screening program at British Columbia Women’s Hospital. 11 The sensitivity and specificity of the ISCC in British Columbia were reported to be 83% and 99.9%, respectively (Table 1).
The probabilities of having a KP and having a liver transplant were obtained from the published literature (Table 1). The proportion of individuals that receives a KP by age group was obtained from a Canadian study that grouped ages 31–90 days together, and so it was assumed that the estimates were the same for age groups 31–60 and 61–90 days after birth. 2 These estimates are in accordance with a large national French study that reported the age at KP for 685 patients. 14 The probability of dying whilst waiting for a liver transplant was taken from a study examining the post-transplant mortality and graft failure among patients with BA. 16 Age- and sex-specific background mortality rates were taken from the Canadian life tables, to reflect underlying mortality. 17
The costing perspective was that of the publicly funded health care system, and so out-of pocket costs to the patient were not considered. Costs are reported in 2015 Canadian dollars and discounted by 1.5%, according to conventional Canadian guidelines. 18 Where applicable, costs were inflated to 2015 Canadian dollars using the Consumer Price Index for health care services in Canada. 19
The costs considered in this analysis are reported in Table 2. The cost of conjugated bilirubin testing was obtained from the British Columbia Ministry of Health schedule of fees for laboratory services (fee codes 91245 and 91250). 20 This fee of $3.19 included the total cost of procurement, testing, and interpretation. The cost of the home-based ISCC program consisted of card production as well as the administrative costs of two clerical assistants. The annual administration cost and card printing cost was $28,000 per 43,000 births, corresponding to $0.65 per patient.
Costs.
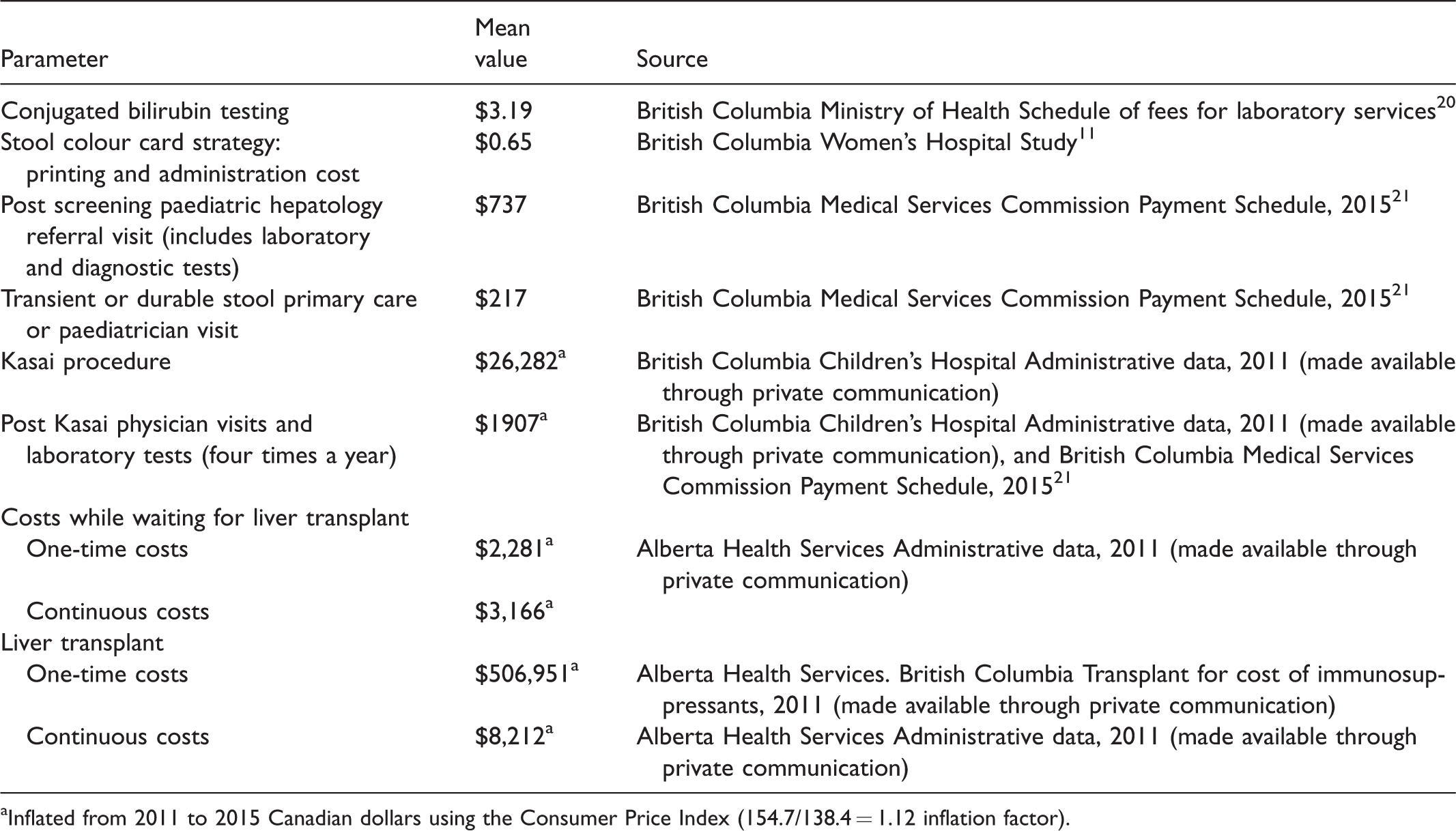
aInflated from 2011 to 2015 Canadian dollars using the Consumer Price Index (154.7/138.4 = 1.12 inflation factor).
For patients with an abnormal stool colour card, the cost of a health care visit as well as laboratory tests were considered, to determine whether the patient was BA positive. A proportion of those with false-positive tests will have a transient or durable abnormal stool colour. 9 For those who have a transient stool colour, a cost of $217 was applied for a physician visit to confirm that they do not have BA. 20 For those who have a durable stool colour, a cost of $737 was applied for a subspecialty physician visit as well as laboratory and diagnostic tests. These costs were obtained from the British Columbia Medical Service Commission Payment Plan and schedule of fees for laboratory services.20–22 For patients who were BA positive and test positive (true positive), there were no additional follow-up diagnostic costs, as these cases would proceed to KP.
The costs associated with the KP were obtained from the administrative records of British Columbia Children’s Hospital and included costs associated with the surgery, inpatient stay, medications, and radiology. The cost of post KP physician visits and laboratory tests were also considered, at a frequency of four times per year.
The cost of awaiting liver transplantation was obtained from the administrative databases of Alberta Health Services and included a one-time cost for a transplant assessment and a continuous cost (yearly) for physician visits. Liver transplantation costs included a one-time cost of organ procurement and liver transplantation. A continuous cost (yearly) of immunosuppression therapies was obtained from British Columbia Transplant, and outpatient physician visits as well as laboratory work were obtained from Alberta Health Services. Alberta Health Services costs were used for liver transplantation because paediatric liver transplants for British Columbia patients are performed in Alberta.
For the base case, a probabilistic analysis was conducted to assess the uncertainty of all parameters. A deterministic one-way sensitivity analysis was conducted on key model parameters such as sensitivity and specificity, administration costs, card utilization rate, liver transplant cost, conjugated bilirubin testing cost, and time horizon. To assess the robustness of the model, the upper and lower estimates were used, rather than median values. The published literature was used to determine the upper and lower estimates for each probability estimate. In the absence of published literature, the upper and lower estimates for each cost parameter were based on clinical input. The threshold value at which the policy results would change is also reported. A probabilistic analysis was run as the base case, applying, where possible, a relevant distribution for each model parameter. A beta distribution was used for binomial data, and a Dirichlet distribution was used for multinomial data. Through Monte Carlo simulations (sampling 10,000 times), values were selected at random from assigned distributions. The expected costs and life-years gained were estimated for a Canadian population of 392,902 births per year.
The incremental cost-effectiveness ratio was calculated for each screening strategy. In judging the cost-effectiveness and value-for-money, we used a conventional Canadian willingness to pay threshold of $50,000 per life year gained. We estimated the likelihood that each strategy was more favourable across a range of Willingness-to-pay thresholds, using cost-effectiveness acceptability curves.
Results
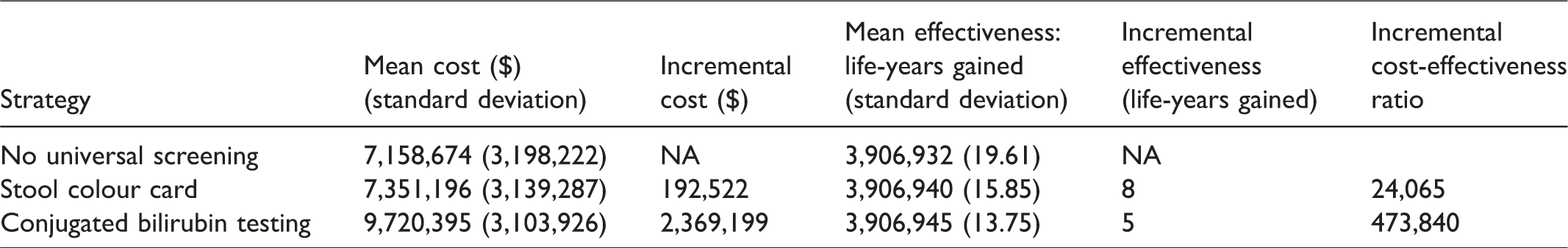
Table 3 reports the expected costs, life-years gained, and incremental cost-effectiveness ratio for each screening strategy. The mean cost of the no universal screening strategy was $7,158,674 and the mean life-years gained were 3,906,932. The costs were comprised of the KP and liver transplantation costs. The ISCC strategy cost $192,522 more compared with no screening but led to eight more life-years gained (ICER = $24,065 per life year gained). The screening strategy with conjugated bilirubin testing versus the ISCC cost $2,369,199 more, and led to five more life-years gained (ICER = $473,840 per life year gained). The results indicate that compared with no screening, using an ISCC was cost-effective.
Expected incremental costs and life years gained of universal screening per 392,902 births in Canada.
Supplemental Table 1 reports the number of transplants for each strategy. The conjugated bilirubin testing resulted in four fewer liver transplants than no universal screening. The home-based ISCC strategy resulted in two fewer liver transplants.
Figure 2 shows the cost-effectiveness acceptability curve, which illustrates the uncertainty surrounding the estimate of cost-effectiveness. The plot indicates a 75% probability that the passive universal screening strategy is cost-effective at a willingness to pay threshold of $50,000 per life year gained. Further, it shows that at any threshold value above $30,000 per life year gained, ISCC screening is cost-effective compared with no universal screening.
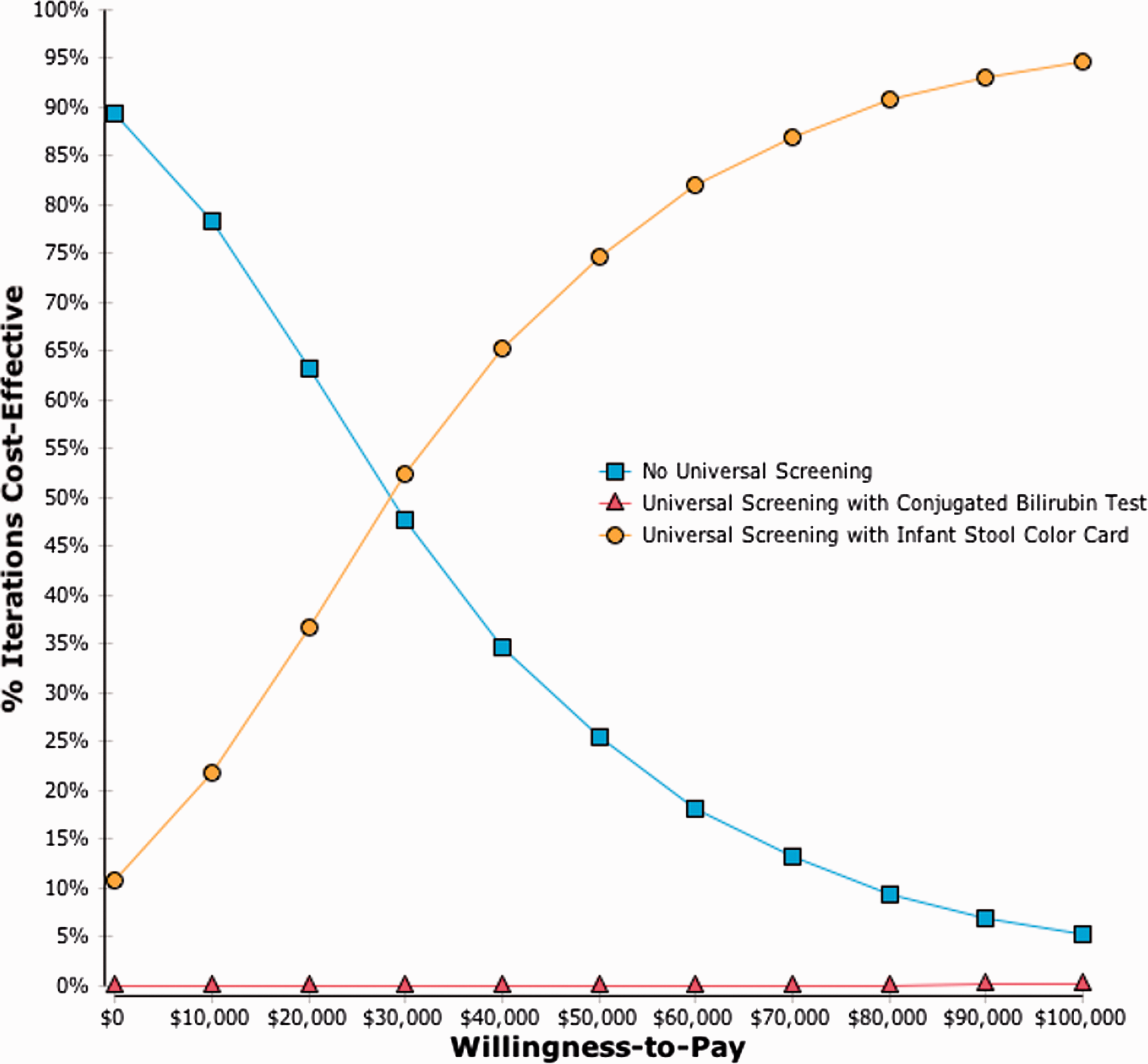
Cost-effectiveness acceptability curve.
Supplemental Table 2 reports the results of the one-way sensitivity analysis. The results were sensitive to the ISCC specificity. If the specificity is reduced to 0.98, the universal home-based ISCC screening strategy is no longer cost-effective. If the specificity of the conjugated bilirubin testing was increased to 100%, this screening strategy would still not be a cost-effective option (and therefore no policy change). If the cost of the conjugated bilirubin testing was reduced to $1.58, this strategy would not be cost-effective. The one-way sensitivity analysis indicated that the model results were not sensitive to variations in the card utilization rate, sensitivity of the ISCC, sensitivity and specificity of the conjugated bilirubin test, liver transplantation cost, time horizon, administration costs, and conjugated bilirubin test cost.
Discussion
We used a decision tree and Markov model to evaluate whether a universal screening program for BA, using conjugated bilirubin testing or a home-based ISCC, was cost-effective compared with no universal screening. 11 The results indicate that an ISCC screening strategy with a passive card distribution at maternity discharge is highly cost-effective. The ISCC screening design is home-based and family centred, without the need to send biological specimens. It appears fairly inexpensive to administer, and any costs are offset by savings in liver transplant costs. The conjugated bilirubin testing did not appear to be cost-effective compared with no universal screening, due, in part, to the high costs of the test that is applied to every newborn.
A strength of our study is that the model was developed in consultation with clinical experts in hepatology, and thus closely represents clinical reality. In addition, the model underwent an extensive validation process, with comparison made of model predictions and screening outcomes.
Despite these strengths, there are five limitations of the model. (i) Parameter estimates were based on best available evidence on the costs and the effects. Estimates for having a second liver transplant, as well as waiting for a first liver transplant when there was no KP performed, were based on expert opinion, as these estimates were not available in the literature. There was consensus across clinicians who reviewed the model that these estimates were reasonable. (ii) While the colour card screening program at British Columbia Women’s Hospital may be representative of the reported British Columbia provincial wide screening program, it may not be representative of the general population of Canada. However, we conducted a one-way sensitivity analysis on the card utilization rates to test the robustness of the results to variation in input parameters. The one-way sensitivity analysis, with the utilization rate of 0.70 to account for any potential recall bias, indicated that that the model results were not substantially different due to variations in the card utilization rate (i.e. there was no policy change). We also varied the sensitivity of the ISCC from 0.65 to 0.97 and found that the ISCC remained cost-effective. (iii) The time horizon was restricted to 10 years, as we were modelling a paediatric population. Increasing the time horizon beyond 10 years would have required us to obtain data on costs and outcomes for adult populations and, as an example, the reported outcomes for those who had a KP and liver transplantation in adulthood were not readily available. (iv) We applied the same costs to all patients irrespective of age, not taking into consideration the costs of liver transplantation for younger infants, who may require more intensive care. However, in jurisdictions where the liver transplantation is more intensive and expensive in younger patients, the screening programs would be even more economically attractive. In addition, we used a 10-year time horizon in order not to model liver transplantations that may take place in adulthood. (v) The cost of having a false-positive result may be overestimated, as a physician visit cost was applied. While a false-positive stool result from a transient stool change might be assumed to generate more costs than a single physician visit, our experience with the program suggests that the vast majority of such cases are readily handled by the screening centre by telephone and do not require a physician visit or any additional laboratory testing. Furthermore, should a family choose to see their doctor rather than contact the screening centre, most family physicians and paediatricians in Canada would request limited investigations for such cases. If we exclude the cost of a physician visit for these cases, the ISCC strategy would become even more economically attractive. In contrast, we may have underestimated the cost of the conjugated bilirubin screening, as the current screening process may include a second stage bilirubin test for those newborns who initially screened positive before maternity discharge. 13
There have been published cost estimates of the KP, as well as liver transplantation for newborns diagnosed with BA. Schreiber and Kleinman calculated the cost of hospitalization and surgical care in the first year of life after the KP to be between $17,500 and $20,000 (US), 23 and the average cost of a transplant to be $225,000 (US). These cost estimates are lower than ours, as they do not include the cost of medical care after the KP and pharmaceutical costs. A further study from France estimated the costs of liver transplant in the first year to be 100,000 Euros ($113,416 US) with annual follow-up charges of 20,000 Euros ($22,683 US). 14
Two studies have examined the cost-effectiveness of a screening strategy for BA. The first study was our own, assessing only the cost-effectiveness of an ISCC in Canada. 11 A second study examined the cost-effectiveness of a hypothetical stool colour card screening strategy in the United States 12 and found that nationwide screening with an ISCC was less costly and more effective than no screening. The study was conducted from a societal perspective, which included costs to the patient. The ISCC production cost was $0.03, with the program administration (for registry and paid staff) and education costs at $125,000 and $25,000, respectively. The cost of the ISCC program was $0.07 per patient, based on the approximate 4.2 million annual births in the USA. Our cost per patient was $0.65, which included the cost of card printing and administration. While the USA ISCC program costs are considerably less than in Canada, it is important to note that our costs came from a study implementing the ISCC. If this lower cost was applied to our model, it would make the ISCC strategy even more economically attractive.
Conclusion
The results of our study suggest that universal screening for BA using an ISCC with a passive card distribution can be highly cost-effective, on the assumption that it can be administered at a low cost and can achieve screening performance levels comparable with those reported in British Columbia. Screening will reduce the number of late referrals for the KP, and subsequently, the number of transplants needed. In addition, because this strategy may obviate the need for liver transplantation, it will lower the burden of organ availability. This screening strategy may help to lower waitlist times and mortality across the spectrum of liver disease, as it will free up organs to children with other diseases. The impact of this secondary gain makes the screening strategy even more economically attractive.
Supplemental Material
Supplemental material for Universal screening of newborns for biliary atresia: Cost-effectiveness of alternative strategies
Supplemental Material for Universal screening of newborns for biliary atresia: Cost-effectiveness of alternative strategies by Lisa Masucci, Richard A Schreiber, Janusz Kaczorowski, JP Collet and Stirling Bryan in Journal of Medical Screening
Footnotes
Acknowledgements
The authors thank Dr Mei Hwang Chang for her input on the model structure and parameters, and Dr Mohsen Sadatsafavi for his generous statistical support and guidance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Supplemental Material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.