Abstract
Objective
Beta thalassaemias are a group of hereditary red cell disorders resulting in a reduced or absent production of the main adult haemoglobin, adult haemoglobin. In England, the NHS Sickle Cell & Thalassaemia Screening Programme recommends reporting newborn beta thalassaemia disease as an incidental finding when detected whilst screening for sickle cell disease. The current action value to initiate further investigations is 1.5% adult haemoglobin, using high-performance liquid chromatography or capillary electrophoresis. We examined the reliability of this action value.
Methods
A 44-month country-wide prospective study using data from 13 newborn screening laboratories in England.
Results
There were 81 cases reported with an adult haemoglobin of 1.5% or less at first-line screen, of which nine were lost to follow-up. The six false-positive results were all of 32 weeks’ gestation or less. Of the 66 true-positives, 36 had confirmatory molecular results (11 of these cases also have results from tandem mass spectrometry), 19 had clinical confirmation and 11 had the results of both parents available which were consistent with the screening result. There was one false-negative, a confirmed beta thalassaemia major case with an adult haemoglobin of 1.7%, above the action value at first-line screen but known to be at risk from parental results and therefore referred into clinical care by the laboratory.
Conclusions
This study demonstrates a positive predictive value of 91.7%, with a specificity of 99.9% and a sensitivity of 98.5%. These results confirm the reliability of the current action value.
Keywords
Introduction
Beta thalassaemias are a group of hereditary red blood cell disorders resulting in a reduced or absent production of the main adult haemoglobin, Hb A. Although not specifically screened for, the disease state of these conditions is reported as an incidental finding if detected when testing the dried blood spot in screening for sickle cell disease. In England, this approach has been supported by the National Screening Committee. 1 Guidelines have been published for standards of clinical care, recommending early and accurate diagnosis as soon as possible after birth to ensure close monitoring of the child. 2 There are limited reports on the benefits of this approach; however, clinical manifestations are reported to appear in infancy and include severe anaemia, jaundice, failure to thrive, irritability and decreased activity. Hepatosplenomegaly and frontal bossing are usually observed in untreated cases. 3
The amount of adult haemoglobin at birth can be highly variable and affected by a number of factors. Significantly reduced levels are seen in premature babies and may cause false-positives. This may also occur in cases with haemoglobin variants where the normal adult fraction is reduced because it is divided into normal and variant fractions.
In a previous study, the NHS Sickle Cell & Thalassaemia Screening Programme reviewed 170 babies with an Hb A of 1.5% or less and reported a positive predictive value of 87% and sensitivity of 99%. The study combined prospective and retrospective data and was limited by coverage, data availability, and numbers of positive cases. 1 Here we aim to review the recommended action value.
Methods
To review the recommended action value of less than or equal to 1.5% Hb A, a prospective country-wide study was established in 2012 to collect Hb A values in all cases. DNA results were included where available. During the course of this study, tandem mass spectrometry (MSMS) was approved for use in the UK.4,5 One laboratory submitted results using this technique. Prior to introduction, studies were carried out to ensure consistency of action values between the existing and new techniques. 4
Anonymised data on all babies with an Hb A of less than 3% and a haemoglobin pattern of Hb FA or Hb F only were requested from all newborn screening laboratories in England (n = 13) from April 2012 to December 2016. Data collected included first- and second-line screening results, the Hb A%, and the gestational age, with MSMS, DNA, parental results and clinical outcome if available. The average annual birth rate during this time was 699,619. 6 All Hb A values of less than or equal to 1.5% were reviewed. Samples were tested following the protocols outlined in the NHS Sickle Cell & Thalassaemia Screening Programme Handbook for Laboratories in use at this time. 7 Results for Hb A% were generated by either high-performance liquid chromatography (HPLC) or capillary electrophoresis. Results from MSMS are expressed using the gamma:beta wild-type ratio, as previously described.4,5 All MSMS cases had corresponding HPLC results.
Results
Although collected, the results between 1.5 and 3% have not been included in this report (n = 440), unless infants were suspected to have beta thalassaemia disease (n = 1) or subsequently presented clinically (n = 0). A total of 81 cases were reported with a first-line screen Hb A of 1.5% or less; values ranged from 0 to 1.5%, with a median of 0%. In 36 cases, DNA analysis was performed, and they were shown to be affected. All cases had homozygous or compound heterozygous beta thalassaemia mutations (see Table 1), and 11 of these cases also had results of the MSMS T2 wild-type gamma/beta ratio. All MSMS results were above the designated action value of 4.0, 5 range 8.1 to 89.4, median 41.1.
Mutation results.
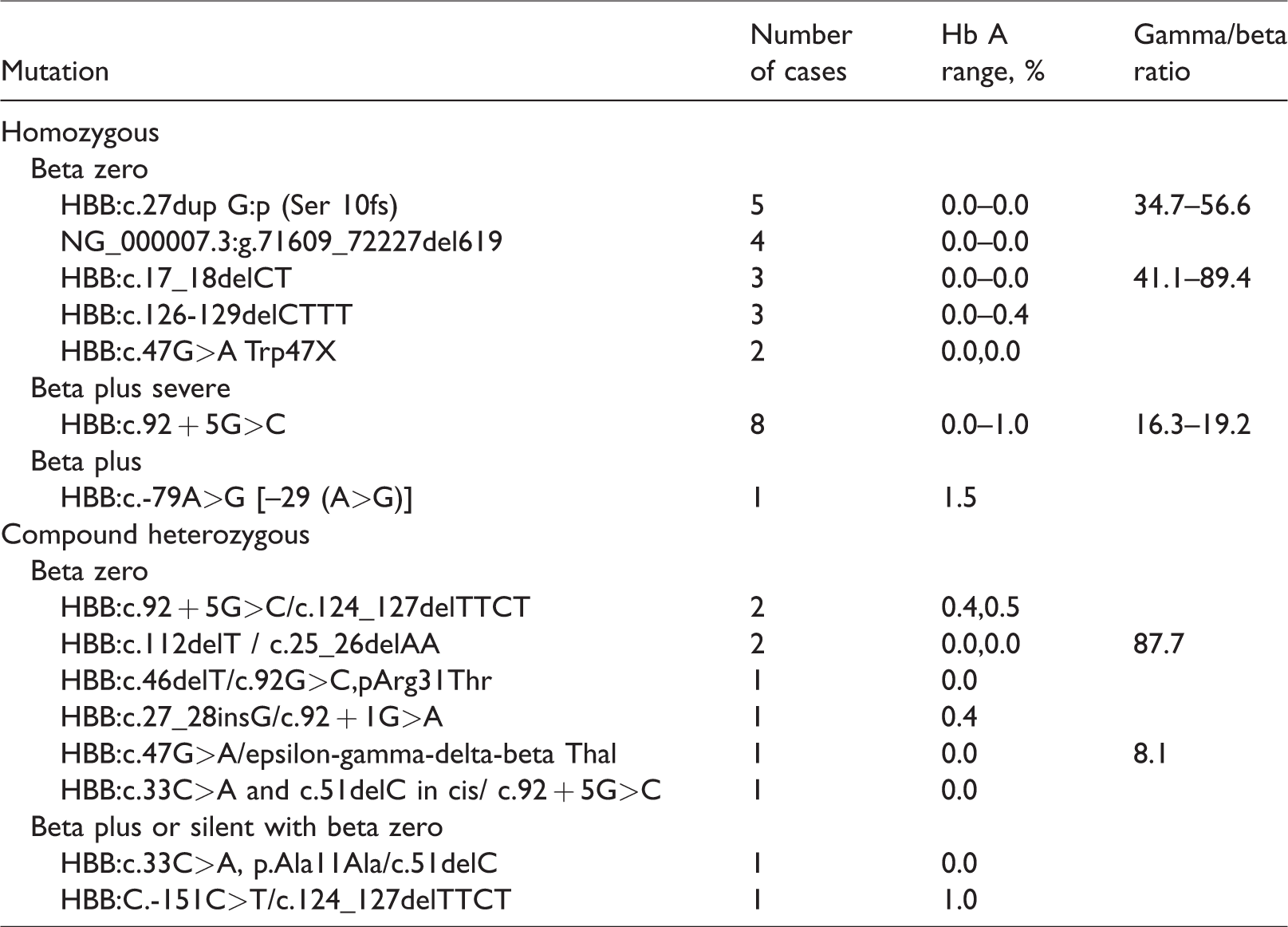
Of those without DNA, 19 cases had clinical confirmation of beta thalassaemia disease, 11 of these with supporting parental results. A further 11 had the results of both parents available, and parental results were all consistent with the screening result of beta thalassaemia disease. Nine were lost to follow-up.
There were six false-positives (see Table 2), all at 32 weeks’ gestation or less. The two lowest Hb A% values obtained were a set of twins born at 32 weeks (Hb A = 0.4%). Although other twin cases were reported in this category, the results were not available for both siblings.
False-positive screening results.

There was one false-negative, a confirmed beta thalassaemia major case with an Hb A of 1.7%, above the action value at first-line screen but known to be at risk from parental results. This case was passed for second-line testing, referred to clinical care and subsequently received a bone marrow transplant. No further false-negative cases have been reported to the Screening Programme. Results are summarised in Figure 1.
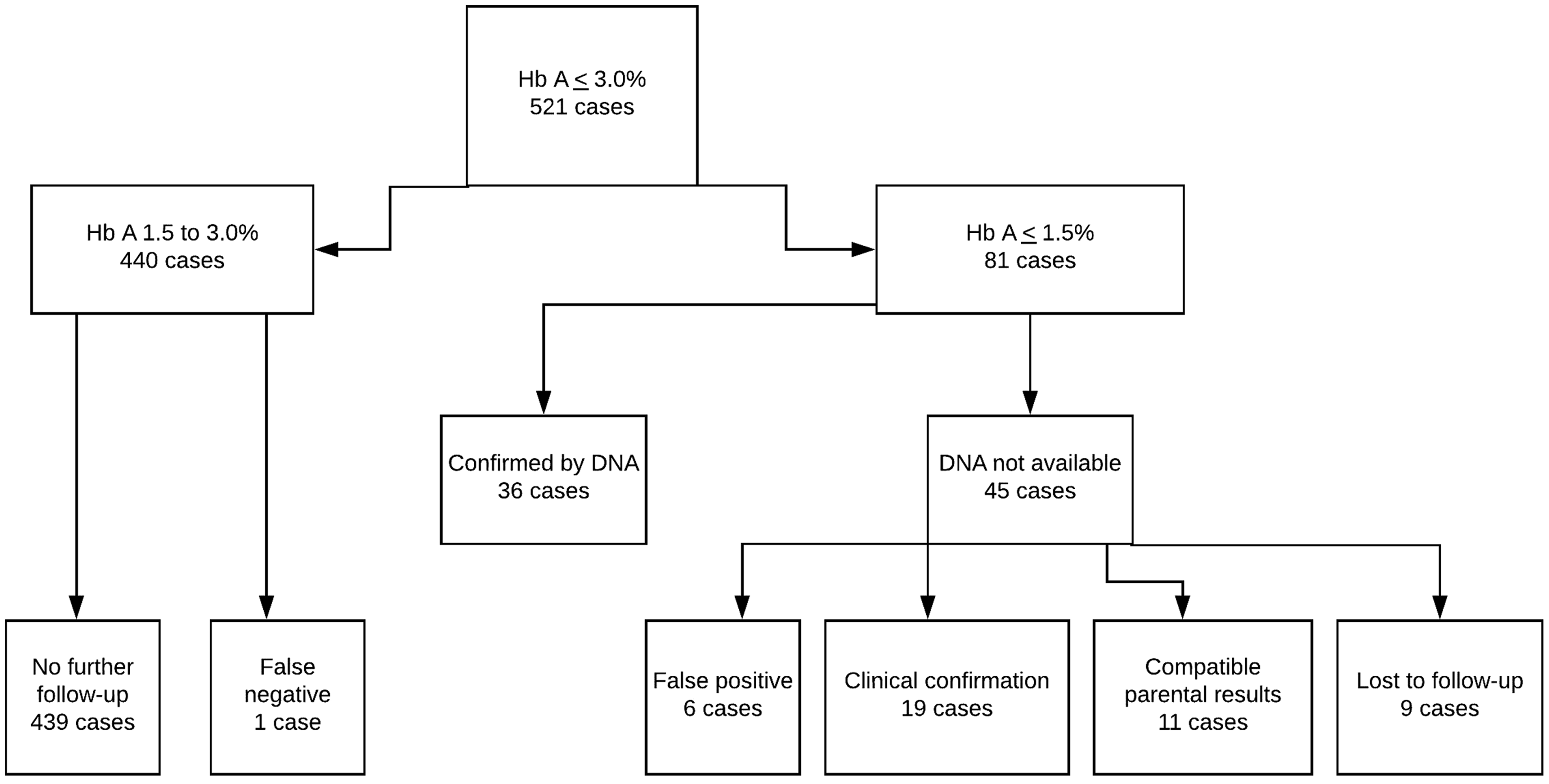
Flow chart of cases.
Excluding cases lost to follow-up, the estimated positive predictive value of the screening test is 91.7%, with specificity of 99.9% and a sensitivity of 98.5%.
Discussion
Due to the low incidence of beta thalassaemia disease in England, evidence to support the reliability of the designated Hb A% action value has been limited. Robust figures on the prevalence of beta thalassaemia disease in England are not available. Annual data reports 6 from the NHS Sickle Cell and Thalassaemia Screening Programme show 74 unconfirmed beta thalassaemia disease screen positive cases in the last three years. These figures correlate well with the cases reported in this 44-month study.
Our previous study investigated 170 babies who were screen positive for beta thalassaemia disease and reported a positive predictive value of 87% and sensitivity of 99%. 1 The findings suggested that an Hb A of 1.5% or less was a reliable screening test for these conditions. This current study, which includes molecular results where available, was undertaken to prospectively test this value and increase the body of evidence. Unlike the previous study, Hb A% was available in all cases; however, limitations remain: it was not possible to obtain molecular confirmation on all results, and some babies were lost to follow-up. The positive predictive value of 91.7% obtained in this study is higher than that of the previous study 1 and compares favourably with other conditions, such as Medium-chain acetyl-CoA dehydrogenase deficiency, for which values of 77% have been reported. 8
In contrast to the previous study, 1 no cases presented unexpectedly or were identified through incident reporting, suggesting there were no false-negatives during this period. However, as previously, there was a single case with an Hb A of 1.7%, just above the action value, but known to be at risk from parental results. This case was referred into clinical follow-up and confirmed as beta thalassaemia major. Such cases illustrate the benefits of a linked antenatal and newborn programme.
Screening protocols are designed to maximise the detection of true-positive cases and minimise numbers of false-positive and false-negative results. The action value of 1.5% Hb A is at the limits of sensitivity for the methods in use and is affected by the foetal to adult haemoglobin switch, with premature babies known to give false-positive results. This study identified six false-positive results, all of 32 weeks’ gestation or less; however, prematurity cannot be used as exclusion criteria, as it is possible for a premature baby to also have beta thalassaemia disease. Historically, missed cases have been reported, but no missed cases have been reported to the screening programme during this study.
The Hb A% observed in the positive cases was variable and did not appear to correlate well with the mutational status; however, the action value is at the limits of testing sensitivity for the instruments. Instrument manufacturers do not report the limits of sensitivity for Hb A, but for haemoglobin variant detection, this is reported as 1.0%. 9
Due to increased uptake of MSMS newborn screening for sickle cell disease in England, approximately half of the population is now screened using this method. Although work has been carried out to ensure consistency of action values with existing methods, at this time, cases detected by MSMS first-line testing have HPLC for their second test. Further validation of the designated T2 gamma/beta ratio action value is required to ensure that at least the same level of reliability is maintained in the future.
Conclusion
This study adds a further 66 cases to support the action value of an Hb A of 1.5% or less, with one additional case which, although detected by the laboratory, is by screening criteria a false-negative. The study increases the available evidence for the use of the action value, and the high positive predictive value and sensitivity and specificity confirm the reliability. These data support the use of the 1.5% or less Hb A value for newborn screening for beta thalassaemia disease.
Footnotes
Acknowledgements
We thank all the newborn laboratories which provide data to the NHS Sickle Cell & Thalassaemia Screening Programme on an annual basis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
