Abstract
Objective
Several European countries are implementing organized colorectal cancer (CRC) screening programmes using faecal immunochemical test (FIT) and/or flexible sigmoidoscopy (FS), but the cost-effectiveness of these programmes is not yet available. We aimed to assess cost-effectiveness, based on data from the established Piedmont screening programme.
Methods
Using the Piedmont programme data, a Markov model was constructed comparing three strategies in a simulated cohort of 100,000 subjects: single FS, biennial FIT, or sequential strategy (FS + FIT offered to FS non-responders). Estimates for CRC incidence and mortality prevention were derived from studies of organized screening. Cost analysis for FS and FIT was based on data from organized programmes. Incremental cost-effectiveness ratios (ICER) between the different strategies were calculated. Sensitivity and probabilistic analyses were performed.
Results
Direct costs for FS, and for FIT at first and subsequent rounds, were estimated as €160, €33, and €21, respectively. All the simulated strategies were effective (10–17% CRC incidence reduction) and cost-effective vs. no screening (ICER <€1000 per life-year saved). FS and FS + FIT were the only cost-saving strategies, with FS least expensive (€15 saving per person invited). FS + FIT and FS were the only non-dominated strategies. FS + FIT were more effective and cost-effective than FS (ICER €1217 per life-year saved). The residual marginal uncertainty was mainly related to parameters inherent to FIT effectiveness and adherence.
Conclusions
Organized CRC screening programmes are highly cost-effective, irrespective of the test selected. A sequential approach with FS and FIT appears the most cost-effective option. A single FS is the least expensive, but convenient, approach.
Introduction
Colorectal cancer (CRC) represents a major cause of morbidity and mortality in Western countries, 1 resulting in substantial costs for surgery, chemotherapy, and palliative treatment. 2 CRC screening, using faecal tests or endoscopy, has been shown to be effective in reducing CRC incidence and/or mortality,3,4 and several European countries are implementing organized programmes adopting guaiac faecal occult blood test (gFOBT) or faecal immunochemical test (FIT), or flexible sigmoidoscopy (FS). 3 For a population-based intervention, assessment of the cost-effectiveness of available strategies is crucial to ensure the largest expected net benefit (NB). While most cost-effectiveness analyses are based on the reimbursement rates of the tests, 5 organized programmes involve additional costs, including the resources necessary to implement active invitation procedures for the whole target population, and to organize and monitor the programme, including information technology investment. Extensive data on the detection rate (DR) of polyps and already developed CRC, costs, and long-term outcomes of FS, gFOBT, and FIT from ongoing population based programmes3,6–9 provides a unique opportunity to assess their expected cost-effectiveness. This analysis aimed to compute the actual cost of organized FS and/or FIT programmes, and to assess their cost-effectiveness, based on real data extracted from an established screening programme.
Methods
The Piedmont screening programme combines FS and biennial, single sample, FIT (positivity cut-off: 20 µg Hb/gr. faeces). People aged 58 are offered FS once in the lifetime, and biennial FIT is offered until age 69 to those who did not respond to the FS invitation. 10 Piedmont data are representative of the CRC prevalence and screening DR in Northern-Central Italy.6,11,12
To assess the direct costs of FS and FIT, we calculated the yearly direct cost of FS and FIT programmes, performing an analytical computation of the different cost components of our regional population based programme: (i) programme organization and evaluation, (ii) endoscopy staff, (iii) equipment, (iv) endoscopy accessories, (v) disposable material, (vi) hospital furniture, (vii) FIT costs, and (viii) other components. We then divided these cost estimates by the expected number of subjects examined within one year. We ran separate estimates for first and subsequent FIT rounds, to account for the different positivity rates (i.e. total colonoscopy workload). The costs of FIT and FS scenarios included the costs of FS- and total colonoscopy-related polypectomies (including histology), based on the polyp DR assessed within the Piedmont region FS programme. Thus, although polyps were not considered for the efficacy assessment in our model, they were included in the cost analysis.
Detailed description of the cost analysis is reported in online Appendix 1.
We simulated the screening impact on a cohort of Piedmont residents aged 58 to 70 (Table 1). A simple Markov model was constructed, including three health statuses in the natural history compartment: no CRC, CRC, or death (CRC-related, or non CRC related). Age- and gender-specific CRC incidence and mortality rates, as well as CRC stage distribution in the non-screened population, were available from the Piedmont cancer registry. 13 These data were used to model the expected CRC incidence and mortality in the absence of screening. Age- and gender-specific all-cause mortality according to official Piedmont life-tables were applied for population attrition. 14
Model characteristics.
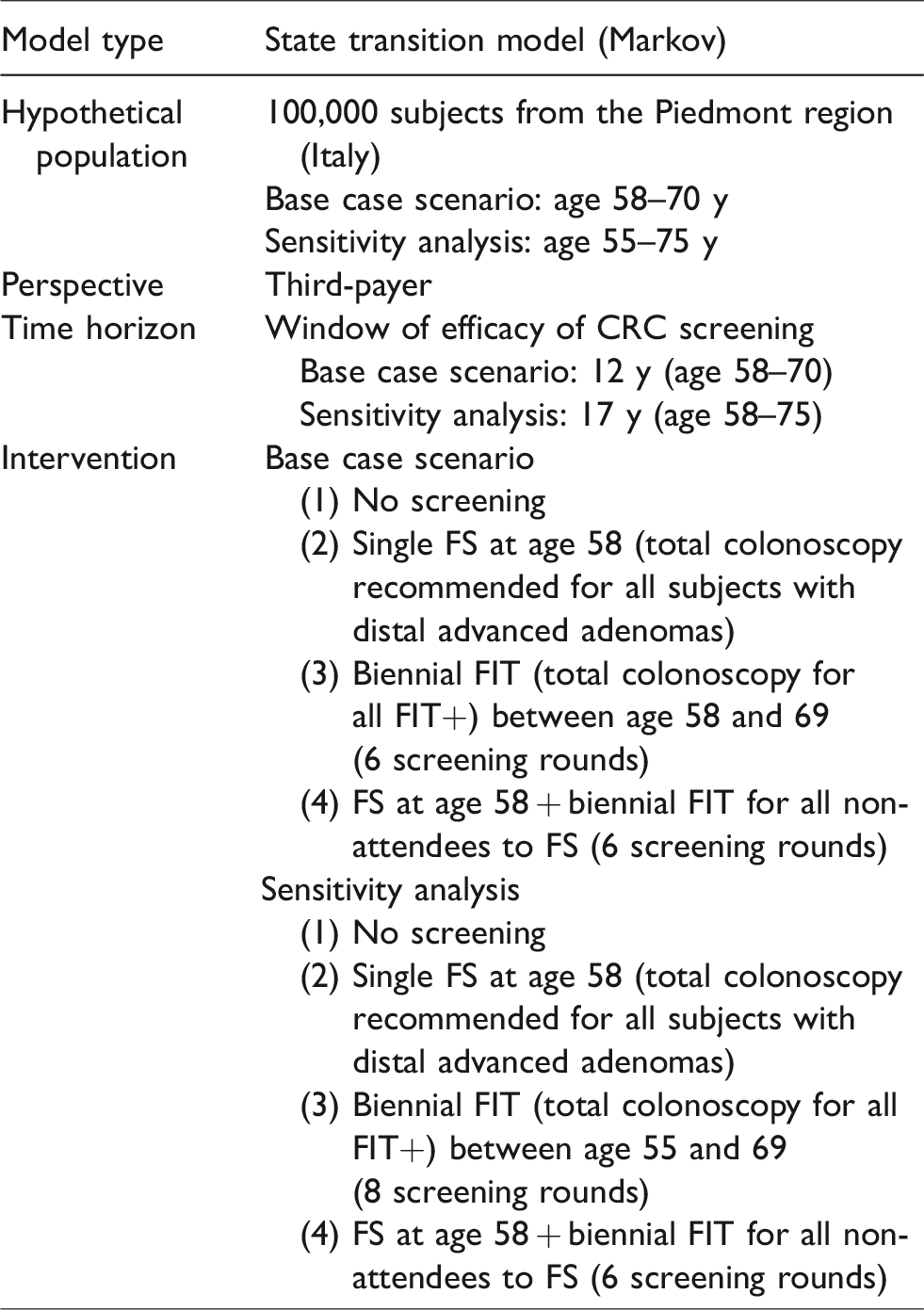
The health interventions superimposed on the natural history model were: (i) single FS at age 58, (ii) six rounds of biennial FIT between ages 58 and 69, (iii) combined strategy: single FS at age 58 with non-responders to FS invited to FIT every other year until age 69. All inputs and relative ranges are reported in Table 2. Online Appendix 2 provides a detailed discussion of the rationale for the model assumptions.
Parameters used for the reference case and probabilistic sensitivity analyses, reporting point estimates and ranges of the parameter distributions.
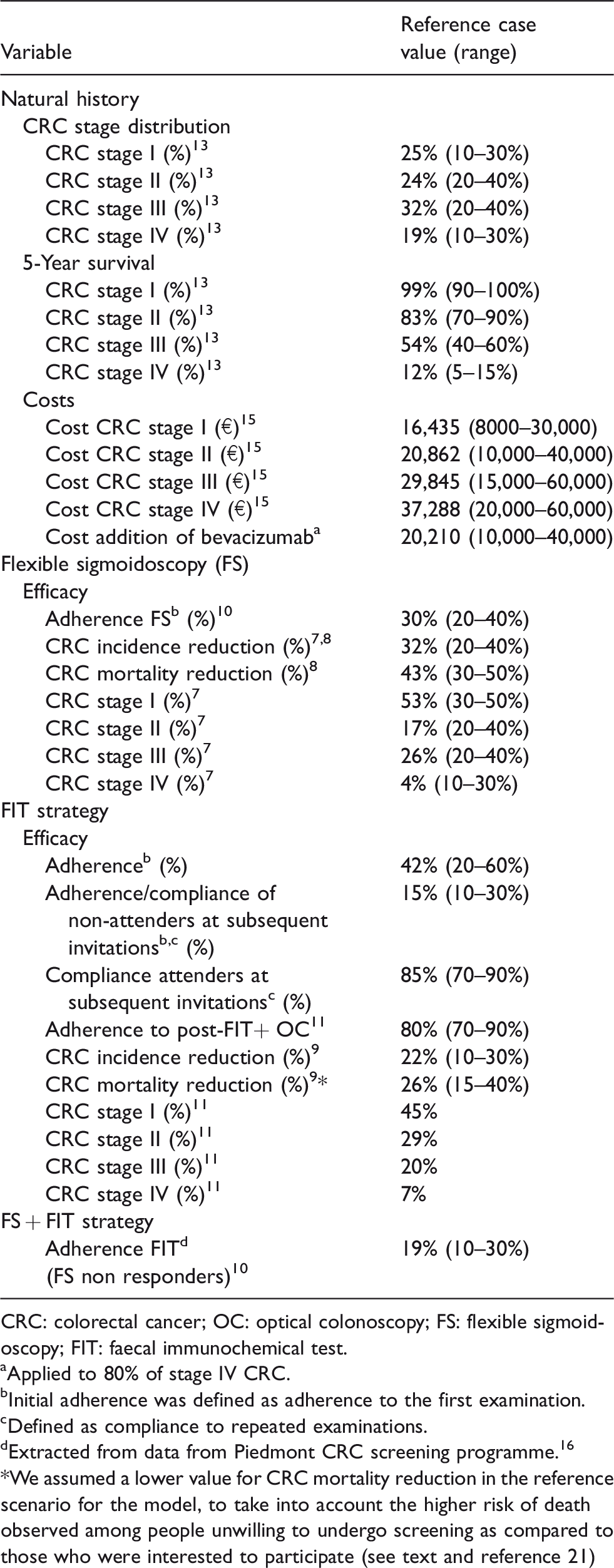
CRC: colorectal cancer; OC: optical colonoscopy; FS: flexible sigmoidoscopy; FIT: faecal immunochemical test.
aApplied to 80% of stage IV CRC.
bInitial adherence was defined as adherence to the first examination.
cDefined as compliance to repeated examinations.
dExtracted from data from Piedmont CRC screening programme. 16 *We assumed a lower value for CRC mortality reduction in the reference scenario for the model, to take into account the higher risk of death observed among people unwilling to undergo screening as compared to those who were interested to participate (see text and reference 21)
The time interval of the simulation was set at 12 years in the reference scenario, as it corresponds to the duration of the follow up of the FS screening trials,7,8 and of the only cohort study assessing FIT effectiveness. 9 As it also corresponds to the actual screening window in the Piedmont region, this time interval allowed us to base our simulation on real inputs from the programme.
The attendance rate in the reference scenario was set at 30% for FS, 42% for FIT, and 19% for the FIT invitation amongst FS refusers, as observed in the Piedmont programme. 10 In a recent Italian cohort study within an organized FIT based programme, 17 responders in each invitation round showed an 85% compliance at each subsequent invitation, while non-responders in previous rounds showed a 15% compliance with any of the subsequent invitations. Our model simulated cumulative adherence as the sum of the initial and repeated adherence over subsequent rounds.
To estimate FS screening effect amongst attendees, we used per protocol estimates, adjusted for non-compliance, of CRC incidence and mortality reduction reported in the European FS trials.7,8 In the model simulation, these effects started at the time of FS examination for screening attendees.
Although available evidence suggests that FIT has a higher sensitivity than gFOBT, both for CRC and advanced adenomas, 18 and it shows a high advanced adenoma DR over several screening rounds, 17 direct experimental evidence of a reduction of CRC mortality is available only for gFOBT. 4 To our simulated cohort FIT attendees we applied the effectiveness estimates derived from a prospective study conducted within an Italian FIT programme 9 reporting 22% and 41% reductions in CRC incidence and mortality, respectively, over 12 years amongst screening attendees, as compared with never attendees. These data of efficacy were applied to screening attendees in the simulated cohort. The simulated effect started at the beginning of the simulation for those who were adherent to the initial invitation, and from the date of their first test for those who accepted the invitation only at subsequent rounds. In this study, attendees had performed on average four tests, so that the estimates already included the cumulative effect of multiple tests. Screening efficacy was simulated to be equivalent throughout the follow-up time, as previously reported.19,20
FIT mortality reduction was derived taking into account down-staging of screen-detected CRCs amongst attendees. Based on the results of a recent study in our setting, 21 documenting a 30% lower risk of CRC death amongst unscreened people willing to participate in screening, compared with unscreened subjects not interested in screening, the impact of down-staging was reduced by 30%.
The only costs incurring in the ‘natural history’ compartment were CRC treatment costs, while for the screening compartment we computed the yearly direct cost of FS and FIT programmes (see above and Table 3), including costs for polypectomy and post-polypectomy surveillance. Recent Italian estimates of stage-specific CRC treatment costs 15 were integrated, including the offer of 10 bevacizumab therapy cycles for metastatic CRC, not yet adopted at the time of that analysis. 15
Clinical effectiveness of screening was measured in terms of life-years (LYs) gained, or down-staging of CRC. Both future costs and future LYs saved were discounted using a 3% annual rate. More costly and less effective strategies were ruled out by simple dominance. Strategies that were more costly and less effective than a combination of other strategies were ruled out by weak dominance. The relative performance of the remaining strategies was measured using the incremental cost-effectiveness ratio (ICER). A 50,000$ICER per LY gained was used as willingness-to-pay threshold to differentiate an efficient from an inefficient procedure. 22 Expected NB for each strategy was calculated as: number of LYs gained × willingness-to-pay − cost. The strategy with the highest NB in the reference scenario represents the optimal strategy with the available information.22,23
One- and two-way sensitivity analyses were performed for all the variables of the model; all efficacy and cost parameters were allowed to vary in the Monte Carlo analysis, according to the limits reported in brackets in Table 2, and the most relevant results were reported. We also performed a sensitivity analysis focused on adherence, or model structure (i.e. by varying the adherence rate, or the time horizon and the duration of protective effect of screening). The higher FS uptake rates reported in Norway, 24 England, 25 The Netherlands, 26 and Italy 10 were used in the sensitivity analysis. The time horizon of our simulation was extended to 75 years, assuming both for FS and FIT strategies the same protective effect as in the first 12 years. For FS, this assumption is supported by the recent update of the UK trial follow-up, showing that the protective effect of a single FS screening was not reduced at the 17-year follow-up. 27 For FIT, it is supported by the long-term follow-up of the gFOBT randomized trials,19,20 showing the persistence of the protective effect up to 30 years after the end of screening. We then compared a cohort aged 55–75, undergoing eight FIT rounds between ages 55 and 69, with a cohort undergoing a single FS at age 58.
To estimate the distribution of expected costs and efficacy of the screening strategies dependent on the uncertainty in the input parameters, we adopted the approach of value-of-information analysis,28,29 which estimates and combines the likelihood of making a wrong decision, and the associated foregone benefit (see also online Appendix 3). The expected value of perfect information (EVPI) estimates the value of removing all uncertainty related to the decision problem, representing the upper limit of the amount the decision maker should be willing to pay to eliminate all uncertainty in the decision. Monte Carlo simulation was used to repeatedly sample from the distributions assigned to all the uncertain parameters shown in Table 2–3. The expected value of a decision taken with perfect information (i.e. the identification of the optimal test) was found by averaging the maximum NB over the joint distribution of the included parameters. The difference between the maximum NB with perfect information and the maximum NB with current information corresponds to the total EVPI per subject. An analysis of the EVPI associated with particular subsets of model parameters was also conducted (partial EVPI), to indicate what type of additional evidence would be most valuable. The model was simulated by using Excel spreadsheets (Microsoft Corp., Redmond, Washington) and @risk 5.0 (Palisade Corp., Ithaca, NY).
Costs a of the screening strategies, modelling assumptions, and parameters for sensitivity analysis and assumptions.
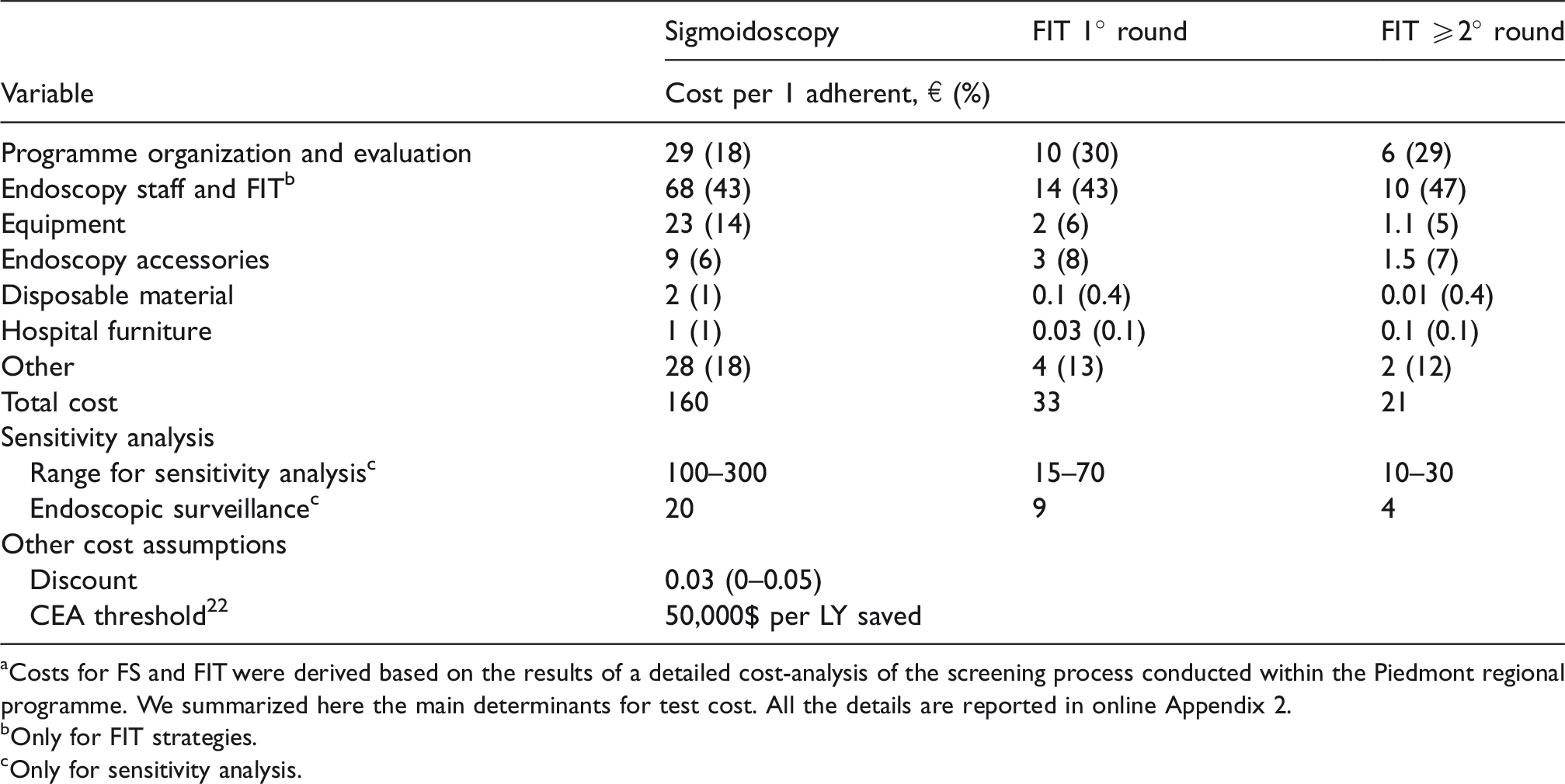
aCosts for FS and FIT were derived based on the results of a detailed cost-analysis of the screening process conducted within the Piedmont regional programme. We summarized here the main determinants for test cost. All the details are reported in online Appendix 2.
bOnly for FIT strategies.
cOnly for sensitivity analysis.
Results
Table 3 shows the costs of FS and FIT. The direct cost of FS was estimated at €160 per adherent. The largest contribution related to the cost of the endoscopy staff and equipment (€68), while a minor contribution was due to the programme organization. The direct cost of the FIT programme was €33 and €21 per adherent at first and subsequent rounds, respectively. This difference was mainly explained by the higher FIT positivity at the first compared with subsequent rounds.
Table 4 shows screening efficacy. FS screening resulted in a 10% and 13% reduction in CRC incidence and mortality, respectively, totalling 1207 discounted LYs gained (4.4 days per person invited). FIT alone resulted in a reduction of CRC incidence and mortality of 14% and 16%, respectively. FIT appeared to be more effective than FS, resulting in additional 253 (0.9 days per person invited) discounted LYs gained. The FS + FIT strategy appeared to be the most effective option, associated with 17% CRC incidence and 22% CRC mortality reduction, resulting in an additional 752 (2.7 days per person invited) discounted LYs gained, compared with FS alone, and an additional 499 (1.8 day per person invited) LYs saved, compared with the FIT alone scenario.
Cost, effect and net benefit for all the included strategies for a cohort of 100,000 subjects invited for screening.
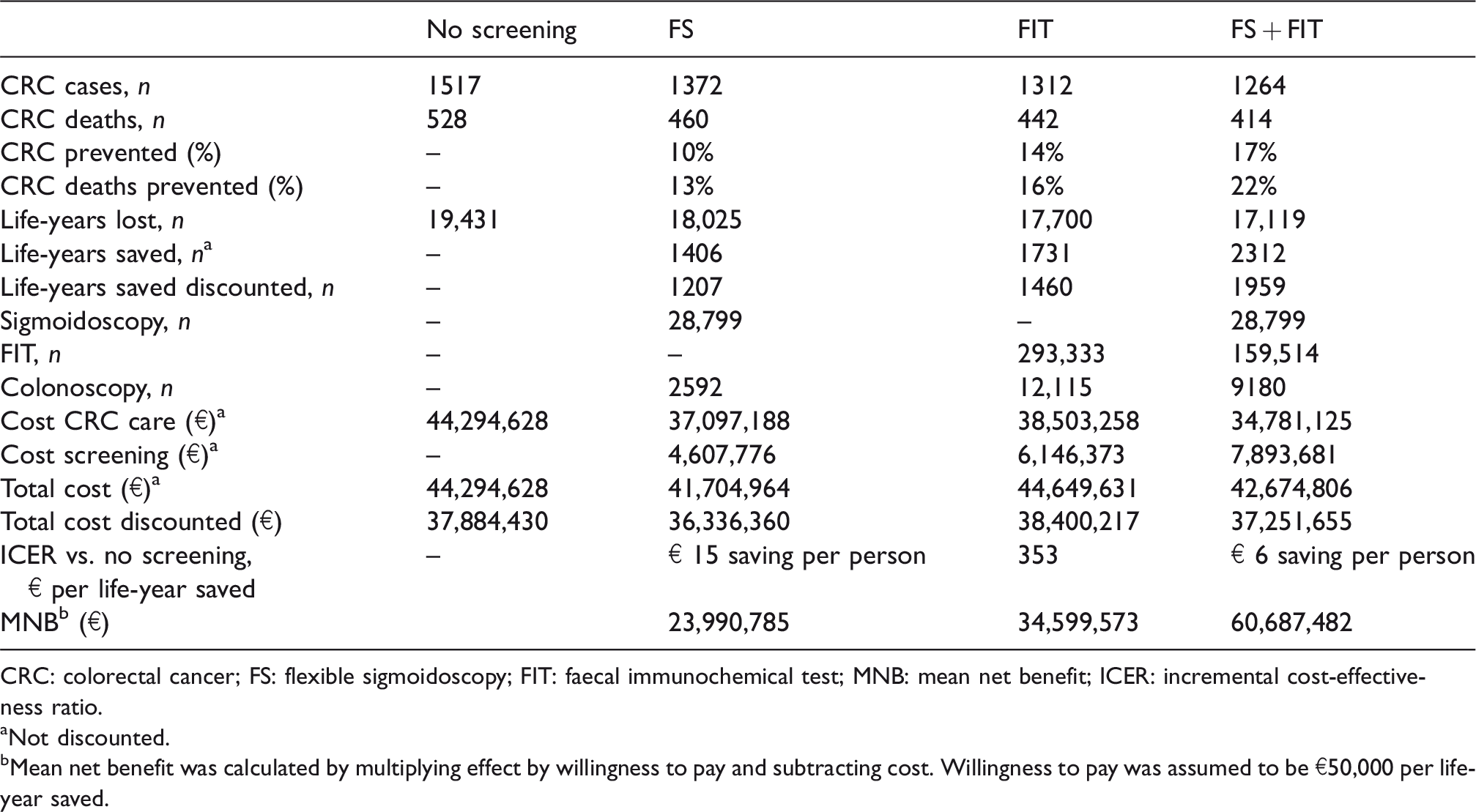
CRC: colorectal cancer; FS: flexible sigmoidoscopy; FIT: faecal immunochemical test; MNB: mean net benefit; ICER: incremental cost-effectiveness ratio.
aNot discounted.
bMean net benefit was calculated by multiplying effect by willingness to pay and subtracting cost. Willingness to pay was assumed to be €50,000 per life-year saved.
All the screening strategies were associated with a substantial reduction in CRC costs, ranging between 13% and 21%, corresponding to a €58–€95 undiscounted saving per person invited, compared with no screening. Cost reduction was related to the expected screening impact on CRC incidence, which was substantially higher using FS than using FIT alone. This was offset by the cost of screening, ranging between €46 and €79 (undiscounted) per person invited. The overall discounted cost was lower than in the no screening scenario for FS and FS + FIT, but higher using FIT alone.
When comparing screening with no screening, all strategies appeared to be cost-effective, with an ICER below €1000 per LY saved (Table 4); FS and FS + FIT strategies also resulted in savings, of €15 and €6, respectively. When ranking the simulated strategies, FS appeared to be the least expensive cost-effective option, being more effective and less costly than no screening (Table 5). FS + FIT appeared to be more cost-effective than FS alone, with an ICER of €1217 per LY saved. The FIT only strategy was dominated by the combined strategy, despite representing a cost-effective alternative to FS alone (Figure 1, scenario 1).
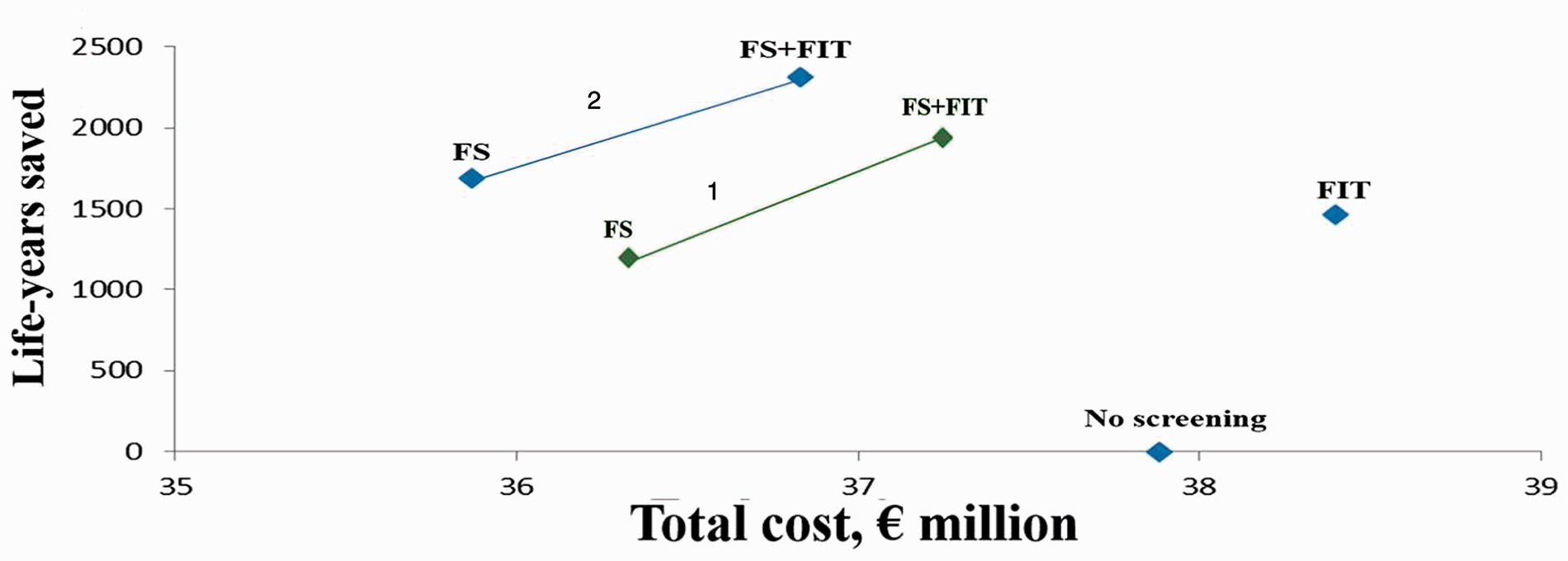
Cost-effectiveness of the different strategies. Scenario 1 (reference) – adherence: FS = 30%; FIT alone = 42%; FS + FIT − FS: 30% − FIT (FS–): 19%. Scenario 2 – adherence: FS = 42%; FIT alone = 42%; FS + FIT − FS: 42% − FIT (FS−): 19%. Non-dominated strategies are connected by a continuous line.
Cost-effectiveness of the different strategies for CRC screening in the simulated cohort of 100,000 subjects. a
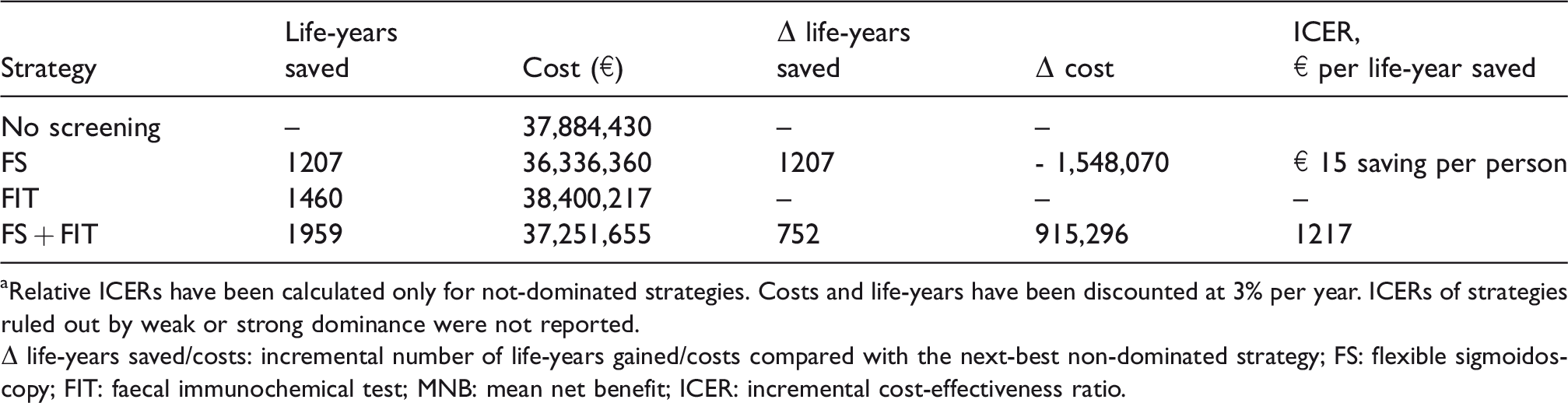
aRelative ICERs have been calculated only for not-dominated strategies. Costs and life-years have been discounted at 3% per year. ICERs of strategies ruled out by weak or strong dominance were not reported.
Δ life-years saved/costs: incremental number of life-years gained/costs compared with the next-best non-dominated strategy; FS: flexible sigmoidoscopy; FIT: faecal immunochemical test; MNB: mean net benefit; ICER: incremental cost-effectiveness ratio.
The main results of our analysis were not affected when assuming equal adherence for all strategies, although FS appeared to be more effective (1689 vs. 1460 LYs) and less costly (€359 vs. €384 per person invited) than FIT (Figure 1, scenario 2; online Appendix 4). Only a reduction of FS attendance from 30% to 17%, or an increase in the attendance with the FIT-only strategy from 42% to 70% when considering the overall uptake, or from 15% to 35% when considering the response rate to the re-invitation for subsequent FIT amongst non-attendees in previous rounds, could reverse the superiority of FIT + FS over FIT alone. When assuming 100% adherence, corresponding to the expected benefit for a screening attendee, the gain resulting from the expected reduction in CRC incidence and mortality with FS and FIT alone scenarios could be estimated as 15 and nine days per person, respectively.
When expanding the time horizon of the simulation to 55–75 years (biennial FIT between age 55 and 69, and FS at age 58), the main results of the model did not change (Figure 2). However, when assuming equal adherence, FS was less effective than FIT alone, due to the additional FIT tests performed between 55 and 57. When simulating the persistence of the test efficacy up to 75 years, FS + FIT remained the most cost-effective option, being also cost-saving. Even assuming a persistence of the effect for FIT alone, but not for FS, FS + FIT remained the most effective and cost-effective option. Similar results were observed when extending the time horizon for screening to 50–75 years (online Appendix 5).
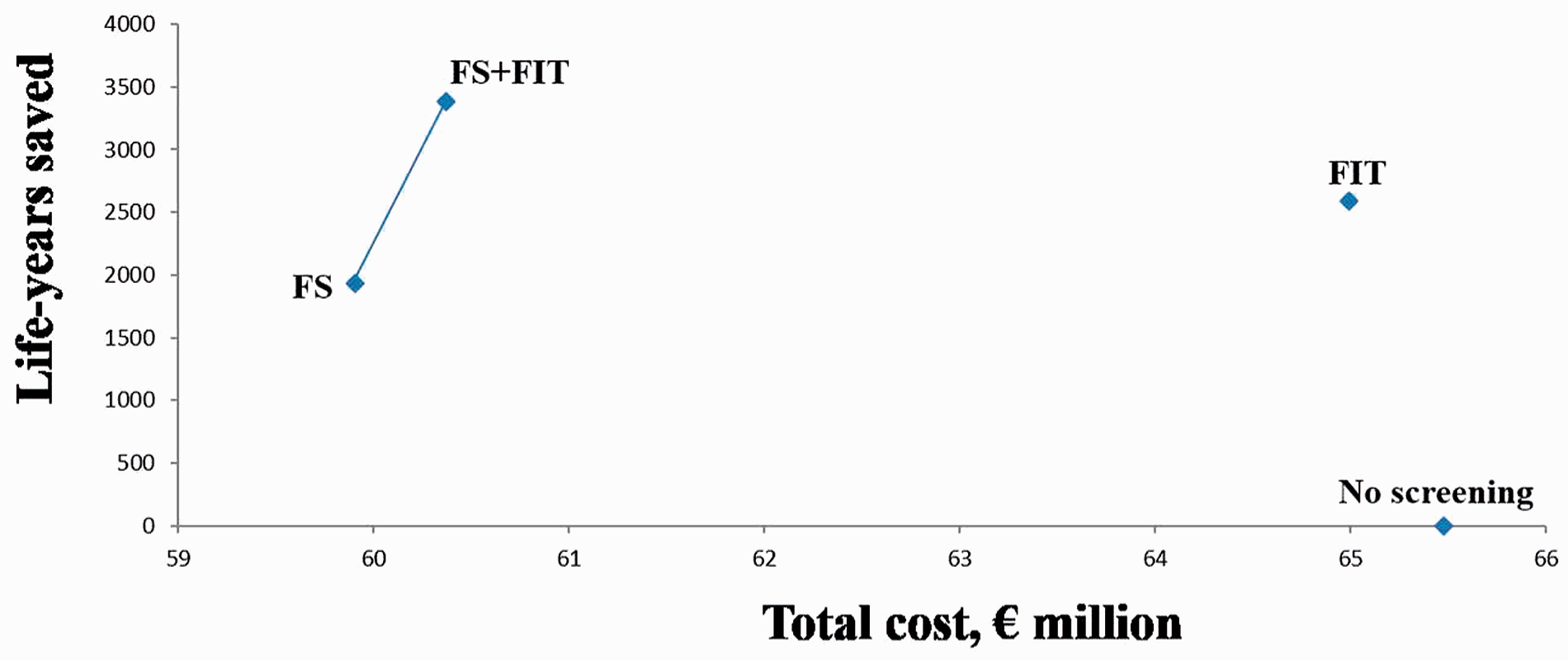
Cost-effectiveness amongst the different strategies when assuming 55–75 years as horizon time, with screening between ages 55 and 69 for FIT and at age 58 for FS and FS + FIT strategies (sensitivity analysis). Adherence: FS = 30%; FIT alone = 42%; FS + FIT − FS: 30% − FIT (FS−): 19%. Non-dominated strategies are connected by a continuous line. In this condition, both FS and FS + FIT strategies were cost-saving.
The superiority of sequential strategy (FS + FIT) over the remaining options was also robust to even large changes in the FS or FIT costs. When simulating a lower mortality reduction with FIT, from 26% to 21%, FS alone (with 30% adherence) appeared to be more cost-effective than FIT alone, and less cost-effective than FS + FIT. An increase from 26% to 45% in CRC mortality reduction was required for FIT to become more cost-effective than FS + FIT.
The probabilistic sensitivity analysis showed a marginal degree of uncertainty, with FS + FIT achieving the highest NB in 83% of the iterations, while FIT alone and FS alone strategies were associated with the maximum NB in 15% and 2% of the cases, respectively. When exploring the partial EVPIs, uncertainty appeared to be mainly related to efficacy of FIT (partial EVPI: €12 per subject) and adherence (partial EVPI; €5 per subject), whilst costs did not play any significant role (partial EVPI: €1 per subject).
Discussion
In our simulation study, a sequential approach, stipulating FS with re-invitation to FIT for participants non-adherent to FS, was the most cost-effective approach for an organized CRC screening programme. When financial or medical resources are limited, FS appears the most convenient option.
The superiority of the combined approach is explained by the intrinsic convenience and efficacy of FS as initial test, and the ability of the sequential re-invitation to FIT to shift a substantial proportion of non-attendees to another cost-effective screening option. Regarding the former point, when assuming equal adherence, FS appeared to be more effective and also less costly than FIT alone strategy. Regarding the latter point, the impact of the lower compliance with the initial FS invitation was reversed – in the combined strategy – by the progressive gain in participation amongst non-attendees, due both to the initial adherence to FIT and to the repeated compliance to sequential FIT re-invitation. The superiority of the sequential FS + FIT approach was robust at sensitivity analysis; even large variations in FS or FIT parameters were unlikely to reverse its superiority. In particular, a strategy of FIT alone would appear to be more cost-effective compared with the sequential FS + FIT approach only when assuming fairly large differences (≥40% absolute increase) in the response rate to FS compared with FIT invitation. Smaller differences have been reported from comparative studies of FS and FIT strategies.11,26
The results of our study are relevant for several reasons. First, the findings of the probabilistic analysis, showing that the marginal uncertainty present in the model is mainly dependent on the assumptions about the relative compliance of FS and FIT, not only confirmed the indications of sensitivity analysis, but would also suggest that comparative cost-effectiveness estimates, derived from simulation models assuming 100% compliance for each tested screening modality, 5 might not be directly relevant for the purposes of screening planning. Our modelling results, based on observed data of compliance, positivity and DR from strategies already in place within organized screening programmes,9,10 and combining the expected compliance with the effectiveness and costs of the screening strategies of interest, are instead directly informative for policy-makers. Second, we directly derived the costs of screening and assessment tests from an ongoing organized programme adopting the simulated strategies. These analytical estimates are more likely to approximate the real costs of an organized programme than estimates based on procedures fees charged by Medicare, or other medical institutions.5,30 Moreover, we included costs related with the organizational and evaluation phases, that are not generally included in CRC modelling.5,30 Thus, our model is informative for policy-makers, also on the side of the cost evaluation. Third, both FS and combined FS + FIT strategies appeared cost-saving, providing incentive for any health system to implement any of them, even when no additional resources are available.
As we did not introduce a natural history component in our model, we were not able to analyze other screening options. However, our approach also minimized uncertainty, mainly related to assumptions about unknown transition rates for the different classes of adenomas. Indeed, a validation exercise 31 against the outcomes of the UK FS trial of the three models currently used for informing screening policy decisions showed that, as a result of using different assumptions about natural history parameters, each model missed some target, some of which (i.e. cumulative incidence and screen detected CRCs) were likely to influence the cost-effectiveness of screening interventions. Similarly, a recent editorial commenting on the usefulness of simulation models for breast cancer screening 32 noted that the accuracy of model predictions tends to be limited by uncertainty in the initial risk estimates and trends, or about future clinical and diagnostic scenarios, and by imprecise knowledge about outcome utilities, natural history of the disease, and validity of intermediate endpoints.
We also limited the time horizon of our simulation, at age 70 in the reference scenario and up to age 75 in the sensitivity analysis, based on available evidence about the duration of the protective effect FS and FIT. All the included strategies appeared to be already cost-effective in this limited period, so that additional data on long-term efficacy are unlikely to add evidence to further justify the opportunity of organized programmes for CRC screening. Indeed, any prolongation and/or age-anticipation appeared to similarly affect all the included strategies at sensitivity analysis, without affecting the main results. Also, the limited time-window considered in our analysis reflects the target age range for CRC screening adopted in the UK (60–74), Finland (60–69), or The Netherlands (55–74). Although our estimates of relative cost-effectiveness were not changed when extending the time horizon to age 75, our approach might still result in an underestimate of screening benefit. In particular, with FIT, the benefits of the excision of advanced adenomas detected at older age will not be reaped until later. However, extending the time horizon up to 80 or 100 years would require additional assumptions about the persistence of the screening effect, as well as about the impact on CRC risk of birth cohort effects, population ageing, and competitive mortality. The above mentioned editorial 32 suggested that uncertainties about these parameters would limit the accuracy and validity of extrapolations beyond observed evidence. Model-based estimates of the size of CRC screening protective effect are indeed showing wide variability beyond 10–15 years. 33
We derived the estimates of FS screening effectiveness from the results of the Italian and UK trials,7,8 adopting a similar protocol to the Piedmont programme. Even if showing consistent results, the Norwegian 24 and US 34 trials adopted a different protocol, and therefore their results could not be directly generalized to our setting. We adopted Italian sources to estimate the costs of screening tests, so these estimates may not be immediately reproducible in other countries. However, the main results of our analysis were robust to even large variations in the input costs, so that eventual differences are unlikely to affect the model outputs. There are no reasons for which the Italian health service should have relative costs different from other health systems, when considering that reimbursement is similarly based on code of diagnosis. In addition, we calculated the number of tests to be performed under different scenarios, which allows derivation of an estimate of the costs in different countries, applying the local costs for those procedures. The model is available on request, so the simulation may be re-run using costs from other jurisdictions.
Conclusion
Organized CRC screening programmes are highly cost-effective, irrespective of the test selected. A sequential approach with FS and FIT appeared to be the most cost-effective option. In the case of limited resources, a single FS screening would appear the most convenient approach.
Supplemental Material
Supplemental material for Cost-effectiveness of colorectal cancer screening programmes using sigmoidoscopy and immunochemical faecal occult blood test
Supplemental material for Cost-effectiveness of colorectal cancer screening programmes using sigmoidoscopy and immunochemical faecal occult blood test by Carlo Senore, Cesare Hassan, Daniele Regge, Eva Pagano, Gabriella Iussich, Loredana Correale and Nereo Segnan in Journal of Medical Screening
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LC is employee of i-m3D Spa; CS, CH, EP, DR, GH, NS have no conflict of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was conducted in the context of a comparative trial of FS and CT Colonography, funded by the Piedmont Region Health Authority and Innovation and Research Authority and by i-m3D Spa. The funding institutions had no role in study design or in data collection, analysis, and interpretation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
