Abstract
Objective
To investigate the outcomes of biennial guaiac faecal occult blood test (gFOBT) screening after once-only flexible sigmoidoscopy (FS) screening.
Methods
Between 1994 and 1999, as part of the UK FS Screening Trial (UKFSST), adults aged 55–64 were randomly allocated to an intervention group (offered FS screening) or a control group (not contacted). From 2006, a subset of UKFSST participants (20,895/44,041 intervention group; 41,497/87,149 control group) were invited to biennial gFOBT screening by the English Bowel Cancer Screening Programme. We analysed gFOBT uptake, test positivity, yield of colorectal cancer (CRC), and positive predictive value (PPV) for CRC, advanced adenomas (AAs), and advanced colorectal neoplasia (ACN: AA/CRC).
Results
Uptake of gFOBT at first invitation was 1.9% lower (65.7% vs. 67.6%,
Conclusions
Uptake, positivity and PPV of gFOBT screening were reduced following prior offer of FS screening. However, a quarter of FS screened participants receiving a diagnostic examination after positive gFOBT were diagnosed with ACN.
Introduction
In England, a biennial guaiac faecal occult blood test (gFOBT) screening programme, the Bowel Cancer Screening Programme (BCSP), began in 2006. 1 In addition, roll-out of a programme of once-only flexible sigmoidoscopy (FS) screening at age 55 started in 2013. 2
Screening for colorectal cancer (CRC), using either FS or the gFOBT, has been demonstrated to reduce CRC cause-specific mortality in randomized controlled trials.3–7 Colonoscopy, FS, gFOBT, and faecal immunochemical tests (FITs) have all been included in various screening programmes. 8 Each screening modality has a different profile in terms of cost, sensitivity, specificity, and burden on endoscopy services. FS is more sensitive than gFOBT and FIT for distal advanced colorectal neoplasia (ACN).9–15 It is the only screening modality that has been shown to reduce CRC incidence in multiple randomized controlled trials.16,17 However, a single FS does not offer complete protection against distal CRC. Furthermore, as the maximum reach of FS is the splenic flexure, proximal colonic neoplasia is only detected if the findings at FS warrant further investigation by colonoscopy. Achieving high uptake of either FS or gFOBT screening is challenging.1,18 The effect of adding once-only FS to biennial gFOBT screening in England has been modelled, with predictions that it may prevent an additional 10,000 cases of CRC and 2000 deaths by 2030. 2 The true impact is not known and will depend on the uptake, positivity, and yield of CRC from biennial gFOBT following FS, outcomes which are uncertain.
We investigated the impact of prior FS on the outcomes of biennial gFOBT screening using data from participants of the UK Flexible Sigmoidoscopy Screening Trial (UKFSST) who were invited to gFOBT screening through the English BCSP. While there have been a number of studies examining the performance of FS relative to gFOBT, to our knowledge, this is the only study to investigate the performance of biennial gFOBT screening in patients previously randomized to either screening FS or to no screening.
Methods
The UKFSST (ISRCTN28352761), a multi-centre randomized controlled trial of once-only FS screening, 3 has been previously described in detail.3,17,19 Ethics approval for the trial was obtained from local research ethics committees. Men and women aged 55–64 were eligible for the trial if they were registered to a participating GP practice and did not meet any of the following exclusions: inability to provide informed consent, severe or terminal disease, history of CRC, adenomas or inflammatory bowel disease, life expectancy less than five years, or sigmoidoscopy or colonoscopy within the previous three years. Individuals who met these eligibility criteria were sent information about CRC and FS, and were asked “If you were invited to have the bowel cancer screening test [FS], would you take up the offer?” Individuals who indicated that they would take up the offer of screening were invited to participate in the trial, and those who agreed to participate and provided written informed consent were randomized in a ratio of 2:1 to either a control group (not contacted), or an intervention group (offered FS). During FS, small polyps were removed by polypectomy. Participants were referred for colonoscopy if polyps met any of the following criteria: diameter 1 cm or larger, three or more adenomas, 20 or more hyperplastic polyps above the distal rectum, tubulovillous or villous histology, severe dysplasia, or malignancy. 19
As part of the English BCSP, adults aged 60–74 (previously 60–69) registered with a GP practice are invited every two years to complete a gFOBT kit (Hema-Screen®), which includes six windows for two samples from three separate stools. Participants with a positive gFOBT outcome (i.e. definitive abnormal) are referred to a specialist screening practitioner and offered colonoscopy or, in a small proportion of individuals (typically <3%), another diagnostic investigation, such as computed tomography colonography. Individuals with three or more adenomas, or one or more large adenomas (≥1 cm), enter colonoscopic surveillance following polypectomy. 20 Due to age restrictions on gFOBT screening and an extended, rather than immediate, roll-out of the screening programme, only a subset of English UKFSST participants were invited to gFOBT screening. Some individuals aged 75 or older entered the BCSP, for example, as over-age self-referrals. These individuals were excluded from our analyses, as they are not representative of typical invitees to the screening programme.
To obtain information on the outcomes of gFOBT screening in included individuals, the UKFSST cohort was matched with BCSP data covering the period 1 August 2006 to 3 March 2016. To ensure sufficient follow-up of at least six months post-invitation, only invitation rounds where the gFOBT invitation was sent before 3 September 2015 were included in this analysis.
CRCs were identified either through BCSP data or in cancer registry data within six months of a positive gFOBT. Incorporating cancer registry data allowed identification of CRCs in UKFSST participants who did not attend diagnostic investigation following positive gFOBT, who had a diagnostic investigation performed by a private healthcare provider, or where the diagnostic investigation missed the cancer. Though registry data were available only up until 31 December 2014, rather than to 3 March 2016, we do not expect that this will have affected the results considerably, as in any given year only one or two CRCs not present in the BCSP data were identified in the registries within six months of a positive gFOBT. Dates of death up to 31 December 2015 were obtained from the Office for National Statistics via NHS Digital.
Adenomas were classified as advanced if either endoscopy or histology data indicated a size greater than or equal to 10 mm, if villous or tubulovillous histology was present, or if there was high-grade dysplasia. We categorized patients by their most advanced neoplastic finding. Hence, patients with CRC and advanced adenomas (AAs) were defined as having an outcome of CRC. Colonic findings distal to the splenic flexure were categorized as distal. ACN was defined as an outcome of AA and/or CRC.
Outcomes of gFOBT screening are presented by UKFSST randomization status and by compliance with the offer of FS screening. Uptake at first invitation is defined as the proportion of invitees who were adequately screened by gFOBT (i.e. gFOBT outcome of positive (definitive abnormal) or negative (definitive normal) at first invitation). Uptake of any gFOBT kit is defined as the proportion of invitees who were adequately screened by gFOBT in at least one invitation round.
Test positivity is defined as the proportion of participants adequately screened by gFOBT who had a positive gFOBT outcome. Yield of CRC is defined as the proportion of participants adequately screened by gFOBT who had a positive gFOBT and were diagnosed with cancer. Test positivity and yield of CRC are presented both at first gFOBT screen, and over all observed gFOBT screening rounds (i.e. the proportion of gFOBT screened participants who tested positive in at least one screening round; and the proportion of gFOBT screened participants who were diagnosed with CRC in at least one round).
We also present the positive predictive values (PPVs) for CRC, AAs, and ACN, at first gFOBT screen. These are defined as the proportion of participants attending diagnostic investigation following a positive gFOBT, who were diagnosed with the specified outcome. We excluded registry-only CRC diagnoses from these calculations, as we did not have diagnostic examination information for cancers identified outside the BCSP. In online supplemental tables, we provide PPVs and yield of CRC stratified by gender, as well as the stage distribution of CRCs detected at gFOBT screen. Cancer staging was determined using cancer registry data and data collected by UKFSST trial staff.
Results
In the UKFSST, men and women (
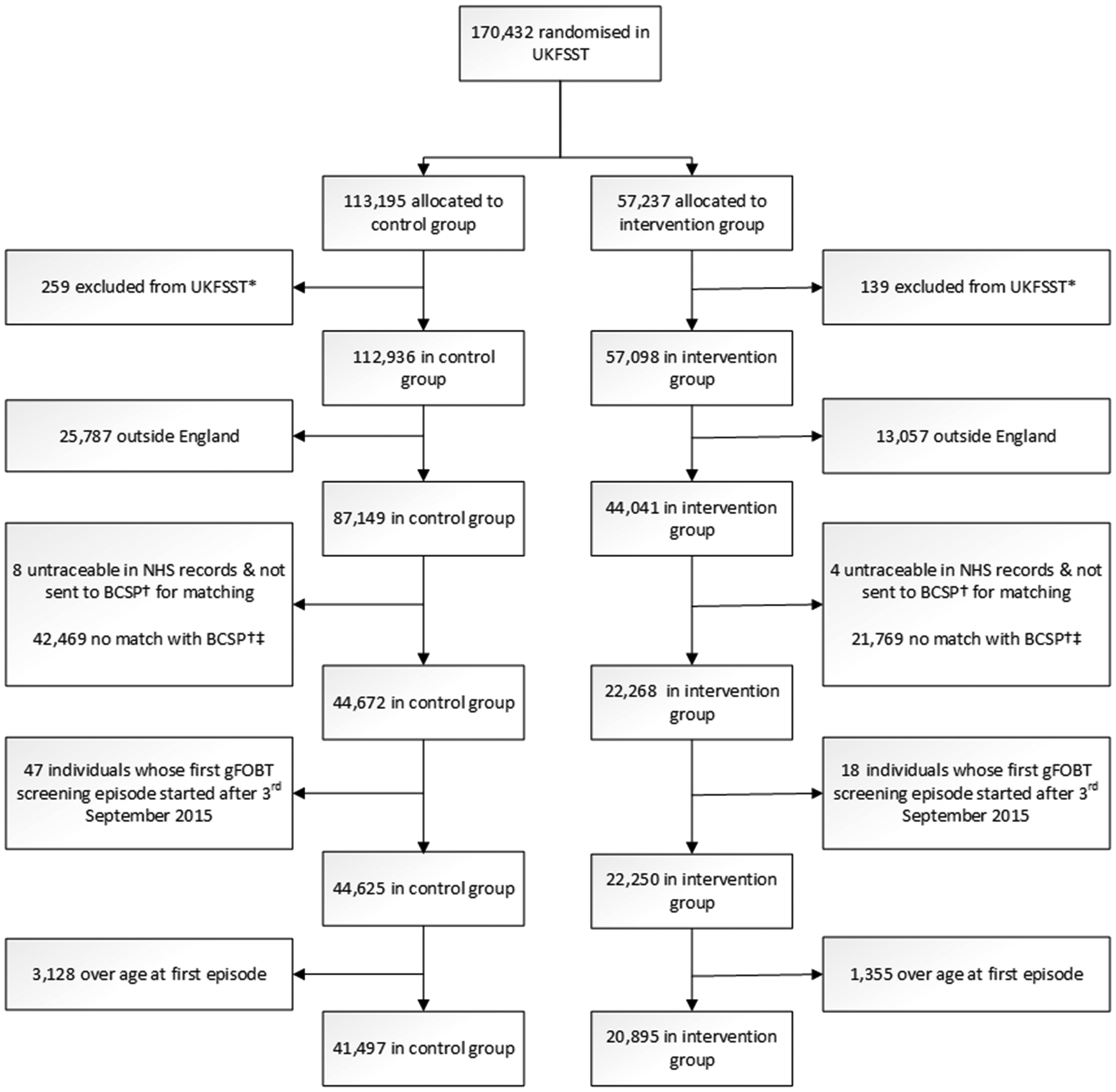
Flow chart of UKFSST participants included in analyses.
At the initiation of the BCSP, on 1 August 2006, 91.7% (40,389/44,041) of the English UKFSST intervention group and 91.6% (79,870/87,149) of the English UKFSST control group were alive (Table 1). Survival was lower among English UKFSST participants who refused FS (88.1%, 10,866/12,337) than in those who accepted FS (93.1%, 29,523/31,704) or the control group. Furthermore, survival was lower in men (89.4%, 57,522/64,328) than women (93.8%, 62,737/66,862).
Characteristics of English UKFSST participants.a
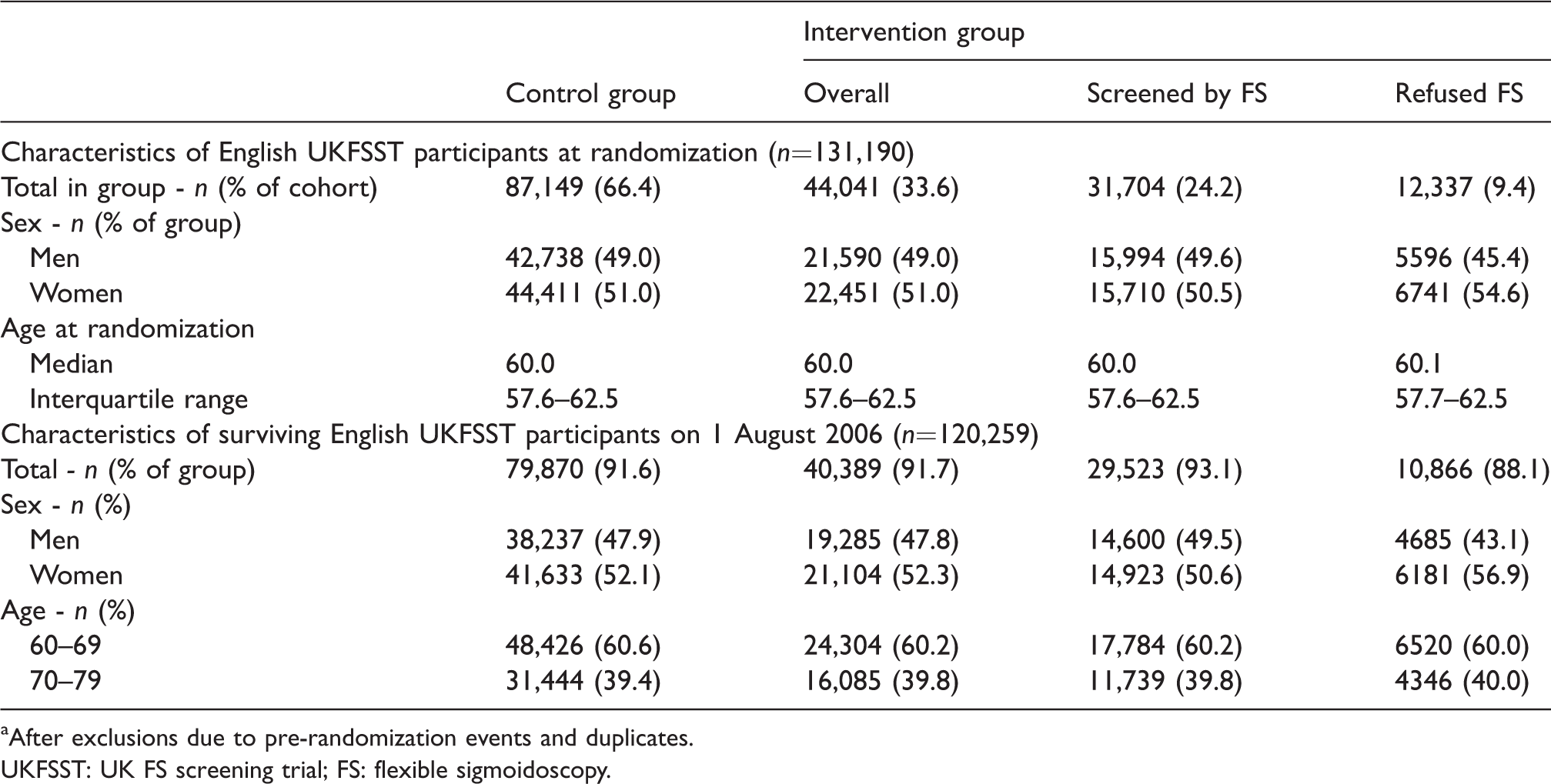
aAfter exclusions due to pre-randomization events and duplicates.
UKFSST: UK FS screening trial; FS: flexible sigmoidoscopy.
Between August 2006 and September 2015, 62,392 English UKFSST participants aged 60–74 were invited to gFOBT screening through the BCSP. This included 20,895 of 44,041 (47.4%) and 41,497 of 87,149 (47.6%) from the English UKFSST intervention and control groups, respectively (Figure 1). A lower proportion of those who refused FS (44.8%, 5532/12,337) were invited to gFOBT screening than those who accepted FS (48.5%, 15,363/31,704) or those in the control group. In comparison with the overall English UKFSST cohort of 131,190 UKFSST participants, those subsequently invited to gFOBT screening were younger (aged 57.8 vs. 60.0 at randomization) and a higher proportion (52.6% vs. 51.0%) were female (Tables 1 and 2).
Characteristics of English UKFSST participantsa invited to gFOBT screening (
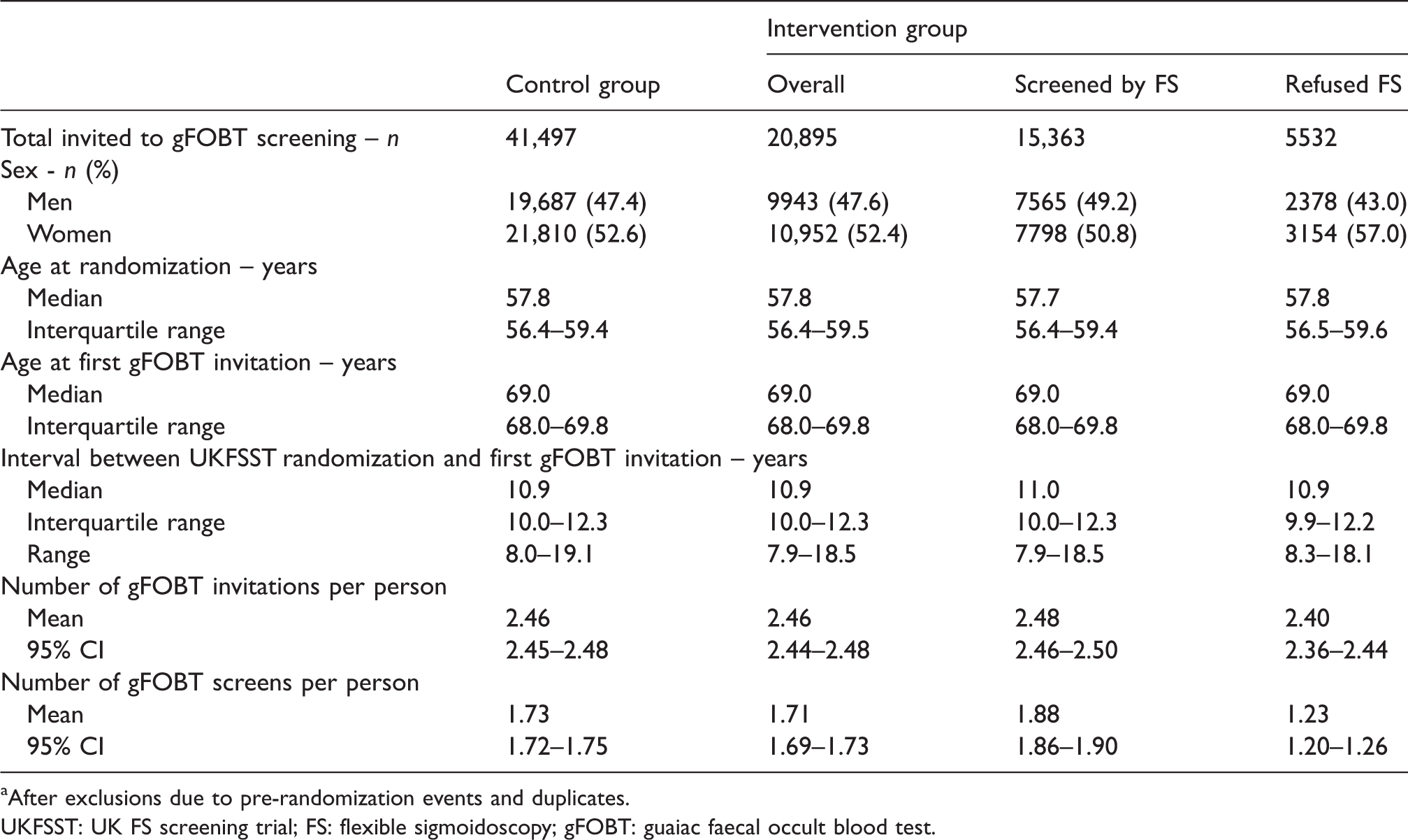
aAfter exclusions due to pre-randomization events and duplicates.
UKFSST: UK FS screening trial; FS: flexible sigmoidoscopy; gFOBT: guaiac faecal occult blood test.
UKFSST participants invited to gFOBT screening from the intervention group were similar, on average, to those invited from the control group (Table 2). In both groups, the median age at first gFOBT invitation was 69.0, just over half of invitees were women, a median of 10.9 years had elapsed between UKFSST randomization and first gFOBT invitation, and the mean number of gFOBT invitations sent over the study period was 2.46.
There were, however, differences among intervention group gFOBT invitees, between those who had refused and those who accepted FS screening (Table 2). A greater proportion of women refused FS than men, and for this reason a higher proportion of gFOBT invitees who had refused, rather than accepted FS, were women (57.0% vs. 50.8%). Furthermore, the average number of invitations sent and the average number of gFOBT screens were lower among those who had refused, rather than accepted, FS (2.40 vs. 2.48 invitations, and 1.23 vs. 1.88 screens, respectively).
Uptake of gFOBT screening was lower in the intervention group than in the control group at first invitation (65.7% vs. 67.6%,
Uptake of gFOBT screening in English UKFSST participants invited to gFOBT screening (
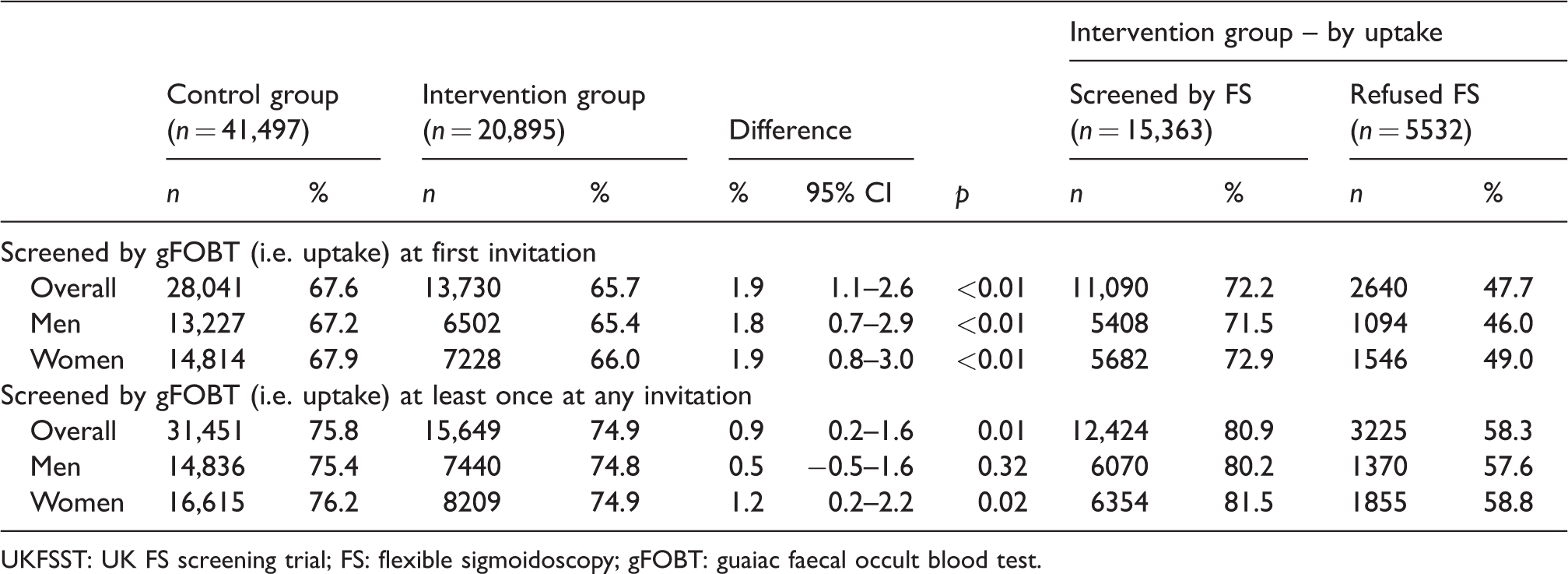
UKFSST: UK FS screening trial; FS: flexible sigmoidoscopy; gFOBT: guaiac faecal occult blood test.
Of the 62,392 invited to gFOBT screening, 47,100 were screened by gFOBT at least once (Table 4). Test positivity was lower among intervention group than control group gFOBT screenees at first gFOBT screen (2.0% vs. 2.4%,
Outcomes of gFOBT screening in English UKFSST participants who were screened by gFOBT at least once (
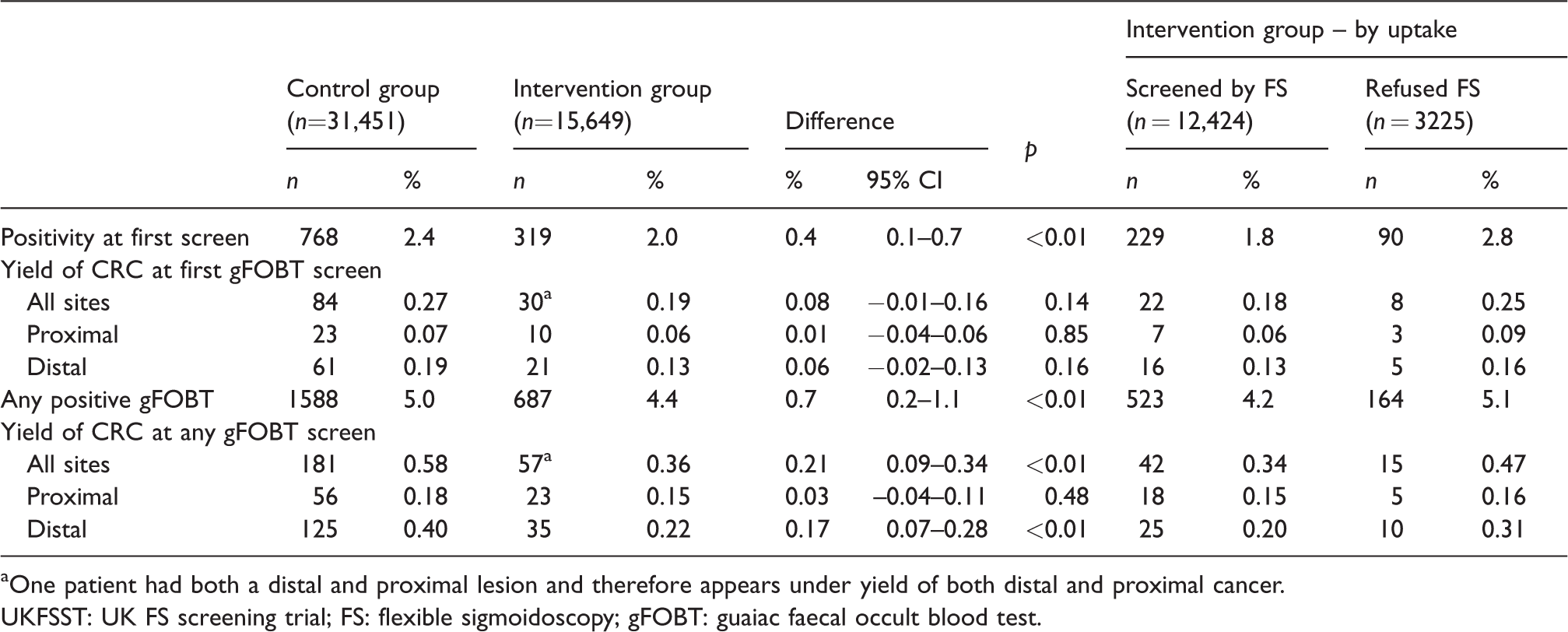
aOne patient had both a distal and proximal lesion and therefore appears under yield of both distal and proximal cancer.
UKFSST: UK FS screening trial; FS: flexible sigmoidoscopy; gFOBT: guaiac faecal occult blood test.
The yield of CRC in those screened by gFOBT at least once was lower in intervention group than in control group gFOBT screenees, at first (0.19% vs. 0.27%,
Outcomes among those attending diagnostic investigation after positive gFOBT at first screen (
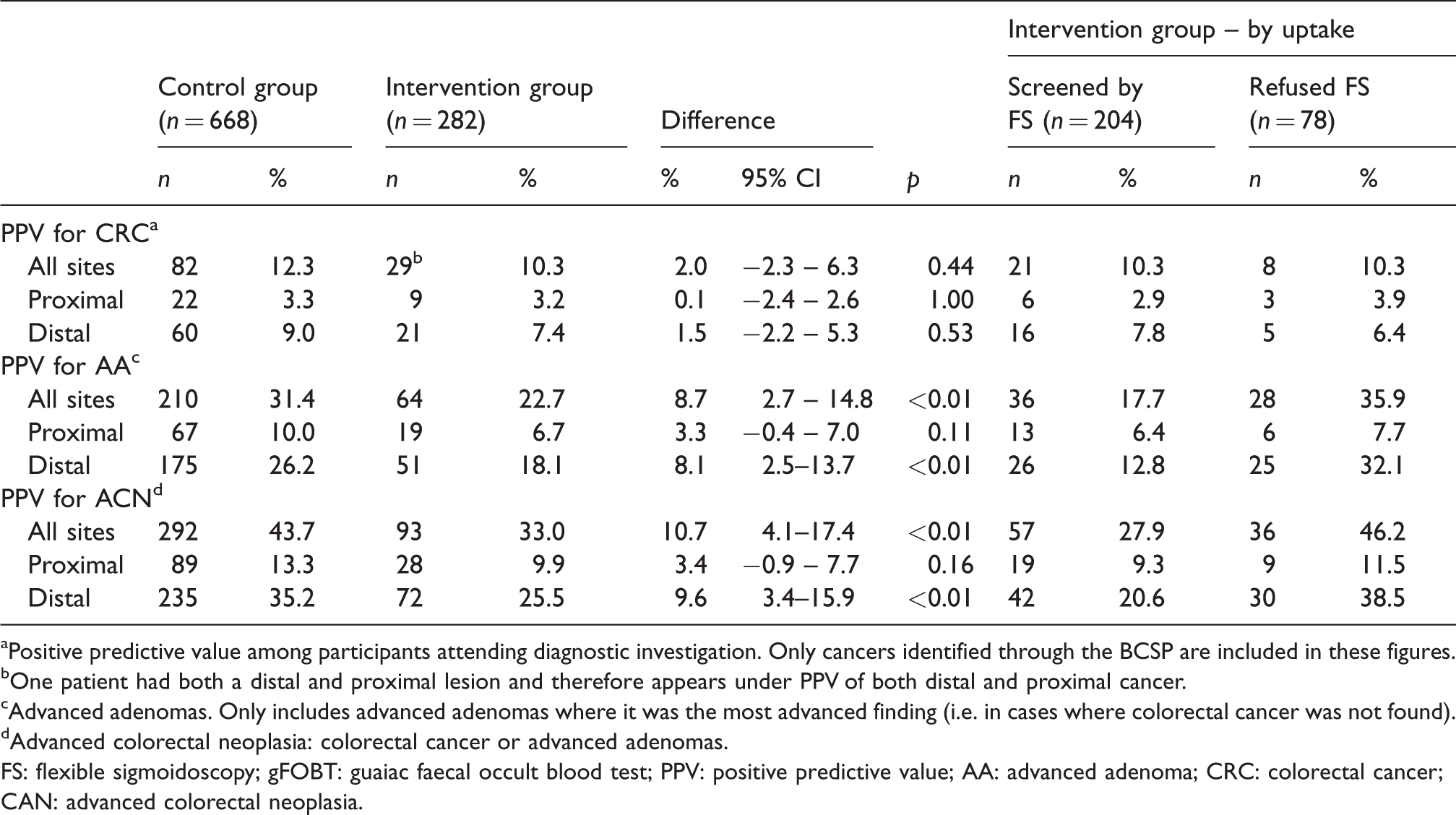
aPositive predictive value among participants attending diagnostic investigation. Only cancers identified through the BCSP are included in these figures.
bOne patient had both a distal and proximal lesion and therefore appears under PPV of both distal and proximal cancer.
cAdvanced adenomas. Only includes advanced adenomas where it was the most advanced finding (i.e. in cases where colorectal cancer was not found).
dAdvanced colorectal neoplasia: colorectal cancer or advanced adenomas.
FS: flexible sigmoidoscopy; gFOBT: guaiac faecal occult blood test; PPV: positive predictive value; AA: advanced adenoma; CRC: colorectal cancer; CAN: advanced colorectal neoplasia.
The PPV for distal cancer at first gFOBT screen was lower in the intervention group (7.4%, 21/282) than in the control group (9.0%, 60/668) diagnostic investigation attendees (
Discussion
At gFOBT, test positivity and yield of CRC were slightly lower among those who had previously been offered, or had received, an FS. One potential explanation for this is that prior FS, through the removal of adenomas, reduced the number of CRCs and adenomas that could be found by gFOBT screening. Reduced incidence of CRC in the UKFSST intervention group at 17 years post-randomization, as reported elsewhere, supports this explanation. 17 Furthermore, as would be expected given that FS is a screening examination of the distal colon and rectum, the difference in yield of gFOBT screening in the UKFSST intervention group versus the control group was greater for distal than for proximal cancer.
Though the yield of CRC was lower among UKFSST intervention group gFOBT screenees, gFOBT screening after FS could still be of potential benefit. However, given the effectiveness of FS at reducing CRC incidence for at least 17 years after screening, and given minimal randomized trial evidence that gFOBT or FIT reduce incidence, it is likely that extensive faecal testing would be required to achieve a benefit similar to that accomplished already by once-only FS.7,17
Achieving high uptake is crucial to a successful screening programme. Uptake of gFOBT at first invite was lower in participants previously offered FS (65.7%) than in those allocated to the control group (67.6%) in the UKFSST. While uptake was lower, the absolute difference was only 1.9%. Furthermore, in participants who had been offered and had accepted FS, uptake of gFOBT screening was high (72.2%). In general, uptake was considerably higher than typically observed in the BCSP, but this is to be expected given that only individuals who expressed an interest in screening were eligible for the UKFSST.1,21
Though uptake was lower than in other groups, many participants who had refused FS were screened by gFOBT (uptake of 47.7% at first, and 58.3% at any, gFOBT invitation). A number of researchers have examined the uptake of FIT in FS non-responders. Hol et al. 22 offered FIT to 4407 FS screening non-attenders and found that 25% attended FIT screening. Similarly, Senore et al. 23 found that among 37,691 FS screening non-responders, uptake of FIT offered six months following non-response was 19.3%. Subsequent faecal occult blood testing (either gFOBT or FIT) could be used to increase screening uptake above that observed with FS alone.
The differences identified between participants who had refused and those who had accepted FS may partly reflect the impact of FS, but may also be due to FS refusers being a less healthy group (i.e. healthy user bias). 24 Survival was lower, and PPV for ACN from gFOBT screening was higher, in the FS refusers than in the control group.
Though the sample size of our study is relatively large, only a subset of participants tested gFOBT positive, and only a subset of these had an outcome of interest, such as CRC. The low frequency of events limited statistical power, particularly for PPVs and yield.
Many UKFSST participants were not invited to gFOBT screening. Reasons for non-invitation include mortality, emigration, and being over the eligible age limit. Differential CRC incidence and mortality due to FS may have affected, to some extent, the composition of the intervention and control group invited to gFOBT screening. However, at gFOBT invitation, the two groups were broadly similar on many characteristics, including age and sex.
Generalizability of our findings to the current English BCSP is weakened by patient and screening characteristics unique to the study cohort. Only individuals who expressed an interest in screening were eligible for UKFSST, and for this reason we would have expected higher uptake of biennial gFOBT in this group than in the national BCSP or in other studies. The average interval between gFOBT screening and FS was 10.9 years. This is considerably longer than the current 5-year interval in the BCSP, where FS is being offered at age 55 and gFOBT screening from age 60. This longer interval means that there was more time between screenings for ACN to develop. UKFSST subjects invited to gFOBT screening were older than average BCSP invitees, with a median age at first gFOBT invitation of 69.0. Prevalence of ACN is known to increase with age. 25
While gFOBT has been the test used in the English BCSP, a different stool test, FIT, is being introduced in 2018. Both gFOBT and FIT are mailed tests for occult blood in faeces. At low faecal haemoglobin positivity thresholds FIT is more sensitive than gFOBT, and uptake of FIT is expected to be slightly higher than gFOBT. 26
Though there are a number of limitations, our study also has considerable strengths. Randomization applied in the UKFSST means that, except for differential attrition, at gFOBT invitation the intervention and control groups should be similar. Another strength of the study is that by using cancer registry data, we have been able to check for cancers diagnosed outside the BCSP diagnosed shortly after a positive gFOBT.
The findings of this study indicate that the combination of gFOBT screening and FS could be beneficial in terms of screening uptake, yield of CRC, and yield of AAs. FS is highly sensitive for neoplasia located in the distal colon and rectum, more so than gFOBT or FIT.9,27–29 However, the ability of FS screening to detect cancer or adenomas in the proximal colon is limited and depends on the criteria determining referral for colonoscopy. 30 There is evidence to suggest that the performance of gFOBT and FIT is also limited in the proximal colon, with lower sensitivity than for findings in the distal colon and rectum. 31 Given the apparent limitations of both techniques in detecting proximal findings, there remains a need for a low cost non-invasive screening test that is highly sensitive for proximal colon cancer.
Conclusion
Uptake, positivity, PPV and yield of gFOBT screening were lower among UKFSST participants who had been offered FS than in those who had not. Among FS screened participants attending diagnostic investigation following a positive gFOBT, ACN was found in just over a quarter of individuals. The findings of this study indicate that, dependent on cost-effectiveness, a combination of FS and faecal occult blood test screening might be beneficial.
Supplemental Material
Supplemental material for Effect of once-only flexible sigmoidoscopy screening on the outcomes of subsequent faecal occult blood test screening
Supplemental material for Effect of once-only flexible sigmoidoscopy screening on the outcomes of subsequent faecal occult blood test screening by Jeremy P Brown, Kate Wooldrage, Ines Kralj-Hans, Suzanne Wright, Amanda J Cross and Wendy S Atkin in Journal of Medical Screening
Footnotes
Acknowledgements
We thank Urvi Shah, Jim Summers, Eva Morris, James Thomas, Julia Snowball, and Claire Nickerson for their work in obtaining the data. Data for this study were based on information collected and quality assured by the Public Health England (PHE) National Cancer Registration and Analysis Service. Access to the data was facilitated by the PHE Office for Data Release.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Wendy Atkin has received non-monetary research support from Eiken Chemical Co. Ltd for reagents and lease of a machine for processing immunochemical faecal occult blood tests for the FIT for Follow-up study. All other authors declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The UKFSST has received funding from the Medical Research Council (MRC), National Health Service R&D, Cancer Research UK, and KeyMed. The trial is currently funded by the National Institute for Health Research [16/65/01]. The work of the Cancer Screening and Prevention Research Group is supported by Cancer Research UK [Grant No. A25004]. Infrastructure support for this work was provided by the National Institute for Health Research (NIHR) Imperial Biomedical Research Centre (BRC). The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Supplemental material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
