Abstract
Objective
To investigate trends in breast cancer mortality in New Zealand women, to corroborate or negate a causal association with service screening mammography.
Method
Cumulated mortality rates from breast cancer deaths individually linked to incident cases diagnosed before and after screening commencement were compared, in women aged 50–64 (from 2001) and aged 45–49 and 65–69 (from 2006). Trends and differences in aggregate invasive breast cancer mortality (1975–2013) were assessed in relation to introduction of mammography screening targeting women aged 50–64 and 45–69. Joinpoint analysis was also undertaken.
Results
The reduction in incidence-based cumulated breast cancer mortality before and after the introduction of screening was
Conclusion
Breast cancer mortality declines occurring since the advent of screening mammography in New Zealand are consistent with other incidence-based and aggregate studies of screening mammography in populations, individual-based cohort studies, and randomized controlled trials.
Introduction
Breast cancer is the most common cancer in New Zealand (NZ) women, accounting for approximately 28% of all female cancers. 1 After lung cancer, it is the second most common cancer cause of death, but for women aged 25–64 it is the leading cancer cause of death. 1
Most aggregate studies of service screening, employing a wide variety of methodologies, indicate a breast cancer mortality reduction of around 25–30%;2,3 however, the results of some have been disputed because many of the screening programmes were introduced contemporaneously with significant breast cancer treatment improvements in the late 1980s–early 1990s. This study is not confounded by concurrent treatment improvements, as service screening commenced in NZ in 1999.
At the initiation of service screening in 1999, BreastScreen Aotearoa (BSA), the national, publicly funded screening programme, targeted women aged 50–64 for screening mammography. All eligible women were offered screening simultaneously. In July 2004, the target age group was extended to include women aged 45–69. BSA offers bilateral, two-view, mammography screening without direct charge to age-eligible women. All mammograms are independently read by two radiologists, with arbitration by a third to resolve conflicting findings. Screening mammography in NZ is delivered through a national network of providers, each covering a defined geographical catchment area. Recruitment is via self-initiated enrolment or referral from a medical practitioner. Once enrolled, invitations for repeat mammography are offered biennially. The programme aims for 70% coverage of screening target age groups, 4 with the objective of achieving a 30% reduction in breast cancer mortality, in line with the findings of Tabár et al. 5 The latest available biennial coverage for women aged 50–69, for June 2015 to May 2017, was 72%. 6
An individual-based cohort study of screening mammography in NZ women has reported breast cancer mortality reductions in the order of 34% attributable to screening. 7 It is, nevertheless, important to determine whether congruent findings are evident from incidence-based and aggregate analyses of population breast cancer mortality data in relation to screening mammography. In this study, trends in breast cancer mortality were analysed in relation to the introduction of population-wide service screening mammography in 1999 for women aged 50–64, and its age extension in 2004 to include women aged 45–69, employing incidence-based and aggregate breast cancer mortality analyses.
Methods
Annual female deaths from breast cancer over 1948–2013 by five-year age group were obtained from the NZ Ministry of Health, 8 with corresponding estimated denominator populations. Mortality was coded using International Classification of Diseases ICD-9-CM-A for deaths prior to 2000, and ICD–10-AM 6th edition for deaths from 2000. 9 Cancer deaths are cross-checked between the NZ Cancer Registry and the Mortality Collection, to ensure accurate recording of the primary cancer site, and the Cancer Registry or the Mortality Collection is modified, if required. The NZ Cancer Registry collects all incident cases of cancer nationally, with mandatory pathology notification since 1994. 8 The incidence-based mortality analysis was based on unit records of incident breast cancers linked to breast cancer mortality. These latter data extend over 1996–2011 and were supplied to the authors in de-identified form by the NZ Cancer Registry.
Incidence-based breast cancer mortality was examined using breast cancer deaths cumulated from breast cancers diagnosed in four predefined population cohorts: women aged 50–64 diagnosed in 1996–98 (before service screening), and in 2001–3 (after screening introduction), women aged 45–49 and 65–69 (extended target age ranges) diagnosed in 2001–3 (before introduction of screening for these ages), and in 2006–8 (after introduction of screening). Breast cancer mortality was cumulated to eight years in the 50–64 age group, up to 2004–6 for the group diagnosed in 1996–98 prior to service screening, and to 2009–11 for the 2001–3 diagnosis group following the introduction of screening. Breast cancer mortality was cumulated for three years in the 45–49 and 65–69 age groups, to 2004–6 for the 2001–3 diagnosis group prior to screening for these ages and to 2009–11 for the 2006–8 diagnosis group following introduction of screening. The incidence cohorts and breast cancer mortality follow-up periods are shown in Figure 1.
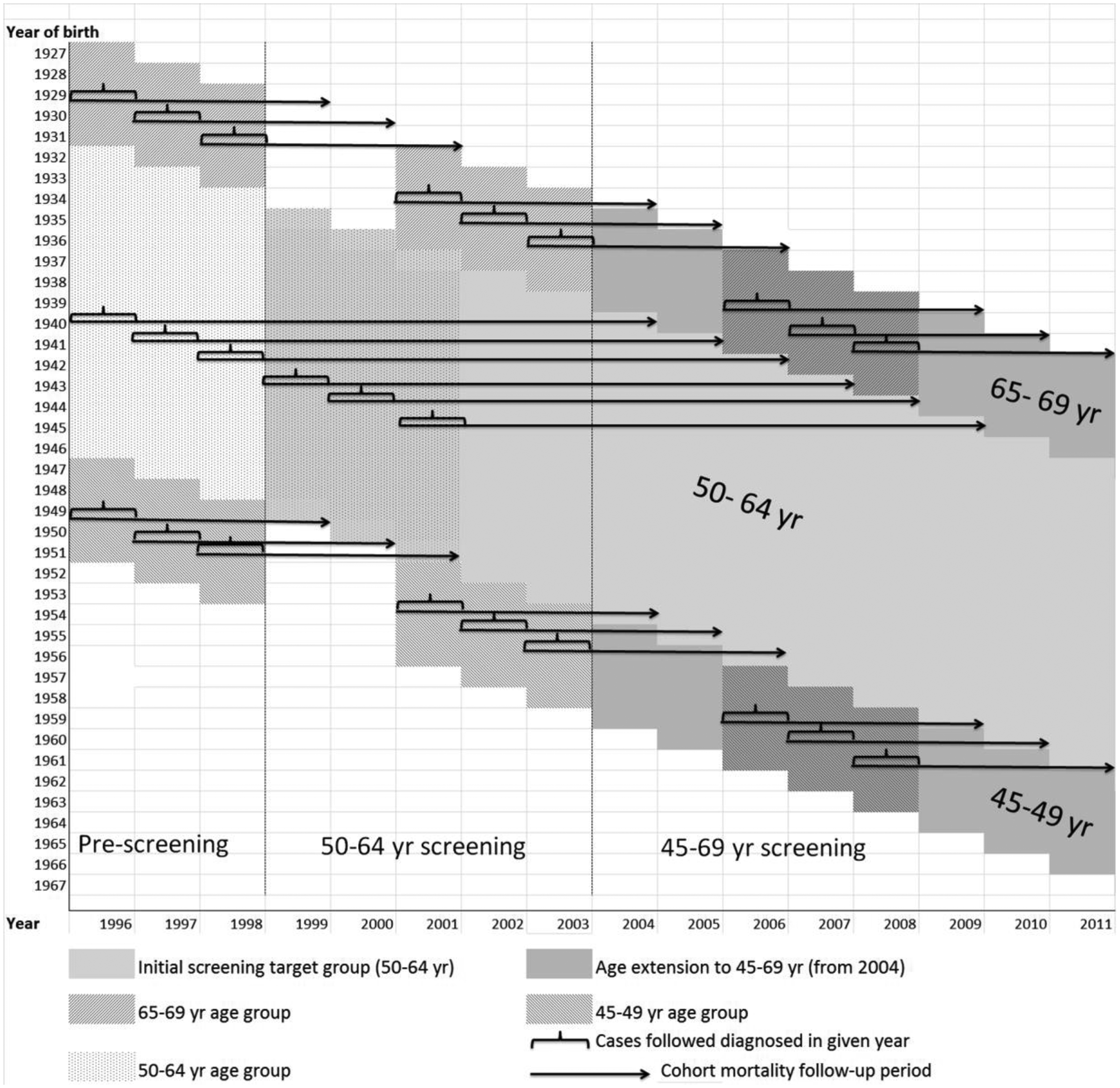
Incidence-based breast cancer mortality follow-up in relation to exposure to service screening mammography, New Zealand women, by birth cohort (1927–66) and period (1996–2011) for screening age groups.
The diagnosis groups following introduction of service screening were chosen two years after screening commencement for each target age range, to exclude the prevalent breast cancers diagnosed in the initial first round of screening, where a relatively high proportion from previously unscreened women are diagnosed. 10 The time windows for choosing these initial diagnostic cohorts were, by necessity, narrow. The control group before screening for screening-exposed women aged 50–64 was the three-year diagnostic period from when pathology notifications to the cancer registry became mandatory (1996), and prior to when screening was introduced in 1999 (i.e. 1996–98). The same three-year diagnostic period was applied to equivalent screening-exposed women aged 50–64 after screening was fully established (i.e. 2001–3). The smallest maximum equivalent available follow-up time for comparing cumulative breast cancer mortality in women aged 50–64 was eight years, as available incidence-based mortality data extended to 2011 only. Each breast cancer death occurring within 2922 days (eight years) of diagnosis was classified as a breast cancer death within eight years’ follow-up, and counted towards the eight-year cumulated mortality. Similar considerations applied to the age 45–49 and 65–69 initial diagnostic cohorts, where the controls not offered screening comprised those diagnosed from 1996 to 1999 and from 2001 to 2003, and the group exposed to service screening comprised those diagnosed in 2006–8, with the smallest maximum available follow-up time now limited to three years for each cohort. Each breast cancer death occurring within 1096 days (three years) of diagnosis was classified as a breast cancer death within three years follow-up, and counted towards the three-year cumulated mortality. We estimated three-year cumulative breast cancer mortality in women aged 45–49 and 65–69 diagnosed with breast cancer in 1996–98 to compare with that in the 2001–3 period prior to service screening for this age group, after screening commenced in 1999 and offered initially only to women aged 50–64.
Aggregate (non-incidence based) breast cancer mortality is analysed by age group by significant periods: 1980–91 before population service screening and prior to the effects of improvements in breast cancer therapy from the early 1990s, 1992–98 before service screening but after the introduction of improvements in breast cancer therapy from the early 1990s, 1999–2004 service screening offered to women aged 50–64 only, and 2005–2013 following age extension of screening to ages 45–69 to the most recent data available.
Trends in annual (log) age-standardized overall breast cancer mortality rates (age 50–64) are also examined to estimate mean annual percentage changes through joinpoint regression, 11 using JoinPoint v. 4.3.1.0 software. 12 Breast cancer mortality in the age group 50–64 is directly age standardized to the 2006 NZ census female population using three five-year age groups prior to trend analysis. Confidence intervals (95% CI) for cumulated breast cancer mortality estimates are based on the normal approximation to the binomial. P-values of differences in cumulated rates are calculated using χ2 statistics.
Ethics approval for this study was not required as it relied on de-identified cancer mortality and population data.
Results
The change in incidence-based cumulated breast cancer mortality before and after introduction of service screening for women aged 50–64 of −17% was significant (p = 0.0047). For all women aged 45–69 cumulated breast cancer mortality was −15% lower in those exposed to service screening compared with those not exposed, significant at p = 0.005 (Table 1). Prior to screening targeting women aged 45–49 and 65–69, three-year cumulated breast cancer mortality in those diagnosed in 1996–98 compared with 2001–3 declined by −40% (p = 0.001) in women aged 45–49 and −30% (p = 0.027) in women aged 65–69. Cumulated three-year breast cancer mortality in women aged 45–49 and 65–69 diagnosed in the 2001–3 period prior to service screening for these ages, compared with the 2006–8 period following the introduction of screening, was unchanged for women aged 45–49, and decreased by −19% in women aged 65–69 (non-significant).
Incidence-based cumulated breast cancer mortality from cancers diagnosed three years pre- and post-screening, New Zealand women followed over three and eight years post-diagnosis, 1996–2011.
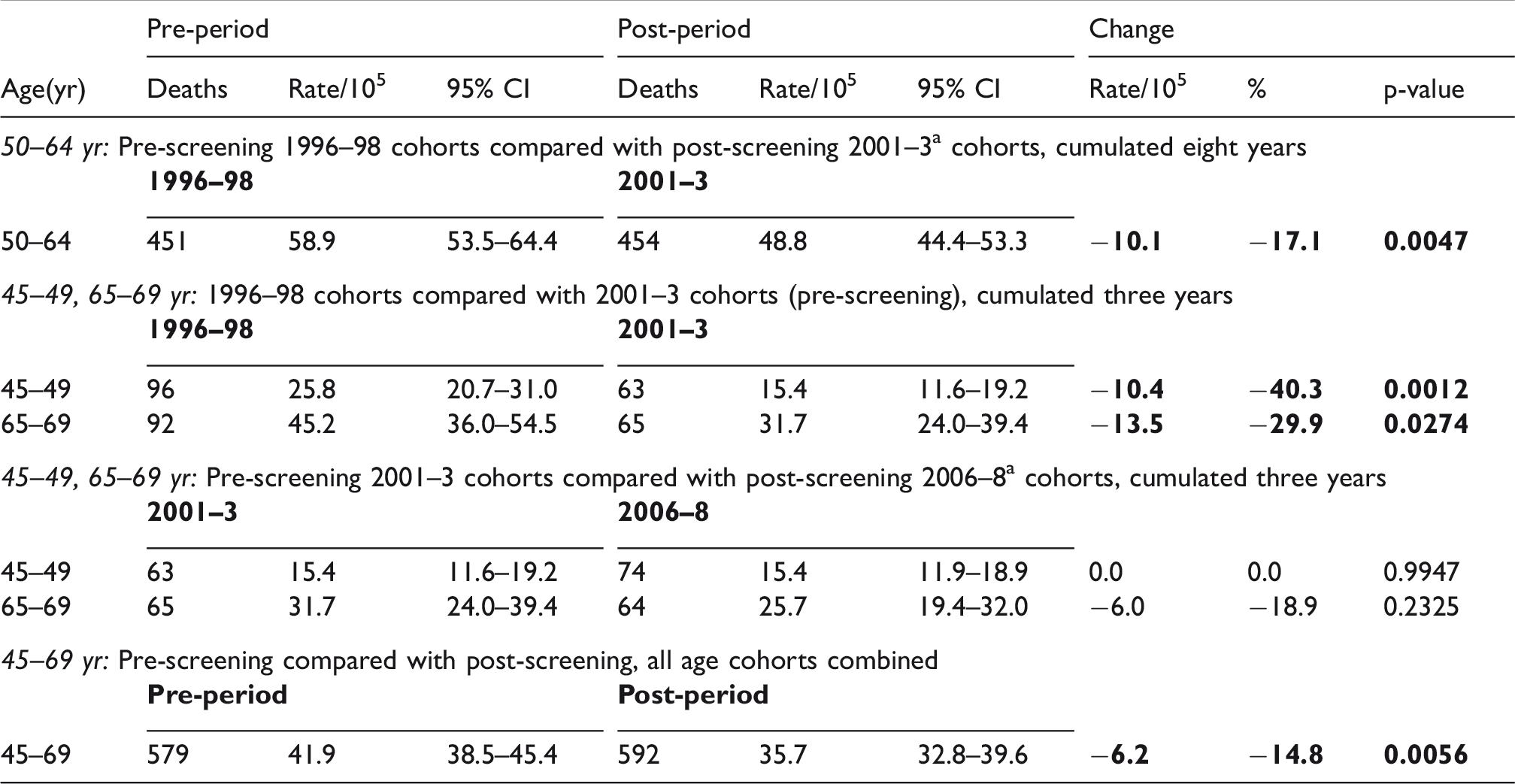
CI: confidence interval.
aPost-screening cohorts selected after two years following screening availability to exclude predominantly prevalent cancers detected by initial screens.
In women aged 50–64, age-adjusted breast cancer mortality declined by −9.8% between 1980–91 (prior to widespread implementation of treatment improvements) and 1992–98, contemporaneously with publication of successful trials (and meta-analyses) of treatment improvements for primary breast cancer (Table 2). After the introduction of service screening in 1999, there was a further decline in breast cancer mortality of −17% to 1999–2004, and −20% in the next period to 2005–13, producing a cumulative proportional mortality decline in women aged 50–64 of −34% since the introduction of service screening.
Aggregate breast cancer mortality in relation to screening by period and age group, New Zealand women 1980–2013.
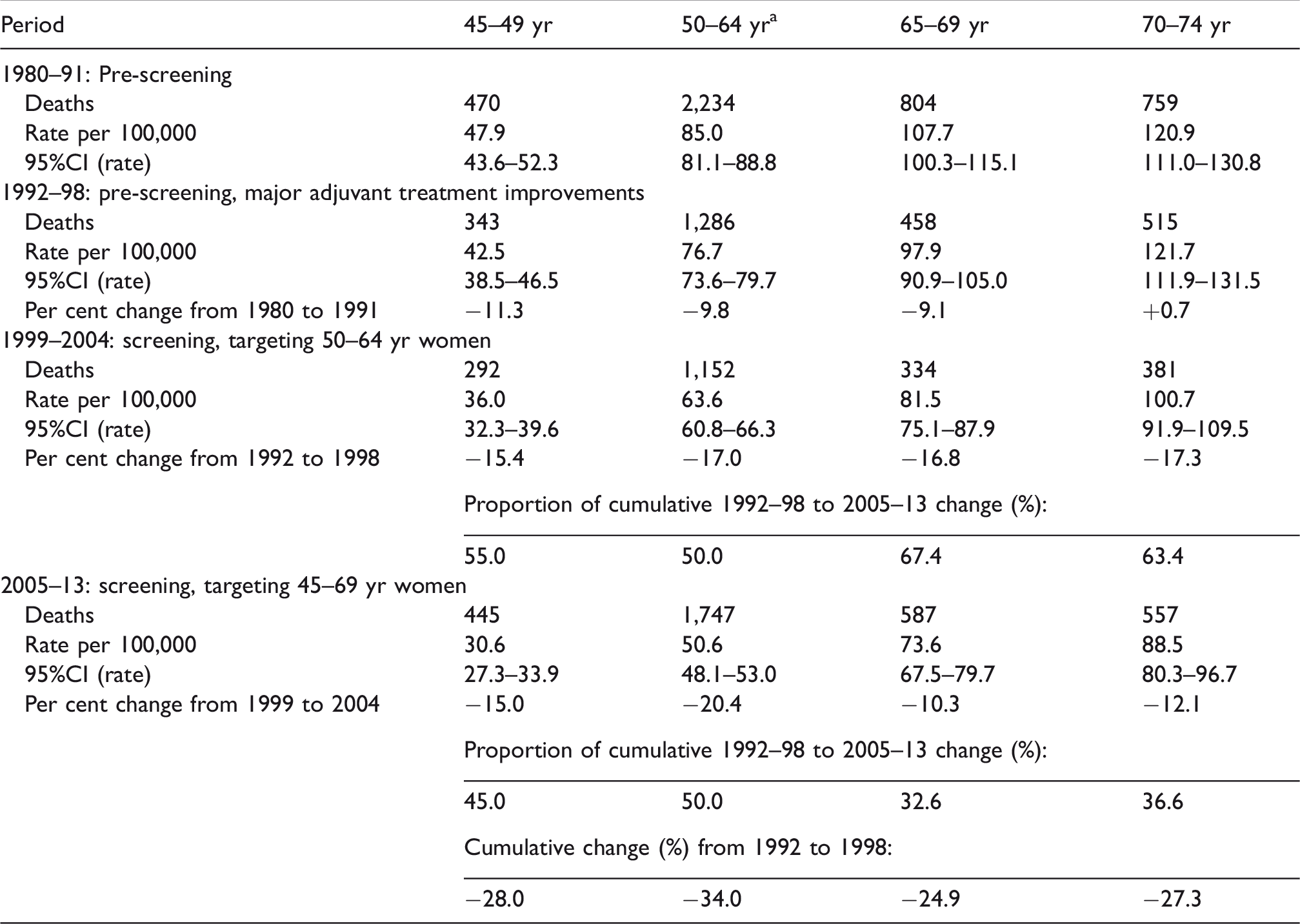
CI: confidence interval.
aAge standardized (direct) breast cancer mortality, standardized to NZ 2006 census population using three five-year age groups.
Breast cancer mortality also declined by −11% for women aged 45–49 and by −9% in women aged 65–69 from 1980–91 to 1992–98. This decline continued following the 1999 introduction of screening, at −15% for women aged 45–49 and −17% for those aged 65–69 to 1999–2004, even though these age groups were not included in the programme during this period. Following the age extension in 2004, the mortality decline to 2005–13 continued similarly in women aged 45–49 (−15%), but slowed in those aged 65–69 (−10%). The screening target group aged 50–64 had the highest cumulated mortality reduction of 34% between 1992–98 and 2005–13.
Breast cancer mortality in the age group 70–74 (never a screening target group) is affected by improvements in therapy, the effects of screening in younger age groups and from previous screening in these women prior to reaching age 70–74. There was no significant change in breast cancer mortality in women aged 70–74 until a −17% decline following the introduction of the national screening programme targeting women aged 50–64 from 1996–98 to 1999–2004. Following extension of screening to age 69 in 2004, breast cancer mortality rates in the 70–74 age group declined by a further −12% to 2005–13.
In the age groups 45–49 and 65–69, most of the 1992–89 to 1999–2004 mortality improvement occurred prior to screening, whereas in the 50–64 age group, 50% of the improvement occurred in 1999–2004. Conversely, after screening was introduced for women aged 45–49 and 65–69 in 2004, proportionally most of the mortality reduction from 1992–98 occurred subsequent to screening introduction, during 2005–13. Notably, when the mean cumulative mortality decline for the 45–49 and 65–74 age groups from 1992–98 to 2005–13 of 26.7% is compared with the mortality decline of 34.0% in women aged 50–64, then the service screening effect may be approximated as 27%.
From joinpoint analysis of women aged 50–64, a turning point was identified around 1987, which marked the beginning of the decline in breast cancer mortality from previously increasing annual rates (of +1.2% per annum 1975–87) to a decline of −1.8% per annum from 1987 to 1997. Another joinpoint was evident around 1997, with a steepening of the decline in breast cancer mortality to −3.1% per annum over 1997–2011 (Figure 2).
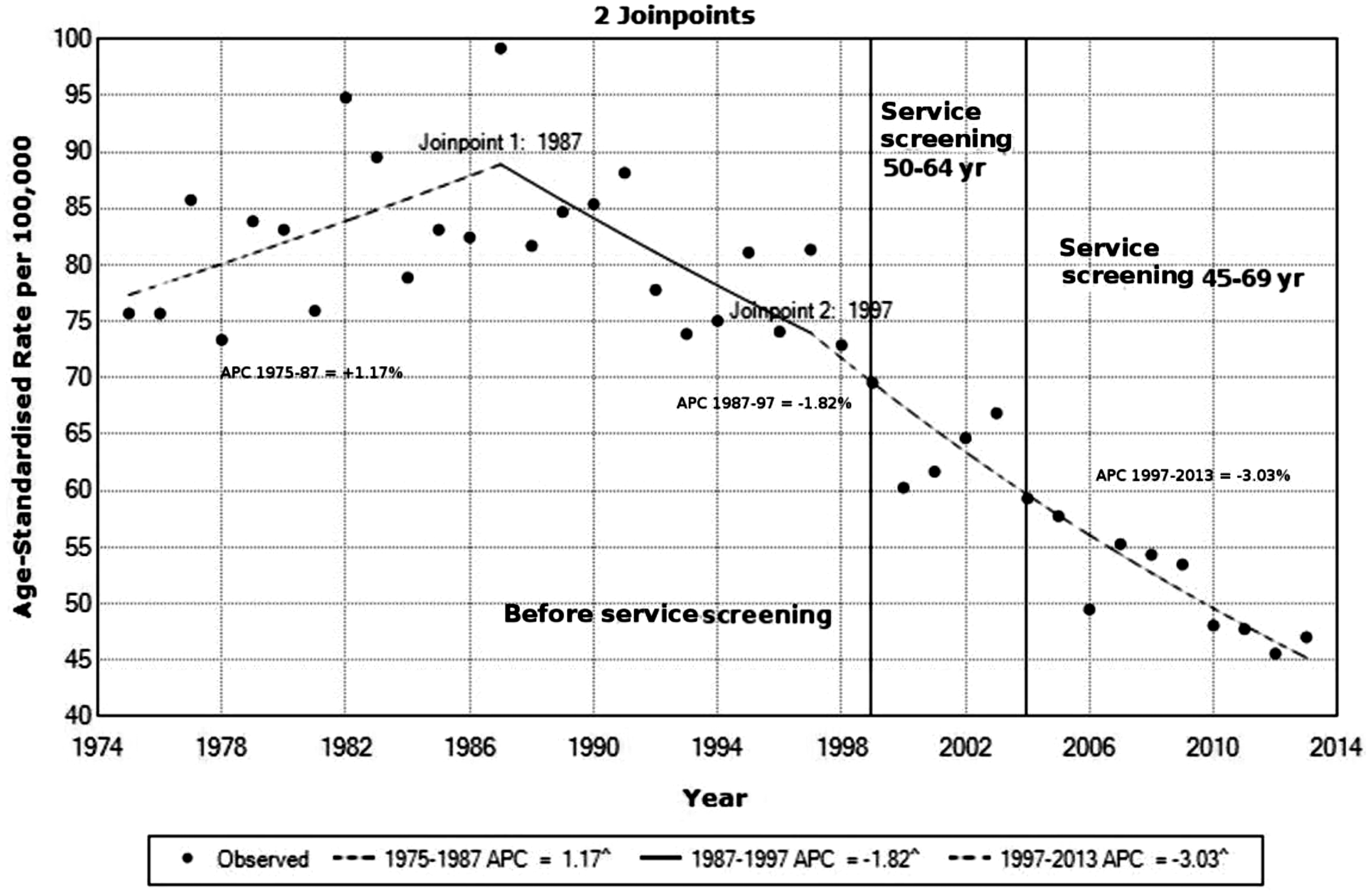
Annual aggregate trends in age-standardised breast cancer mortality in relation to screening mammography, 50–64 year New Zealand women 1975–2013: Jointpoint analysis. Statistically significant (p ⩽ 0.05) in relation to slop = 1. APC: annual percentage change.
Discussion
Both incidence-based and aggregate analyses of declines in breast cancer mortality in NZ in relation to service screening show results consistent with effects of the mammography screening programme. For women aged 50–64, breast cancer mortality reduction was −17.1% for incident breast cancers in 1996–98 compared with 2001–3 (each with eight-year follow-up), and −17.0% for aggregate breast cancer mortality 1992–98 compared with 1999–2004. Women aged 45–49 and 65–69 were not officially targeted following service screening commencement during 1999–2003, yet both incidence-based breast cancer mortality (diagnosed in 1996–98 compared with 2001–3, with three year follow-up), and aggregate breast cancer mortality 1992–98 compared with 1999–2004, declined substantially.
Assessment of changes in overall (aggregate) breast cancer mortality with respect to the advent of service mammography screening, without knowledge of when the cancers are diagnosed, is likely to produce results that, to varying degrees, cannot be wholly attributed to screening, as a proportion of breast cancer mortality during the screening epoch will have originated from cancers diagnosed before the introduction of screening. While all cancer mortality prior to a screening programme occurs in cases diagnosed before screening, this is not the case for the screening epoch. During the initial screening period of 1999–2004, aggregate breast cancer mortality in women aged 50–64 would have included breast cancer deaths from cancers incident prior to introduction of screening, and would have comprised a relatively high proportion of prevalent breast cancers detected by screening in previously unscreened women, especially during the first screening round of 1999–2000. Consequently, mortality results associated with screening are likely to be biased towards null effect of screening.
Population service screening was introduced around 1990 in Europe, Canada, and Australia, contemporaneously with augmented adjuvant therapy of early breast cancer, 13 which has rendered attribution of observed secular reductions in breast cancer mortality to screening difficult. 14 Examination of exclusively secular trends in such circumstances will produce, at best, uncertain estimates of the relative contributions of service screening and treatment improvements to documented breast cancer mortality reductions. Some have attributed most, if not all, of the observed breast cancer mortality decline to treatment rather than screening, for example, in Australia 15 and the UK. 16 In NZ, screening mammography was introduced in 1999, after the advent of major improvements in breast cancer adjuvant treatment around the late 1980s to early 1990s, and consequently the association of service screening with breast cancer mortality reductions in NZ cannot so easily be discounted by confounding from major treatment improvements.
In NZ, prior to the introduction of screening mammography in 1999, breast cancer mortality declines of 9–11% occurred in women aged 45–69, coinciding with adjuvant treatment improvements (1990–98), similar to women aged 50–64 (10%), but with no observed improvement in women aged 70–74 (Table 2). The highest cumulative aggregate mortality decline from 1992–98 (before service screening) to 2005–13 (following screening introduction) occurred in the target age group 50–64 (34%), but mortality declines were also noted in other screening age groups (25–28%), and in the age category 70–74 (27%).
With the introduction of national mammography screening for women aged 50–64 in 1999, there were probably improvements in breast cancer diagnosis and treatment across all age groups, not only those targeted for screening, especially as the NZ programme explicitly included treatment for screen-detected cancers. 17
For women aged 45–49 and 65–69 not targeted for screening from commencement of service screening (initially offered to women aged 50–64 in 1999) to the commencement of age extension in 2004, both incidence-based mortality (1996–98 to 2001–3, Table 1) and aggregate breast cancer mortality (1992–98 to 1999–2004, Table 2) show substantial reductions in breast cancer mortality.
Major mortality benefits from screening at a population level, if these were to occur, would be expected to manifest when a large segment of the target population offered screening participates, and when incident breast cancers in previously screened women predominate as the screen-detected cancers. When aggregate breast cancer mortality is cumulated from 1992–98 to 2005–13, the reduction was indeed larger in the target women aged 50–64 than in the previously non-targeted age groups, proportionately by 34%, and in line with other studies of breast cancer mortality changes in relation to screening mammography.
Studies that appear to negate the effects of service mammography screening on breast cancer mortality using population trend analyses are inconsistent with the far stronger observational evidence for the effectiveness of service screening emanating from cohort and case–control studies from Europe, UK, Australia, and NZ2,7,18–23 or evidence from more detailed small-area or temporal aggregate studies conducted in Australia 24 (including an incidence-based study 25 ) and elsewhere, and the evidence from randomized controlled trials.
Incidence-based breast cancer mortality is not amenable to trend analyses, as mortality needs to cumulate from the commencement of equivalent comparison periods before and after the introduction of service screening for differences to emerge. Incidence-based mortality analysis is a well-established method for evaluation of mammography screening service.26,27 Prevalent cancers detected at the commencement of a screening programme in the first ‘prevalent’ round of previously unscreened women will contribute to higher mortality than from subsequent rounds and are usually excluded from screening evaluation. As the screening interval in NZ is two years, cancers diagnosed during the initial rounds (i.e. 1999–2000 for women aged 50–64 and 2004–5 for women aged 45–49 and 65–69) were excluded, to minimize bias from this source.
A notable finding in this study was the decline in cumulated breast cancer mortality (from incidence-based analyses) in women aged 45–49 and 65–69 diagnosed in 2001–3 compared with those diagnosed in 1996–98, both periods preceding the offer of screening for these women. No significant decline was found from cases among women aged 45–49 and 65–69 diagnosed in 2001–3 compared with cases diagnosed after age extension (2006–8), although the magnitude of decline for women aged 65–69 was consistent with the decline among women aged 50–64. None of these periods were characterized by major improvements in treatment methods, but the significant breast cancer mortality decline did occur in age groups 45–49 and 65–69 when screening was already being offered to women aged 50–64 (from 1999). A component of the mortality decline in women aged 65–69 would emanate from lower incidence in this age group from cancers detected earlier than they would otherwise have been, at age 60–64, with subsequent lower breast cancer mortality in women aged 65–69. Evidence of screening-related incidence reduction is in the accompanying article on screening mammography and NZ breast cancer incidence in this issue of the journal (Figure 2). 28 The lack of a breast cancer mortality decline in women aged 45–49 subsequent to them being offered screening warrants further investigation.
The breast cancer mortality declines in women aged 45–49 and 65–69 from 1996–98 to 2001–3 may also have been a consequence of the advent of service screening in 1999, where numerous assessment and breast cancer treatment pathways were established, and women and their general practitioners would have been aware of the screening programme, and such protocols may have been applied to the cases that clinically presented in women aged 45–49 and 65–60. Furthermore, some women in these age groups may have availed themselves of ad hoc mammography or de facto screening in the private sector, as recruitment for service screening mammography for women aged 50–64 was in full force. However, there was no prominent incidence inflation in these age groups until age extension in 2004. 28 It has been anecdotally reported that, in NZ, breast accredited surgeons rather than generalists became the norm after the advent of service screening, and it became routine for women with breast cancer to see both medical and radiation oncologists to discuss recommendations from the entire multidisciplinary management team, not just the surgeon. Additionally, increasing sentinel node biopsies were being performed based on results from the Sentinel Node biopsy versus Axillary Clearance trial. 29 This led to increased numbers of women with detected micro-metastases and subsequent chemotherapy for node positive disease (Gregory M, 2018, personal communication). Thus, some stage shifting to higher grade occurred, coupled with a downward impact on breast cancer mortality, especially in metastatic cases, which would have manifested 3–5 years after sentinel node biopsies became prevalent. After 2005, a number of lower impact treatment improvements were implemented in NZ. Herceptin was approved and funded for use in 2007. Taxanes were funded also at the same time. Aromatase inhibitors were approved and funded for use in 2005 if a woman was unable to tolerate tamoxifen, and became available fully funded as first-line treatment in 2007 (Gregory M, 2018, personal communication). These treatment improvements on breast cancer mortality would also be expected to manifest 3–5 years after implementation and would have impacted on cohorts of women diagnosed in 2006–8 examined in the present study. Finally, ad hoc screening was anecdotally reported in NZ during the late 1990s, particularly in Auckland and Christchurch, coinciding with breast awareness campaigning by non-government organizations. Actual data concerning these activities, which could have influenced breast cancer mortality outcomes, are not available.
The causal evidence for breast cancer mortality reductions resulting from screening mammography comes from randomized trials conducted in the 1970s and 1980s on populations invited and not invited to screening.18,30 Screening recommendations have been based on the results of such trials from Sweden, Edinburgh, New York, and Canada. Meta-analyses of these trials 31 have suggested reductions in breast cancer mortality of 24–2932,33 and 20–30%18,30 in those invited to screening.
Service studies of mammography screening employing a variety of methodologies in the United Kingdom,34,35 Holland, 36 Finland, 37 Sweden,18,30,38 and Australia22,24,25,39 have indicated lower mortality associated with screening compared with non-screened populations, although not all results reached statistical significance. Australian studies have shown significant breast cancer mortality reductions associated with screening mammography using different study designs and analytical approaches.24,25,39 Moreover, there is evidence that reductions in breast cancer mortality from screening increase with longer follow-up times. 40 Most of these studies have adjusted for screening selection bias and found the mortality reduction to persist.
A strength of this study is that data are sourced from complete and accurate national cancer registration and death recording systems. In addition, as screening mammography commenced in NZ in 1999, after the introduction of augmented adjuvant treatment for early breast cancer around 1990, the effects of introduction of screening are temporally separate from effects of introduction of major adjuvant treatment improvement, and separated effects can therefore be delineated more readily. A weakness of the study is lack of information on private mammography outside BSA, which may include de facto screening, particularly prior to 1999. In NSW, Australia, prior to the introduction of screening mammography, private mammography rates in women aged 50–69 were approximately 12% for 1989–92, and thereafter declined sharply to 7% by 1996, after screening mammography in NSW reached full geographic coverage. 41 Another weakness of this study is the difficulty of quantifying the impacts of treatment improvements on breast cancer mortality after the advent of service screening, and of the impact of screening in general on breast cancer treatment for women outside the screening target age groups.
Conclusion
Since the early 1990s, breast cancer mortality in all NZ women has declined, and a further steepening of breast cancer mortality reduction occurred after the introduction of service mammography screening in 1999 in the age group 50–64. Incidence-based analyses confirm the association between screening mammography and breast cancer mortality, and these results are consistent with those from individual-based trial and observational studies.
Footnotes
Acknowledgement
The authors would like to acknowledge one of the anonymous peer reviewers of this paper whose suggestions improved it considerably.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
