Abstract
Objective
To investigate the impact of population mammography screening on breast cancer incidence trends in New Zealand.
Methods
Trends in age-specific rates of invasive breast cancer incidence (1994–2014) were assessed in relation to screening in women aged 50–64 from 1999 and 45–69 following the programme age extension in mid-2004.
Results
Breast cancer incidence increased significantly by 18% in women aged 50–64 compared with 1994–98 (p<0.0001), coinciding with the 1999 introduction of mammography screening, and remained elevated for four years, before declining to pre-screening levels. Increases over 1994–99 incidence occurred in the 45–49 (21%) and 65–69 (19%) age groups following the 2004 age extension (p<0.0001). Following establishment of screening (2006–10), elevated incidence in the screening target age groups was compensated for by lower incidence in the post-screening ⩾70 age groups than in 1994–98. Incidence in women aged ⩾45 was not significantly higher (+5%) after 2006 than in 1994–98. The cumulated risk of breast cancer in women aged 45–84 for 1994–98 was 10.7% compared with 10.8% in 2006–10.
Conclusions
Increases in breast cancer incidence following introduction of mammography screening in women aged 50–64 did not persist. Incidence inflation also occurred after introduction of screening for age groups 45–49 and 65–69. The cumulated incidence for women aged 45–84 over 2006–10 after screening was well established, compared with 1994–98 prior to screening, shows no increase in diagnosis. Over-diagnosis is not inevitable in population mammography screening programmes.
Introduction
The population of New Zealand (NZ) in 2013 was 4.2 million. 1 Breast cancer is the most common cancer in NZ women, accounting for 28% of all female cancers, and is the second most common cause of cancer death in women after lung cancer. 2 For women aged 25–64, breast cancer is the leading cause of cancer death. 2 Accurate breast cancer incidence from complete cancer registration is available in NZ from 1994. 3
Reflecting trends in other developed countries,4,5 the incidence of invasive breast cancer in NZ may have risen over the past half century from changes in risk factors such as obesity,6,7 fertility patterns,8,9 use of hormone replacement therapy, 10 and the introduction of mammography screening.4,5,11 A component of screening-related incidence may include over-diagnosed cancers, which is evident in some screening programmes,5,12–14 but not others.15,16 Immediately following the introduction of a mammography screening programme, there is generally a rise in recorded breast cancer incidence, known as the ‘prevalence peak’,11,16,17 from diagnosis of prevalent cancers in the initial screening round, and earlier diagnosis of incident cases. Following this initial increase, the overall incidence is expected to return to pre-screening levels as incident cancers are redistributed to younger ages compensated for by lower incidence at older ages. This expected pattern has been observed in mammography screening programmes in Denmark and Spain,16,18 but not in Australia,5,11 the United Kingdom,4,19 and Sweden, 12 where incidence has remained elevated over an extended period of time and raises the possibility of over-diagnosis, where cancers detected by screening would never have manifested clinically over a lifetime.
This investigation aimed to determine the extent to which the introduction of mammography screening in NZ has been associated with initial incidence inflation, and whether incidence has returned to background levels (as expected), or whether any incidence inflation has continued, suggesting over-diagnosis. The analyses here are of annual and biennial trends in breast cancer incidence in relation to screening, for age groups targeted and not targeted by service screening, to be directly comparable with other studies of breast cancer incidence and screening, including those conducted in New South Wales (NSW), Australia, 5 and South Australia. 20
BreastScreen Aotearoa (BSA), the national, publicly funded mammography screening programme, commenced in 1999, initially targeting women aged 50–64. In July 2004, the target age group was extended to 45–49 and 65–69. BSA offers bilateral, two-view mammography screening to age-eligible women without direct charge. All mammograms are independently read by two radiologists, with arbitration of conflicting findings by a third. Recruitment is via self-initiated enrolment or referral from a medical practitioner, then invitation by the programme, with a target of 70% coverage for screening age groups. 21 The latest biennial coverage for women aged 50–69 screened June 2015–May 2017 is 72%. 22
Methods
Annual female breast cancer registrations by five-year age group were obtained from the NZ Ministry of Health for 1994–2014, using data reported to the NZ Cancer Registry,2,23 which collects all incident cases of cancer nationally with mandatory pathology notification since 1994. Only incident invasive breast cancers are analysed. Population estimates are based on successive quinquennial censuses (most recent 2013), with intervening annual totals estimated by interpolation, and by projection for post-2013 populations. Mammography screening rates for BSA are obtained from de-identified unit record data made available to the authors by BSA. 24 Coverage is expressed as eligible women with one or more mammograms over two years divided by the average annual population (biennial rate).
Annual age-specific breast cancer incidence rates (women aged ⩾45) are calculated by five-year age group. Incidence rates are aggregated by age groups 45–49, 50–64, and 65–69 and by screening regime. Incidence rates for women aged 50–64 are directly age standardized to the 2006 NZ Census female population by five-year age group. 25 Confidence intervals (CIs) (95%) of incidence rates are from the normal approximation of the binomial. 25 Age-specific analysis of breast cancer incidence, aggregated over five years prior to the introduction of screening in 1999 (1994–98), and for five years following the first prevalent screening round after age extension in 2004 (2006–10), is used to assess evidence for over-diagnosis associated with screening mammography, as indicated by incidence inflation persisting in screening target age groups after the establishment of screening without compensating lower incidence in older post-screening age groups. Cumulative risks with 95% CI 26 are estimated for women aged 45–84 to compare the overall difference between the two incidence plots. Areas under these incidence curves are also estimated and compared directly, using a simple Riemann sum approximation, 27 and also from definite integrals of six-degree polynomials fitted to each plot, each estimated over the 45–⩾85 age range. Two-yearly incidence rates in target populations are used for comparison with corresponding biennial screening coverage in trend analyses. Annual incidence by age groups is compared between the periods prior to and after the introduction of service screening: 1994–98 compared with 1999–2003 when women aged 50–64 were targeted for screening, and 1994–98 compared with 2004–14 following age extension of screening to ages 45–49 and 65–69.
Ethics approval for this study was not required as it relied on de-identified screening data and publicly available aggregate cancer incidence and population data.
Results
Breast cancer incidence in relation to service mammography screening in New Zealand 1994–2014.
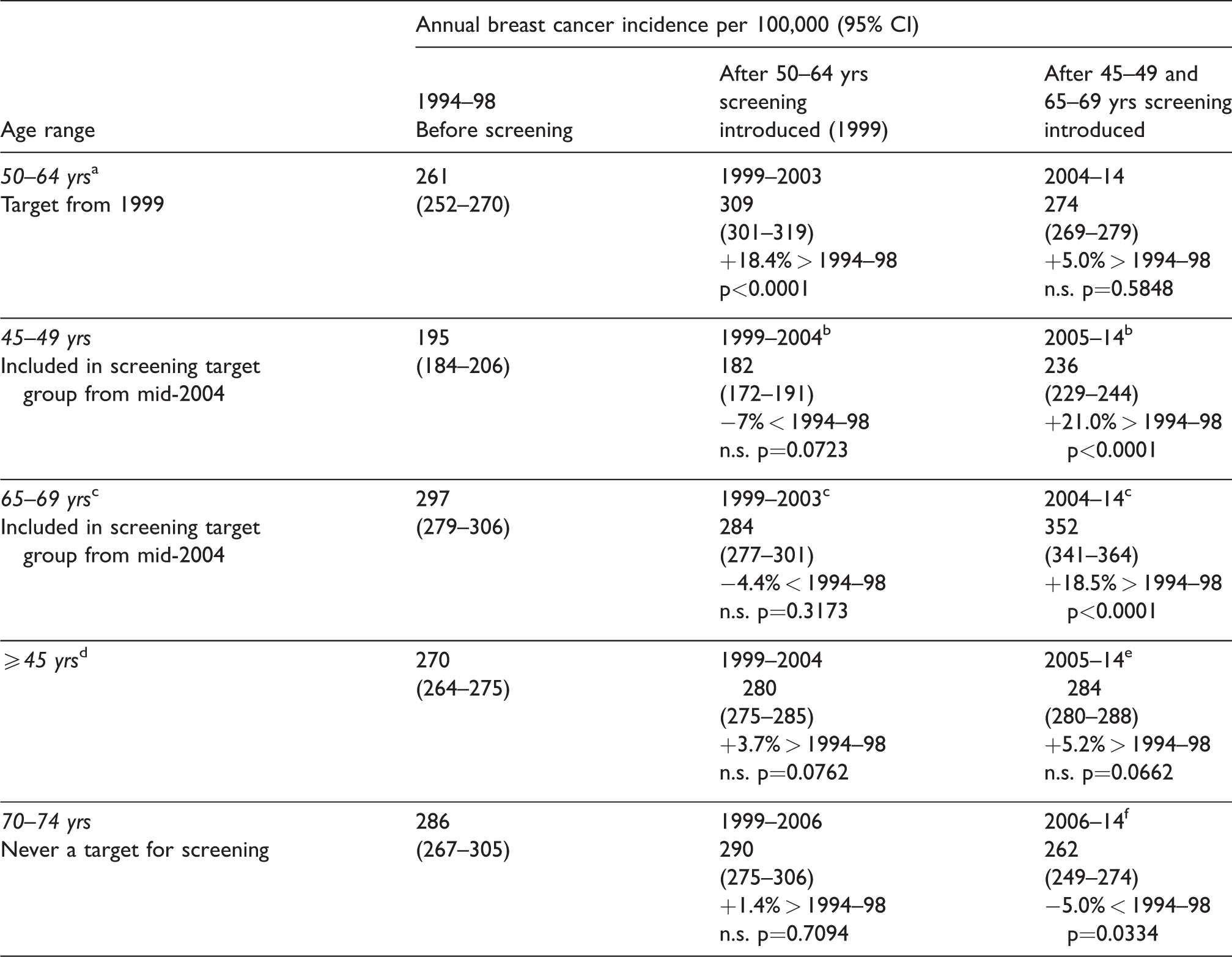
CI: confidence interval.
aAge standardized (direct) breast cancer incidence using three five-year age groups.
bScreening participation in 45–49 year age group was low until 2005.
cScreening participation in 65–69 year age group increased with age extension in mid-2004 because participants were already enrolled with the screening service.
dAge standardized (direct) breast cancer incidence using eight five-year age groups and age group 85+.
ePeriod following target age extension in 2004 and detection of predominantly prevalent breast cancers in previously un-screened women aged 45–49 and 65–69.
fA proportion of these women would have received screening mammography previously when in target age group in 1999–2005.
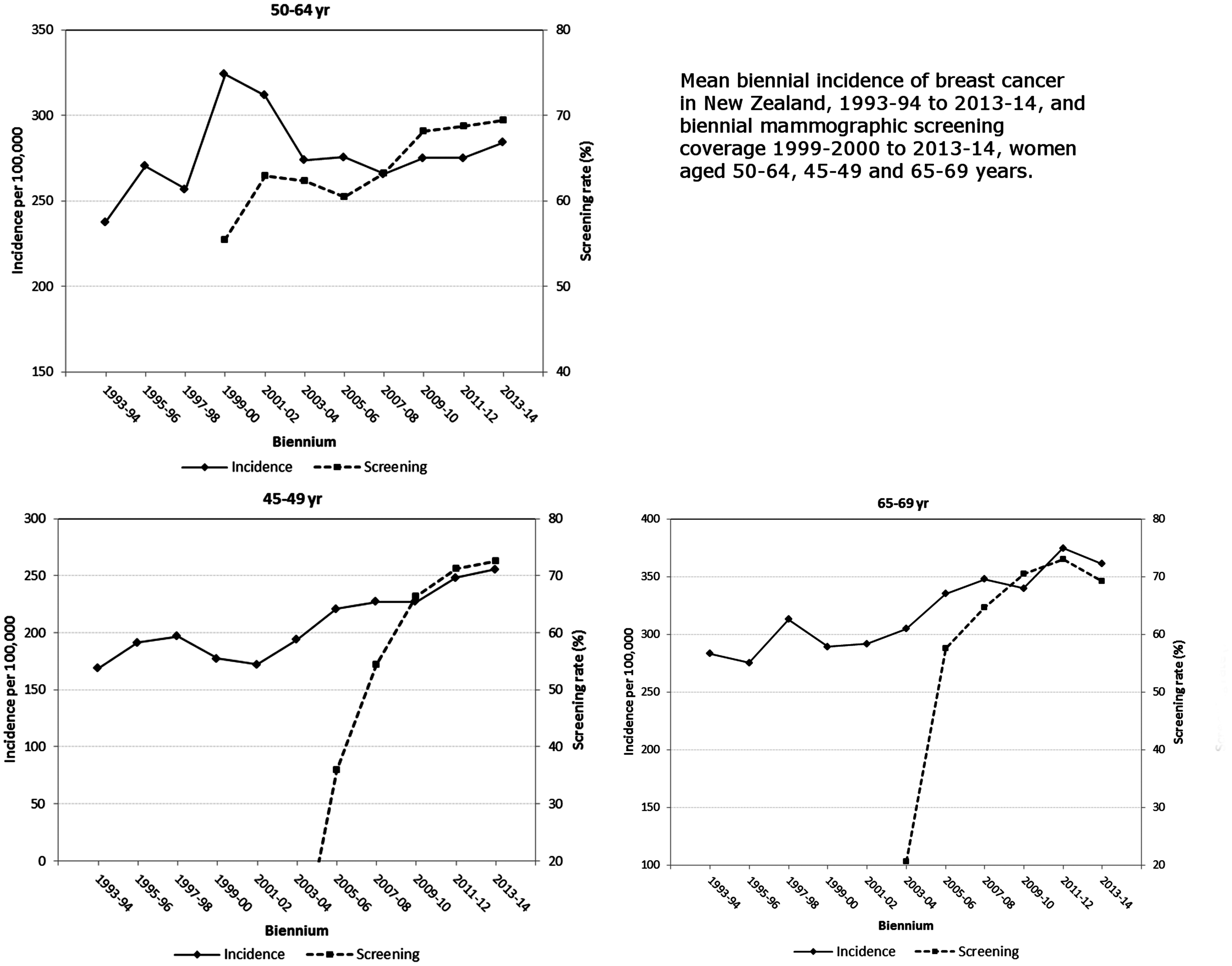
Incidence of breast cancer in New Zealand and biennial mammography screening coverage: 50–64 yrs, 45–49 yrs, and 65–69 yrs.
Biennial screening in women aged 50–64 rose steadily, from 54% in 1999–2000, to 69% by 2014 (Figure 1). Most of the screening-related breast cancer incidence increase occurred during 1999–2002, followed by a rapid return to pre-screening incidence levels by 2003–4.
With the extension to age 45–69 in mid-2004, screening participation in women aged 45–49 rose from 0.5% in 2003–4 to 36% in 2005–6, and exceeded 70% by 2013–14. Breast cancer incidence in these women correspondingly rose from 195/10
From 2006, when screening was well established with sufficient screening participation and predominantly incident breast cancers detected, age-specific breast cancer incidence for 2006–10 was higher in the screening target age groups than in 1994–98 prior to the establishment of screening, and lower for women aged 70 and older compared with the pre-screening period (Figure 2). These trends show inflated screening-related incidence from cancers detected earlier through screening than they would otherwise have been being compensated for by lower incidence in older age groups, suggesting that little or no over-diagnosis has occurred. The differences in the areas under the incidence curves in women over ages 45 to ⩾85 indicate total incidence differences between populations offered and not offered screening. From the Riemann approximation, the area under the 2006–10 plot was 2335 per 10
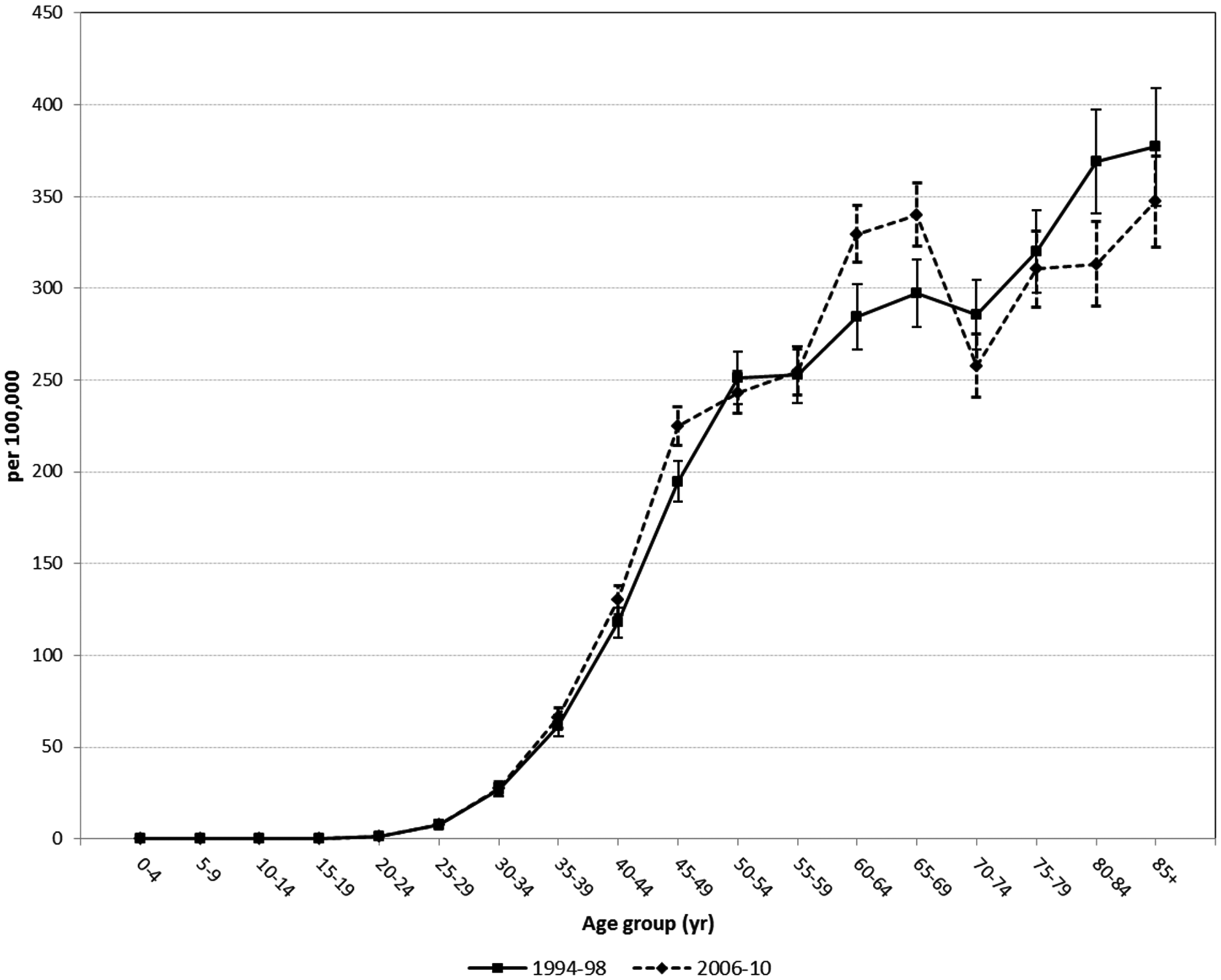
Age-specific breast cancer incidence in New Zealand women 1994–98 and 2006–10.
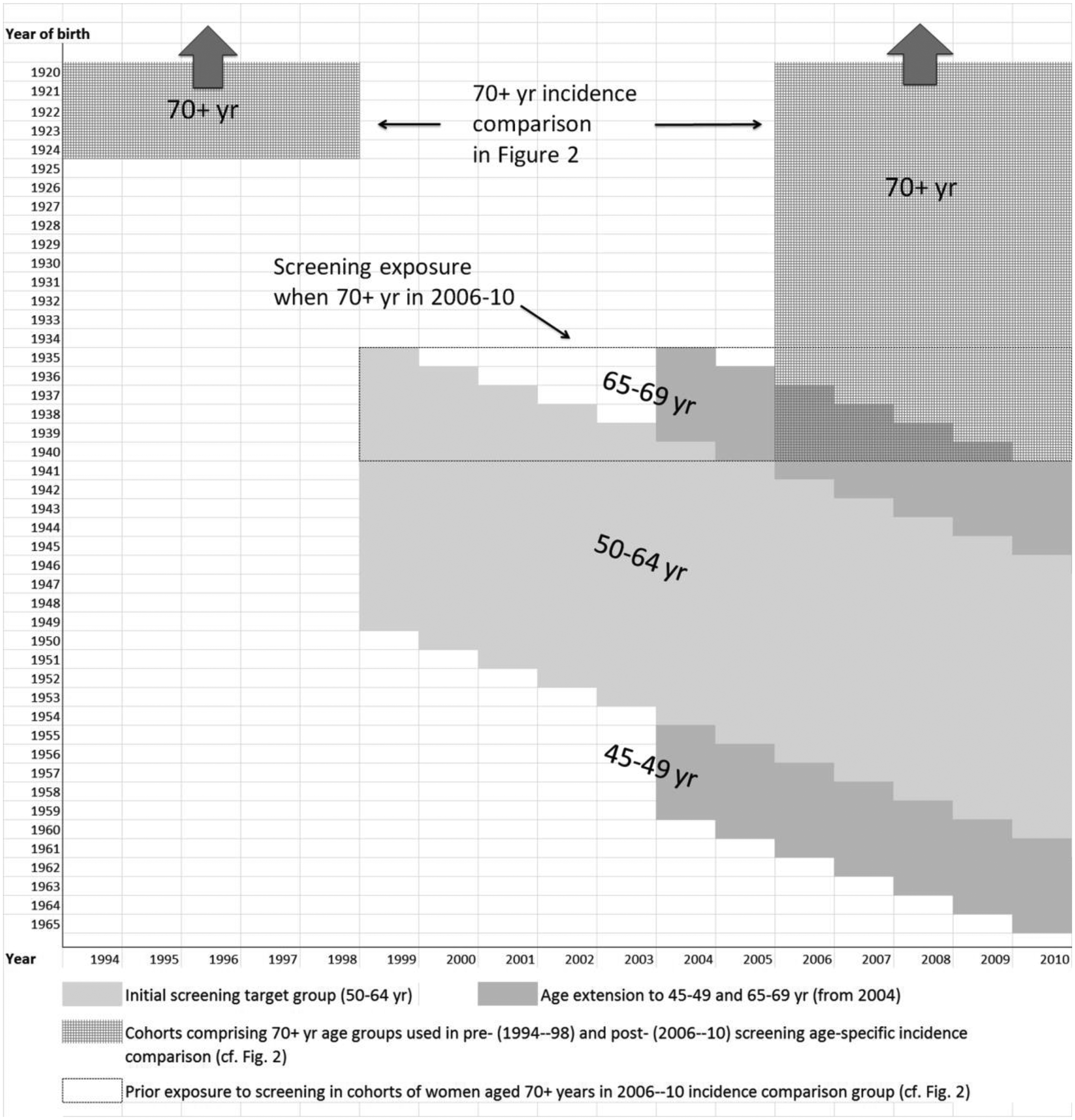
Screening coverage by (birth) cohort and period for screening age groups, and previous exposure to service screening in ⩾70 year women in 1994–98 and 2006–10, for the age-specific incidence comparison in Figure 2.
For all women aged 45 and older, breast cancer incidence (age standardized) was not significantly higher in 1999–2005, during the establishment of screening and before age extension, than prior to service screening (1994–98). In 2006–14, following the 2004 age extension, incidence in women aged ⩾45 was not significantly higher than in 1994–98. Despite higher incidence in women aged 45–49 and 65–69 extending for a longer period compared with women aged 50–64, significantly higher breast cancer incidence overall for women aged ⩾45 is not manifest. No sustained rise attributable to screening mammography was observed.
Discussion
The introduction of screening mammography in 1999 for women aged 50–64 coincided with a subsequent 1999–2002 increase in the incidence of invasive breast cancer. When screening became available to women aged 45–49 and 65–69 in 2004, an increase in breast cancer incidence also occurred in these women.
The subsequent fall in incidence rates by 2003 in women aged 50–64, to levels close to those observed in the period before service screening, represents evidence against any substantial over-diagnosis as a feature of organized screening in NZ for these women. This is highlighted by the comparison of age-specific incidence before and after the advent of service screening, showing the expected higher incidence in women aged 50–64, followed by compensatory lower incidence in the older ⩾70 age groups compared with the period before service screening (Figure 2). Even though women aged ⩾70 are not targeted for screening, by 2006–10 some of these women would have been screened when younger (see Figure 3). This represents a virtual textbook example of how screening should affect breast cancer incidence, by detecting cancers earlier than they would otherwise have been detected, but not producing a significant rise in overall incidence, which is confirmed by before-and-after comparisons of cumulative incidence, areas under the age-specific incidence curves, and by lack of difference in age-adjusted incidence in women aged ⩾45 in 2006–14 compared with 1994–98. Further, the lower incidence rates following the establishment of screening in those aged ⩾70, who experienced screening exposure at younger ages, illustrate the population effects of earlier diagnosis of breast cancer.
Evidence of over-diagnosis would manifest as increased incidence in the screening age group persisting across these age groups after the establishment of the screening programme, and also in elevated overall breast cancer incidence, as has occurred in some service screening programmes, such as in NSW Australia, 5 Norway, 14 and Sweden. 12 However, the quantification of the extent of over-diagnosis is difficult, and estimates can be sensitive to the approach used, especially so in the example of Norway. 28
The less immediate increase in breast cancer incidence in women aged 45–49 following the age extension of screening in mid-2004 may be attributed to the need for initial recruitment and enrolment with the screening service of these women, who had not previously been offered service screening. Incidence increased in 2005, coinciding with increased mammography coverage in women aged 45–49. In contrast, many women aged 65–69 had previously been enrolled in mammography screening when aged 60–64 and were already recruited and screened with consequent higher initial coverage than in women aged 45–49, thus manifesting a correspondingly more immediate and pronounced incidence increase in 2004. In NZ, unscreened women are recruited to screening through health providers and the general promotion of screening, while in other countries unscreened women may be recruited through personal invitations sent from the screening programme when they reach the screening target age range. In NZ, once women are recruited and on the BSA screening register, they are sent routine screening invitations and reminders.
Initial increases in incidence coinciding with the introduction of screening are expected, as prevalent cancers in asymptomatic women produce incidence inflation from earlier diagnosis.11,16 This finding is consistent with incident increases seen in Sweden (45%) and Norway (50%) during the early years of screening. 12 In Spain, incidence increased by 20–30% in target age groups, 18 and in NSW, Australia, the incidence increase was 33%. 11 In NZ, initial incidence rates in women aged 50–64 returned to levels slightly above rates before service screening after four years, but not beyond those expected as a result of screening detecting incident cancers before they become clinically manifest. It is expected that after most prevalent cases are detected, breast cancers detected by screening will continue to be diagnosed earlier in predominantly already-screened women, producing incidence rates somewhat above those in populations not offered screening, and that are expected to be compensated for by lower incidence in older women who age into groups no longer offered screening, as observed in NZ.
The observed NZ increases in breast cancer incidence in women aged 45–49 and 65–69 coinciding with screening have not yet declined to levels similar to those prior to the age extension in 2004 (see Figure 1). It is possible that, for women aged 45–49 and 65–69, higher proportions of previously unscreened women with prevalent cancers are being screened over a more extended period. Nevertheless, incidence in these age groups should not be expected to return to pre-screening levels, as cancers are being detected earlier. In the absence of over-diagnosis, raised screening-related incidence in women aged 45–49 would be compensated for by lower incidence in women aged ⩾50, as women aged 45–49 age through the screening target age groups. Similarly, raised incidence in women aged 65–69 should be compensated for by lower incidence in women aged ⩾70, who are out of the screening target age range, as has been observed.
Strengths of this study are that incidence data are sourced from a complete and accurate national cancer registry, and service screening participation rates originate from complete and accurate national aggregations of BSA data. Deficiencies include the lack of information on mammography in the private sector, which may include ad hoc mammography and de facto screening. Anecdotal information indicating that screening mammography is infrequent outside of government service screening is supported by the most recent programme screening participation rates of 72%. Screening participation in NZ is similar to that of the screening trials, and in Australia such screening rates are approached only if private mammography de facto screening is included.
11
There is no suggestion of breast cancer incidence increases occurring just prior to the actual introduction of service screening in NZ, coincident with publicity concerning the imminent introduction of screening, that could be attributable to increased private screening, as was documented in NSW, Australia.
11
On these grounds, evidence for substantial private mammographic screening in NZ is meagre, although there may have been some private mammography screening before 1999, prior to the introduction of service screening. Although lack of complete cancer registration before 1994 poses some issues of data completeness, baseline incidence was available for five years prior to the introduction of screening in women aged 50–64, where incidence varied between 250 and 275/10
Reduction in overall breast cancer incidence following the prevalence peak has been observed in some other mammography screening programmes. The initial breast cancer incidence increase of 20–30% in Spain following the gradual introduction of mammography screening for women aged 45–64 and then 45–69 during the 1990s 18 began to fall in all age groups ⩾45 within five years following achievement of high participation rates. Similarly, in Denmark a pronounced peak in breast cancer incidence in screened age groups after the introduction of screening around 1992 was followed by a decline, to levels compatible with those expected from this population in the absence of screening. 16 In contrast, several countries have experienced a sustained breast cancer incidence increase following the introduction of service screening. Breast cancer incidence in NSW, Australia, rose by 33% between 1992 and 1994 in the mammography screening target age group, coincident with increased mammography coverage. 11 Australian data for 2009 indicate that incidence in women aged 50–69 has remained elevated well above levels prior to service screening, 29 and incidence in women aged ⩾70 (not a target for screening) has not fallen below the incidence in women aged ⩾70 in the period prior to service screening. 5 Breast cancer incidence in Sweden rose by 45% during 1986–2000 following the introduction of screening, and in Norway by 54% during 1998–2001 following screening introduction in 1996. 12 No decline in Sweden was observed by 2004, 30 and by 2008, incidence in Norway remained elevated in all screening age groups, well above levels observed prior to service screening. 31
In England, mammography screening was initiated between 1988 and 1993, and breast cancer incidence rates in the initial screening target age group of women aged 50–64 rose sharply over 1989–92, from 200 to nearly 300/10
Sustained increased breast cancer incidence following the introduction of population mammography screening suggests over-diagnosis. Estimation of over-diagnosis involves separating the increased incidence due to lead-time effects from earlier pre-clinical detection, from the effects of underlying secular trends in breast cancer risk factors. Estimates of rates of over-diagnosis in mammography screening programmes have varied widely, depending on the programme studied and the methodology used, from negligible to more than 50%. 34 A review of the UK screening programme in 2012 estimated over-diagnosis to be, on average, 19%, 35 although it may be inappropriate to produce average estimates of screening-related over-diagnosis when such over-diagnosis appears not inevitable, as exemplified by the present study and other reports.34,36
A factor that may account for differences in over-diagnosis between different screening mammography programmes is the medico-legal environment. In NZ, the no-fault insurance system through the Accident Compensation Corporation may mean that there is less medico-legal pressure on histopathologists. In Australia, the medical compensatory system is fault based, subject to tort liability, and medical practitioners, including pathologists, are required to obtain (mandatory) medical indemnity insurance cover. 37 Histopathologists may tend to err on the side of caution to avoid misclassifying cancer as a benign entity and then be subject to legal consequences. There seems little doubt that mammography-related breast cancer over-diagnosis is evident in the Australian and several international service screening programmes. A possible, if unlikely, scenario is that over-diagnosis was already present in clinical practice in NZ prior to the advent of screening, in which case this study has shown no overall increase in breast cancer incidence and over-diagnosis associated with the advent of service screening mammography.
Breast cancer mortality reductions of the order of 30% have been shown to be associated with screening mammography in meta-analyses of randomized controlled trials38–41 and reviews of service studies.42,43 Breast cancer mortality reductions of −34% are evident for ever-screened NZ women, 44 and are 25–34% in aggregate analyses of NZ breast cancer mortality and −17% in incidence-based analyses before and after introduction of service screening. 45 The present study shows that expected breast cancer mortality reductions in NZ have occurred with the introduction of organized service screening mammography, without evidence of over-diagnosis. This study provides evidence that screening-related over-diagnosis of breast cancer is not inevitable, nor necessary, in successful population mammography screening programmes. Further, breast cancer over-diagnosis thus may be remedied by health service interventions.
Footnotes
Acknowledgement
The authors would like to acknowledge one of the anonymous peer reviewers of this paper whose suggestions improved it considerably.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
