Abstract
Objectives
Low-dose computed tomography lung cancer screening has been shown to reduce lung cancer mortality but has a high false-positive rate. The precision medicine approach to low-dose computed tomography screening assesses subjects’ benefits versus harms based on their personal lung cancer risk, where harms include false-positive screens and resultant invasive procedures. We assess the relationship between lung cancer risk and the rate of false-positive LDCT screens.
Methods
The National Lung Screening Trial randomized high-risk subjects to three annual screens with low-dose computed tomography or chest radiographs. Following the completion of National Lung Screening Trial, the Lung CT Screening Reporting and Data System (Lung-RADS) classification system was developed and retrospectively applied to National Lung Screening Trial low-dose computed tomography findings. The rate of false-positive screens (by Lung-RADS) and the resultant invasive procedures were examined as a function of lung cancer risk estimated by a model.
Results
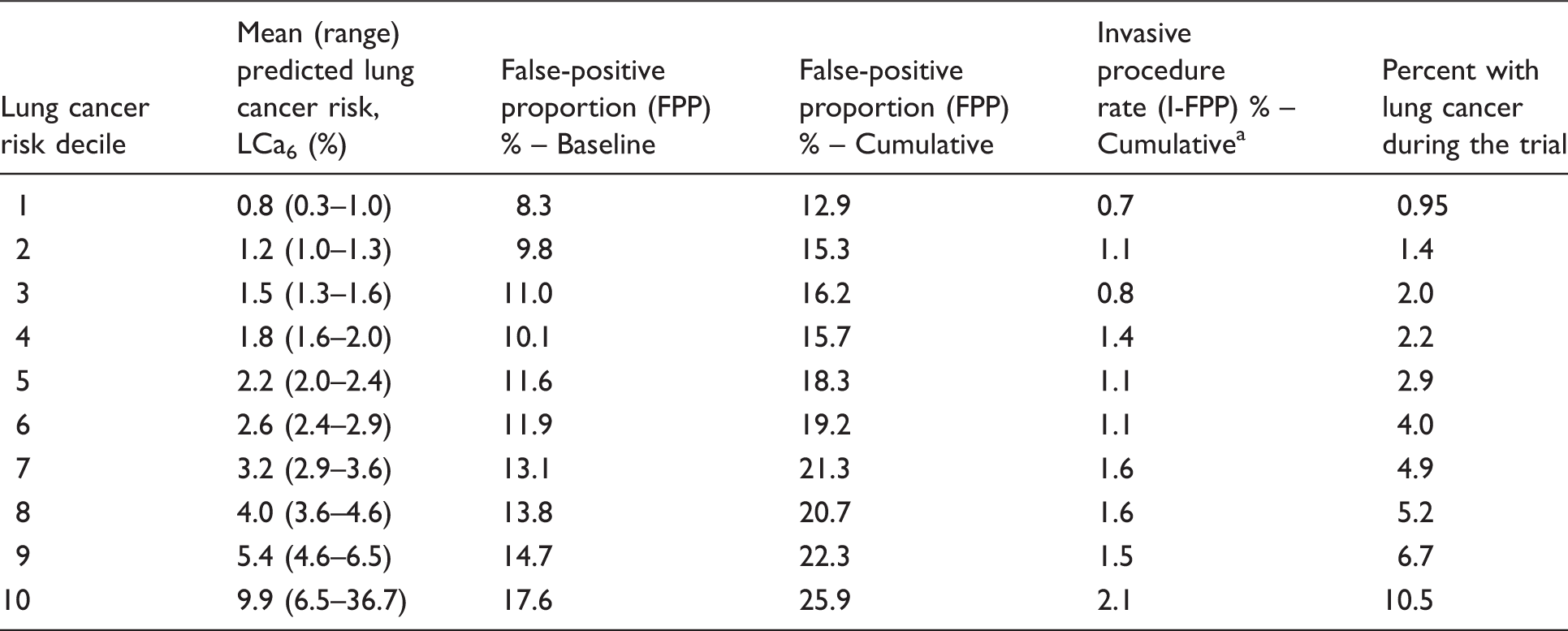
Of 26,722 subjects randomized to the low-dose computed tomography arm, 26,309 received a baseline screen and were included in the analysis. The proportion with any false positive over three screening rounds increased from 12.9% to 25.9% from lowest to highest risk decile, and the proportion with an invasive procedure following a false positive also significantly increased from 0.7% to 2.0% from lowest to highest risk decile.
Conclusion
These findings indicate a need for personalized low-dose computed tomography lung cancer screening decision aids to accurately convey the benefits to harm trade-off.
Introduction
The National Lung Screening Trial (NLST) of low-dose computed tomography (LDCT) screening 1 demonstrated a reduction in lung cancer mortality but with an average false-positive rate (FPR) of 23% in each of the three screening rounds. In 2014, the American College of Radiology released the Lung CT Screening Reporting and Data System (Lung-RADS) scoring system, which had the potential to reduce lung cancer screening false positives. 2 A retrospective analysis of NLST showed that employing Lung-RADS reduced the FPR on baseline and postbaseline screening by 50% and 75%, respectively, but false positives remained substantial. 3 The American College of Radiology recommends standardization in LDCT screening interpretation using Lung-RADS. 2
The NLST eligibility criteria included having a 30+ pack-year smoking history, and either current smoking or having quit within the past 15 years. 1 These criteria, with minor adjustments, have been used by various guideline bodies, including the US Preventive Services Task Force, in recommending lung cancer screening. 4 An alternative approach, under the rubric of precision-screening, is to utilize a lung cancer risk model to determine who should be screened and also to assess subjects’ benefits versus harms of screening.5,6
A prior analysis of NLST, based on the original NLST screen positivity criteria, not on Lung-RADS classifications, showed that the number needed to screen to prevent one lung cancer death decreased with estimated lung cancer risk, but the FPR increased with risk. 7 It would also be of interest to examine whether invasive procedures following false positives increase with lung cancer risk. Here, we analyze the relationship between lung cancer risk and the Lung-RADS FPR and resultant invasive procedure rate in NLST.
Methods
The NLST randomized subjects aged 55–74 years with the previously described smoking history to LDCT or chest X-ray screening. 1 Subjects were enrolled at 33 United States screening centers during 2002–2004 and received screens at baseline and annually for two more years. Participants completed an eligibility verification and baseline questionnaire, which covered smoking history, medical history, family history, and demographics. All subjects provided informed consent, and the trial was approved by Institution Review Boards at each screening center. Radiologists in the NLST reported nodule characteristics, including size and attenuation. At postbaseline screens they reported, based on examining prior images, whether any abnormality was preexisting or new, and if preexisting, whether it had grown or changed attenuation since the past screen(s). Other abnormalities, including adenopathy or effusion, were also recorded. Investigators tracked diagnostic procedures and cancer diagnoses, and subjects were followed with annual surveys to ascertain incident cancers in the absence of a positive screen. All reported cancers were verified with medical records.
An earlier analysis retrospectively assigned Lung-RADS scores to NLST CT scans based on recorded nodule characteristics. A false-positive screen was defined based on Lung-RADS categories, with a category of three or four denoting a positive screen. A Lung-RADS positive screen not followed by lung cancer diagnosis within three years was defined as a false positive. An invasive procedure following a false positive was defined as a Lung-RADS false-positive screen with an invasive procedure within one year. 3
Using the Lung-RADS criteria, we computed the proportion of all screened subjects receiving a false-positive result denoted as the false-positive proportion (FPP). The FPP differs from the standard FPR as the denominator for FPP is all subjects screened, not only those without lung cancer as is the case for FPR. The FPP is a better metric for comparing the actual risk of experiencing a false positive among screening groups with different lung cancer risk levels. FPP was computed for the baseline screen and cumulatively over the three rounds. Similarly, we computed the proportion (of all screened subjects) who received an invasive procedure following a false positive (I-FPP), at baseline and cumulatively.
The Tammemagi lung cancer risk model predicts six-year lung cancer risk (LCa6) in ever-smokers utilizing smoking history, medical history, and demographic variables. 5 The model included the smoking-related variables of smoking duration, cigarettes per day, current smoking status, and years since quit, along with age, race/ethnicity, family history of lung cancer, chronic obstructive pulmonary disease (self-reported), personal history of cancer, education, and body mass index. We applied the model to each NLST subject to generate their LCa6 value.
All subjects receiving the baseline LDCT screen were included in the analysis. LCa6 values were categorized by decile for descriptive analyses; a p value for trend was computed using decile rank. We developed logistic regression models of FPP (baseline and cumulative) and I-FPP by LCa6. Goodness of fit analysis, using the Hosmer–Lemeshow test, demonstrated that log(LCa6) provided a better fit than LCa6 for all three models, with each model showing adequate fit; therefore, the final models utilized log(LCa6). Additionally, major individual risk factors comprising the Tammemagi model were examined for their relation to FPPs.
Results
False-positive proportion (FPP) and invasive procedure proportion (I-FPP) by lung cancer risk decile.
Invasive procedure following a false positive.
Note: All decile levels had between 2630 and 2632 subjects; p value for trend <0.001 for baseline and cumulative FPP and I-FPP.
The logistic models showed odds ratios of 1.26 (95% confidence interval (CI): 1.21–1.30; p < 0.001), 1.26 (95% CI: 1.22–1.30; p < 0.001), and 1.31 (95% CI: 1.18–1.44; p < 0.001) per a doubling of lung cancer risk for baseline FPP, cumulative FPP, and cumulative I-FPP, respectively.
With respect to specific risk model factors, cumulative FPP was significantly associated with pack-years (20.1% for 50+ vs. 17.6% for <50 pack-years), smoking status (19.6% for current vs. 18.0% for former smokers), history of chronic obstructive pulmonary disease (23.3% for history vs. 17.8% for no history), and age at baseline (23.2% for 65+ vs. 17.2% for under 65 years).
Discussion
In this analysis of NLST data, using current diagnostic criteria (Lung-RADS), the occurrence of false-positive screens substantially increased with predicted lung cancer risk, roughly doubling from the lowest to highest risk decile. The occurrence of invasive procedures following a false positive also substantially increased with predicted risk. There are several reasons why false positives may increase with lung cancer risk, including increased risk of nodules with increased cumulative smoking burden 8 and age. 9 Our findings demonstrated increased FPP with increased age and pack-years and concomitant chronic obstructive pulmonary disease. Although the use of Lung-RADS was shown in NLST to decrease the FPR, it also modestly decreased sensitivity (i.e., increased the false-negative rate). The impact of this change on the mortality reduction of LDCT is unknown and needs further study.
Our findings have important implications for lung cancer screening. Acknowledging the potential harms of screening, the Centers for Medicaid & Medicare requires clinicians to conduct a shared decision-making visit with a patient prior to the screening event. 10 As seen in our analysis, as individuals’ risk of lung cancer increased from lowest to highest decile, their risk of a false-positive screen and having a resultant invasive procedure doubled and tripled, respectively. Therefore, accurately conveying the potential risks and benefits of lung cancer screening will require personalized decision aids.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
