Abstract
Objective
To evaluate the association between repeated faecal occult blood testing and advanced colorectal cancer risk at population level in Canada.
Methods
A retrospective cohort study of all Ontario residents aged 56–74 diagnosed with colorectal cancer from 1 April 2007 to 31 March 2010, identified using health administrative data. The primary outcome was stage IV colorectal cancer, and primary exposure was faecal occult blood testing use within five years prior to colorectal cancer diagnosis. Patients were categorized into four mutually exclusive groups based on their exposure to faecal occult blood testing in the five years prior to colorectal cancer diagnosis: none, pre-diagnostic, repeated, and sporadic. Logistic regression was utilized to adjust for confounders.
Results
Of 7753 patients (median age 66, interquartile range 61–70, 62% male) identified, 1694 (22%) presented with stage I, 2056 (27%) with stage II, 2428 (31%) with stage III, and 1575 (20%) with stage IV colorectal cancer. There were 4092 (53%) with no record of prior faecal occult blood testing, 1485 (19%) classified as pre-diagnostic, 1693 (22%) as sporadic, and 483 (6%) as repeated faecal occult blood testing. After adjusting for confounders, patients who had repeated faecal occult blood testing were significantly less likely to present with stage IV colorectal cancer at diagnosis (Odds ratio 0.46, 95% Confidence Interval 0.34–0.62) than those with no prior faecal occult blood testing.
Conclusions
Repeated faecal occult blood testing is associated with a decreased risk of advanced colorectal cancer. Our findings support the use of organized screening programmes that employ repeated faecal occult blood testing to improve colorectal cancer outcomes at population level.
Introduction
Colorectal cancer (CRC) is the third most common cause of cancer death in males and the second in females, with approximately 1.2 million new cases and over 600,000 deaths estimated worldwide in 2008. 1 CRC screening using faecal occult blood testing (FOBT) has been shown to reduce CRC mortality in randomized controlled trials (RCTs) by 15–33%.2–6 However, generalizability of these findings may be limited, as compliance to FOBT screening 7 and endoscopic follow-up for FOBT positive patients 8 outside of clinical trial settings is variable.
Data from The United Kingdom 9 and France 10 suggest that organized screening programmes can enhance screening participation, and lead to the detection of CRC at an earlier stage. In 2007, the province of Ontario instituted Canada’s first organized colorectal screening programme, ColonCancerCheck (CCC). Since programme inception, participation with FOBT screening has increased, from 18.9% in 2005–2006 to 29.9% in 2013–2014.11,12 However, the success of a population-based screening programme may depend on continued participant adherence, 13 as repeat testing can increase CRC detection and survival. 14 The purpose of this study is to determine the association between repeated use of FOBT and the detection of advanced CRC at the population level in Ontario. We hypothesized that repeated use of FOBT is associated with a decreased risk of advanced CRC.
Methods
We performed a retrospective cohort study to examine the association between repeated FOBT use and the detection of advanced CRC, using five Ontario administrative healthcare databases, linked using unique encoded identifiers and analysed at the Institute for Clinical Evaluative Sciences.
The Ontario Cancer Registry (OCR) captures all incident cancer cases diagnosed in Ontario since 1964. Site of primary CRC was determined from pathology reports and discharge summaries, and collected within the registry. Since 2007, Cancer Care Ontario has captured the stage of CRC with the use of imaging, surgical, and histological data from hospitals where the diagnosis was made. Cancer stage was available for > 90% of cases. The Tumour Node Metastases staging methods were used to establish stage of CRC. 15 The Ontario Health Insurance Plan (OHIP) database contains information since 1991 on claims billed for physician and laboratory services, including FOBT and endoscopic procedures. The Registered Persons Database is a roster of OHIP beneficiaries and maintains age, sex, postal code information, and vital statistics for all Ontarians with a valid OHIP number. The Corporate Provider Database provides information on demographic data on all practising physicians in Ontario. Approximately 74% of Ontarians are registered in a primary care enrolment model (PEMs), in which patients are rostered to a single primary care physician. 16 There are different types of PEMs, which vary in terms of their remuneration structures (ranging from more fee-for-service to more capitation) and in the extent of patient services offered. The Client Agency Program Enrolment (CAPE) is an electronic record of the type of PEM and the linkages between physicians and their rostered patients. The Canadian Institute of Health Information (CIHI) Hospital Discharge Abstract Database includes information on all patients discharged from a hospital or same day surgery unit in Ontario since 1988.
Through the CCC programme, 17 individuals aged 50 to 74 and at average risk for CRC are offered guaiac FOBT; those at increased risk (having one or more first degree relatives with a diagnosis of CRC) are offered colonoscopy. Eligible participants are asked to visit their primary care physicians (PCPs) to be triaged for screening. Although the programme now systematically invites by mail to all eligible persons in Ontario to participate in CRC screening, 18 at the time of the study, these invitations had not yet been implemented. PCPs receive financial incentives to screen their eligible patients. The small proportion (<5%) of individuals without a PCP can obtain an FOBT kit by calling a 1-800-number (INFOline) to have a kit mailed to them if they are deemed to be at average risk. Otherwise they are referred for a diagnostic workup, or assigned to a PCP willing to accept new patients. For average risk individuals with a positive FOBT, defined as one or more positive samples out of six, colonoscopy is recommended within eight weeks.
Using the OCR, we identified a cohort of individuals aged 56–74 who were diagnosed with CRC (International Classification of Diseases, Tenth Revision, codes C18, C18.2–C18.8, C19 and C20) from 1 April 2007 to 31 March 31 2010. The date of CRC diagnosis was defined as the index date. We excluded those with a previous diagnosis of CRC, inflammatory bowel disease, or colonic resection. To ensure that the cohort was eligible for screening with stool based tests, we excluded persons who had a colonoscopy 6 months to 10 years prior, or a flexible sigmoidoscopy 6 months to 5 years prior to being diagnosed with CRC. It was assumed that large bowel endoscopy performed within six months prior to the index date led to the diagnosis of CRC.
The primary outcome was stage IV CRC. The primary exposure of interest was use of FOBT within five years of the index date. It was not possible to determine the indication for the FOBT, but in Ontario, over 85% of FOBTs are performed for screening purposes as part of the CCC programme. 19 To distinguish diagnostic FOBTs, we separated one time FOBTs performed within nine months of the index date from those performed from nine months to five years prior to the index date. The nine months’ time period was selected to encompass published wait times between a positive FOBT and colonoscopy in Ontario.20,21 The five-year exposure period was chosen to allow for the nine month pre-diagnostic period and to encompass two biannual cycles of FOBT as defined by screening guidelines. 22 Patients were categorized into four mutually exclusive groups based on their exposure to FOBT during the five-year period prior to the index date as follows: (1) none – no record of FOBT; (2) pre-diagnostic – only one FOBT performed within nine months of diagnosis; (3) repeated – two or more FOBTs where the latest FOBT was performed within nine months of diagnosis and a second was performed 12–24 months prior to the latest FOBT; and (4) sporadic – all other cases.
Data on age, sex, comorbidity, income (classified as rural or by median neighbourhood income quintile 23 for persons living in urban areas), and immigration status were collected. Comorbidity was determined using the Johns Hopkins Adjusted Clinical Groups Case Mix System. 24 Ontario inpatient (CIHI) and outpatient (OHIP) diagnosis codes from the year prior to the index date were used to estimate the case mix with the Johns Hopkins algorithm. We adjusted for comorbidity using aggregated diagnosis groups (ADGs), which are clinically meaningful groupings of diagnoses. Diagnoses within a given ADG are similar in terms of disease severity and anticipated duration. 24 Comorbidity was categorized according to the number of ADGs (0, 1 to 8 and > 8). Neighbourhood income quintile assignment was restricted to urban areas, as they are smaller and more homogenous than rural areas, resulting in a stronger correlation between area based and individual characteristics. 23 Date of OHIP enrolment was used as a proxy for immigration. Recent immigrants were defined as those who enrolled in OHIP within five years of their diagnosis of CRC. Long-term residents were those who were enrolled for five years or longer. 25
To estimate access to colonoscopy services, we identified the health region for each member of the study cohort and assigned their region’s annual colonoscopy rate (colonoscopies per 1000 persons among those aged 56–74) for the one-year period prior to the date of diagnosis. CRC site was classified as right sided (proximal to and excluding the splenic flexure), left sided (from the splenic flexure to the rectum), or site unknown (site of CRC could not be determined or the data were missing).
Each patient was assigned a usual provider of care (UPC) using his or her CAPE registration status. For patients not registered in CAPE (approximately 26% of Ontarians), the physician with the greatest proportion of patient encounters over the two fiscal years prior to the index date was the designated UPC. Continuity of care was defined as the extent to which patients concentrated their primary care with the same healthcare provider and was estimated by dividing the number of visits to the UPC by the total number of visits with any primary care physician over the two-year period prior to the index date. The UPC score was dichotomized as high and low based on a cut-off of 0.75. 26
Physician age, sex, location of medical training, and practice group setting were considered. Multiple PEMs exist in Ontario, differing in organizational structure, level of interdisciplinary care, and physician reimbursement method. We categorized physician practice setting based on PEM. In the Family Health Group model, physician reimbursement is mainly fee-for-service. The Family Health Network and Family Health Organization models utilize blended fee-for-service and capitation reimbursement. The Family Health Team reimbursement process is similar to the Family Health Network and Family Health Organization models; however, they include an interdisciplinary team, funding for an executive director, and electronic medical records. Patients enrolled in other PEMs as well as those not registered in a PEM were categorized separately. 16
Exposure to FOBT, patient, and physician characteristics are reported for all patients diagnosed with CRC. Patients with stage IV CRC were then compared with those with all other stages. For the descriptive analyses, where appropriate, X2 test was used to compare proportions of nominal or categorical variables. The Wilcoxon Mann–Whitney test was applied to compare medians. Multivariate logistic regression modelling was used to evaluate the relationship between exposure to FOBT and the diagnosis of stage IV CRC, adjusting for patient and physician factors. Model parameters were estimated using the generalized estimating equation to account for clustering of patients within physicians. Statistical analyses were conducted using SAS Version 9.13 (SAS Institute, Cary, NC).
This study was approved by the institutional review board at Sunnybrook Health Sciences Centre, Toronto, Canada.
Results
Patient-level cohort characteristics at the time of colorectal cancer diagnosis – Overall and by stage of cancer.
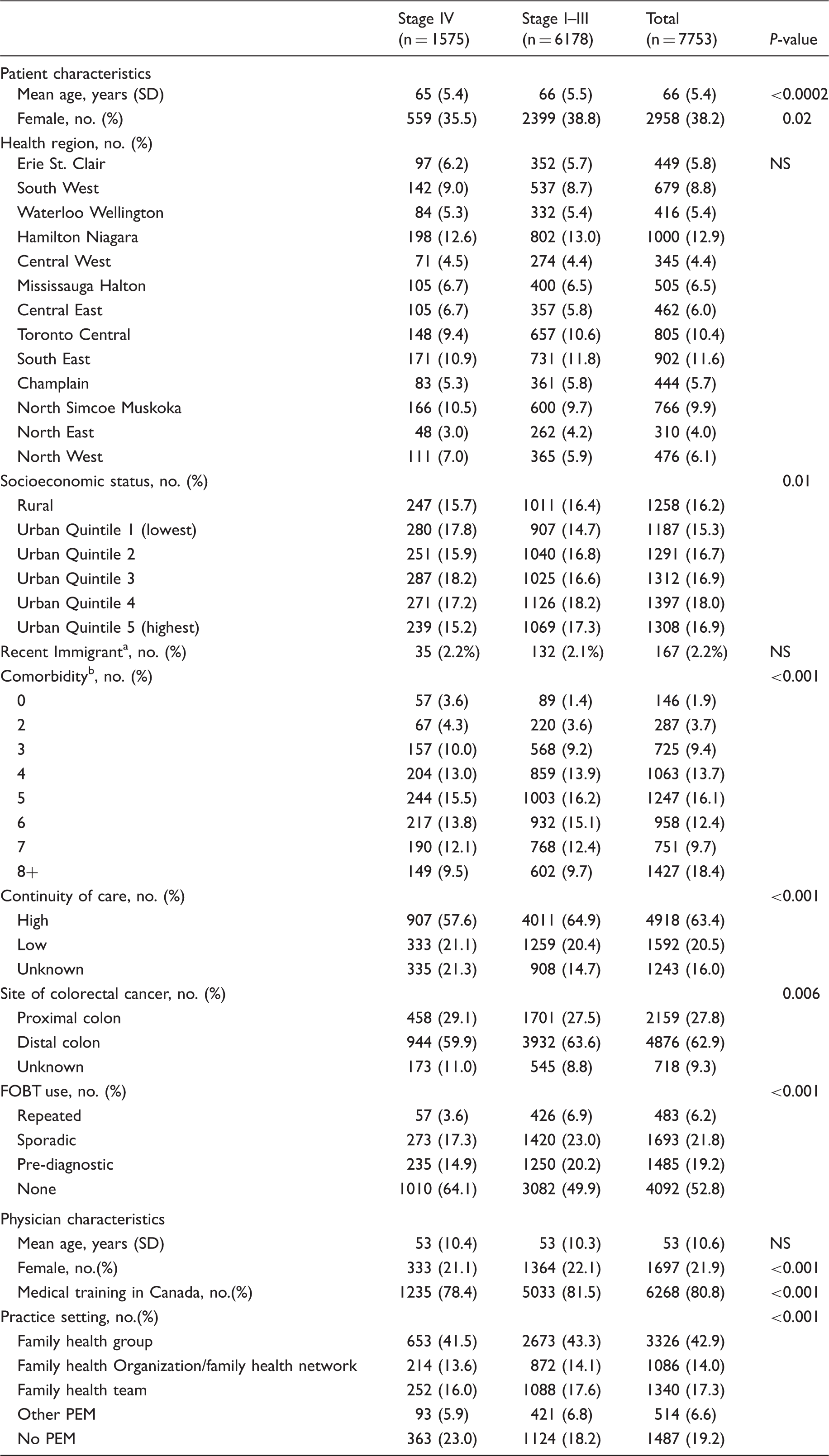
Recent immigrant defined as OHIP registration < 5 years prior to index date.
Comorbidity scored using the number of aggregated diagnosis groups applying the Johns Hopkins Case Mix System.
NS: not significant; SD: standard deviation; PEM: primary enrolment model
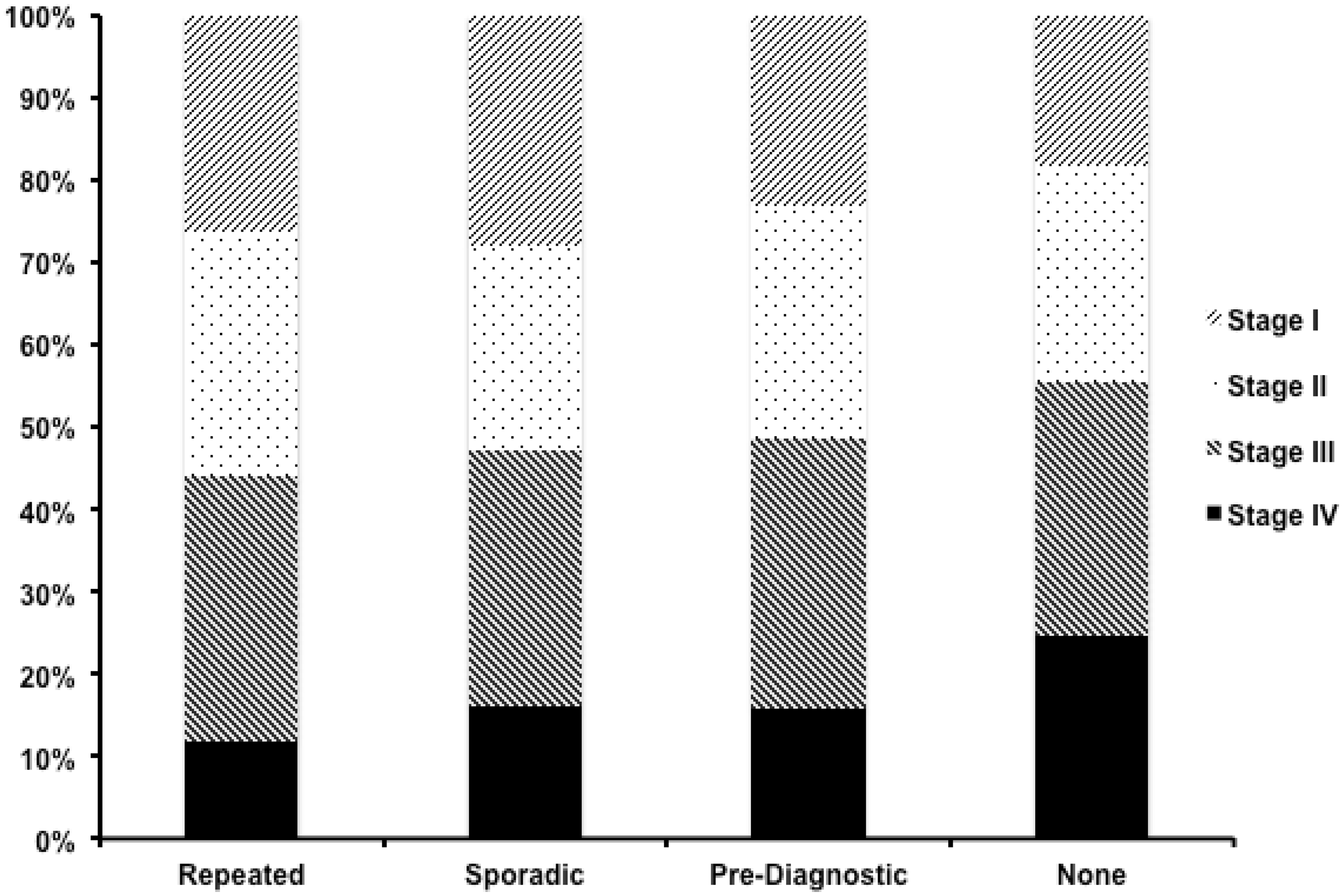
Distribution of colorectal cancer by stage and FOBT exposure group.
In univariate analyses, younger age at diagnosis, male sex, low continuity of care, proximal CRC location, and no prior exposure to FOBT were factors associated with an increased risk of stage IV CRC at diagnosis. In addition, patients not in a PEM, who had male physicians, and whose physicians were trained outside of Canada were at increased risk of stage IV CRC at diagnosis (Table 1).
Multivariate logistic regression analysis of patient and physician characteristics associated with the diagnosis of stage IV colorectal cancer.
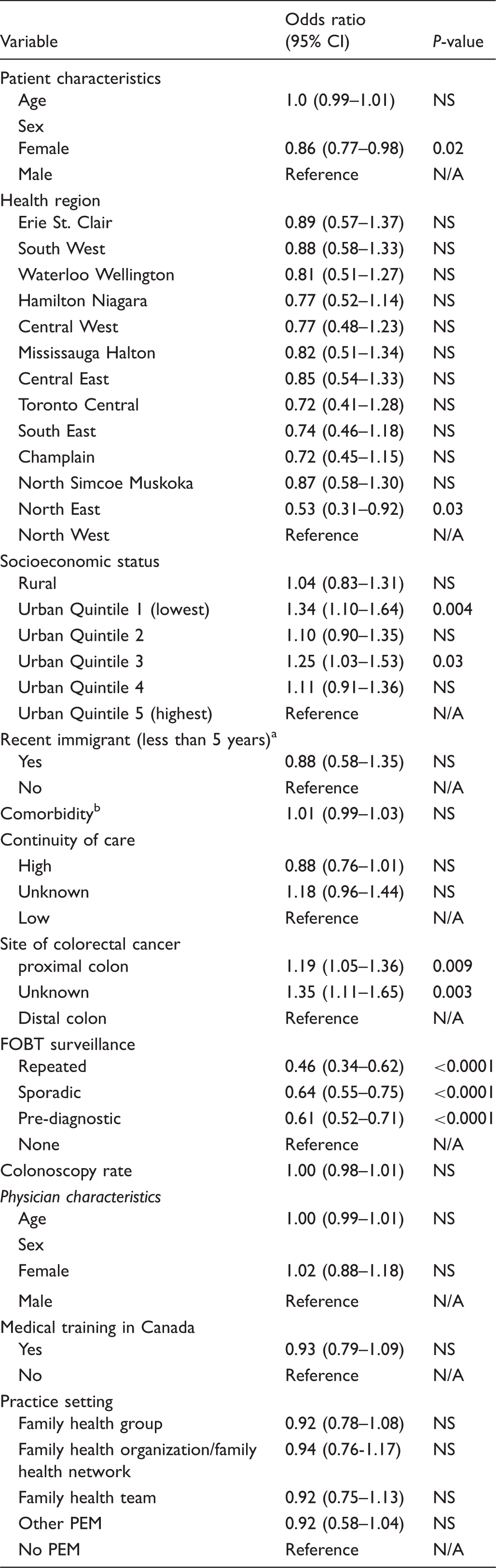
Recent immigrant defined as OHIP registration < 5 years prior to index date
Comorbidity scored using the number of aggregated diagnosis groups applying the Johns Hopkins Case Mix System.
NS: not significant; N/A: not applicable; PEM: primary enrolment model; PEM: primary care enrolment model
Discussion
This study demonstrates that repeat FOBT is associated with a reduced risk of being diagnosed with advanced stage CRC. Although any exposure to FOBT prior to diagnosis resulted in a reduction in risk, the association was stronger when repeat FOBTs were utilized prior to diagnosis. These findings are consistent with RCTs27,28 and confirm the population level benefit of repeat FOBT outside of a clinical trial setting.
The contribution of FOBT screening to reducing CRC mortality has been clearly demonstrated in RCTs. In a systematic review of RCTs by Hewitson et al., 27 FOBT screening was associated with a 16% reduction in CRC mortality. In a recent follow-up study of one of the RCTs by Shaukat et al., 28 the benefit of FOBT screening on reducing death from CRC was shown to persist after 30 years. Adherence is an important determinant of the effectiveness of CRC screening programmes, as repeated FOBT screening is associated with a further reduction in the risk of CRC mortality. In a trial of biennial FOBT screening, Jorgensen et al. 29 demonstrated a relative risk of death of 0.70 (95% CI 0.58–0.85) after one and 0.66 (95% CI 0.54–0.81) after seven rounds of screening compared with controls. This is consistent with our findings. The adjusted odds of being diagnosed with advanced (Stage IV) CRC were 54% lower among those exposed to repeated FOBT compared with those not exposed to FOBT (Figure 1).
There are several possible explanations for this observation. First, repeated FOBTs increase the test’s sensitivity (sometimes referred to as ‘programmatic sensitivity’), as prevalent cancers have a greater chance of being detected. Second, the longer follow-up may allow for de novo cancers to be detected. 9 Third, those who adhere to repeated FOBT may be more likely to have access to and seek treatment when CRC is detected. 9 Finally, if patients who undergo repeated FOBT testing are being screened in the context of an organized screening programme, other programme features, such as timely access to follow-up colonoscopy, may contribute to improved outcomes.
Although RCTs are widely considered the gold standard for evidence regarding efficacy, factors such as adherence, continuity of primary care, and access to endoscopic evaluation, may influence the effectiveness of FOBT in a real world setting. Other regions that have implemented FOBT-based CRC screening programmes also provide insight into the effectiveness of repeated FOBT screening. Using data collected prospectively from a pilot screening programme in Scotland, Steele et al.9,14 observed that the percentage of screen detected CRC at Duke’s Stage A and D decreased with sequential rounds of FOBT screening. They suggested that repeated screening helps in the detection of both new cancers and prevalent cases missed by the initial FOBT. Similar benefits were observed for faecal immunochemical test (FIT) screening in a longitudinal cohort study from Japan. 30 Sakata et al. 30 found that the risk of CRC is 63% lower on repeated vs. initial FIT screening. Greater overall survival was also observed for FIT screened subjects compared with patients presenting with clinical symptoms.
In our multivariate analyses, patients in the lowest neighbourhood income quintile were at the greatest risk of being diagnosed with stage IV CRC. This is consistent with previous studies demonstrating that low income individuals are at an increased risk of CRC 31 and tend to have lower adherence to repeat FOBT testing than higher income individuals. 7 Data from Canada suggest that targeted interventions could alleviate such disparities 32 and that social inequalities in CRC incidence have improved. 33 Our observations highlight the ongoing importance of directing screening towards low income individuals to further reduce social disparities in the CRC mortality.
We found that advanced CRC was more likely to be identified in the proximal colon. This is consistent with previous studies demonstrating that interval post-colonoscopy CRC diagnosis has been found to be more common in the proximal colon. 34 Screening-related reasons attributed to this finding include missed lesions on colonoscopy, incomplete polypectomy, and interval development of new lesions. 35 Additionally, FOBT may be less sensitive for lesions in the proximal colon; 36 however, this has not been consistently demonstrated.37,38 Colonic tumour growth rates may differ based on their location in the colon, with proximal colon cancers progressing more rapidly to advanced disease. 37 Improvements in CRC screening training, technology, and quality assurance will be essential to maximizing the detection and management of CRC lesions throughout the colon.
Our study has several limitations. First, the indication for FOBTs performed could not be definitively determined from the databases, and up to 15% of these tests may not have been performed for screening purposes. To address this, we categorized cases where a single FOBT was performed within nine months of diagnosis separately, as this group was more likely to have FOBT testing for symptoms or other non-screening purposes. Second, retrospective cohort studies are vulnerable to confounding by unmeasured factors. Although we considered age, sex, geographic regions, socioeconomic status, and co-morbidity, we were not able to adjust for factors that may be associated with CRC deaths or FOBT such as healthy lifestyle 39 , diet,39,40 and family history.39–41 Third, guaiac-based FOBT (gFOBT) kits were used during the study period, so our results may not directly apply to screening programmes that utilize FITs. However, with the improved test characteristics 42 and adherence rates associated with FIT compared with gFOBT, 43 there may be an even stronger association between CRC screening and the early detection of CRC if FIT is used. Finally, we assume that the detection of advanced stage CRC is associated with CRC mortality. Lead and length time biases may occur in observational studies; however, this is more likely to occur when survival is the primary outcome. 44 Using stage IV CRC as our primary outcome may reduce these biases, as this outcome is closely tied to mortality. 45
Conclusion
In our Ontario-based cohort, repeated use of FOBT was associated with a reduced risk of advanced CRC. Our findings indirectly support the use of organized CRC screening programmes as their design and infrastructure promotes adherence, thereby increasing the likelihood that participants will benefit from the use repeated FOBT.
Footnotes
Acknowledgements
Jill Tinmouth held a Canadian Institute of Health Research New Investigator Salary Award. Paul James was supported by a Canadian Association of Gastroenterology and Canadian Institute of Health Research Fellowship Award. The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES, the Ontario, MOHLTC or Cancer Care Ontario is intended or should be inferred. Parts of this material are based on data and information compiled and provided by Canadian Institute for Health Information (CIHI). However, the analyses, conclusions, opinions and statements expressed herein are those of the author, and not necessarily those of CIHI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Institute for Clinical Evaluative Sciences (ICES), which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC) and by Cancer Care Ontario.
Ethical approval
The research ethics board at Sunnybrook Hospital in Toronto, Ontario, Canada approved the study.
