Abstract
Objectives
To compare preoperative staging, multidisciplinary team-assessment, and treatment in patients with screening detected and non-screening detected colorectal cancer.
Methods
Data on patient and tumour characteristics, staging, multidisciplinary team-assessment and treatment in patients with screening and non-screening detected colorectal cancer from 2008 to 2012 were collected from the Stockholm–Gotland screening register and the Swedish Colorectal Cancer Registry.
Results
The screening group had a higher proportion of stage I disease (41 vs. 15%; p < 0.001), a more complete staging of primary tumour and metastases and were more frequently multidisciplinary team-assessed than the non-screening group (p < 0.001). In both groups, patients with endoscopically resected cancers were less completely staged and multidisciplinary team-assessed than patients with surgically resected cancers (p < 0.001). No statistically significant differences were observed between the screening and non-screening groups in the use of neoadjuvant treatment in rectal cancer (68 vs.76%), surgical treatment with local excision techniques in stage I rectal cancer (6 vs. 9%) or adjuvant chemotherapy in stages II and III disease (46 vs. 52%). Emergency interventions for colorectal cancer occurred in 4% of screening participants vs. 11% of non-compliers.
Conclusions
Screening detected cancer patients were staged and multidisciplinary team assessed more extensively than patients with non-screening detected cancers. Staging and multidisciplinary team assessment prior to endoscopic resection was less complete compared with surgical resection. Extensive surgical and (neo)adjuvant treatment was given in stage I disease. Participation in screening reduced the risk of emergency surgery for colorectal cancer.
Introduction
Because screening programmes can identify patients with early stage disease, there is a risk that the reduction in mortality achieved by screening could come at the cost of over-treatment for some patients. It is important to ensure that patients with screening detected colorectal cancer (CRC) are neither over- nor under-treated. Thorough preoperative clinical staging (cTNM) should correctly identify early stage cancer (stage I), and guide selection and sequencing of treatment. Therapeutic alternatives should be evaluated in a multidisciplinary team (MDT) setting, to optimize treatment for the individual patient. For early CRC, therapeutic alternatives to major abdominal resection surgery include endoscopic resections techniques, such as endoscopic mucosal resection and endoscopic submucosal resection, and local excision techniques, including transanal endoscopic microsurgery (TEMS) with or without preoperative (chemo) radiotherapy.1,2 A non-surgical strategy with chemo radiotherapy alone for distal rectal cancer has, in selected cases, given a complete response, leaving no residual tumour. 3 For early rectal cancer patients scheduled for surgical treatment, the use of neoadjuvant therapy (i.e. radiotherapy) may not balance the benefit of reduced local recurrence rates and the negative side effects of increased morbidity and impaired function. This study aims to evaluate the completeness of preoperative staging and MDT-assessment, and the subsequent treatment of patients with population-based screening detected CRC compared with patients with non-screening detected CRC.
Methods
In the Stockholm/Gotland region (2.3 million inhabitants), population-based CRC screening began in 2008, progressively adding two birth cohorts every year until all of the target population had been included. Residents aged 60–69 were invited to biennial screening with guaiac-based faecal occult blood testing (FOBT) (Hemoccult®). Invitations were initiated centrally, without primary health care physician involvement. A test-kit with instructions was posted, and a reminder sent after 8 weeks if no results were registered. Patients with positive tests (at least one out of three cards) were immediately referred for colonoscopy and booked for examination within 2 weeks. CRC detection was followed by referral to one of the region’s nine surgical colorectal units.
Data were collected from the Stockholm–Gotland screening register and the Swedish Colorectal Cancer Registry. All data on the screening process, colonoscopy procedure and findings, including the histopathological diagnosis, were prospectively collected in the Stockholm–Gotland screening register (previously described 4 ). The Swedish Colorectal Cancer Registry is a national quality register collecting data on all patients with adenocarcinomas in the colon and rectum. The registry started in 1995 including patients with rectal cancer, and in 2007 patients with colon cancer were added. Data are prospectively and continuously collected to ensure quality control and adherence to national guidelines, and are also used for research and clinical studies. Detailed information on preoperative clinical staging (cTNM), (neo) adjuvant treatment, the surgical procedure and findings, pathological staging (pTNM) according to the International Union Against Cancer/American Joint Committee on Cancer tumour node metastasis classification, complications according to the Clavien-Dindo classification and follow-up until 5 years after diagnosis are recorded.5,6 The registry has a 98% coverage of all cases of CRC in Sweden,7,8 collected data on rectal cancer has demonstrated a high validity, 9 and validation of the colon cancer part of the registry is ongoing. 10
Data on all individuals in the screening programme with a positive FOBT from 1 January 2008 to 31 December 2012 were retrieved. On all patients with a screening detected and histopathologically verified CRC, data regarding patient and tumour characteristics, preoperative work-up and treatment were further analysed.
Corresponding data were retrieved from the Swedish Colorectal Cancer Registry on all patients aged 60–69 with a histopathologically verified CRC diagnosed from 1 January 2008 to 31 December 2012. The group of patients with screening detected CRC was then excluded from this cohort to generate a control group of all patients with non-screening detected CRC in Sweden, matched by age to those with screening detected CRC.
Chi-square test and Fisher’s exact test were used as appropriate to determine significant differences between proportions. The p-values < 0.05 were considered statistically significant. Stata 12.0 (StataCorp, 4905 Lakeway Drive, Collage Station, TX 77845, USA) was used for the statistical analysis. The study was approved by the Regional Ethics Committee in Stockholm.
Results
During the study period, 205,842 individuals received at least one screening test kit. Of the 131,671 (64%) individuals who participated, 4618 had one positive (guaiac based) FOBT, and of these, 4080 (88%) complied with the assessment colonoscopy. The number of histopathologically confirmed screening detected CRCs was 249, yielding a cancer detection rate in screened individuals of 0.2%.
Pre-therapeutic assessment
Staging and multidisciplinary team (MDT)-assessment of patients with screening and non-screening detected colorectal cancer planned for elective surgery.
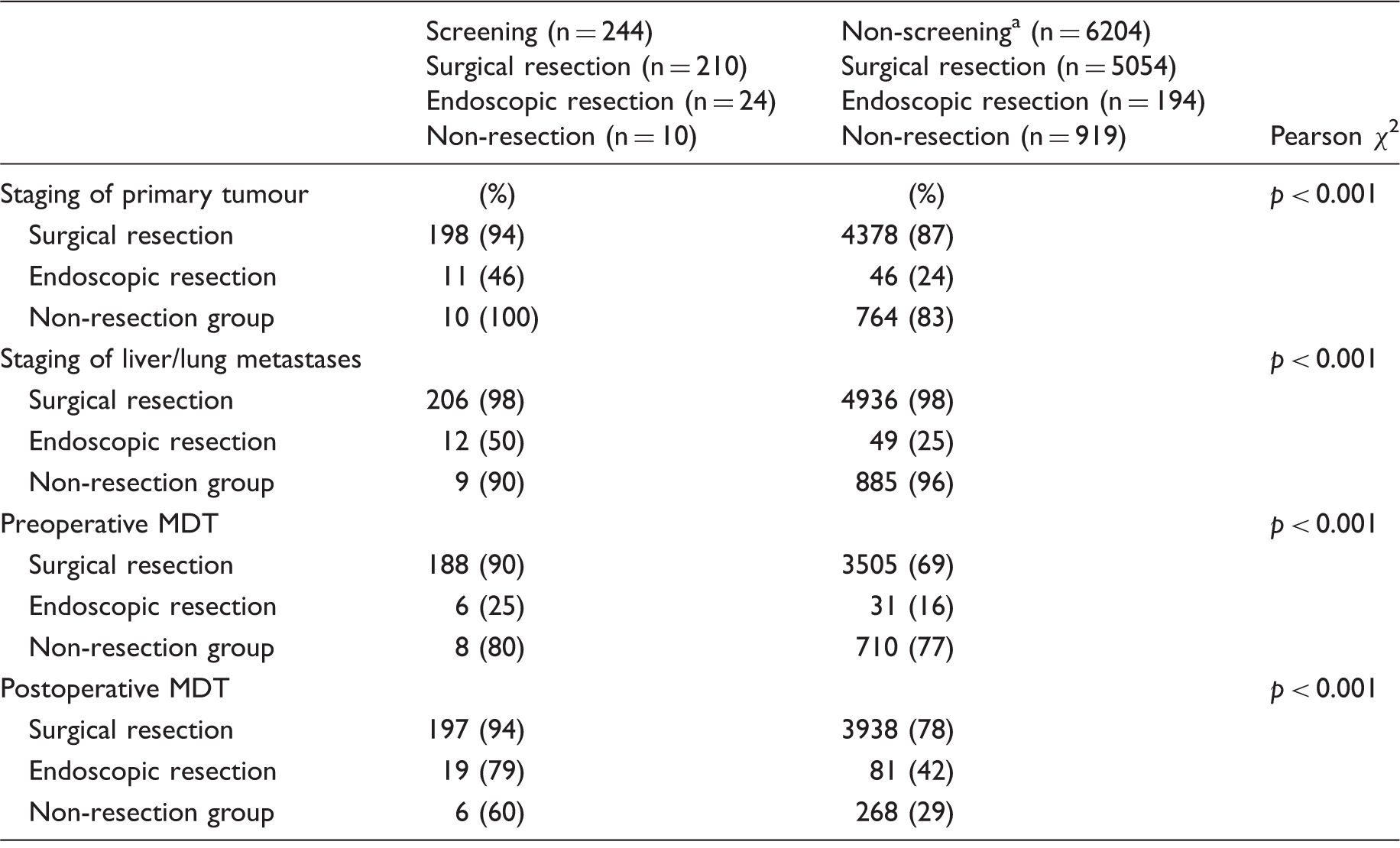
Thirty-seven cases not included in the analysis: missing (n = 12), unspecified operation (n = 16) and appendectomy (n = 9).
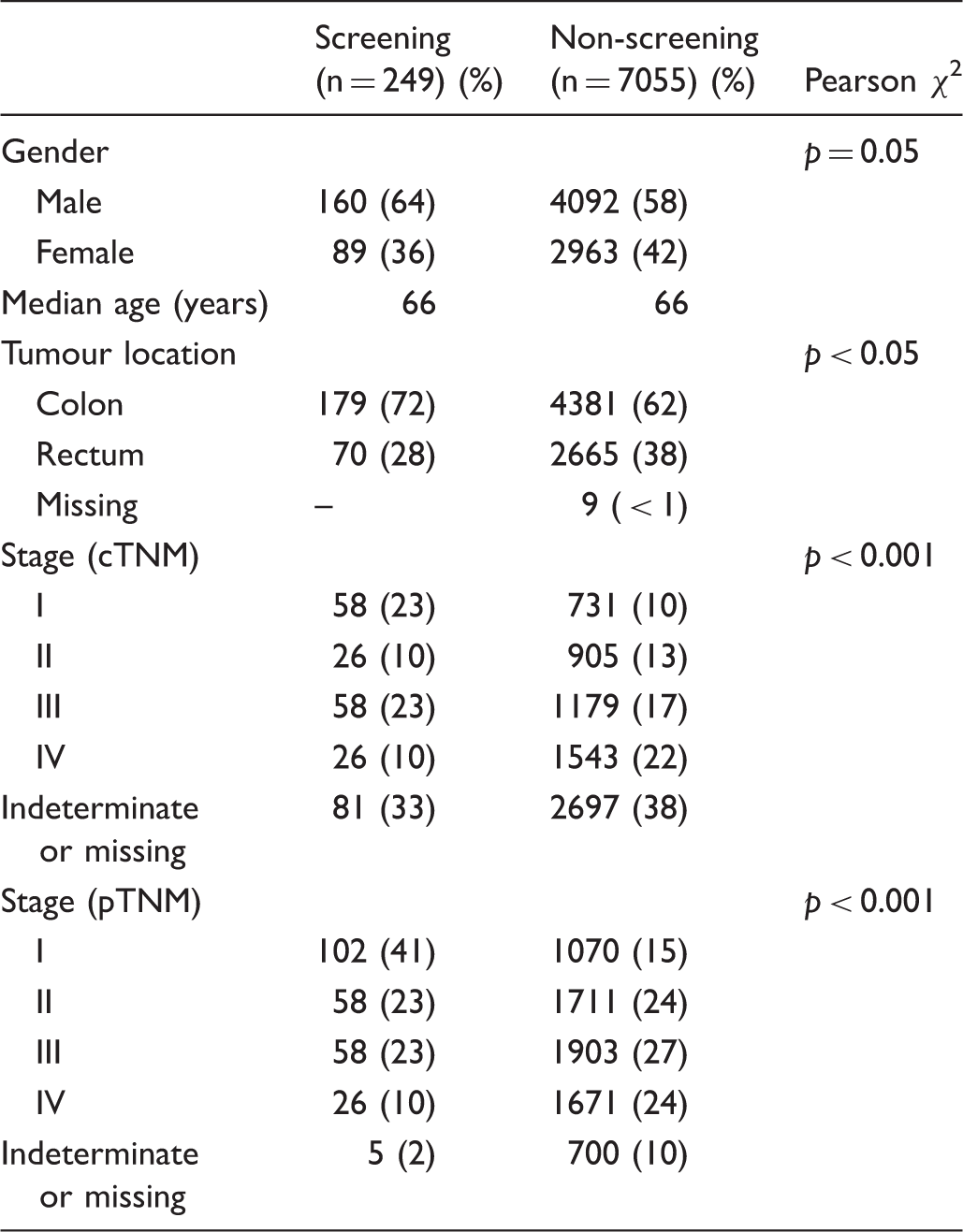
Baseline patient and tumour characteristics of all patients with screening detected vs. non-screening detected colorectal cancer diagnosed 2008–2012.
Treatment
Among patients with screening detected CRC, 244 (98%) underwent elective interventions (Figure 1). The majority, 210 patients (86%), underwent surgical resection; 24 patients (10%) underwent endoscopic resection. In 10 patients (4%), the decision following an MDT, or at laparotomy, was not to attempt of resection. Of the patients undergoing surgical resection, 209 (99.5%) underwent abdominal resection. The corresponding numbers among patients with non-screening detected cancers were 6204 (88%) underwent elective treatment, of whom 5054 (81%) underwent surgical resection; 4992 (99%) of these were abdominal resections. Endoscopic resection was performed on 194 (3%) patients and non-resectional therapy on 919 (15%) patients. The proportion of emergency interventions in CRC cases among those participating in screening was 4%, among individuals with a positive FOBT attending screening it was 2%, and among those with a negative FOBT attending screening it was 6%. Among those invited to, but not attending screening the proportion of emergency cases was 11%, and in those not invited to screening, 12% (p < 0.001).
Overview of interventions in patients with screening and non-screening detected colorectal cancer.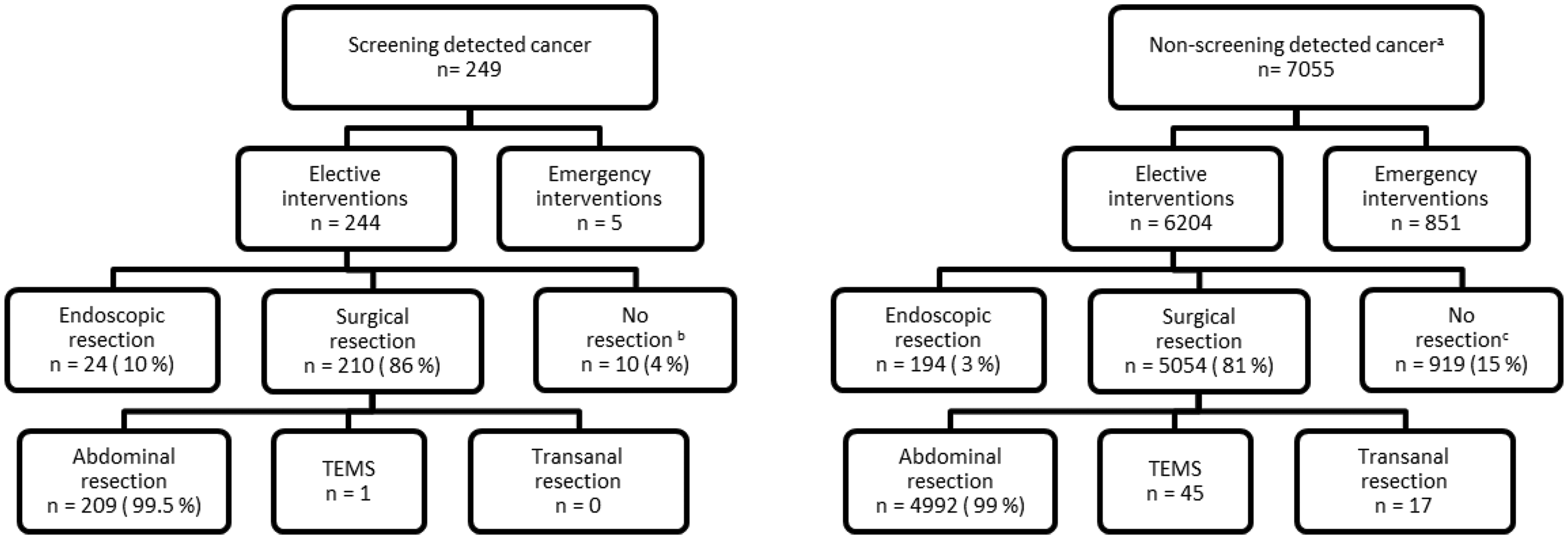
Rectal cancer
In the screening group, 16 patients (23%) with electively treated rectal cancer had (cTNM) stage I disease, among whom one patient underwent endoscopic resection, and none was resected with TEMS or transanal excision. In the non-screening group, 369 patients with electively treated rectal cancer had (cTNM) stage I disease (14%), of whom 23 (6%) underwent TEMS or transanal excision. Endoscopic resection of the tumour was performed in 9 (2%) patients. There was no statistically significant difference between the groups in the use of local excision techniques, i.e. TEMS, transanal excision or endoscopic resection (p = 1.0).
(Neo)adjuvant therapy
Among patients with rectal cancer selected for abdominal surgery, the proportions assigned for neo adjuvant (chemo) radiotherapy were 68% and 76% in the screening and non-screening groups, respectively (p = 0.14) (Figure 2). In stage I (cTNM), 40% of patients in both groups were assigned for neo-adjuvant treatment. Adjuvant chemotherapy after elective abdominal resection surgery in patients with stages II and III (pTNM) CRC was given to 46% of patients in the screening group and 52% of patients in the non-screening group (p = 0.15).
Neoadjuvant (chemo) radiotherapy in relation to stages I–IV(cTNM) in patients with screening detected (S) and non-screening detected (NS) rectal cancer prior to elective abdominal resection surgery.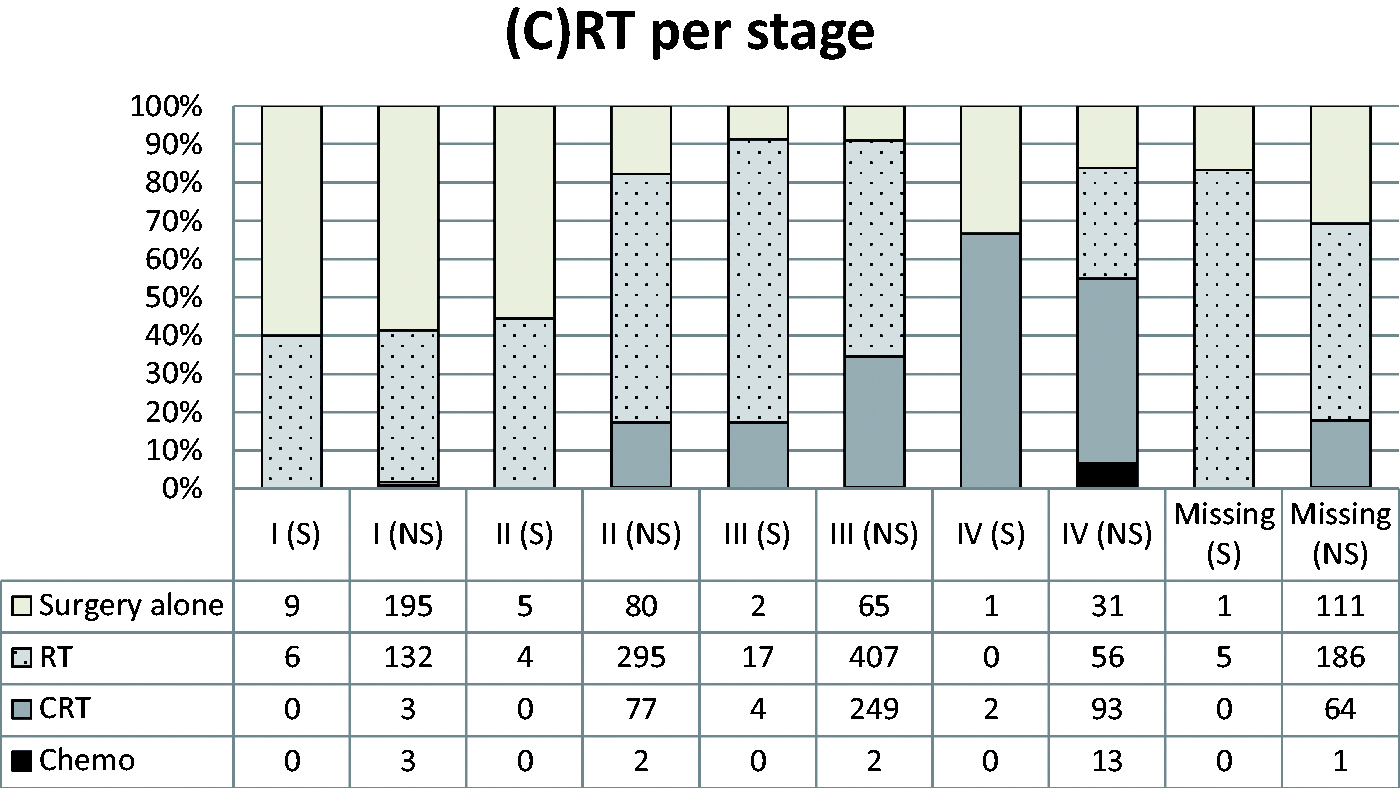
Discussion
Patients in our study with screening detected CRC had a more complete preoperative staging of the primary tumour and distant metastases, had more early stage tumours, and underwent MDT assessment to a higher extent than the patients with non-screening detected CRC. No difference was seen between the groups in the use of abdominal resection surgery and (neo)adjuvant therapy. Screening participants were at low risk of emergency interventions.
MDT assessments are important for staging and planning of surgery, and also for the timing, sequencing and strategy of any (neo)adjuvant treatment. Studies have demonstrated that MDT conference assessment improves staging and survival and reduces involved resection margin.11–13 The Swedish board of health and Welfare strongly advocates the use of MDT conferences for CRC patients. 14 The high proportion of patients with complete staging and MDT-assessments in the screening group may reflect that these patients come from the Stockholm region, which has a higher completeness of staging and preoperative MDT-assessment in colon cancer patients than other Swedish regions. 7 Restricting the comparison to the Stockholm region may eliminate these differences, but higher proportions of complete staging and MDT assessments are seen over time in all Swedish regions, thereby reducing regional differences.7,8 In rectal cancer, the regional differences in staging and MDT-assessment are very small. 8
The differences in staging and MDT assessment in patients treated with endoscopical resection compared with surgery were extensive. Although not desirable, the low rate of preoperative staging and MDT assessment is logical in a case of a polypectomy resulting in an unexpected cancer diagnosis. However, an endoscopic resection of a cancer-suspected lesion without a complete preoperative staging and MDT-assessment may be unsatisfactory management. With the introduction of new techniques (e.g. endoscopic mucosal and submucosal resection) as treatments, both for adenomas and as an option for early cancers, preoperative radiological staging and the MDT-assessment must be complete to ensure the correct selection of patients and tumours most suited to undergo resection.
The benefits of local excision for rectal cancer are recognized, in terms of low mortality, low morbidity and good functional results.15,16 Limited use may be caused by concerns over local recurrence and impaired survival, although local recurrence rates following TEMS in small T1sm1 tumours (i.e. tumours extending to the most superficial third of the submucosal layer of the bowel wall) without adverse features are low, 17 and 5-year overall survival is comparable with that using conventional radical surgery.18,19 In this study, none of the patients with screening detected electively treated rectal cancer in (cTNM) stage I disease was treated with TEMS or transanal surgery, but in the non-screening group, the corresponding number was 23 out of 369 (6%). By comparison, 2010 data from the US National Cancer Database show that over 50% of patients with stage I rectal cancer were treated with local excision. 20 In attempting to avoid overtreatment of early stage cancer, this study suggests that there is potential to extend the use of local excision techniques, at least in rectal cancer, taking into consideration individual risk profile and patient preference.
In the present study, preoperative (chemo) radiotherapy was used extensively in rectal cancer patients from both the screening and non-screening detected groups; there was no statistically significant difference between the groups. Forty percent of patients with both screening detected and non-screening detected stage I rectal cancer underwent preoperative irradiation, a result in concordance with recent data on the extensive and increasing use of preoperative (chemo) radiotherapy in Stockholm. 21 The side effects of irradiation, including impaired genito-urinary and bowel functions, increased risk of anastomotic leakage, and the risk of secondary pelvic insufficiency fractures, are well documented.22–24 In this study, it was not possible to observe any possible benefit of reducing the use of pre-operative irradiation in patients with screening-detected early-stage rectal cancer.
Many studies have demonstrated that patients undergoing emergency surgery for CRC have more advanced tumours and a higher rate of obstruction and perforation at presentation. Previous studies have also shown that emergency surgery is associated with higher perioperative mortality, longer admissions, more readmissions, impaired disease-free survival and higher rates of loco regional recurrence.25–27 In this study, there was a significant reduction of emergency interventions among individuals participating in screening (4% emergency interventions) compared with those not participating (11% emergency interventions), and those not invited to screening (12% emergency interventions). Similar observations have been made in England, where the introduction of CRC screening was followed by a reduction in the emergency surgery rate, falling to 12% in the screening age group compared with 16% overall. 28 In Scotland, a reduction of emergency admissions from 20% to 11% was observed in screening participants compared with controls. 29 Also in Scotland, a reduction from 20% to 13% of patients with emergency presentation of CRC prior to surgery was observed over time after the introduction of CRC screening. 30
The early detection benefits of screening will also have an impact on short- and long-term outcome by the reduction of emergency interventions.
Conclusion
Tumour staging and MDT-assessment were more complete in patients with screening detected compared with non-screening detected CRC. However, within both these groups, patients undergoing endoscopical resection were not staged and MDT was not assessed to the same extent as those treated with surgery. Overall, patients with early stage disease received extensive treatment, both in terms of surgery and (neo)adjuvant treatment. Screening participation reduced the risk of emergency surgery.
Footnotes
Acknowledgements
We thank Robert Johansson (MSc - Regional Cancer Centre North, Department of Radiation Science, Oncology, Umeå University, Sweden), Sini Kilpeläinen (Statistician, Regional Cancer Centre Stockholm–Gotland, Sweden) and Tongplaew Singnomklao (Data manager, Swedish Colorectal Cancer Registry, Regional Cancer Centre Stockholm–Gotland, Sweden) for their contributions in data extraction.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was provided through the Regional Agreement on Medical Training and Clinical Research (ALF) between the Stockholm Community Council and Karolinska Institutet, and also by the Swedish Cancer Society and the Stockholm Cancer Society.
