Abstract
Objective
Prior work on the disparities among women in breast cancer screening adherence has been methodologically limited. This longitudinal study determines and examines the factors associated with becoming adherent.
Methods
In a cohort of Canadian women aged 50–74, a three-state transitional model was used to examine adherence to screening for breast cancer. The proportion of time spent being non-adherent with screening was calculated for each woman during her observation window. Using age as the time scale, a relative rate multivariable regression was implemented under the three-state transitional model, to examine the association between covariates (all time-varying) and the rate of becoming adherent.
Results
The cohort consisted of 2,537,960 women with a median follow-up of 8.46 years. Nearly 31% of women were continually up-to-date with breast screening. Once a woman was non-adherent, the rate of becoming adherent was higher among longer term residents (relative rate = 1.289, 95% confidence interval 1.275–1.302), those from wealthier neighbourhoods, and those who had an identifiable primary care provider who was female or had graduated in Canada.
Conclusion
Individual and physician-level characteristics play an important role in a woman’s adherence to screening. This work improves the quality of evidence regarding disparities among women in adherence to breast cancer screening and provides a novel methodological foundation to investigate adherence for other types of screening, including cervix and colorectal cancer screening.
Keywords
Introduction
Breast cancer screening is valuable for detecting early stage cancer and reducing breast cancer mortality.1–3 Canadian cancer screening guidelines recommend mammography screening for breast cancer (free of charge for all Canadian residents) every two to three years for average-risk women aged 50–74.4,5 In Ontario, mammograms can be obtained either through physician referral or through the Ontario Breast Screening Program (OBSP), for which referral is not required and women are invited by mail to book their next mammogram when it is due. 4 Despite free universal healthcare, the OBSP, and the guidelines and the well-known benefits of breast cancer screening, there remains a large proportion of screen-eligible but unscreened women.4,6,7
Underscreened populations in Canada include new immigrants, women living in poverty, and women without a physician.8–12 Although physician referrals are not required to obtain a mammogram, primary care providers (PCPs) often order mammograms, and a physician’s advice or recommendation to have mammograms enhances their receipt. 13 Non-adherence to regular screening mammography increases the risk of a delay in breast cancer diagnosis, which may affect treatment options and outcomes.14–16 Conservative approaches to treatment and increased benefits of surgical, medical, and radiation treatments are more likely to be possible for regular screeners with early stage cancers compared with the more extensive treatments often required for advanced cancers among non-regular screeners.14–16
The important association between breast screening adherence and the risk of delay in breast cancer diagnosis has been previously examined in a variety of ways. Studies in the UK have investigated ethnic, cultural, and socio-economic factors related to breast screening coverage, using cross-sectional analyses, or area-level analyses conducted during a particular year.17,18 Some studies calculated the proportion of age-eligible women who were up-to-date with breast screening at a specific point in time,8,19,20 and others categorized women as adherent or non-adherent according to their prior screening history. 6 In reality, however, a wide range of screening patterns exists among non-adherent women. As some women may be non-adherent for only a small portion of their age-eligible years, while others may be so for a significant period, categorizing both groups as non-adherent may be an oversimplification.
We here propose a multistate modeling approach for examining adherence to breast cancer screening. Multistate models are particularly useful for understanding the complexities of event processes where individuals can transition between well-defined distinct states during observation. The states represent the condition of the individual at any given time (for example, either adherent or non-adherent), and the transition times are the points at which changes in condition occur (i.e. the times at which events occur). Multistate models are sufficiently flexible to accommodate varying lengths of follow-up, right-censoring, delayed state entry (which can occur when individuals are observed to go in and out of being at risk of a particular transition), prior history of the event process (for example, number of previous states visited, or how long a person spent in a previous state), instantaneous changes in the event status over time, instantaneous changes in the covariate measures over time, and the occurrence of events, such as death, that alter future risk of other events.
We believe this study is the first individual-level analysis conducted on a province-wide cohort to longitudinally examine a woman’s pattern of being adherent with breast cancer screening over time, using a multistate model. As women can go in and out of being adherent numerous times during their age-eligible years, this rigorous longitudinal approach is well suited for examining adherence to screening. Under the multistate model framework, we identify populations of women who have lower rates of becoming adherent. Our work will improve the quality of evidence regarding disparities among women in being adherent with breast cancer screening over time, and our proposed methodology will provide a foundation for understanding patterns of being adherent with various types of cancer screening, including cervix and colorectal cancer screening.
Methods
Study population and observation window
This retrospective cohort study used linked population-based databases of women in Ontario, Canada’s most populous province. As healthcare is universal and free of charge in Canada, the cohort consisted of all women who belonged to the Ontario Health Insurance Plan (OHIP). To be age-eligible for breast cancer screening, each woman was aged 50–74 at some point during the accrual period, from 1 January 2001 to 31 December 2012. Information on health insurance eligibility and age was obtained from the OHIP database and the Registered Persons Database (RPDB). Women were excluded if they had a previous diagnosis of cancer (data obtained from the Ontario Cancer Registry), previous bilateral mastectomy (data obtained from OHIP and Canadian Institute for Health Information database, CIHI), or previous breast implant (data obtained from OHIP and CIHI). Follow-up was terminated if any of these events occurred during the observation window, or otherwise on the day before the 75th birthday, the date of OHIP ineligibility, the date of death (obtained from the RPDB), or on the study end date (31 December 2013), whichever occurred first.
Variable definitions
Outcome definition
Screening status could transition between non-adherent and adherent several times during a woman’s observation window. The outcome for each woman was defined as the times of transitions between these screening status states. At any given time during a woman’s observation window, she was considered adherent if she had a screening mammogram within the last 36 months, and non-adherent if she had not. The same criteria determined initial state at study entry. Screening mammogram dates were retrieved from OHIP codes and the OBSP data. 21 The 36-month interval was based on current Canadian cancer screening guidelines, and although the recommendation has changed over the past decade, this definition was more conservative for deriving a woman’s adherence status and has also been used in prior work.5,22
Individual-level characteristics
For each woman, at the start of her observation window, we determined her age, socio-economic status, immigration status, and level of healthcare resource utilization. Socio-economic status was captured by linking postal and residential codes in RPDB to census data and then assigning each study individual to one of five quintiles of median household income (urban), or to a rural categorization.22,23 Immigration status, retrieved from the Citizenship and Immigration Canada database, was measured as a 4-level categorical variable; women who immigrated within the last five years were recent immigrants, within the last 5–10 years were intermediate immigrants, over 10 years were long-term immigrants, and those not found in the CIC database were considered long-term residents. 24 Healthcare resource utilization was measured using the Johns Hopkins Adjusted Clinical Groups System25,26 in which counts of Aggregated Diagnosis Groups (ranging from 0 (no diagnosis) to 32 distinct diagnosis groups) over a two-year look-back window indicated the level of comorbidity. The Johns Hopkins system is one of the best performers for predicting health service utilization in primary care settings.22,27 For every woman, the measurement of each individual-level characteristic was updated at the start of each year (using the look-back windows above) to capture any changes.
Physician-level characteristics
We determined whether each woman, at the start of her observation window, had an identifiable PCP in the previous two years. Using billing data from OHIP, a physician was defined as a woman’s PCP if they had claimed the maximum dollar value (from a set of 18 comprehensive primary care billing codes) during the previous two years. This set of billing codes and look-back window have been used in previous studies, and we maintained this definition for consistency.22,28 If there were no relevant claims in the previous two years, the woman was considered not to have an identifiable PCP. The PCP’s age, sex, and country of graduation (Canada, United States, or other) were retrieved. As a woman’s PCP may change over time, we also updated all physician-level characteristics at the start of each year (using the look-back windows above).
Statistical analyses
We implemented a time non-homogeneous multistate model under Markov assumptions
29
to study the patterns of being adherent with breast cancer screening over time. The technique used to assess whether the Markov assumption was reasonable is discussed below. At any given time, a woman’s status was classified as one of three states: State 1 – Non-adherent with breast cancer screening, State 2 – Adherent with breast cancer screening, or State 3 – Absorbing event. The absorbing event included the occurrence of breast cancer, bilateral mastectomy, breast implant, or death (whichever occurred first). The rationale is that the occurrence of any of these events changes the future meaning and risk of screening adherence and that considering these events as part of an absorbing state is more appropriate than censoring, particularly when examining transition risks over time. A woman could make transitions back and forth between being non-adherent and being adherent, until the absorbing event occurred. Our three-state model is illustrated in Figure 1, where the arrows indicate the directions in which instantaneous transitions are possible. Multistate models are governed by transition intensity functions, which represent the instantaneous incidence rate of moving from one state to another at a specific point in time.29–31 For context, a transition intensity function is equivalent to a hazard function in a survival model (a survival model is simply a unidirectional two-state model).
Three-state model for examining adherence to breast cancer screening.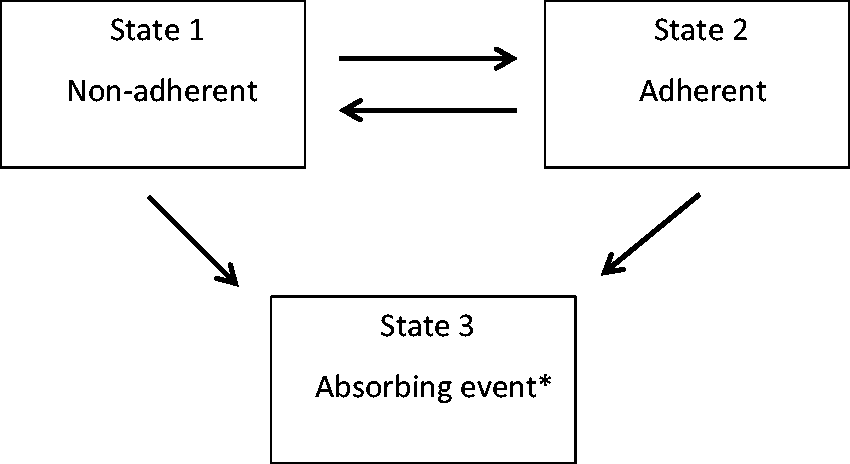
The multistate modeling framework was used to conduct three analyses. First, we conducted descriptive analyses by computing each woman’s total person-years spent in the non-adherent and adherent states, and the proportion of time spent being non-adherent throughout her observation period. Median and interquartile ranges of these measures were provided for the cohort characteristics. Second, we implemented a relative rate (RR) regression model within our multistate framework, to examine which individual and physician-level factors were associated with a higher transition intensity or rate of becoming adherent (from the non-adherent state). This was a semi-parametric approach that used a partial likelihood function to estimate the association parameters. 30 All covariates in the regression model were included as time-varying measures. The data were setup based on a counting process structure. Age was used as the time scale in the analysis and was therefore not included in the regression model as a covariate. Third, we estimated the cumulative transition intensity function from the non-adherent to the adherent state. This was done to plot the mean cumulative number of visits to the adherent state (from the non-adherent state) over time among women with and without an identifiable PCP in the prior two years (where information was updated on a yearly basis, as described earlier). Women could move in and out of contributing to both curves, depending on how their PCP exposure status changed over time, but at any specific time could only contribute to one of the curves. The corresponding 95% confidence interval (CI) for each curve was also plotted.
The Markov assumption was assessed by incorporating aspects of the event process history as a covariate in our regression models.30,32 Specifically, the prior proportion of time spent being non-adherent (total prior time spent in the non-adherent state since study entry/total time since study entry) was included as a time-varying covariate in our model, updated yearly for each woman. This measure was not a significant factor in multivariable regression analyses, a reasonable explanation being that the current state of adherence for an individual along with information on their individual and physician-level characteristics were the driving factors in understanding the transition rates between adherent and non-adherent states. We therefore proceeded with the Markov assumption. The study was approved by the Research Ethics Board at Sunnybrook Hospital. All analyses were conducted using SAS version 9.3 and R statistical software version 3.2.0 (R Development Core Team, Vienna, Austria). 33
Results
Distributions of cohort characteristics (N = 2,537,960).
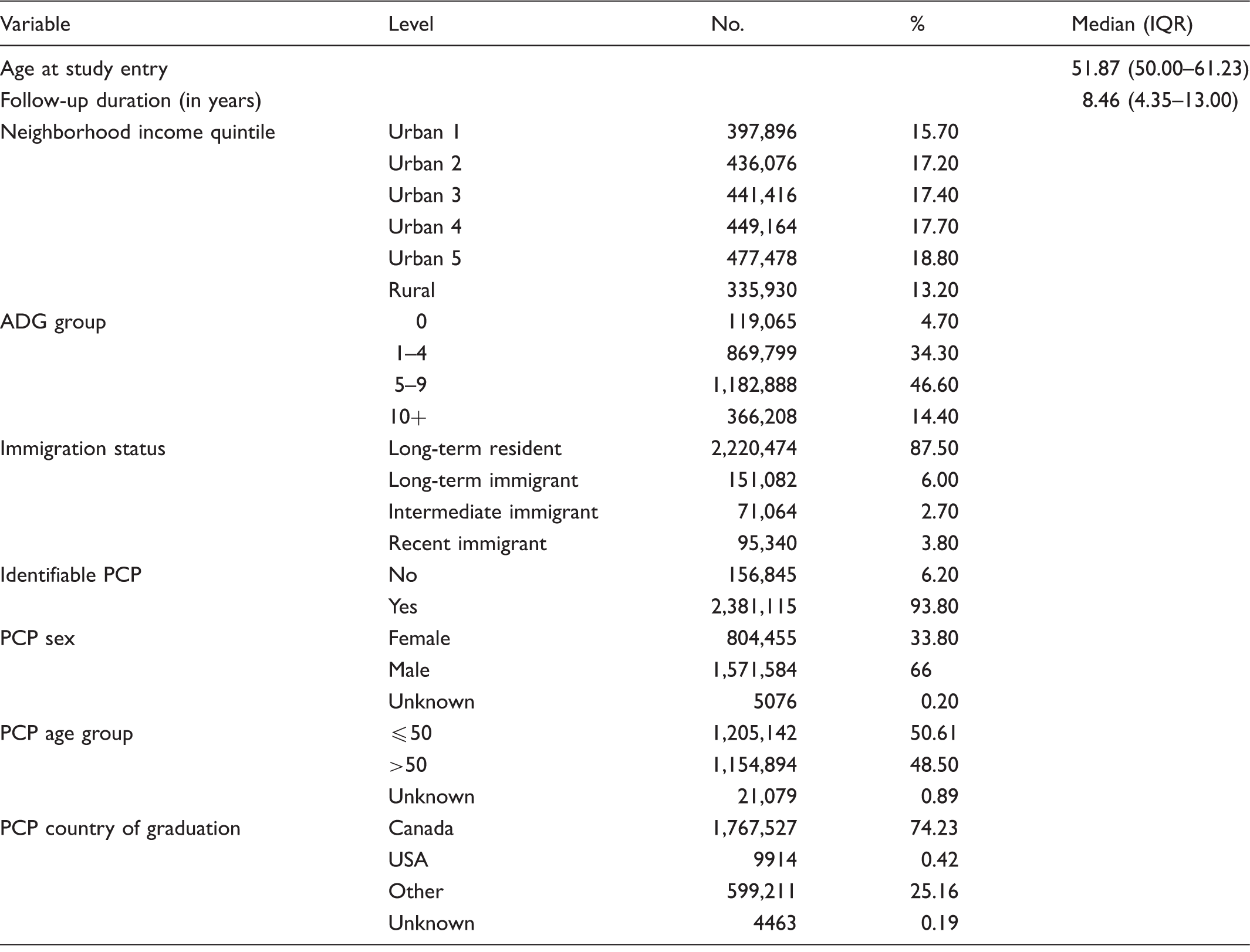
Note: Neighborhood income quintile ‘Urban 5’ implies the richest; ADG group ‘0’ implies the least amount of healthcare resource utilization.
Distributions of PCP sex, PCP age group, and PCP country of graduation are only among women with identifiable PCP = yes.
PCP: primary care provider.
Total person-years in the non-adherent and adherent states, and the percentage of time spent non-adherent, by cohort characteristics.
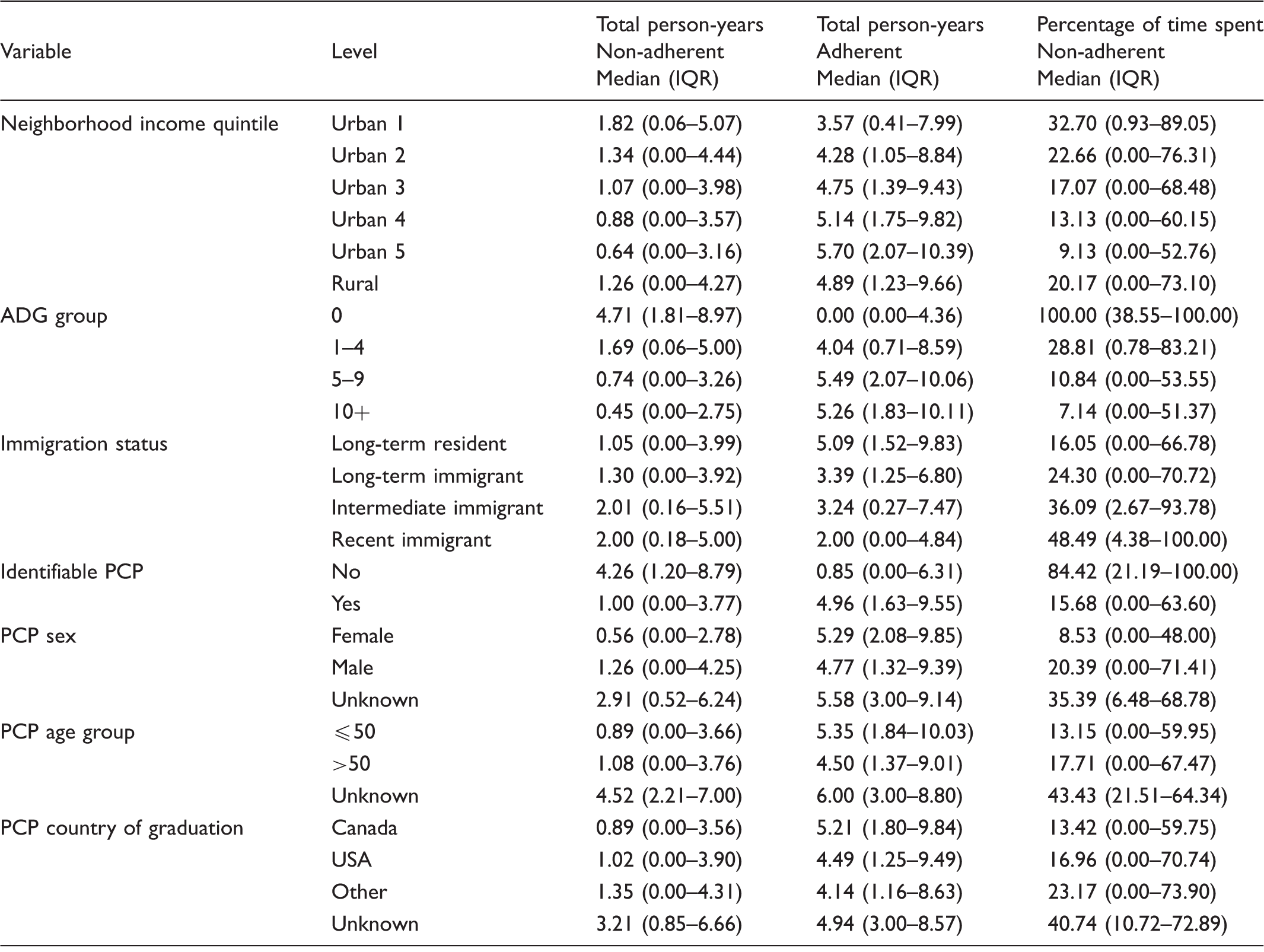
Note: Neighborhood income quintile ‘Urban 5’ implies the richest; ADG group ‘0’ implies the least amount of healthcare resource utilization.
Distributions of PCP sex, PCP age group, and PCP country of graduation are only among women with Identifiable PCP = yes.
PCP: primary care provider.
Results from the relative rate regression model for the transition from the non-adherent state to the adherent state.
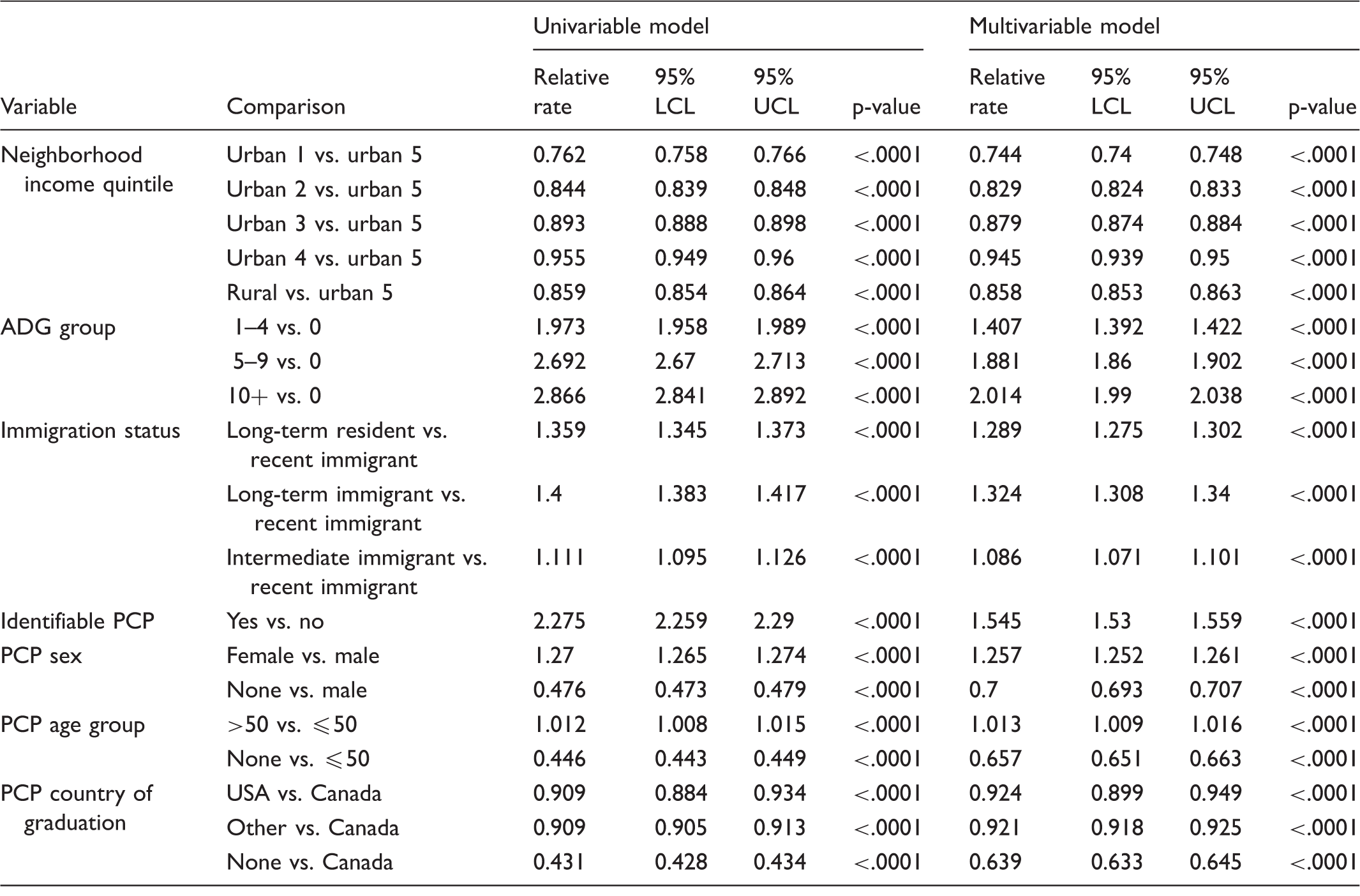
Note: Neighborhood income quintile ‘urban 5’ implies the richest; ADG group ‘0’ implies the least amount of healthcare resource utilization.
PCP: primary care provider.
Figure 2 provides the estimated mean cumulative number of visits to the adherent state (from the non-adherent state) over time among women with and without an identifiable PCP in the prior two years. A woman could transition back and forth between these curves, depending on her PCP status at a given time. The figure illustrates that, at any given time, the expected cumulative number of transitions to the adherent from the non-adherent state is higher among women who had an identifiable PCP than those who had not; this difference was significant, as shown by the non-overlapping 95% CIs and remained consistent over time.
Estimated mean cumulative number of visits/transitions to the adherent state (from the non-adherent state) over time, with 95% confidence intervals, among women with and without an identifiable PCP.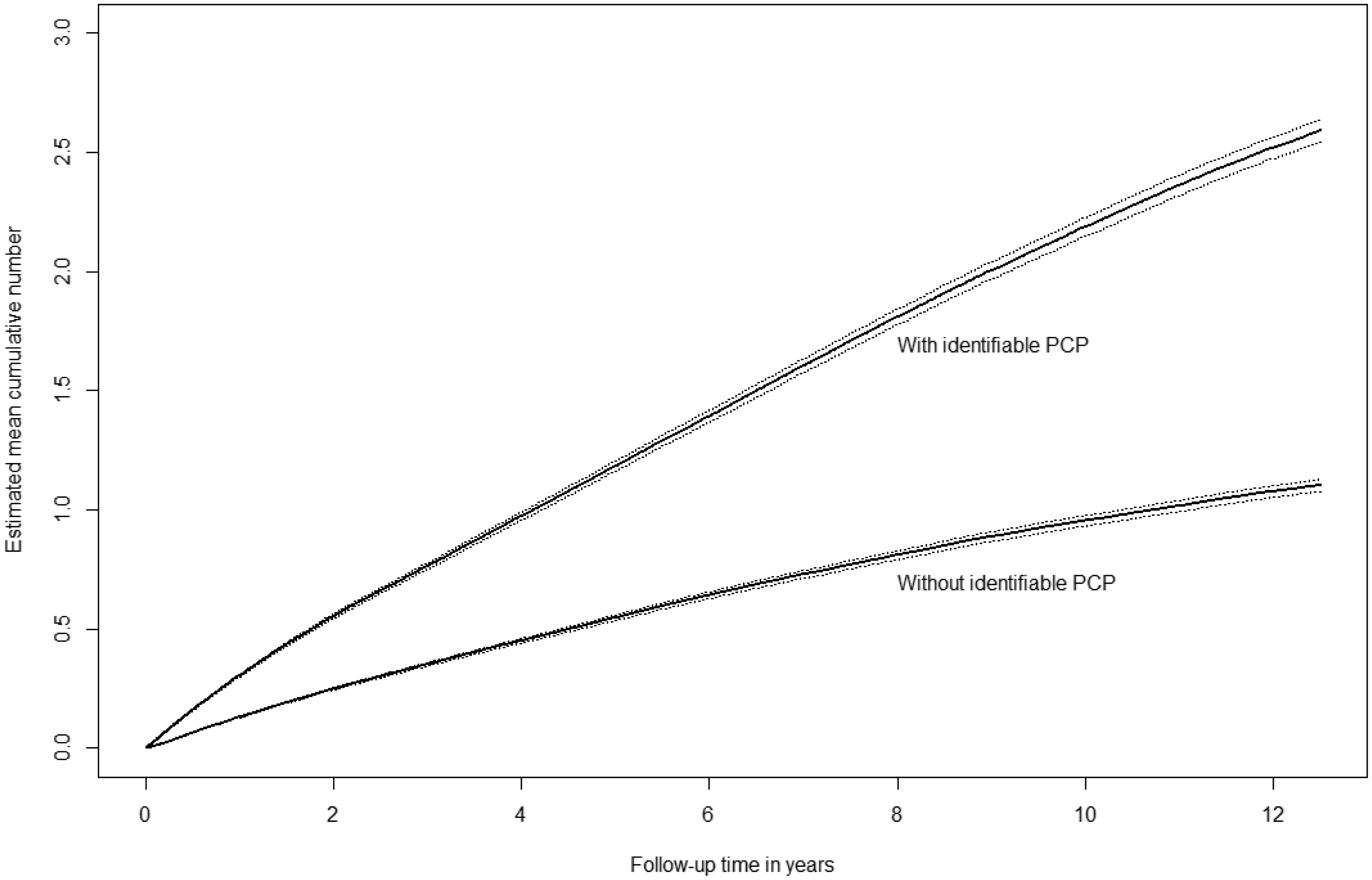
Discussion
In studying adherence to breast cancer screening over 13 years among a province-wide cohort of over 2.5 million screen-eligible women, a longitudinal analysis was conducted using multistate transitional models. The results indicated faster rates of transition from the non-adherent to the adherent state among women living in wealthier neighbourhoods, with more healthcare resource utilization, longer duration of residence in Canada, and an identifiable PCP who was female and graduated from Canada.
Women residing in Ontario, Canada can receive a mammogram either through physician referral or without referral through the OBSP. Even with these two systems in place, our study demonstrated the continued importance of having a PCP, both to minimize the duration of time spent non-adherent with screening and to increase the rate at which women become adherent. Our results provide a message similar to that from prior work, showing that having a PCP is important in the uptake of periodic mammograms. 22 This is supported by other research findings that recommendations from PCPs can increase participation in public breast screening programs, and that women who have a PCP are more likely to receive these recommendations.33–35
For the majority of women, factors associated with adherence to screening mammography were more PCP-related rather than patient-related. 36 Although the value of screening is well accepted among physicians, prior work has demonstrated that female PCPs were more likely to encourage screening than their male colleagues,37–39 and that PCPs trained in Canada were more likely to provide appropriate screening and preventive care than international medical graduates.40,41 Our results further support this finding by showing that women with female physicians or physicians who were Canadian medical graduates spent a shorter proportion of time in the non-adherent state, and also experienced a faster rate of becoming screen-adherent. It is possible that female physicians hold a stronger orientation towards screening and preventive care, and have greater confidence in the effectiveness of mammography.22,42
Our study design and analytic methodology had several strengths. It was a population-wide study over 13 years of all age-eligible women in the province. This was the first individual-level longitudinal analysis to implement a multistate transitional model to examine a woman’s pattern of adherence to breast cancer screening over time. The multistate transitional model was particularly well suited for the longitudinal structure of the data, as it could accommodate repeated transitions between non-adherent and adherent states, and it accounted for the time-varying nature of each covariate. Study limitations include the inability to control for possible confounders, specifically inability to capture information on ethnic background or individual barriers to screening, as this was not available from administrative databases.
This study makes a novel contribution to improving the quality of evidence on disparities among women in adherence to breast cancer screening over time. Our findings should be used to target appropriate populations to minimize the time spent not adherent. Our methodology also provides a foundation for understanding patterns of adherence for other types of cancer screening, including cervix and colorectal cancer screening.
Footnotes
Acknowledgements
Parts of this material are based on data and information compiled and provided by Canadian Institute for Health Information (CIHI), by Citizenship and Immigration Canada (CIC), and by Cancer Care Ontario (CCO). These datasets were linked using unique encoded identifiers and analyzed at the Institute for Clinical Evaluative Sciences (ICES).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Canadian Centre for Applied Research in Cancer Control (ARCC) to Rinku Sutradhar as the Principal Investigator. This study was supported by the Institute for Clinical Evaluative Sciences (ICES), which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES, the MOHLTC, CIHI, CIC, or CCO is intended or should be inferred.
