Abstract
Objective
To determine whether visits to a primary care provider (PCP) are associated with the uptake of periodic mammograms as women get older.
Methods
The cohort consisted of 2,389,889 women resident in Ontario, Canada, aged 50 to 79 at any point from 2001 to 2010, who were cancer-free and eligible for the Ontario Health Insurance Plan prior to study entry. Non-parametric estimation was used to describe the mean cumulative number of periodic mammograms for women with and without recent exposure to a PCP, as a function of age. Using age as the time scale, a recurrent event regression model was also implemented to examine the association between exposure to a PCP and rate of periodic mammograms, adjusted for income quintile and comorbidity.
Results
The mean observation window was 7.0 years. Uptake of periodic mammograms was significantly higher for women with recent exposure to a PCP compared with those without. This trend remained consistent as women aged, and the magnitude of the association increased for women aged 65 or older. The relative rate of periodic mammograms was lower than 1 and consistently decreased as women from lower income quintiles were compared with women from the wealthiest quintile.
Conclusion
Visits to a PCP play an important role in uptake of periodic mammograms, and this association increases as women age.
Keywords
Introduction
Screening for breast cancer is an important tool in detecting cancer at an early stage or preventing cancer by identifying precancerous lesions.1–3 Canadian cancer screening guidelines for average-risk women recommend that women aged 50–74 undergo mammographic breast cancer screening every 2–3 years.4,5 Despite this, every year there remains a large proportion of screen-eligible women who have not been screened.4,6,7 Studies indicate that those who do not attend regular screening mammograms are at increased risk of delayed breast cancer diagnosis, which may affect treatment options and outcomes.8–10
Under universal health care in Canada, mammograms are free for all residents. Before the 1990s, mammograms in the province of Ontario were obtained through the Ontario Health Insurance Plan (OHIP) and required a referral from a physician. In the early 1990s, the Ontario Breast Screening Program (OBSP) was introduced, to provide free, high-quality breast cancer screening services 11 , without a physician’s referral, for Ontario residents. Despite this programme, and universal health care, the under-screened populations in Ontario include women without a family physician, and women living in poverty.12–16 Previous studies show that having a primary care provider (PCP) is instrumental for breast cancer screening.12–18 Although referrals are not necessary in the OBSP, mammograms are often ordered by PCPs, and a physician’s advice or recommendation to have mammograms enhances uptake. 18
Most studies have examined the role of PCPs in screening for breast cancer in a cross-sectional manner12,17, and none has examined whether the PCP role remains strong and consistent as women become older. To our knowledge, this is the first individual-level analysis of a province-wide cohort to longitudinally assess whether visits to a PCP are associated with the uptake of periodic mammograms as women age. We aim to improve the quality of evidence on the role of PCPs in breast cancer screening, and our methodology may provide a foundation for understanding the importance of PCPs for other types of cancer screening.
Methods
Study population
In this retrospective longitudinal cohort study, we identified all Ontario female residents aged 50–79 at any point from 1 January 2001–31 December 2010, using the Registered Persons Database. These women were then linked, using unique, encoded patient health care identifiers, to other population-based administrative databases at the Institute for Clinical Evaluative Sciences (ICES). Women eligible for health coverage through OHIP at the time of study entry were included. Using the Ontario Cancer Registry and Canadian Institute for Health Information Discharge Abstract Database, we excluded women who had a cancer diagnosis, mastectomy, or breast implant procedure prior to study entry. Women were followed until the day before their 80th birthday, or their date of cancer diagnosis, mastectomy, breast implant procedure, death, first date of OHIP ineligibility (typically due to moving out of the province), or until 31 December 2010, whichever happened first. The study complied with the ICES data confidentiality and privacy guidelines and was
Outcome and covariate definitions
The outcome for each woman was defined as the times of bilateral periodic mammograms occurring during their observation window (retrieved from the OBSP database). Only bilateral periodic mammograms received through the OBSP, which do not require PCP referral, were considered an outcome. A mammogram was considered periodic if it occurred 11–36 months following a previous mammogram; the previous mammogram may have occurred through the OBSP or through OHIP. As our study period was from 2001–2010, to construct this conservative 11–36 month window for periodic mammograms, we selected the lower cut–off at 11 months based on the 2001 Canadian task force recommendation for mammograms every 1–2 years, and the upper cut-off at 36 months based on the 2011 Canadian task force recommendation for mammograms every 2–3 years. 5 Mammograms occurring outside this window were not considered periodic, but either follow-up mammograms (if occurring within 11 months) or spurious mammograms (if occurring after 36 months).
The main covariate in our study was exposure to a PCP. Using billing data from OHIP, a physician was defined to be a woman’s PCP if they claimed the maximum dollar value (from a set of 18 comprehensive primary care billing codes) during the two years prior to the time point of interest. This set of billing codes and look-back window have been used in previous studies 19 and we maintained this definition for consistency. If there were no relevant claims in the previous two years, the woman was considered not to have visited a PCP. Other covariates included comorbidity and socioeconomic status. Comorbidity was measured using the Johns Hopkins Adjusted Clinical Groups System20,21, in which counts of Aggregated Diagnosis Groups (ranging from 0 [no diagnosis] to 32 distinct diagnosis groups) over a 2-year look-back window indicated the level of comorbidity. The Johns Hopkins system is one of the best performers for predicting health service utilization in primary care settings. 22 Socio-economic status, retrieved from the Registered Persons Database, was captured using quintiles of neighbourhood median household income via postal code linkage. 23 Information on all covariates was updated at the start of each year (using the above mentioned look-back windows) for every woman throughout their observation period.
Statistical analysis
We examined the distributions of cohort characteristics at study entry, computing proportions for categorical variables, and means and standard deviations for continuous variables. To understand the uptake of periodic mammograms as women aged, we used non-parametric estimation to compute and graph the mean cumulative number of periodic mammograms (also referred to as the expected total number of periodic mammograms) as a function of age, for women who had and those who had not visited their PCP in the last two years (information updated on a yearly basis). 24 Women could therefore go in and out of contributing to both curves, depending on how their PCP exposure status changed over time, but at any specific time could only contribute to one of the curves. The corresponding 95% confidence intervals (CI) for each curve were also plotted.
We examined the association between visits to a PCP and rate of periodic mammograms using an Andersen-Gill multivariable recurrent event regression model 25 , which may be considered an extension to the Cox model as it allows for repeated occurrences of a single type of event over time. Age was chosen as the time scale, as it was more meaningful than selecting an arbitrary time 0 in this study. Thus the event times for each woman were the ages at which they had periodic mammograms. A robust variance estimation approach was used to handle multiple periodic mammograms experienced by the same woman, and the Breslow method was implemented to accommodate any ties in the ages at periodic mammograms across women. 26
A counting process data structure was used to execute the recurrent event model 26 , because it allowed us to capture the times of each event, as well as changes in a woman’s characteristics over time. The age at each periodic mammogram was updated instantaneously. Exposure to a PCP was the main covariate of interest. This was included as a binary time-varying covariate, and was updated at the start of each year based on whether or not the woman visited their PCP during the previous two years. Comorbidity was captured in the model as a time-varying covariate; the counts of aggregated diagnosis groups (ranged 0 – 32) were categorized into four levels (0, 1–4, 5–9, 10+), with count 0 implying no diagnoses. This categorization was updated at the start of each year based on the 2-year look-back window. We also adjusted for neighbourhood income quintile, which was incorporated as a time-varying covariate with six levels: rural neighbourhood, and urban neighbourhood quintiles 1 to 5, 5 being the richest. This information was also updated at the start of each year based on the woman’s postal code at that time. Age was not included as a variable in the model, as it was chosen to be the time scale in the analyses. We also included an interaction between our main time-varying covariate (PCP exposure) and an indicator for age above 65. This was decided a priori in order to determine whether the impact of PCP exposure on the rate of periodic mammograms changed once women reached age 65 or older. The multivariable model thus concurrently consisted of PCP exposure, neighborhood income quintile, comorbidity group, and the interaction between PCP exposure and an indicator for age above 65. As a sensitivity analysis, we re-defined the outcome to include periodic mammograms obtained through either OBSP or OHIP, rather than only through OBSP. All analyses were completed using SAS 9.2 (Carey, NC) and R version 3.1.0.27,28 Variables with p-values less than 0.05 were considered to be significant.
Results
Distributions of characteristics of the cohort at study entry (N = 2,389,889).
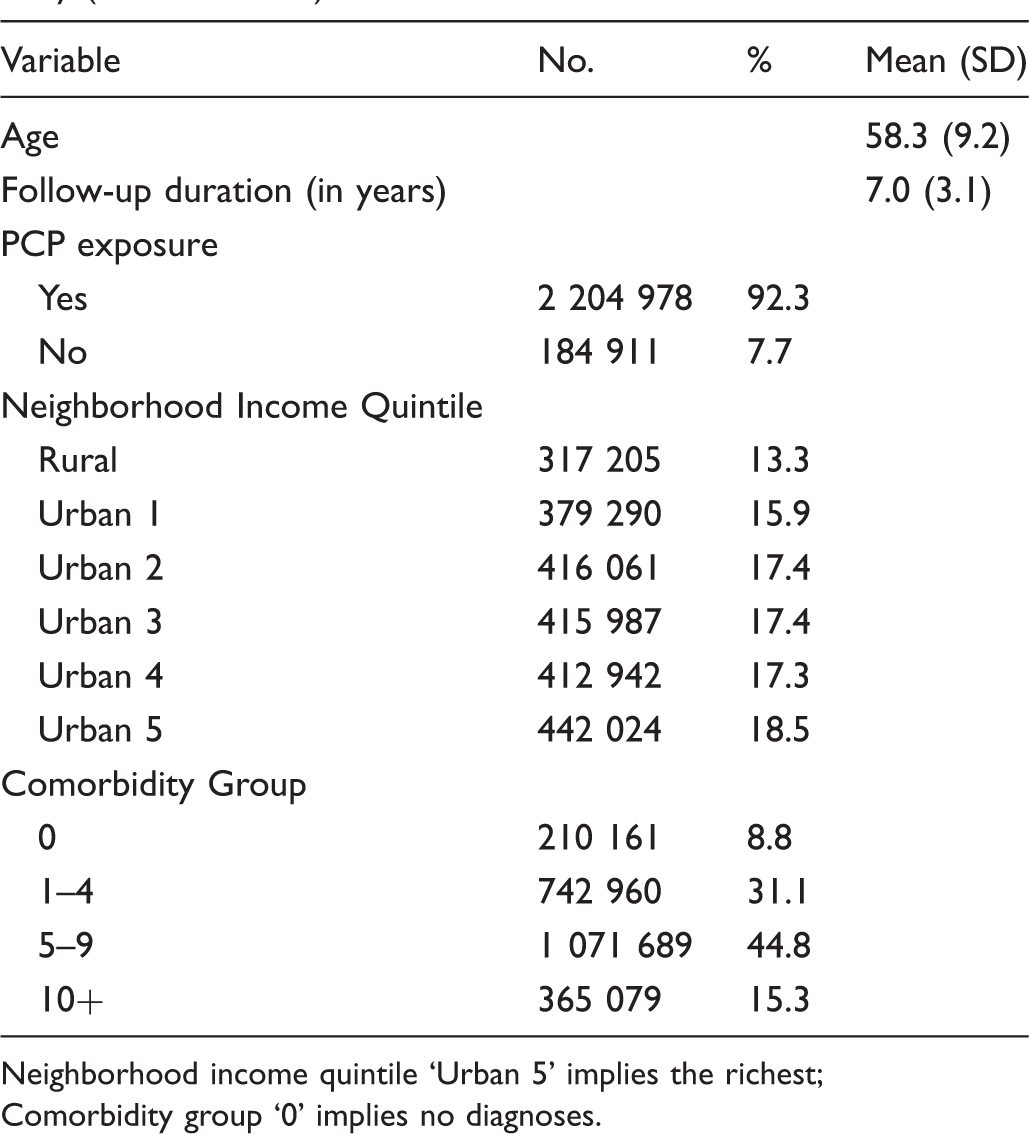
Neighborhood income quintile ‘Urban 5’ implies the richest;
Comorbidity group ‘0’ implies no diagnoses.
Figure 1 illustrates the mean cumulative number of periodic mammograms as a function of age for women had and had not visited their PCP in the last two years. At any given age, the uptake of periodic mammograms was higher for women who had visited their PCP in the prior two years compared with women who had not. This difference in uptake was significant, as seen by the non-overlapping 95% Cis, and remained consistent as women aged. Figure 1 also shows that, irrespective of exposure to a PCP, the uptake of periodic mammograms levels off after age 75, which is consistent with current Canadian breast cancer screening guidelines.
Mean cumulative number of periodic mammograms (and corresponding 95% confidence intervals) as a function of age, for women with and without recent exposure to a PCP.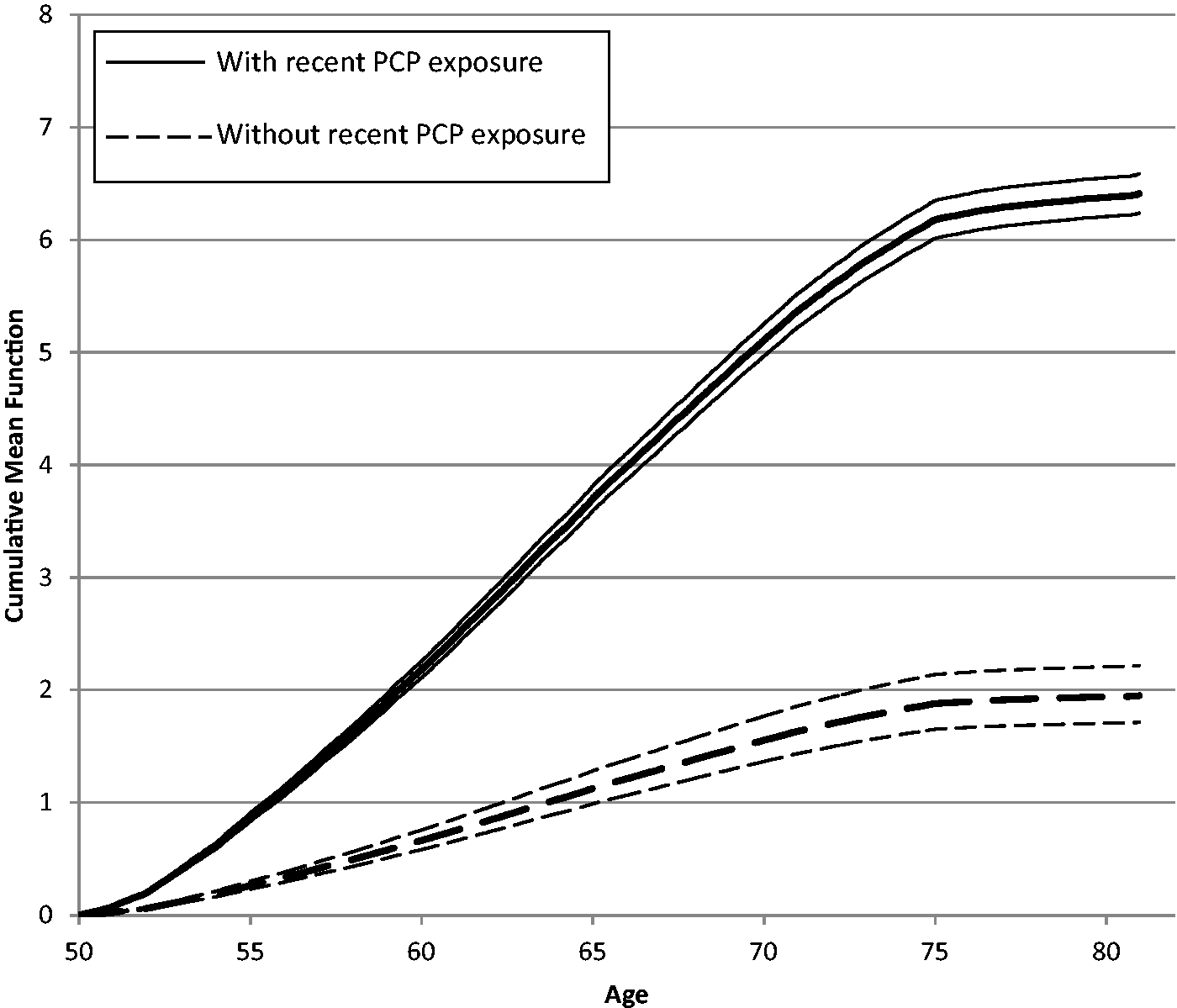
Multivariable recurrent event regression model examining the association between exposure to a PCP and the rate of periodic mammograms.
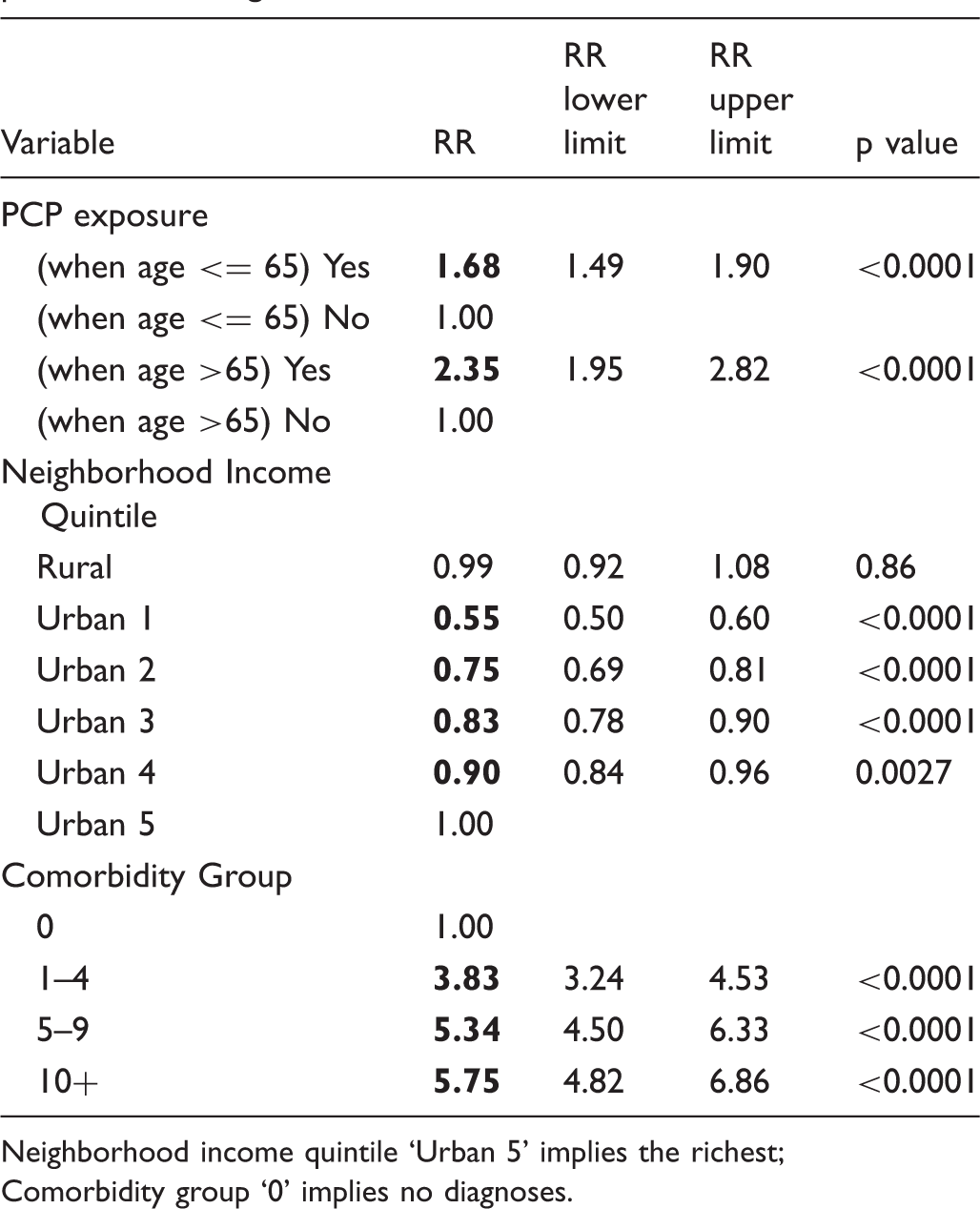
Neighborhood income quintile ‘Urban 5’ implies the richest;
Comorbidity group ‘0’ implies no diagnoses.
There was a progressive decrease in the rate of periodic mammograms among women from the poorer urban income quintiles compared with those from the wealthiest urban income quintile. Compared with women from the wealthiest urban income quintile, the rate of periodic mammograms among women from the poorest urban income quintile was 45% lower (RR = 0.55, 95% CI 0.50–0.60). There was also an increasing trend in the rate of periodic mammograms when comparing women from the sicker comorbidity groups with women from the healthiest comorbidity group.
In a sensitivity analysis, we expanded the outcome to include periodic mammograms obtained through either OBSP or OHIP, rather than only through OBSP. At any given age, the rate of periodic mammograms for a woman who had visited her PCP in the previous two years was 4.24 times higher than a woman who had not done so (RR = 4.24, 95% CI 4.17–4.31). This strong association was expected, as mammograms obtained through OHIP still require physician referral.
Discussion
Since the introduction of the OBSP, women are invited by mail to book when their next screen is due. Results are sent to the woman and to her PCP if she has identified a physician to the programme. Although physician referral is not required for OBSP participation, family doctors in Ontario are encouraged to inform eligible patients about the programme, and the OBSP has a website detailing programme information for both physicians and screen-eligible women. 29 Prior work has shown that recommendations from PCPs increase participation in public breast screening programmes 30 , and it is likely that women who visit PCPs are exposed to more recommendations than those who do not. 29 Although the important impact of PCPs on cancer screening among women has been well studied, prior research has examined this association in a cross-sectional manner.12,17
Our work shows that, although breast screening in Ontario does not require a physician referral, visiting a PCP was associated with a significant increase in the uptake of periodic mammograms among age-eligible women. This association continued to hold true as women became older, and the magnitude of the association further increased once women were aged 65 or older. The significant relationship between PCP exposure and periodic mammograms remained strong even after adjusting for updated neighbourhood income quintile and comorbidity. We believe that this is the first study confirming the instrumental role of PCPs on the rate of periodic mammograms as women age since the establishment of the OBSP.
This study has several strengths. As a population-wide study consisting of all age-eligible women in Ontario, it spanned a 10 year period, and information on the main covariate (PCP exposure) was updated yearly for each woman. We were able to control for neighbourhood income quintile and comorbidity, which were also updated yearly for each woman. The analytic method was suited to the longitudinal structure of the data, and was able to handle the repeated outcomes of periodic mammograms, as well as the time-varying nature of all covariates. The study also had several limitations. We could not control for possible confounders, such as ethnic background or whether or not the woman was a recent immigrant. These measures were not available at the individual level. We are also unable to generalize our findings to populations with operationally different screening programmes.
Although PCPs play a central role in cancer screening, there remain barriers to receiving follow-up screening mammography that are PCP-related rather than patient related in the majority of instances. 31 Prior work suggests that most barriers resulted from PCP failure to recall that a screening mammogram was due. 31 Female PCPs were more likely to encourage screening mammography than their male colleagues32–34, possibly due to stronger orientation towards screening and preventive care, and greater belief in the effectiveness of mammography. 35 In Ontario, PCPs trained in Canada were shown to be more likely to provide appropriate screening and preventive care than international medical graduates.36–38
Public breast screening programmes have provided accessible, high-quality screening services throughout the province. This study demonstrates the continued significance of visiting a PCP on the uptake of periodic mammograms as women age. Future work is needed to longitudinally assess the role of PCPs on other types of screening utilization.
Footnotes
Authors’ disclosures
The author(s) indicated no potential conflicts of interest.
Acknowledgements
This work was supported by a grant of endowed funds from the Research Committee of the Board of Directors of Cancer Care Ontario to Lawrence Paszat as Principal Investigator of the Ontario Cancer Screening Research Network. Richard Glazier is supported as a clinician scientist in the Department of Family and Community Medicine at the University of Toronto and at St. Michael’s Hospital. This study was supported by the Institute for Clinical Evaluative Sciences (ICES), which is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). The opinions, results and conclusions reported in this paper are those of the authors and are independent from the funding sources. No endorsement by ICES or the Ontario MOHLTC is intended or should be inferred. These datasets were linked using unique encoded identifiers and analyzed at the Institute for Clinical Evaluative Sciences (ICES).
