Abstract
Background
Screening participants with abnormal faecal occult blood test results who do not attend further testing are at high risk of colorectal cancer, yet little is known about their reasons for non-attendance.
Methods
We conducted a medical record review of 170 patients from two English Bowel Cancer Screening Programme centres who had abnormal guaiac faecal occult blood test screening tests between November 2011 and April 2013 but did not undergo colonoscopy. Using information from patient records, we coded and categorized reasons for non-attendance.
Results
Of the 170 patients, 82 were eligible for review, of whom 66 had at least one recorded reason for lack of colonoscopy follow-up. Reasons fell into seven main categories: (i) other commitments, (ii) unwillingness to have the test, (iii) a feeling that the faecal occult blood test result was a false positive, (iv) another health issue taking priority, (v) failing to complete bowel preparation, (vi) practical barriers (e.g. lack of transport), and (vii) having had or planning colonoscopy elsewhere. The most common single reasons were unwillingness to have a colonoscopy and being away.
Conclusions
We identify a range of apparent reasons for colonoscopy non-attendance after a positive faecal occult blood test screening. Education regarding the interpretation of guaiac faecal occult blood test findings, offer of alternative confirmatory test options, and flexibility in the timing or location of subsequent testing might decrease non-attendance of diagnostic testing following positive faecal occult blood test.
Introduction
Periodic faecal occult blood testing (FOBt) is a common method of screening for colorectal cancer (CRC).1,2 Meta-analysis of randomized trials demonstrates that guaiac-based FOBt reduces CRC-related mortality by approximately 16%. 3 Such mortality reductions require further colonic testing after a positive FOBt to diagnose CRC and treat smaller cancers or adenomas by endoscopic excision. Maximizing screening completion (i.e. colonoscopy) is crucial for these patients, because up to 10% will have CRC at their first screen. 4
Randomized trials of FOBt screening report non-completion rates of 7–17% after a positive FOBt result.5–8 Similarly analysis of the UK CRC screening pilot 9 and of the national rollout 4 found non-completion rates of 15–18% after positive FOBt. Comparable French data report a 12% rate; 10 in Ontario, Canada, the figure is approximately one in three. 11 Therefore, depending on programme structure, 10–33% of FOBt-positive screenees do not undergo confirmatory testing. Certain patient groups are at higher risk of non-completion, for example, those with lower socio-economic status12,13 or physical/psychological co-morbidity. 14 These ‘epidemiological signals’ suggest that there may be missed diagnostic opportunities 15 in FOBt-based CRC screening at the time of colonoscopy, which might be targeted to improve uptake.
Missed diagnostic opportunities may be due to organizational factors (e.g. insufficient endoscopy resource, poor referral guidelines) or patient factors (e.g. cognitive, emotional, or physical barriers). For example, physicians commonly fail to act on positive FOBt results, 16 either because they never reviewed the result or chose to repeat the FOBt, thereby contravening good practice guidelines.17,18 However, these individual physician-related and organizational factors are not commonly relevant to population screening programmes, in which endoscopy capacity is assured, referral guidelines are established, and the administrative burden is often centralized.4,10,11 Conversely, there are few data regarding patient-specific factors underpinning non-completion in this setting. Lower socio-economic status and physical/psychological co-morbidity are associated with higher rates of non-completion, but we do not know how these risk factors translate to individual decision-making.
FOBt-positive individuals who do not attend for colonoscopy represent a large, high-risk group. There are few patient-level data on why such non-attendance occurs. We investigated this in a population-based screening programme with a centralized call–recall system via retrospective review of detailed screening records.
Methods
This study was conducted within the English Bowel Cancer Screening Programme (BCSP) and was approved by the BCSP Research Committee. Following Health Research Authority guidance, ethical permission was not required for retrospective review of anonymized, routinely acquired data. A preliminary pilot study was approved by the London Harrow Research Ethics Committee, reference 12/LO/1978.
Study population
The BCSP uses biennial FOBt for individuals aged 60–74.
4
Administration and analysis of FOBt kits is coordinated by five regional laboratories. After a positive result, clinical review and colonic testing are conducted at a local ‘screening centre’. Clinical review is led by a trained Specialist Screening Practitioner (SSP). The screening pathway is shown in Figure 1. This study was conducted within two of the 62 screening centres. These centres were selected because they (a) had resources available to support the study, (b) had pre-existing research collaborations with the study team, and (c) were located within a single Hub, simplifying information governance.
Flow chart depicting typical patient flow through the screening programme (thick outlined boxes) and pathways to screening episode closure. Colonoscopy non-participants and SSP clinic non-attenders were eligible for records review.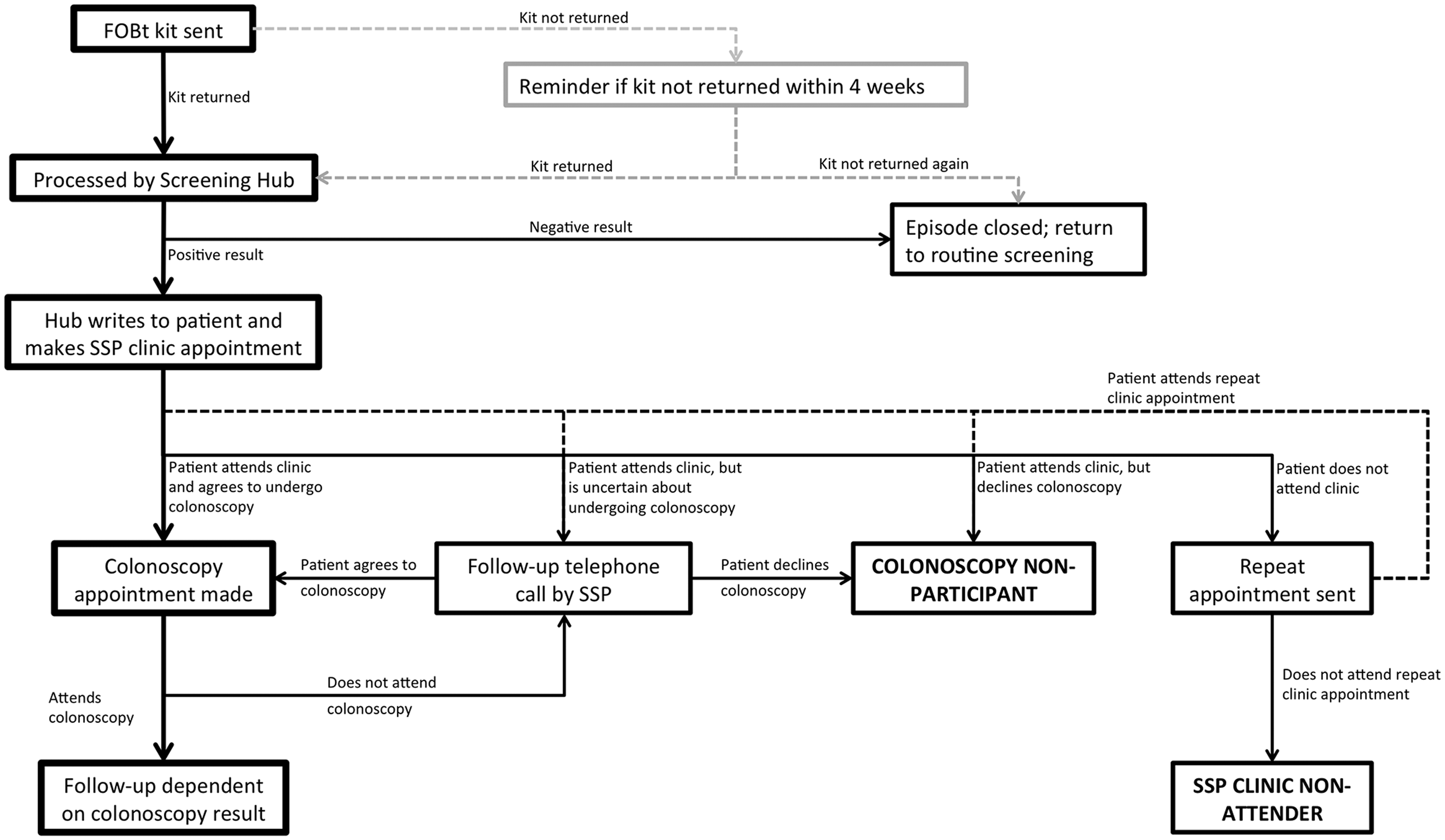
Individuals were eligible for inclusion if they had a positive FOBt result from November 2011 to April 2013 but had not attended a SSP Clinic (to assess fitness for colonic testing and seek informed consent) or an appointment for colonic testing. These individuals were identified using the BCSP in-house database by the Hub Director (SR, who extracted episode notes (including free text entries by screening centre staff) for the researchers conducting the medical record review.
Routine BCSP practice is for two appointment letters to be sent for the SSP clinic. If no contact has been made after this, the screenee is considered a non-attender and the screening episode is closed. Non-participation at colonoscopy is followed by a telephone call (and a letter if non-contactable), inviting the screenee to re-arrange the appointment; non-response by 14 days precipitates episode closure.
Data extraction
We undertook a detailed medical record review of eligible participants to obtain information about non-attendance. The programme records system is a structured Oracle database (Oracle Corporation, Redwood, CA, USA). Each event within a given screening episode (test results, care decisions, or clinical interactions, including telephone consultations) is recorded. Free text entries are encouraged, and such notes are kept meticulously by SSPs. These clinical notes constitute a detailed and valuable resource for monitoring and assessing patient behaviour.
To complete the screening records review, the Screening Hub Director (initially SR, then ND) reviewed the clinical entries of all eligible patients and extracted the following: (i) screenee age and sex, (ii) point of departure from the screening pathway (i.e. non-attendance at the SSP clinic versus colonoscopy), (iii) previous CRC screening history, (iv) subsequent CRC screening history, and (v) free text entries recording reasons for non-attendance. Free text entries were made by screening centre staff and summarized conversation with the patient (or their representative) and screening centre staff (either at the SSP clinic or by telephone). To satisfy research governance permissions, the Hub Director excluded participants who had died, left the country/screening centre, or refused permission for further contact by the screening programme.
Analysis
Free text entries were coded by AG (a psychology researcher) and AAP (a medical practitioner with academic interest in CRC screening). Data were analysed based on established qualitative research methodology.19,20 Initially, each researcher independently reviewed and interpreted the free-text data, and identified broad categories emerging as reasons for non-attendance (e.g. ‘unwilling to have test’). Patients were then coded into all categories that were considered to apply to them. The two researchers then harmonized categories and coding by face-to-face discussion. Category names were discussed to determine whether they could be meaningfully merged with others, renamed, or separated under distinct headings, or grouped under a broader category heading. The independently derived codes for each patient were also discussed, and any disagreements were resolved in consensus, arbitrated by a third researcher (CVW) who was blinded to the originally assigned codes to avoid biasing the decision.
Finally, for each individual subject, the single most important reason for non-attendance was recorded, as judged subjectively in consensus by the raters based on information in the medical records. Data were summarized with descriptive statistics.
Results
Characteristics of study population
During the study period in the two centres, 177,863 individuals were invited for screening, 87,664 completed FOBt screening (49.3%), and 2404 had a positive result (2.7% of those returning a test kit). Records review identified 170 individuals (7.1% of those with a positive result) who ultimately did not undergo colonoscopy prior to screening episode closure (Figure 1 shows routes to episode closure). Of these, 88 individuals (51.8% of all non-attenders) were excluded by the screening Hub Director prior to data extraction, because they had died, left the country, moved to another part of the country, or had requested removal of their contact details from the screening programme database, leaving 82 cases for further analysis. No further data were available for the 88 excluded individuals.
Included individuals had a median age of 64.5 (interquartile range (IQR): 62.2–69.2) and there was an approximately equal gender split (42 females, 40 males). Patients often had a previous history of screening non-adherence: 36 kits had been returned from the 72 previous episodes for which data were available, giving an overall previous guaiac faecal occult blood test (gFOBt) uptake of 50.0%. About half of all non-attenders did not attend the SSP clinic appointment (38/82, 46.3%) and half attended clinic but not colonoscopy (44/82, 53.7%).
Patients frequently made repeated telephone contact with screening services, despite ultimately not attending. The median number of times a non-attending screenee was in contact with the screening centre was 2 (IQR: 1–4). Family members often also telephoned screening centres on the behalf of the patient—this occurred for 15 (18.3%) of the 82 individuals, most commonly to explain non-attendance. An interpreter was requested by eight patients or family members (9.8%).
By the time of data analysis, 39 individuals had been sent a further FOBt kit (i.e. had entered their next biennial round of FOBt screening). Of these, only 17 (43.6%) completed this further round of screening.
Reasons for non-participation at screening colonoscopy
Reasons for non-attendance for further colonoscopy, as stated by 66 individuals: 22 SSP clinic non-attenders and 44 colonoscopy non-attenders. For patients giving more than one reason for non-attendance, all reasons are recorded.

Sixteen further individuals (all of whom did not attend the SSP clinic) had no recorded explanation for non-attendance. Percentages use the total number of reasons given for non-attendance by that category of patient (i.e. SSP-clinic non-attenders or colonoscopy non-attenders) as the denominator.
FOBt: faecal occult blood testing; NHS: National Health Service.
Most patients (43/66, 65.2%) had a single recorded reason for non-participation, 18 (27.3%) had two recorded reasons, and five (7.6%) had three recorded reasons. The 93 explanations for non-participation fell into seven broad categories: unwillingness to have the test (28 reasons, 30.1%), other commitments (21 reasons, 22.6%), belief that the FOBt result was a false positive (16 reasons, 17.2%), another health issue taking priority (14 reasons, 15.0%), already having investigation planned elsewhere (seven reasons, 7.5%), practical barriers (five reasons, 5.4%), and patient errors in bowel preparation/dietary restriction (two reasons, 2.2%) (see Table 1).
Reasons for non-participation were largely similar for either SSP clinic or colonoscopy non-participation, with the exception that SSP clinic non-attenders were more likely to have already arranged colonoscopy outside the programme (SSP non-attenders: 5/23 total reasons for non-attendance, 21.7%; colonoscopy non-participants: 2/70 total reasons, 2.9%, p = 0.0079; Table 1).
When considering only an individual’s most important reason for non-participation, similar patterns were demonstrated: 17 of 66 individuals had other commitments (25.8%), 16 (24.2%) were unwilling to undergo the test, 13 (19.7%) believed the FOBt result was a false positive, 12 (18.2%) had another health issue taking priority, seven (10.6%) were planning treatment elsewhere, and one (1.5%) had a practical barrier (e.g. distance to travel, issues with fasting).
Author interpretations of free-text data entries
Many stated reasons for non-attendance were temporary rather than permanent, for example, short-term illnesses (such as a cold, fever, or a problem with medication), or brief trips away, neither of which would preclude colonoscopy at a later date. In these cases, patients may have subsequently forgotten about their appointment. However, some individuals later refused colonoscopy even after a telephone reminder (e.g. ‘patient said she could not come because she's got a bad cold. She was asked if she wanted to rebook. She said she will call when she feels better … [weeks later] … SSP phoned patient to rebook but she does not want to proceed’; female, 71 years). Another common theme was denial and disbelief that the FOBt result might indicate CRC, and instead must have been a false positive (e.g. ‘Patient opted out – insists results were positive due to a bloody tissue she placed on faeces’; female, 69 years; ‘Patient has piles and is convinced that the bleeding was just due to that’; female, 69 years).
Discussion
In this study, we retrospectively reviewed medical records of patients who had not completed colonoscopy, despite a positive screening FOBt result. We grouped reasons for non-attendance into broad categories, the largest of which were unwillingness to have colonoscopy, other commitments, the belief that the FOBt test result was a false positive, or other health issues taking priority.
Previous research regarding non-attendance for colonoscopy has often focused on its use as a first-line test. 21 Although this provides information regarding colonoscopy-specific barriers, it does not necessarily apply to a screening programme based on FOBt (or faecal immunochemical testing), in which patients testing positive are at higher risk of CRC.22,23 Considering FOBt-positive individuals specifically, Shields et al. 24 found that among patients in a US municipal opportunistic screening programme, those with a positive family history of CRC, greater worry regarding cancer, or with a more strongly positive FOBt result were more likely to undergo colonoscopy. Zheng et al. 25 found that patients who perceived fewer barriers to screening, greater benefits of screening, and had greater knowledge of CRC risk factors reported higher intention to complete screening. More recent data from the Ontario FOBt-based screening programme found that participants with recent prior colonoscopy, hospital admission, or having repeat FOBt were less likely to complete colonic testing. 11 Ferrat et al. 12 found low socio-economic status was associated with non-completion, as were receiving the FOBt kit via post rather than from a Primary Care Physician, and inadequate information regarding colonoscopy. Partin et al. 14 found that older patients, those with limited life expectancy, and dual diagnosis of psychiatric disorder/substance abuse had higher non-completion rates.
We found that the test itself (colonoscopy) constituted a major barrier to screening completion after positive FOBt. This concurs with recent evidence from a vignette-based study in which 11% of respondents would have declined colonoscopy, even if they had symptoms indicating a 10% risk of CRC (similar to after a positive FOBt result). 26 An appreciable proportion of patients clearly find colonoscopy unappealing, even in the face of a high risk of CRC. Some of these concerns may be alleviated by the offer of alternative tests (e.g. CT colonography), which might be perceived as more acceptable. Data from a Dutch randomized trial suggested that non-attendance at colonoscopy was more likely to be due to concerns regarding the test, whereas non-attendance at CT colonography was more likely to be underpinned by lack of time. 27 US data suggest that non-attenders at colonoscopy would accept an offer of CT colonography, 28 and a small randomized study from Italy found that FOBt-positive patients who declined colonoscopy were more likely to attend when offered CT colonography than those who were re-offered colonoscopy. 29
Altering the test used will not always address fundamental reasons for non-attendance. For example, 16 patients felt that there were alternative explanations for their positive FOBt result (including haemorrhoids), or that the result was somehow ‘incorrect’ (e.g. normal previous colonoscopy). Offering an alternative test will not address such misconceptions. Instead, it is important to improve awareness of the principles of CRC screening, particularly with regard to previous colonoscopy (i.e. that a previous normal examination does not always obviate subsequent disease).
Most of the documented reasons for non-completion potentially could have been overcome. For example, temporary fasting or incorrect use of bowel preparation could be resolved by rescheduling. Similarly, while some of the other health issues taking priority were serious, others were not (e.g. temporary medication problems, having a fever, or the common cold), and should not prevent colonoscopy at a later date. It is possible that these stated reasons masked true underlying causes. Previous studies have described patients often presenting superficial explanations for non-attendance that obscure genuine concerns, such as fear of being diagnosed with cancer. 30 Furthermore, for patients who may already be ambivalent about completing screening, an ostensibly small barrier may become relatively more important (as that individual may feel that there is relatively little to gain by completing screening in any case).
Possible approaches to increasing uptake of colonoscopy based on apparent barriers.

FOBt: faecal occult blood testing.
The main strength of this study was that we were able to identify reasons for non-attendance among a particularly difficult-to-access group of individuals, often neglected by prior research. Furthermore, these are patient-triggered case notes, meaning that the contents are likely to align with patients’ own beliefs.
We found a much smaller proportion of patients who did not complete colonoscopy (7.1%) than has previously been reported, both in the UK 4 and internationally,10,11,30 and this is likely to be due to different methods of data extraction, and ‘filtering’ of our dataset by the screening Hub Director to ensure patient confidentiality. It is possible that we have not captured some important reasons for non-attendance. Our study is also limited because we were required to use retrospective reviews of medical records, to overcome the difficulties of contacting and interviewing non-adherent patients. Although detailed, it is possible that these medical records do not capture all relevant reasons, and some richness of the dataset will no doubt be lost. Furthermore, the fact that they have been entered by screening staff (rather than patients themselves) means there is a risk of failure to accurately capture the patients’ original thoughts or intentions. Although one-to-one interviews are an intuitively appealing alternative, we originally invited patients for a telephone interview to explore their reasons for non-attendance and received only a 3% response rate. Such interviews would be neither representative nor practical. Engagement of non-attenders is clearly extremely challenging, although intense recruitment facilitated via primary care might be possible. Additionally, there was a degree of subjectivity in our assessment and coding process, although we reduced this by using two independent coders and resolving disagreements with a third arbitrator. Our relatively small sample size means the estimated prevalence of each barrier to attendance carries some uncertainty. This could be addressed by a larger data extraction in the future, allowing more confident estimates of the importance of each of our major categories of reasons for non-attendance. Finally, the screening centres participating in this study are both urban, with relatively higher socio-economic deprivation and ethnic diversity than the national average.
In summary, the most frequently stated reasons for non-completion of colonoscopy in FOBt-positive patients were unwillingness to have the test, the perception that their FOBt result was a false positive, or other commitments and health issues taking priority. These individuals had low adherence to subsequent FOBt screening, meaning they remain a difficult-to-screen group. Education regarding the nature of FOBt screening and offering alternative tests with flexible scheduling at a range of locations might address some of these concerns.
Footnotes
Acknowledgement
We dedicate this article in memory of Professor Jane Wardle (1950–2015).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was funded in part by a Cancer Research UK Programme Grant (C1418/A14134) and by the Royal College of Radiologists Kodak Fund Scholarship. A proportion of the work was undertaken at University College London and University College London Hospital, which receive funding from the NIHR Biomedical Research Centre Scheme. None of the funding bodies were involved in the design of the study; the collection, analysis, or interpretation of the results; the writing of the manuscript; or the decision to submit for publication. The views expressed are those of the authors, and not necessarily those of Cancer Research UK, the Royal College of Radiologists, the NHS, the NIHR, or the Department of Health.
