Abstract
Objectives
To assess the feasibility and acceptability of offering self-sampling for Human Papillomavirus (HPV) testing to cervical screening non-attenders when they consult primary care for any reason.
Methods
In a pilot implementation study, six general practices in London, UK, offered self-sampling kits during consultation to women aged 25–64 who were at least six months overdue for cervical screening (no cytology test recorded in the past 3.5 years if aged 25–49, or 5.5 years if aged 50–64). Eligible women were identified using an automated real-time search (during consultation) of the general practice electronic medical record system. Women collected samples either in clinic or at home (dry flocked swabs analysed using Roche Cobas®4800).
Results
Of approximately 5000 eligible women, 3131 consulted primary care between January and December 2014 (mean recruitment period 9.5 months). Of these, 21% (652) were offered kits, 14% (443) accepted, and 9% (292) returned a self-sample. The proportion of eligible women offered kits varied considerably among practices (11–36%). Sample return rates increased with kit offered rates (r = 0.8, p = 0.04). Of 39 HPV positive women 85% (33) attended follow-up, including two with invasive cancers (stage 2A1 and 1A1).
Conclusions
Offering self-sampling to cervical screening non-attenders opportunistically in primary care is feasible. Return rates could be increased if more women were offered kits. A large trial is needed to identify how self-sampling is best integrated into the national screening programme, and to identify determinants of uptake.
Introduction
Conventional cervical screening relies on clinician-collected samples, but the introduction of human papillomavirus (HPV) testing presents the possibility of self-sampling (SS). SS can overcome both emotional (e.g. embarrassment, fear of pain) and practical (e.g. difficulty making appointments) barriers to cytological screening. Women can collect a self-sample in private, at a time and place they choose, without being examined, thereby saving both clinician time and clinic costs. HPV testing on self-collected samples is at least as sensitive as good quality cytology, to high grade CIN 1 but slightly less sensitive than HPV testing of clinician-collected samples (with similar specificity). 2 The Dutch screening programme will offer SS to cervical screening non-attendees from 2016, and several other countries are expected to follow suit.
Numerous non-UK studies3–9 have shown that SS can increase screening uptake in non-attenders (∼30–40% response), and that around 80% of women who test HPV positive attend follow-up investigations. The most commonly assessed approach and the only one (to date) to show increased uptake in randomized studies has been sending kits directly to women. An obvious disadvantage is the high number of wasted kits, and the associated costs. Opt-in strategies do not appear to work well.7,10
The UK picture is less clear; two studies11,12 posted SS kits to cervical screening non-attenders, but only 6–8% of women returned a self-sample (i.e. >92% kit attrition and the lowest response rates of all SS studies to date). A series of studies in Dumfries and Galloway found that 5–20% of screening non-attenders returned a self-sample when asked to request SS kits or SS kits were sent directly to women. 13 Further evidence is needed before SS can be recommended for integration into UK screening programmes.
One approach not yet studied is targeted offering of SS to cervical screening non-attenders when they consult primary care for any reason. Key advantages include fewer wasted kits (kits are handed directly to women) and an in-person contact with a population who are difficult to engage.14,15 An audit of electronic primary care records found that of women >6 months overdue for cervical screening, 32% consulted over three months and 60% over one year, 16 suggesting that a large proportion of relevant women could be approached in this way. Here we report on a pilot study to assess the acceptability and feasibility of opportunistically offering SS for HPV testing to cervical screening non-attenders in primary care.
Methods
Six general practices, four from East London and two from South London, took part. Practices were required to have the EMIS web (Egton Medical Information Systems Ltd, 2010) electronic patient record system. We targeted practices with low cervical screening coverage (below 75% of eligible women screened in the previous five years). Eligible women were aged 25–64 who were >6 months overdue cervical screening.
In England, cervical screening is offered to women aged 25–49 every three years and to women aged 50–64 every five years. An automatic search was set up in EMIS web to flag women (in real time) who did not have a cytology entry recorded in EMIS in the last 42 months for women aged 28–49 and 66 months for women aged 50–64. The lower age limit for the search was set to 28 instead of 25 to focus on women who were ‘true’ screening non-attenders (as opposed to still deciding whether or not to respond to their first invitation). An alert message appeared on screen for eligible women when their record was opened. General practitioners, nurses, and health care assistants were asked to offer flagged women a SS kit during any (non-cervical screening) consultation and to record reasons provided for declining kits. Kits contained a flocked swab in a self-sealing tube (FLOQSwabs™, Copan Italia, Brescia, Italy), a plastic specimen bag, an instruction leaflet for SS (written and pictorial), an HPV information leaflet, a study information leaflet, and a prepaid envelope for returning samples. Women were able to choose between taking their sample in clinic (behind a curtain or in the lavatories) or at home. Women who collected samples in clinic handed them to a member of staff for sending to the relevant testing laboratory. Women who collected samples at home posted them to the laboratory using the prepaid envelope. Samples were transported dry and were analysed at one of two laboratories (Barts Health NHS Trust cellular pathology and Preventx Limited commercial laboratory), depending on the general practice catchment area. Both laboratories used Cobas® 4800HPV Test (Roche Diagnostics GmBH) for HPV testing.
HPV positive women were advised in their results letter to book a cytology test in primary care. Those who had not had cytology within six months of testing HPV positive were invited to a study smear/colposcopy clinic, where they were offered cytology with or without colposcopy. Women who tested HPV negative were exited from the study. As self-samples are not currently included in the national cervical screening programme, only cytology tests were able to change the women’s screening status and contribute towards the general practice coverage statistics (which are linked to practice payments and used as an indicator of practice quality). This meant that some women who tested HPV negative on a self-sample also had a subsequent cytology test, either of their own accord or following active encouragement from their general practice.
Feedback from general practices was obtained throughout the study on an informal and ad hoc basis (e.g. by attending clinical meetings, during monitoring visits, or via email).
Statistical analysis
The study aim was to assess the feasibility and acceptability of opportunistically offering SS to cervical screening non-attenders in general practice primary care, as measured by the proportion of eligible women who are offered kits, accept kits, and return a self-sample. The exact number of cervical screening non-attenders over the study period was not known because the dates that women register and leave (unregister) general practices are not reliably recorded in EMIS. We were therefore unable to determine whether women who left during the study were overdue screening before leaving. Instead we estimated the total number of eligible women as the number who were >6 months overdue and were still registered at the end of the study, plus the number >6 months overdue who consulted but left during the study.Response rates were calculated using the proportion of eligible women (>6 months overdue cervical cytology) who consulted that accepted a SS kit (i.e. responders) and that returned a SS kit. Cervical screening status was classified using the time from last recorded cytology before study invitation as (1) ‘late’ – overdue by 6–24 months, (2) ‘very late’ – overdue by at least 24 months, and (3) ‘never’ – no cytology tests recorded. Descriptive statistics were used to compare demographic and cervical screening status between responders and non-responders. For HPV positive women, we calculated the proportion who had a follow-up investigation within six months. The χ2 test was used to determine the differences in proportions between groups. Pearson’s correlation coefficient was used to measure the strength of association between the proportion of women offered kits and the proportion who returned a self-sample at each practice. All analyses were performed using Stata version 13 (College Station, TX: StataCorp LP). A p-value of less than 0.05 was considered statistically significant. All statistical tests were two-sided.
Results
Between January and December 2014, 3131 of approximately 5000 eligible women consulted the general practice primary care. The mean recruitment period was 9.5 months (range 7.8–10.7 months). Of the 3131 women, 20.8% (652) were offered a SS kit, 14.1% (443) accepted, and 9.3% (292) returned a self-sample (i.e. 45% of those offered, returned a sample) (see Figure 1). This is equivalent to an annual rate of screening via SS of about 7% of non-attenders, varying from 4% to 27% among practices.
Participant flow chart.
Characteristics of participating GP practices and women who returned a self-sample by GP practice.
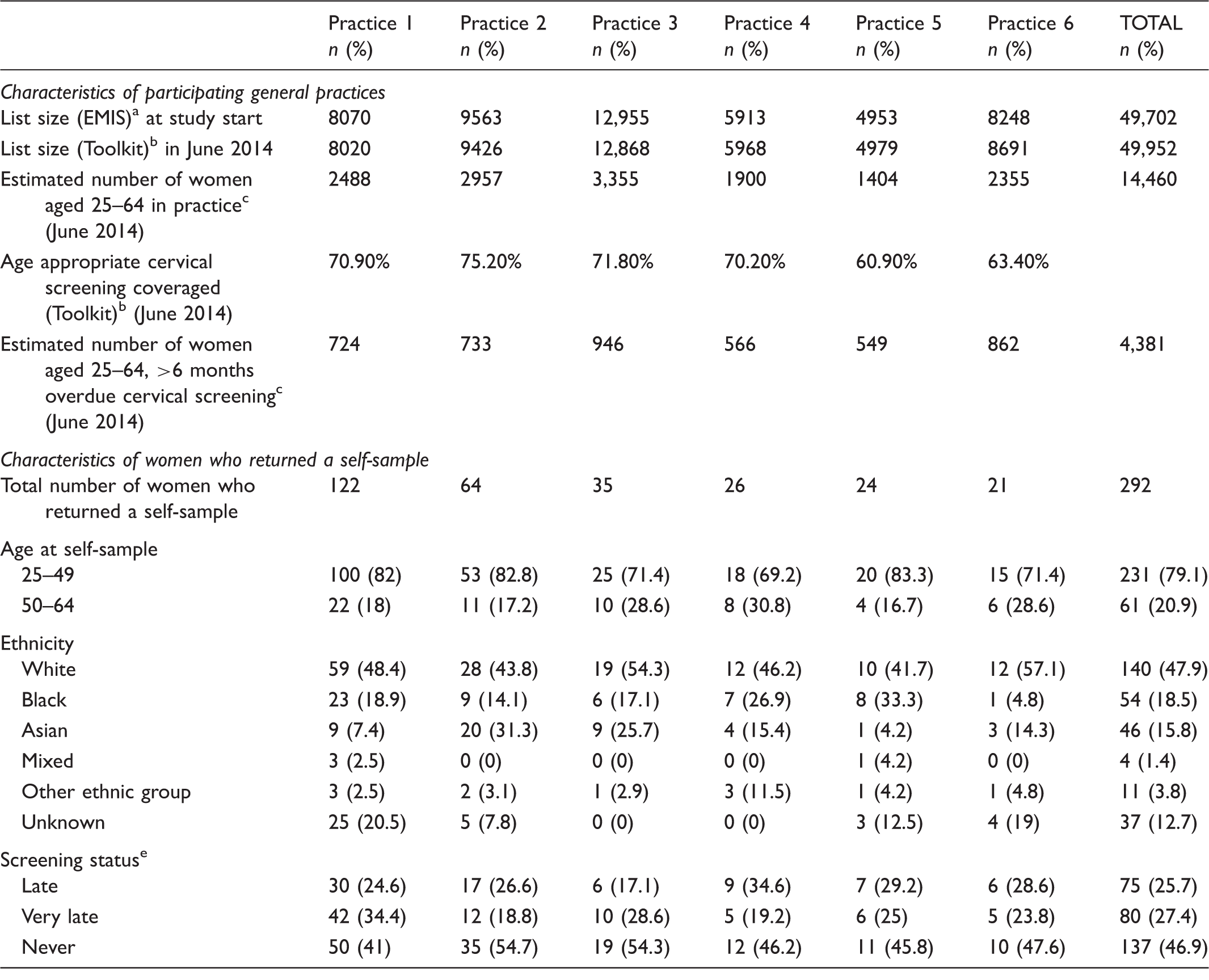
Data from EMIS GP record system at study start.
Data from the NCIN cancer toolkit GP practice profiles (https://www.cancertoolkit.co.uk/Profiles/PracticePublic/Filters). Data in the toolkit were extracted from the Open Exeter system in June 2014, and cover the period 2007/2008–2013/2014.
Estimated from toolkit data by multiplying the number of women aged 25–64 attending cervical screening within target period by (1-coverage).
Age appropriate coverage is the number of women registered at the practice screened adequately in the previous 3.5 years (if aged 24–49) or 5.5 years (if aged 50–64) divided by the number of eligible women on the last day of the review period.
‘Late’ = women who were overdue by 6–24 months, ‘Very late’ = women who were overdue by at least 24 months and ‘Never’ = women with no cytology tests recorded.
Cytology results for women who tested HPV positive on a self-sample.
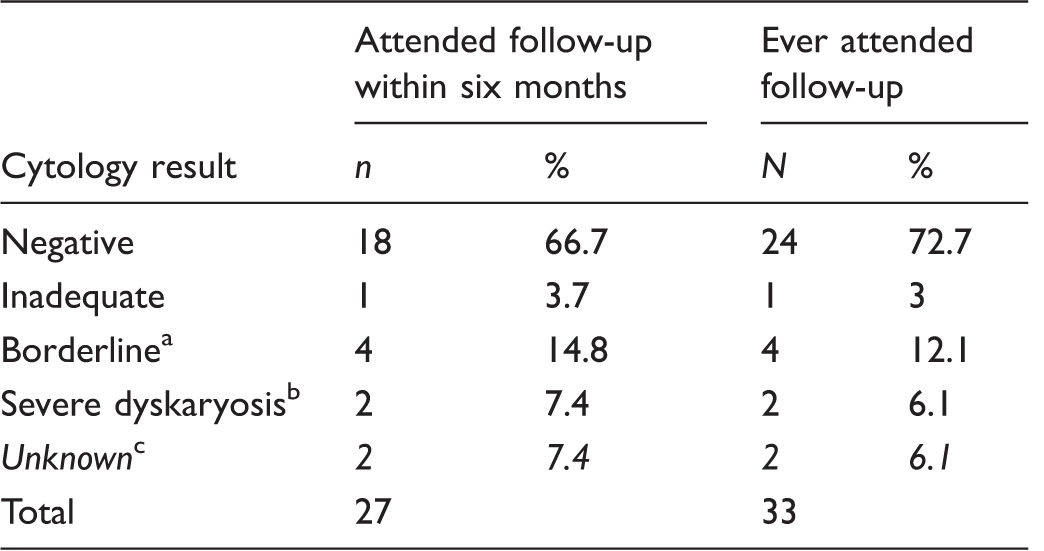
One CIN1 (confirmed on biopsy), three normal on subsequent cytology or colposcopy.
Two invasive squamous cell carcinoma.
One woman was lost to follow up (left the practice), for one woman cytology results were still not received four months after cytology testing.
The most common reason (30.6%) provided for declining an SS kit was that women preferred or intended to book a smear. However, only 52% (33/64) of those who said this had a smear within the subsequent six months. Only 11% (27/247) of women who tested HPV negative had a smear within six months of returning a self-sample.
There was substantial variation among general practices in the proportion of consulting women who were offered kits (ranging between 11 and 36%) and the proportion offered kits who returned a self-sample (30–74%). However, the proportion of eligible women who returned a sample increased with increasing proportion invited (see Figure 2 correlation coefficient r = 0.8, p = 0.04), suggesting that if more women are offered kits, more would return a self-sample. Similarly, there was considerable variation within general practices in offering SS kits. Some clinicians offered none throughout the study, whereas at three practices a single general practitioner offered more than half of all kits offered at the practice. The majority (74.8%) of kits were offered by a doctor (rather than a nurse or health care assistant), although again this varied considerably among practices (range 40–96%). Based on informal feedback from general practices, offering SS kits did not substantially increase consultation time, and was generally acceptable to general practitioners, nurses, and health care assistants.
Proportion of eligible women who returned a sample vs the proportion of eligible women who were invited (i.e. offered kits).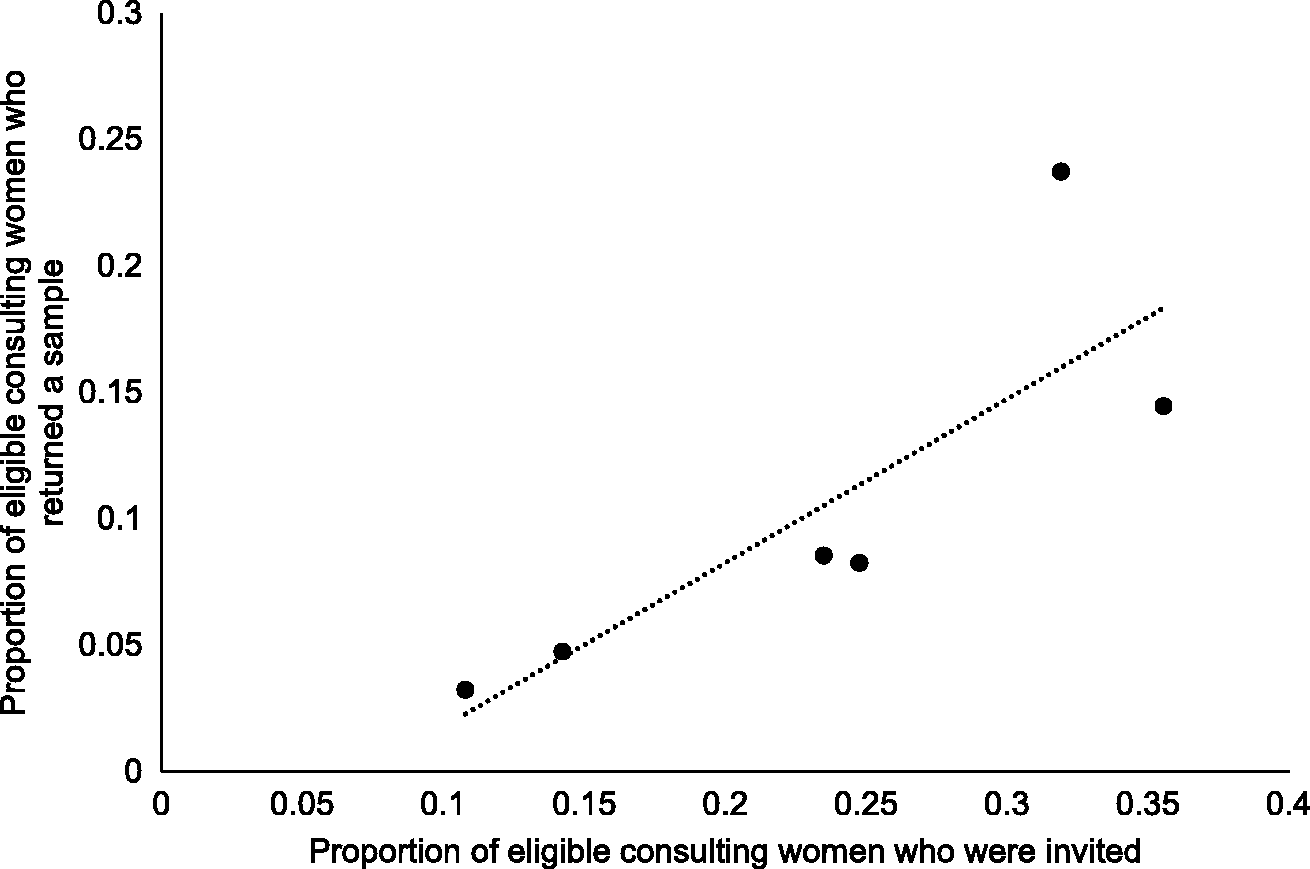
Discussion
Main findings
Targeted offering of SS to cervical screening non-attenders opportunistically in primary care is feasible and acceptable. The potential impact of this approach on screening coverage may be less than expected, as 11% of women who tested HPV negative came for cytology testing in the next six months. However, it may be that offering SS prompts women to re-engage with the cervical screening programme.
The proportion of women offered kits varied substantially both between and within general practices. We were unable to disentangle precise reasons but anecdotally some general practitioners reported having limited time in consultation (particularly if women had complicated or multiple problems) or that they saw no or very few eligible women (usually due to sub-specialty). Some expressed concern that the test was inferior to cervical cytology, that test uptake did not contribute to the annual reward and incentive programme (Quality of Framework, QoF), and some believed that women would eventually come for a smear. The fact that more samples were returned when more kits were offered indicates that there is scope to further increase uptake, and identified the need to motivate individual general practitioners to participate in this intervention. This, taken together with the positive relationship between the proportion of women who returned a sample and the proportion who were invited, implies that differences in uptake were largely determined by individual general practitioners rather than characteristics of the women. Offering kits using this targeted approach seemed to be general practitioner-led. A likely explanation is that a high proportion of nurse consultations will be for cervical screening.
Strengths and limitations
To our knowledge, this is the first study to examine targeted offering of SS for HPV testing in primary care. A key strength of the study is that the design closely resembled current clinical practice, therefore the findings will be informative for future studies and for implementation of the strategy into the screening programme.
A major limitation of the study was that SS was not included as part of the national screening programme, and therefore did not count towards coverage or change the women’s screening status. This was often highlighted as being a disincentive for offering SS, because general practice payments (QoF) are linked to coverage, and because it suggested that SS was inferior to cytology. A further limitation was that we were unable to calculate the exact number of cervical screening non-attenders over the study period. Also, we relied on cytology data in general practice records to identify eligible women, but cervical screening status is not always up to date (particularly for women newly registered at the general practice in the past year). Participating clinicians were asked to confirm eligibility before offering kits, but it is possible that some ineligible women may have been included. Furthermore, over half (59%) of women in the study had been registered at the practice for over one year. The practices in our study opted to take part, and most were from a single area in London, so may not be representative of the rest of England. Indeed, we purposefully targeted some practices because they had low screening coverage; however, we did not find a relationship between invitation rates and coverage (range 59.5–70.6%).
Interpretation in light of other studies
The proportion of women testing HPV positive who attended follow-up within three months was similar to that in a previous UK SS study 11 (56% vs 59%, respectively). However, by the end of our study, this figure was in line with what has been reported in most other studies (81–90%).3,4,6,8,17 Heterogeneity between centres has also been reported in an Italian trial, 10 which suggested that this is driven by underlying logistical and organizational differences. Similarly, in our study the differences in self-sample return rates could relate to whether or not women were encouraged to collect their sample in clinic. Return rates for vaginal SS for chlamydia testing for kits taken home are known to be low. 18 Unfortunately, accurate information on whether or not samples were taken in clinic or at home was not available for our study. The return rates in our study are not directly comparable with those in the two published UK studies, both population-based, which posted SS kits to cervical screening non-attenders 6%12 and 8%11 returned a self-sample). However, it seems reasonable to conclude that we achieved very similar return rates with much fewer wasted kits.
Implications
Cervical screening in England is set to undergo major changes in the coming years, which are likely to include a switch to primary HPV screening. It seems almost inevitable that, given its acceptability, ease of use, and advances in test performance, 2 some form of SS will also be incorporated for cervical screening non-attenders.19–21 Sending SS kits directly to screening non-attenders appears to work well in studies in other countries, but in the UK the optimal approach is likely to be multi-faceted. Targeted offering of SS kits, even in a busy general practice setting, seems to be possible. Using this approach could help minimize costs by reducing kit wastage (two-thirds of kits given out were returned). Other factors requiring further consideration include the possible impact of introducing SS on screening behaviour of women who currently attend screening regularly, 22 and whether there should be a higher threshold for triage for women testing HPV positive on a self-sample. In the future, the uptake of SS could further increase as HPV testing becomes more widely recognized by the public and primary care givers as an effective test for cervical screening. The high diagnostic yield of invasive cancers observed in this study suggests that the women targeted were from a very high risk group, and that SS is valuable in non-attenders. There were fewer than expected CIN2/3 lesions identified, but this may have been due to small numbers (i.e. a chance finding).
Conclusion
Targeted offering of SS to cervical screening non-attenders when they present to general practice primary care is acceptable and feasible. Further research is needed to understand the determinants of uptake by women and by primary care health professionals. The next logical step in the UK is to carry out a large trial or pilot implementation study, which we are currently planning, to assess the impact of SS on coverage and cost effectiveness.
Study registration
ISRCTN: 35286331; NIHR Study ID: 15712
Footnotes
Acknowledgements
We thank all the general practices and women who took part in the study, in particular Ruth Waring (practice manager Blithehale Medical Centre), Sue Neville (Neaman Practice), Dee Holland (Amersham Vale Training Practice), Khurram Shahzad (Hoxton Surgery), Helen Snowden (Chrisp Street Health Centre), Tracy Crossfield (Kingfisher Medical Centre), Sally Knapp (Amersham Vale Training practice) and Christopher Ward (Blithehale Medical Practice). We give special thanks to Jayne Callaghan from the Clinical Effectiveness Group at Queen Mary University for helping to set up the searches and alerts in EMIS web; Preventx Limited for providing HPV testing and self-sampling kits, Mohammed Khan (IT manager at Hoxton Surgery) and Mark Cronin (IT manager at Chrisp Street Health Centre) for additional EMIS support; and to Geeja Srilal, Keziban Rehman and Katie Atkins-Guays for laboratory support.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Preventx Limited provided HPV testing and self-sampling kits for the study free of charge. The authors have no other conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Cancer Research UK (grant no. C8162/A16892).
Ethical approval
Ethical approval for the study was granted by the NRES Committee South East Coast – Brighton and Sussex (13/LO/1441).
