Abstract
Objective
To compute a ratio between the estimated numbers of lives saved from breast cancer death and the number of women diagnosed with a breast cancer that never would have been diagnosed during the woman’s lifetime had she not attended screening (epidemiologic over-diagnosis) in the Norwegian Breast Cancer Screening Program.
Methods
The Norwegian Breast Cancer Screening Program invites women aged 50–69 to biennial mammographic screening. Results from published studies using individual level data from the programme for estimating breast cancer mortality and epidemiologic over-diagnosis comprised the basis for the ratio. The mortality reduction varied from 36.8% to 43% among screened women, while estimates on epidemiologic over-diagnosis ranged from 7% to 19.6%. We computed the average estimates for both values. The benefit–detriment ratio, number of lives saved, and number of women over-diagnosed were computed for different scenarios of reduction in breast cancer mortality and epidemiologic over-diagnosis.
Results
For every 10,000 biennially screened women, followed until age 79, we estimated that 53–61 (average 57) women were saved from breast cancer death, and 45–126 (average 82) were over-diagnosed. The benefit–detriment ratio using average estimates was 1:1.4, indicating that the programme saved about one life per 1–2 women with epidemiologic over-diagnosis.
Conclusion
The benefit–detriment ratio estimates of the Norwegian Breast Cancer Screening Program, expressed as lives saved from breast cancer death and epidemiologic over-diagnosis, should be interpreted with care due to substantial uncertainties in the estimates, and the differences in the scale of values of the events compared.
Keywords
Introduction
The balance between benefits and detriments of mammographic screening is a subject of debate.1–3 Recently, the International Agency on Cancer Research stated that there is sufficient evidence of reduced breast cancer mortality, both in randomized controlled trials and in observational studies. 4 Other benefits of mammographic screening include less aggressive treatment, with subsequently less serious morbidity, due to detection of the cancer in an early stage. On the other hand, studies have proved an increased incidence of breast cancer after implementation of screening programmes, referred to as excess incidence due to lead time. 5 The increased incidence is expected and is a prerequisite for a successful screening programme but involves an unknown proportion of breast cancer that presumably never would have been diagnosed during the woman’s lifetime had she not attended screening, so-called epidemiologic over-diagnosis. Overtreatment is a possible consequence of detection of slow-growing tumours. Other detriments of mammographic screening include false-positive and false-negative screening results. 4
The organization of mammographic screening differs substantially between programmes and countries. Various approaches are thus required to estimate early performance measures and aspects related to benefits and detriments. In addition, study design, age groups, follow-up time, tumour types (ductal carcinoma in situ (DCIS) and invasive cancer), and availability of individualized data are all important for the outcome. Studies presenting estimates on the benefits and detriments differ greatly, even when derived seemingly from the same data. A study by Duffy et al. 6 adjusted the estimates of benefits and detriments to a common screening and follow-up scenario and found a fourfold range between studies in the absolute benefit from screening in contrast to the 20-fold range found in the unadjusted estimates.
A benefit–detriment ratio of mammographic screening is usually aimed at describing the relationship between the number of lives saved from breast cancer death and the number of epidemiologic over-diagnosed cases. The information is targeted on the invited women and the population in general. However, the implications and consequences of these two events are substantially different, and comparison of the two events should be considered with care. Paci 7 used estimates from the EURO-screen group and found the benefit–detriment ratio to be two to one in favour of breast cancer mortality reduction versus epidemiologic over-diagnosis, meaning that the organized mammographic screening in Europe saved two lives for each woman with epidemiologic over-diagnosis among those participating in the programmes. This is in accordance with another study based on data from service screening in Italy. 8 On the other hand, the Marmot report, 9 using results from randomized controlled trials based on invited women (intention-to-treat), estimated that one breast cancer death was prevented for every three women with epidemiologic over-diagnosis. As part of the external evaluation of the Norwegian Breast Cancer Screening Program (NBCSP), it was suggested that five women were over-diagnosed for each woman saved from breast cancer death. 10
Despite the fact that there are published results on mortality and epidemiologic over-diagnosis, there is no published benefit–detriment ratio for the NBCSP of today. We aimed to take advantage of published results on mortality and epidemiologic over-diagnosis to estimate the ratio, including the uncertainties, for women invited and for those participating in the NBCSP.
Methods
The NBCSP started as a pilot project in 4 out of 19 counties in 1996. 11 The programme invites women aged 50–69 to biennial mammographic screening. The target population was nearly 600,000 women by January 2015. Results of early performance measures from the programme have been published regularly since start-up,11–13 while estimates for mortality14–16 and epidemiologic over-diagnosis10,17–20 have been published in recent years. Our benefit–detriment ratio is based on published results using individual level data from the NBCSP,10,14,15,17 i.e., results from studies using aggregated data or simulations were not included. No approval from the ethical committee was needed as the information used was already published. We adapted the approach presented by the EURO-screen group in 2012 to estimate the number of lives saved and the number of epidemiologic over-diagnosed women in our setting. 7 The ratio relates to a hypothetical cohort of women aged 50–51 who were biennially screened until age 69, and then followed for 10 years, until age 79.
Estimates on mortality reduction and epidemiologic over-diagnosis
Benefit–detriment estimates of reduction in breast cancer mortality and epidemiologic over-diagnosis from published studies using individual level data from the Norwegian Breast Cancer Screening Program.
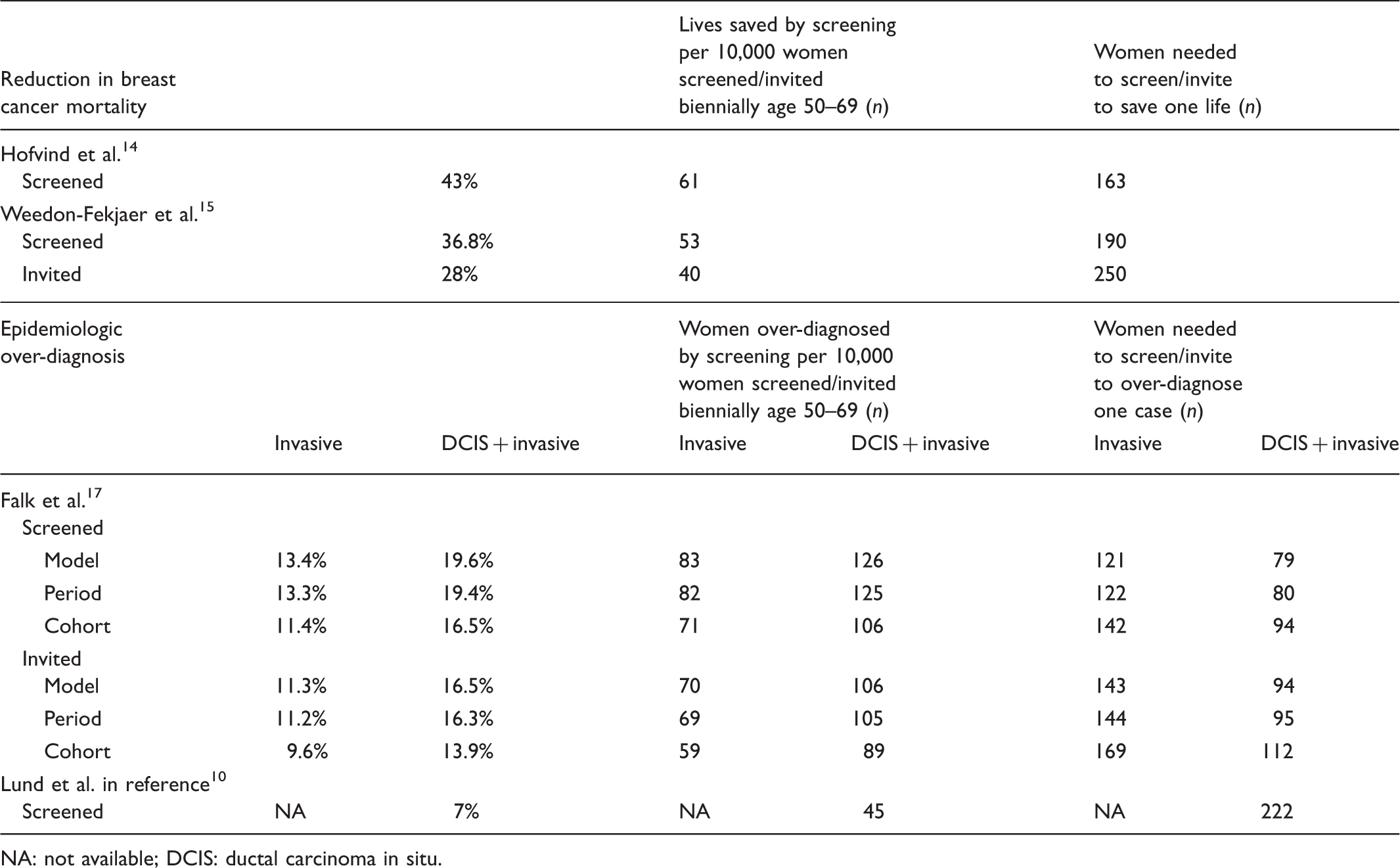
NA: not available; DCIS: ductal carcinoma in situ.
To compute the estimates on epidemiologic over-diagnosis, we used the results from Falk et al. 17 and Lund et al. given in the Research-based evaluation of the NBCSP performed by the Research Council of Norway 10 (Table 1). The study by Falk et al. 17 showed results for DCIS and invasive breast cancer, and invasive cancer alone. Estimates were given for three different methods, varying from 16.5% (95% CI: 9.1%–23.9%) to 19.6% (95% CI: 12.1%–27.1%) for ever versus never screened women diagnosed with DCIS and invasive cancer. The results given by Lund et al. in the report from the Research Council of Norway 10 was also based on ever versus never screened women. They estimated the proportion of epidemiologic over-diagnoses to be 7% (95% CI: Not available–Not available) for DCIS and invasive cancer. Based on these two studies, we computed a weighted average rate of epidemiologic over-diagnosis of DCIS and invasive cancers among women ever screened to be 12.8% ((((16.5 + 19.4 + 19.6)/3) + 7)/2). Both studies express the extent of epidemiologic over-diagnosis as a proportion of breast cancers detected in a situation without screening among women followed from the start of screening and throughout life. This approach is referred to as measure A in the Marmot report. 9
Number of lives saved and epidemiologic over-diagnosis
The cumulative risk of having breast cancer in the absence of screening was defined as the probability that a woman was diagnosed with DCIS or invasive cancer at age 50–79 before the screening programme started. We defined the cumulative risk of breast cancer mortality in the absence of screening as the probability that a woman will die from the disease at age 50–79. To obtain these risks, we used the age-specific incidence and mortality rates for Norwegian women aged 50–79 in 1995 (i.e., before start of the screening programme) obtained from NORDCAN (http://www-dep.iarc.fr/nordcan.htm). The cumulative incidence of invasive breast cancer was 6.18%, while the cumulative risk of breast cancer death was 2.27%. We assumed 4% increase in the cumulative breast cancer incidence due to DCIS. 21
Based on the age-specific incidence rates of invasive breast cancer in the absence of screening, the expected cumulative number of breast cancers per 10,000 women aged 50–79 was estimated to be 618 (0.0618 × 10,000) for invasive cancer and 644 (0.0618/(1 − 0.04) × 10,000) for DCIS and invasive cancer. We estimated 227 breast cancer deaths among 10,000 women (0.0227 × 10,000) aged 50–79 in the period before the programme started, 121 among women aged 50–69 and 106 among those aged 70–79. Based on data from the Cancer Registry of Norway, 25% of breast cancer deaths occurring in the 50–69 age group were diagnosed before age 50, and 51% of breast cancer deaths in women aged 70–79 were diagnosed after age 70, before the screening programme started (1981–1995). 22 The number of breast cancer deaths in women aged 50–79 in which screening may have had a protective effect was thus 143 of 227: 121 × (1 − 0.25) + 106 × (1 − 0.51) = 143. As an example, the average estimate of 39.9% reduction in breast cancer mortality among those screened led to 57 lives saved from breast cancer death due to screening among the 143 women in whom screening might have an effect. The total breast cancer mortality reduction from age 50–79 for a screened population was thus 25% (57/227).
We estimated the number of lives saved and epidemiologic over-diagnosis among 10,000 women (screened or invited) aged 50–69 and further followed for 10 years until age 79. We targeted our results to the incidence of DCIS and invasive cancer altogether, although estimates of invasive cancers alone are presented in the tables. Additionally, we estimated the number of lives saved from breast cancer death, number of epidemiologic over-diagnosed women, and benefit–detriment ratio under different hypothetical scenarios of reduction in breast cancer mortality (20%, 30% and 40%) and proportions of epidemiologic over-diagnosis (5%, 10%, 20% and 30%).
We performed three sensitivity analyses. First, we used cumulative mortality and incidence rates for 1985 instead of 1995, to avoid the possible effect of opportunistic screening prior to the start-up of the NBCSP. Second, because pre-screening rates of incidence and mortality may have changed over time 23 independently of screening, we assumed a 10%, 20% and 30% reduced cumulative incidence, and 10%, 20% and 30% excess cumulative mortality in the 1995 rates. Finally, we assumed a 10% increase instead of 4% in cumulative incidence of breast cancer due to DCIS.
Results
The estimated number of lives saved from breast cancer death per 10,000 women aged 50, who were screened biennially 10 times and further followed until age 79 varied from 53 to 61 (Table 1). The estimated number of women needed to be screened to save one life from breast cancer death varied from 163 to 190. The average estimate of mortality reduction amongst the screened women (39.9%) counted for 57 lives saved from breast cancer death, meaning that 57 fewer women will die from the disease due to participation in screening, 170 instead of 227. In the same time period and in the same cohort of women, 644 were expected to be diagnosed with DCIS or invasive breast cancer without a screening programme. The average estimate of epidemiologic over-diagnosis amongst the screened women (12.8%) counted for 82 additional cases, ranging from 45 to 126. These average numbers gave a benefit–detriment ratio of 0.7 (57/82); for every woman saved from breast cancer death, 1–2 (1.4) women will have an epidemiologic over-diagnosis.
Number of lives saved from breast cancer death, epidemiologic over-diagnosed cases and benefit–detriment ratio by different hypothetical scenarios for women screened.
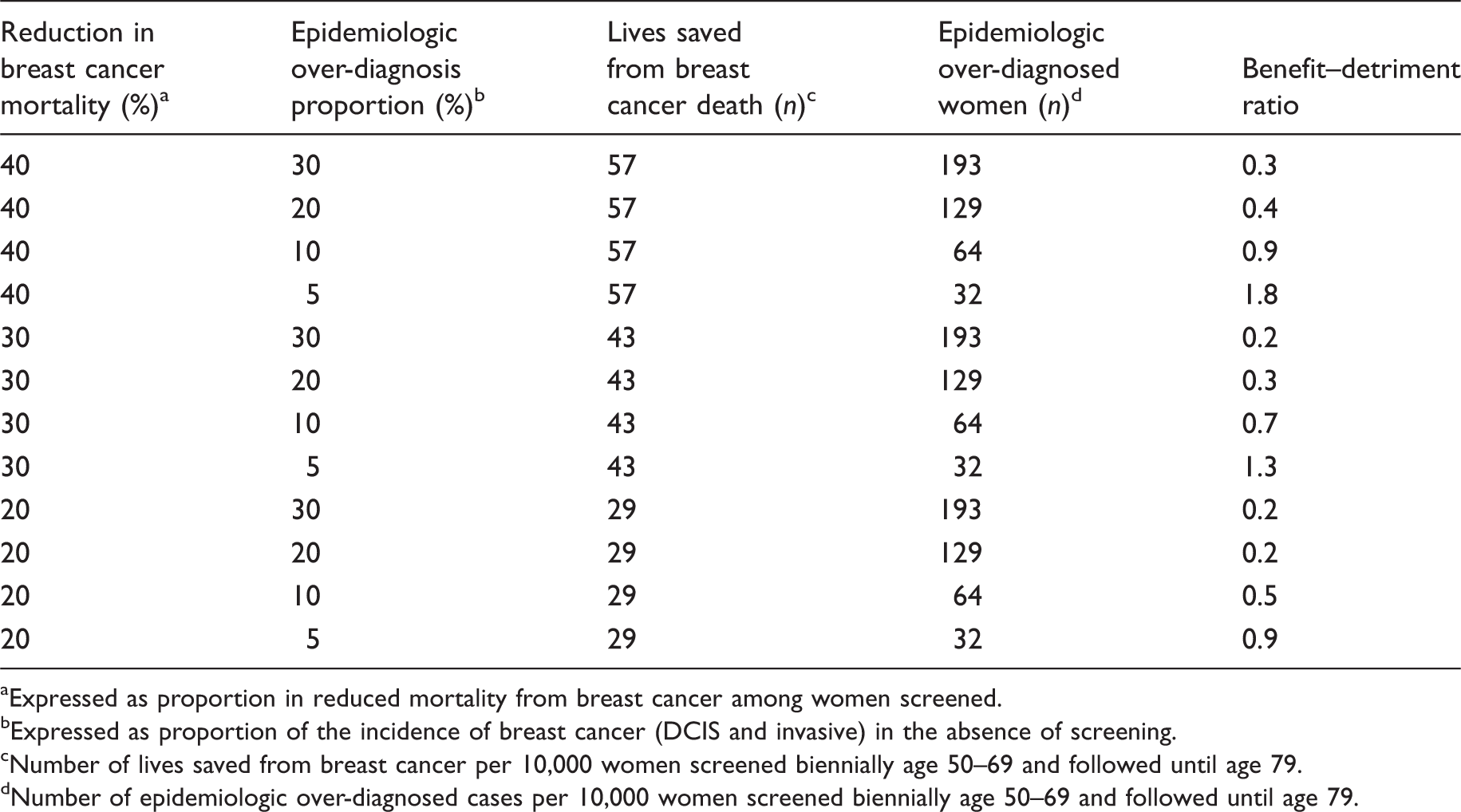
Expressed as proportion in reduced mortality from breast cancer among women screened.
Expressed as proportion of the incidence of breast cancer (DCIS and invasive) in the absence of screening.
Number of lives saved from breast cancer per 10,000 women screened biennially age 50–69 and followed until age 79.
Number of epidemiologic over-diagnosed cases per 10,000 women screened biennially age 50–69 and followed until age 79.
For invited women, 40 lives were saved from breast cancer death, and 250 women were needed to be invited to save one life from breast cancer death (Table 1). The estimated number of epidemiologic over-diagnoses among the invited women ranged from 89 to 106. The number needed to be invited to state an epidemiologic over-diagnose on one woman ranged from 94 to 112 for DCIS and invasive cancer. The numbers result in a benefit–detriment ratio of 0.5 (40/89) and 0.38 (40/106), which means that for every women saved from breast cancer death, 2–3 are diagnosed with epidemiologic over-diagnosis.
The sensitivity analyses, using the cumulative mortality and incidence rates from 1985 resulted in 223 breast cancer deaths and a cumulative 578 breast cancers (DCIS and invasive) per 10,000 women aged 50–79. Using the average reduction in breast cancer mortality rate (39.9%) among screened women and the average epidemiologic over-diagnosis rate (12.8%) resulted in 56 lives saved from breast cancer death and 74 women with epidemiologic over-diagnosis (ratio 0.8, 56/76), compared with a ratio of 0.7 (57/82) when using the cumulative mortality and incidence rates from 1995. Further, we assumed a 10%, 20% and 30% lower cumulative incidence, and 10%, 20% and 30% higher cumulative mortality in 1995, due to time trends, compared with a hypothetical contemporary reference population of non-screened women. Using the average reduction in breast cancer mortality and epidemiologic over-diagnosis among those screened women resulted in 51 lives saved and 87 women with epidemiologic over-diagnosis under the 10% assumption; 46 lives saved and 93 women over-diagnosed under the 20% assumption and 40 versus 98 under the 30% assumption, which lead to benefit–detriment ratios of 0.6, 0.5 and 0.4, respectively. If we assume the pre-screening cumulative incidence in 1995 to be 10% lower than a contemporary reference population, and the cumulative breast cancer mortality to be 10% higher, our estimates showed that 1–2 (1.7) women were epidemiologically over-diagnosed for every women saved from breast cancer death. If we assume a 30% lower cumulative incidence and 30% higher mortality, we estimated that 2–3 (2.5) women were over-diagnosed for every woman saved from breast cancer death. The third sensitivity analysis assumed a 10% increase in incidence due to DCIS. Using the average proportion of epidemiologic over-diagnosed DCIS and invasive cancer (12.8%) resulted in 88 cases (instead of 82) per 10,000 women screened, which translates to 114 (instead of 121) women needed to be screened to state an epidemiologic over-diagnose on one woman.
Discussion
For women screened in the NBCSP, the estimated benefit–detriment ratio indicates that 1–2 women are epidemiologically over-diagnosed for each woman saved from breast cancer death. However, the uncertainties in the estimates are substantial, and the significance of the two events are considerably different.
In general, a benefit–detriment ratio requires equivalent parameters for comparison. Lives saved from breast cancer death and epidemiologically over-diagnosed cases are used without any considerations on the comparability of the two events. The value of a life saved versus an epidemiologically over-diagnosed case, or the consequences of the two different events, are obviously of different magnitude. Further, estimates of epidemiologic over-diagnosis are based on mathematical models, without considerations of the biological aspects, none of these estimates can be applied to an individual level, and the risk of breast cancer death is lower compared with the risk of being diagnosed with breast cancer. Moreover, breast cancer is a heterogeneous disease, and the women diagnosed are thus offered substantial variation in treatment and follow-up and are expected to have different future lives as breast cancer survivors. All these factors indicate that benefit–detriment ratios should be considered and communicated with extensive care.
Our results, based on average mortality reduction and epidemiologic over-diagnosis, are less in favour of screening compared with the results given by the EURO-screen group, 7 but more in favour of screening as the results from the external evaluation of the NBCSP headed by the Research Council of Norway, 10 and the results in the Marmot report. 9 The average number of lives saved per 10,000 women screened was 57 in our study, 70–90 in the EURO-screen group, and 55 in the Marmot report, which equals a 1.6 variation-ratio. The number of epidemiologic over-diagnoses per 10,000 screened women was 82, 40 and 161 (129/0.8), respectively, in our, the EURO-screen, and the Marmot study, which equals a fourfold variation-ratio. Our study and the EURO-screen study deal with results from organized screening programmes and effectiveness and provide over-diagnosis estimates and number of lives saved related to a situation without screening among women followed from the start of screening and throughout life (Measure A in the Marmot report). The estimates in the Marmot report deal with efficacy and are based on results from randomized controlled trials performed in the 1980s.
Based on follow-up of three randomized controlled trials, about 19% of the cancers diagnosed during the period the women received invitations to screening were suggested to represent over-diagnosed cases (measure C). An optimal method to estimate the rate of epidemiologic over-diagnosis is not yet established, and neither randomized controlled trials nor organized screening programmes are designed to answer this issue. In addition, the variation in the methods used to express the epidemiological over-diagnosis reduces the comparability across the published studies, and our results focus solely on lives saved from breast cancer death and epidemiologic over-diagnosis. Other benefits of mammographic screening, such as the less aggressive treatment and less serious morbidity due to detection of early stage breast cancer, should be considered, as well as other detriments, such as false-positive and false-negative screening results.
Our use of a crude benefit–detriment ratio may be misleading, as the consequences of a breast cancer death and an epidemiologic over-diagnosis are of different magnitude. Quality-adjusted live-years (QALY’s) gained from prevention of breast cancer deaths against QALY’s lost due to additional time spent with a diagnosis of breast cancer might be a better approach to communicate the benefit–detriment ratio. Time spent with an epidemiologic over-diagnosed breast cancer is expected to incur less loss of quality of life compared with non-over-diagnosed breast cancer cases, as over-diagnosed cancers are likely to have more favourable prognostic tumour characteristics, less aggressive treatment, and therefore less serious morbidity.
We estimated the benefit–detriment ratio for women participating in a screening programme. Mortality estimates for ever-screened women were combined with proportions of epidemiologic over-diagnosis for women, who actually were screened. Breast cancer mortality is considered to be biased in favour of the screened women, due to self-selection. On the other hand, the reduction in breast cancer mortality among those who actually participate, and have a screen-detected or an interval cancer, is assumed to be higher compared with those ever-screened. The assumption might thus indicate an underestimation of the benefit in the ratio.
The methodological issues regarding the incidence in the reference population will always represent uncertainties, both in studies on breast cancer mortality and epidemiologic over-diagnosis, as it is unfeasible to find a clean and updated reference population of non-screened women. The benefit–detriment ratio of 1:5 published by the Research Council of Norway was based on results from one study only for mortality estimates. The study used a simulation model with data from 2012 to estimate the number of lives saved from breast cancer death due to screening. 10 Our sensitivity analyses, assuming a lower cumulative incidence and higher cumulative reduction in breast cancer mortality in the reference population due to time trends, showed a moderate variation in the number of lives saved from breast cancer death and epidemiologic over-diagnosed women compared with the baseline reference population for 1995. The rate of epidemiologic over-diagnosis in the study by Falk et al. 17 is likely to be somewhat overestimated, due to lack of compensatory adjustment for residual lead time. The wide confidence intervals confirm the uncertainties of the estimates. Estimating epidemiologic over-diagnosis requires at least 10 years of follow-up after the women leave the screening programme.7,24 As the screening programme in Norway became nationwide in 2005, we have to wait almost until 2035 to have information about a complete cohort of women 10 years after they leave the programme. The other study on epidemiologic over-diagnosis included in our benefit–detriment ratio reported not statistically significant results for DCIS and invasive cancer (7%, 95% CI: Not available–Not available). 10 This underscores the substantial uncertainties in our estimations of the benefit–detriment ratio. We did not evaluate the quality of the studies used for the estimations of the benefit–detriment ratio, which represents a limitation of our study. However, we excluded results from studies using aggregated data or ecological studies, due to methodological limitations. 25 The estimates in these studies ranged from 7% to 10% for mortality reduction16,26 and from 15% to 50% for over-diagnosis.18,19,24 Including these estimates would have limited the validity of our study and resulted in even higher uncertainties. Using estimates from studies with different methodological approaches would have reduced the accuracy of our results.
The higher rate of epidemiologic over-diagnosis in Norway compared with other countries could be due to short follow-up time and increased use of hormone therapy in parallel to the implementation of the screening programme.27,28 Use of hormone therapy dropped in Norway, as in most western countries, after the publication of the Womens Health Initiative study in 2002. 29 In addition, use of opportunistic screening is not reported in Norway. We do not know the extent of such activity, but it is assumed to be substantial, particularly before the screening programme started.27,30 Lack of the information described above hamper proper evaluation of the NBCSP.
The impact of opportunistic screening prior to the start of the NBCSP in 1995 is considered to be residual in our reference population. Results from the sensitivity analyses, using 1985 as the reference, showed a reduced impact on the estimates of lives saved from breast cancer death and epidemiologic over-diagnosed women. Similarly, the impact of the proportion of DCIS on the overall incidence of breast cancer prior to implementation of the screening programme was negligible. Assuming a hypothetical proportion of DCIS of 10% resulted in a non-significant increase in the number of over-diagnosed women.
Definitions of epidemiologic over-diagnosis vary, 5 and estimation of the event is a theoretical and mathematical exercise; the real number cannot be exactly identified. In addition, the biological aspect of epidemiologic over-diagnosis represents a challenge, as we really do not know how ‘slow-growing tumours’ will develop for the individual women if left untreated. The discussions about the extent of epidemiologic over-diagnosis will thus remain open until more knowledge about breast tumour progression is gained.
Conclusions
Our estimates suggest that one to two screened women are epidemiologically over-diagnosed for each woman saved from breast cancer death. The results should be interpreted with substantial care because of the uncertainties, both in the numbers used in the estimations, and the final outcome. In addition, the two events include substantially different implications and consequences for the actual women involved.
Footnotes
Declaration of conflicting interests
Hofvind is the head of the Norwegian Breast Cancer Screening program.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author(s) received no financial support for the research, authorship and/or publication of this article.
