Abstract
Objectives
To investigate breast cancer survival and risk of breast cancer death by detection mode (screen-detected, interval, and detected outside the screening programme), adjusting for prognostic and predictive tumour characteristics.
Methods
Information about detection mode, prognostic (age, tumour size, histologic grade, lymph node status) and predictive factors (molecular subtypes based on immunohistochemical analyses of hormone receptor status (estrogen and progesterone) and Her2 status) were available for 8344 women in Norway aged 50–69 at diagnosis of breast cancer, 2005–2011. A total of 255 breast cancer deaths were registered by the end of 2011. Kaplan-Meier method was used to estimate six years breast cancer specific survival and Cox proportional hazard model to estimate hazard ratio (HR) for breast cancer death by detection mode, adjusting for prognostic and predictive factors.
Results
Women with screen-detected cancer had favourable prognostic and predictive tumour characteristics compared with interval cancers and those detected outside the screening programme. The favourable characteristics were present for screen-detected cancers, also within the subtypes. Adjusted HR of dying from breast cancer was two times higher for women with symptomatic breast cancer (interval or outside the screening), using screen-detected tumours as the reference.
Conclusions
Detection mode is an independent prognostic factor for women diagnosed with breast cancer. Information on detection mode might be relevant for patient management to avoid overtreatment.
Introduction
Breast carcinoma is a progressive disease, the development of which can be arrested by screening.1–3 Asymptomatic screen-detected breast tumours are shown to have favourable prognostic and predictive characteristics2,4–7 and better outcome compared with women with symptomatic cancer (interval breast cancer and breast cancer diagnosed among women not invited or not attending screening programmes) in long-term survival analyses.2,5,6,8–15 The point at which tumour progression is arrested is thus crucial. Stage shift by early detection, including lead-time-, length- and over-detection bias, is argued to be the main reason for favourable survival. However, studies have shown that detection mode remains an independent prognostic factor after adjusting for disease stage, and that the above biases cannot fully explain the improved prognosis associated with screening.5,9,10,12–15 The survival benefit among women with screen-detected cancer may therefore relate to biological differences associated with hormonal receptor status, Her2 status, and Ki67 index.5,9,10,13,15–17
Microarray-based technology has provided insights on molecular heterogeneity, and identified clinically relevant molecular subtypes of breast cancer. 18 Different gene expression patterns among the subtypes reflect basic alterations in tumour biology and are associated with significant variation in treatment response and clinical outcome. Including information on molecular subtypes in survival analyses is one possible way to reduce lead-time bias.
Mortality rates are the preferred measure of effectiveness in organized screening programmes to avoid lead-time bias. However, the effect of detection mode on case survival is of considerable interest to clinicians treating cancer patients, and to identify the effect of screening for women diagnosed with interval cancer. The treatment plan for women with breast cancer has become personalized over recent decades, taking age, prognostic, and predictive tumour characteristics into consideration. However, only a few population-based studies have included information on subtypes and detection mode in survival analyses.5,9,13,15,17
To understand more about tumour progression and breast cancer biology we investigated breast cancer survival and risk of breast cancer related deaths by detection mode (screen-detected, interval cancer and breast cancer detected outside the screening programme), adjusting for prognostic and predictive tumour characteristics.
Methods
The Norwegian Breast Cancer Screening Program (NBSCP) started in 1996, expanded gradually and became nationwide in 2005. 19 The programme invites about 580,000 women aged 50–69 to biennial mammographic screening. The Cancer Registry of Norway is responsible for quality assurance and administration of the programme. Cancer reporting is mandatory by law in Norway, and the Cancer Registry incidence database is 99% complete for solid tumours, including breast cancer. 20 Information about vital status, date of emigration, date and cause of death is available from Statistics Norway, and is regularly linked to the incidence database by a unique 11-digit personal identification number assigned to all residents. We received an anonymized data file with information from the incidence database and the screening database, which included information about date of detection of breast cancer, date of breast cancer deaths, detection mode, prognostic and predictive tumor characteristics. No ethical committee approval was required.
Screen-detected cancer (SDC) was defined as breast cancer diagnosed as a result of a positive screening test within four months after screening in the NBCSP. Interval cancer (IC) was defined as breast cancer diagnosed after a negative screening examination, or after a positive screening test including a recall examination which turned out to be negative, within two years after the screening examination. Breast cancer detected outside the screening programme (OS) was defined as breast cancer diagnosed among women never invited, invited but did not attend, or women diagnosed with breast cancer more than 730 days after their last screening examination.
During the study period (2005–2011) 10,309 women aged 50–69 were diagnosed with primary invasive breast cancer. We excluded cases with unknown cause of death (n = 40), detection date after date of emigration (n = 64), no information about residing county (n = 1), stage IV cases (n = 321), and women with pT4 tumours (n = 174). This left 9709 women for subtyping by immunohistochemical analyses (IHC). Information about estrogen (ER) and/or progesterone (PR) receptor status, and Her2 status was used to classify the women into five subtypes: Luminal A (ER+/PR+/Her2-), Luminal B Her2- (ER+/PR-/Her2-), Luminal B Her2+ (ER+/PR±/Her2+), Her2+ (ER-/PR-/Her2+), and Triple Negative Breast Cancer (TNBC) (ER-/PR-/Her2-), as a modification of the recommendation from the St Gallen panel in 2013. 21 Hereafter, the terms “Luminal A-like”, “Luminal B-like Her2-”, “Luminal B-like Her2+”, Her2 positive and TNBC are used for the subtypes based on IHC assessment. Information on subtypes was available for 8344 tumours (86%, 8344/9709); 4835 cases were SDC, 1644 IC, and 1865 OS. There were 255 breast cancer deaths recorded for the study population at the end of 2011, 61 screen-detected, 81 interval cancers, and 107 outside screening.
Age, tumour size (≤20 mm and >20 mm), lymph node involvement (negative/positive), and histologic grade (I + II, and III) were defined as prognostic tumour characteristics. Tumour size and lymph node involvement were classified according to the TNM classification system, 22 and histologic grade was determined by the Nottingham criteria. 23 For the descriptive analyses, 95% confidence intervals (95% CI) and p-values were calculated to examine the differences between screen-detected cancer, interval cancer, and breast cancer detected outside of screening, within and between the subtypes.
To analyze breast cancer specific survival, women were followed from date of breast cancer diagnosis to date of breast cancer death, death due to other reasons, date of emigration, or end of follow-up (31 December 2011), whichever came first. Six years crude breast cancer survival, overall and stratified by detection mode, was estimated using the Kaplan-Meier method. Survival was calculated according to subtypes, and statistical differences between curves were assessed using log-rank tests.
Cox proportional hazards models were used to estimate the hazard ratio (HR) and 95% CI of breast cancer death associated with detection mode and subtypes, as well as age at diagnosis (continuous), tumour size, lymph node involvement, and histological grade. We included women with missing information in the analyses, by adjusting for missing information as a separate category. The results for those with missing values are not shown in the tables. Three sensitivity analyses were performed: a simple correction for lead-time bias (described by Duffy et al. 24 ), testing for interactions between subtypes and the covariates included in the Cox regression model (tumour size, histologic grade, and lymph node status), and Cox proportional hazard model for ever versus never screened women. We included women with at least one screening examination performed in the NBCSP in the “ever screened” group. No attendance registered in the programme was classified as “never screened”. There were 6982 women with breast cancer in the ever screened group, and 1362 in the never screened group. The number of breast cancer death was 187 and 68, in the ever screened and never screened groups, respectively.
Statistical significance was defined as 95% CI not overlapping and two-sided p-value < 0.05. SPSS, version 19 and R version 3.0.2 were used for the analyses.
Results
Distribution of prognostic tumour characteristics by detection mode (screen-detected, SDC; interval breast cancer, IC; and breast cancer detected outside the screening program, OS), stratified by subtype among women aged 50–69 at diagnosis in 2005–2011.
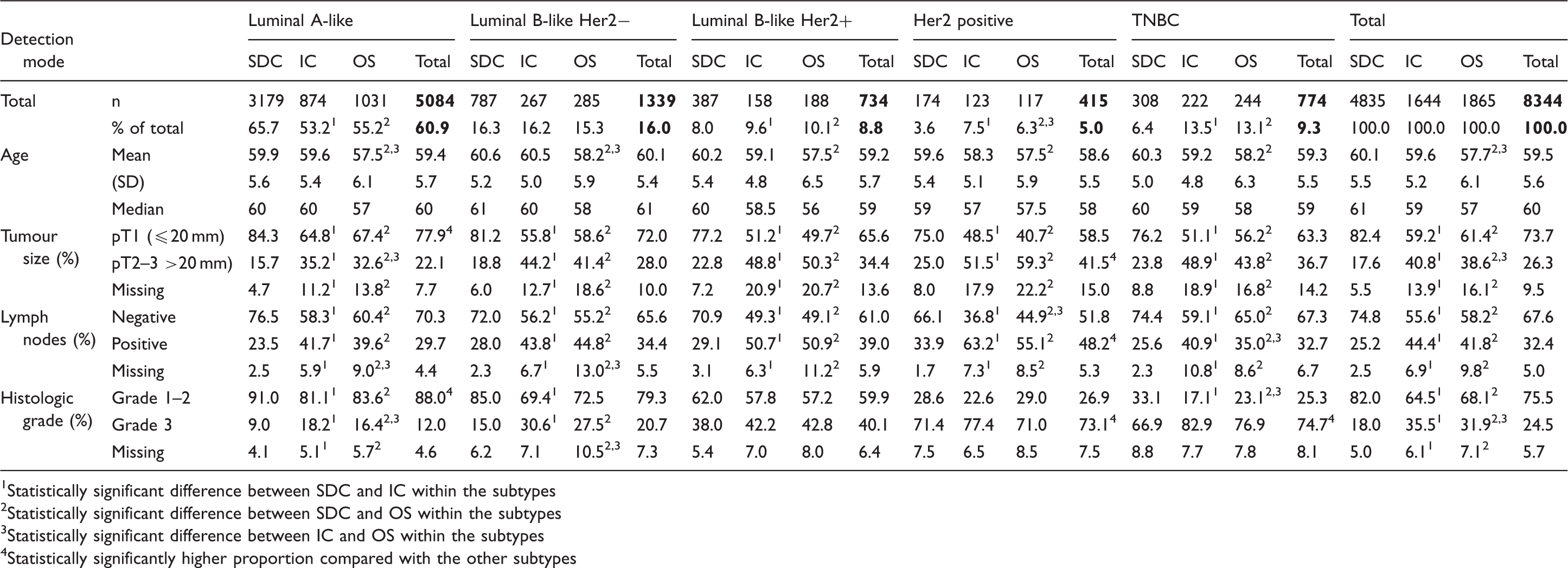
Statistically significant difference between SDC and IC within the subtypes
Statistically significant difference between SDC and OS within the subtypes
Statistically significant difference between IC and OS within the subtypes
Statistically significantly higher proportion compared with the other subtypes
Crude breast cancer-specific survival after six years of follow up was 97.1% (95% CI: 96.3–97.9), 89.9% (95% CI: 87.8–92.0), and 88.0% (95% CI: 85.8–90.3) for women with SDC, IC, and OS, respectively (Figure 1). Luminal A-like SDC had a significantly better six year survival (98.4%, 95% CI: 97.6–99.2) (Figure 2a) compared with IC (92.6%, 95% CI: 89.4–95.9) (Figure 2b) and OS (92.0%, 95% CI: 89.2–94.9) (Figure 2c).
Crude breast cancer-specific survival by detection mode in Norwegian women aged 50–69 at diagnosis in 2005–2011, by detection mode (screen-detected, SDC; interval breast cancer, IC; and breast cancer detected outside the screening programme, OS). Crude breast cancer-specific survival by subtypes among Norwegian women, aged 50–69 at diagnosis in 2005–2011 for (a) screen-detected, (b) interval breast cancer and (c) breast cancer detected outside the screening programme.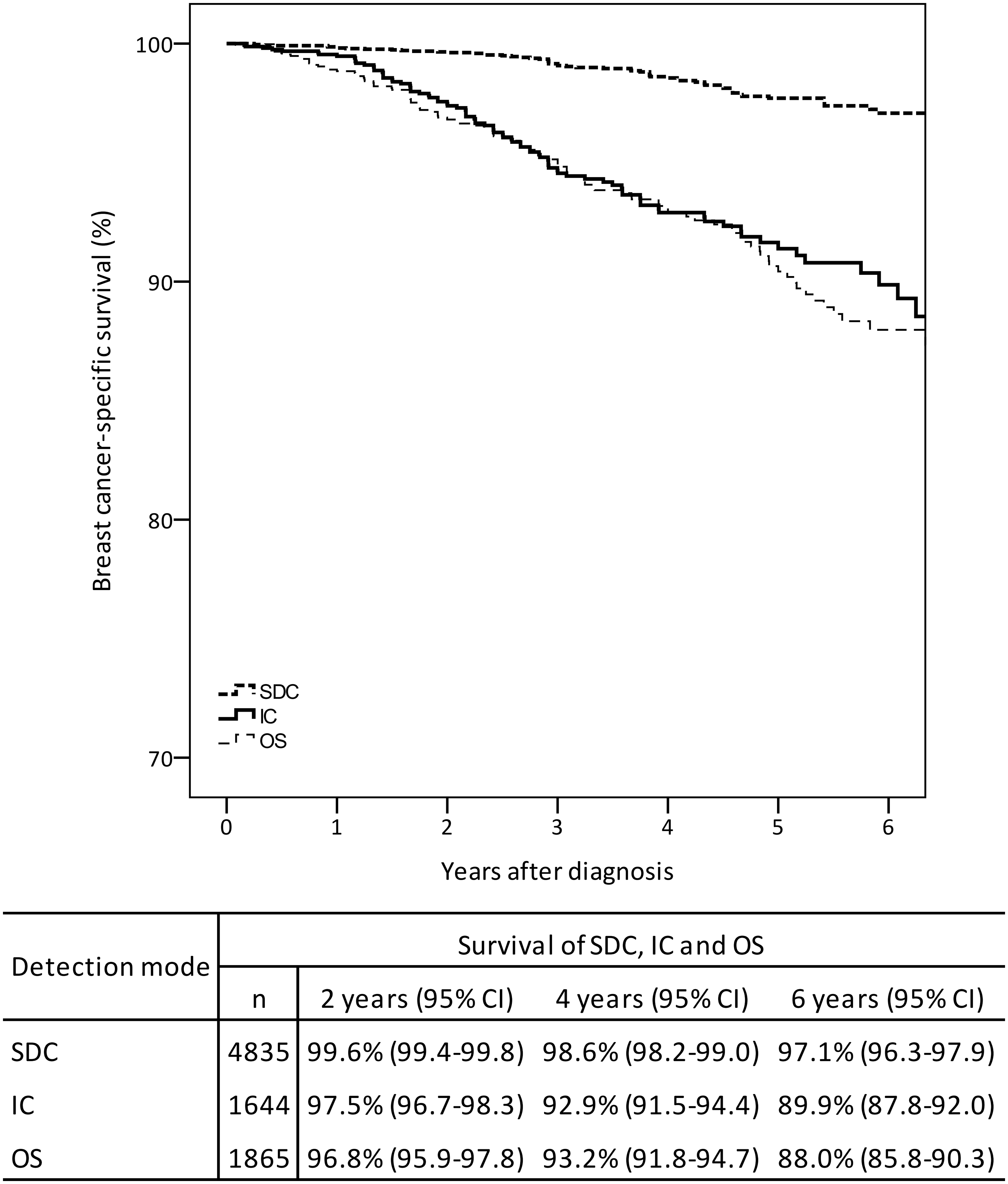
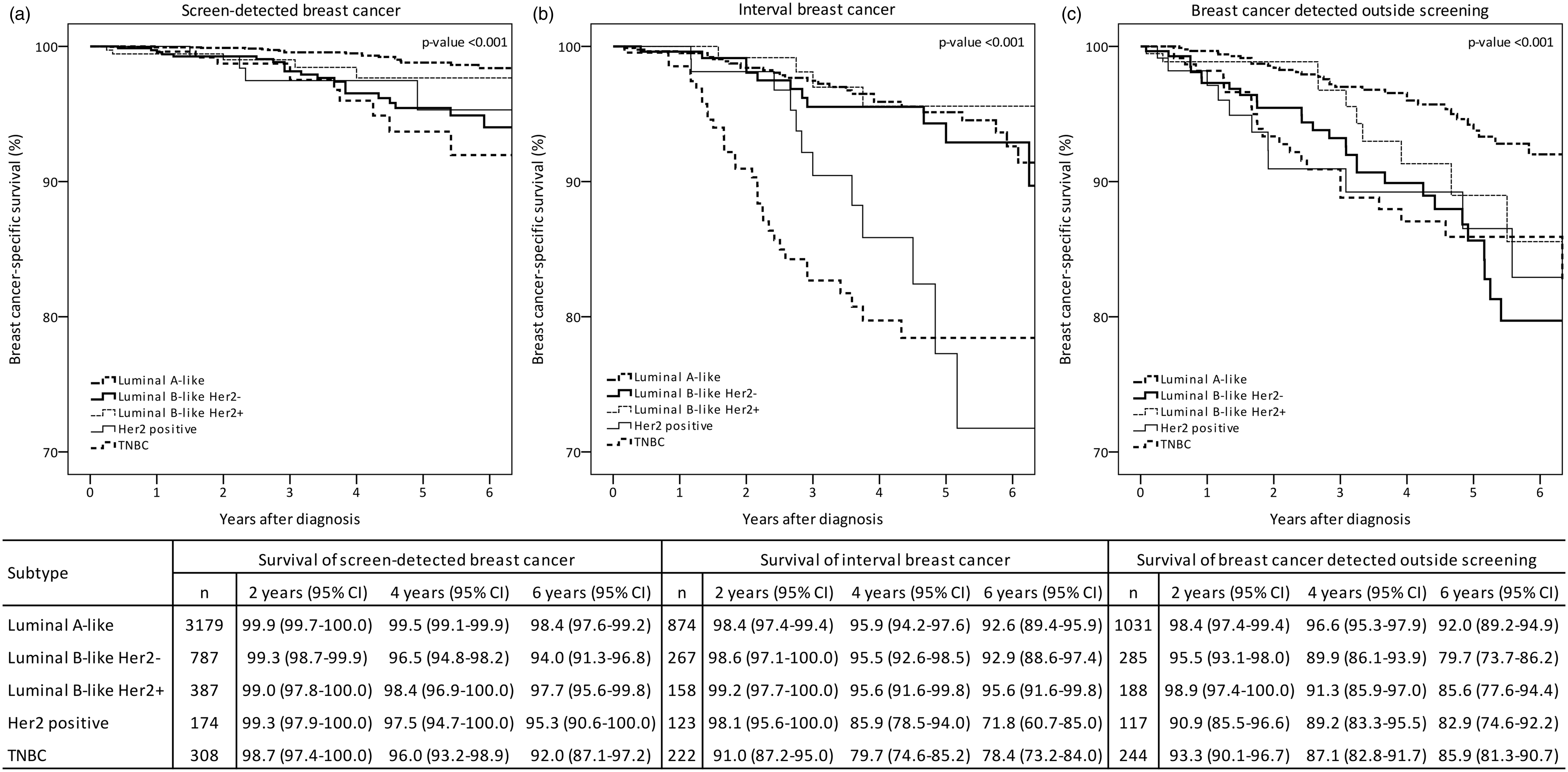
Crude (univariate) and adjusted (multivariate) Cox analyses for breast cancer death, analyses corrected for lead-time using the Duffy method, among Norwegian women aged 50–69 at diagnosis in 2005–2011, by detection mode (screen-detected, interval breast cancer, and breast cancer detected outside the screening program).
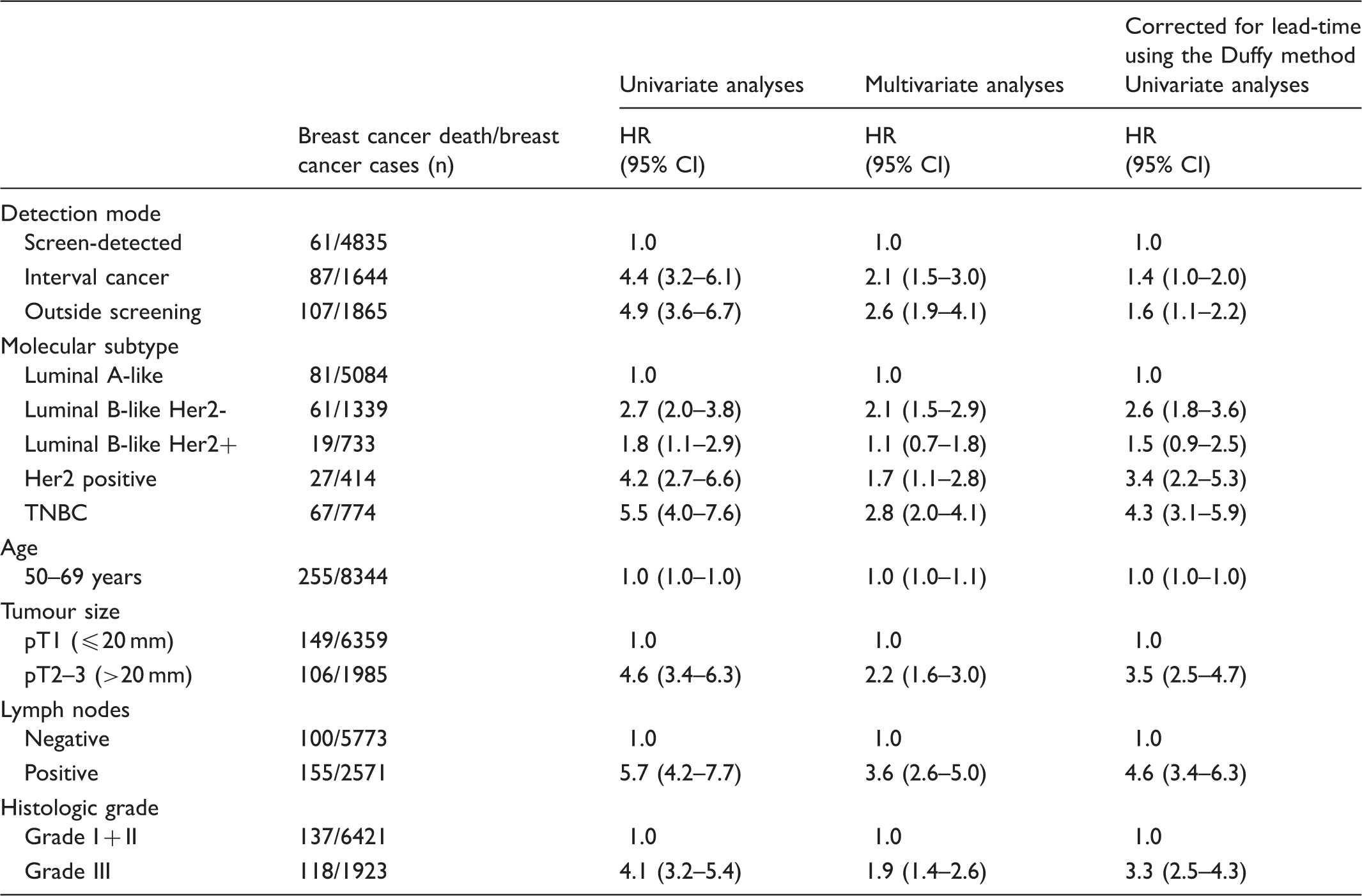
Crude (univariate) and adjusted (multivariate) Cox analyses of breast cancer death among Norwegian women aged 50–69 at diagnosis in 2005–2011, stratified by detection mode.
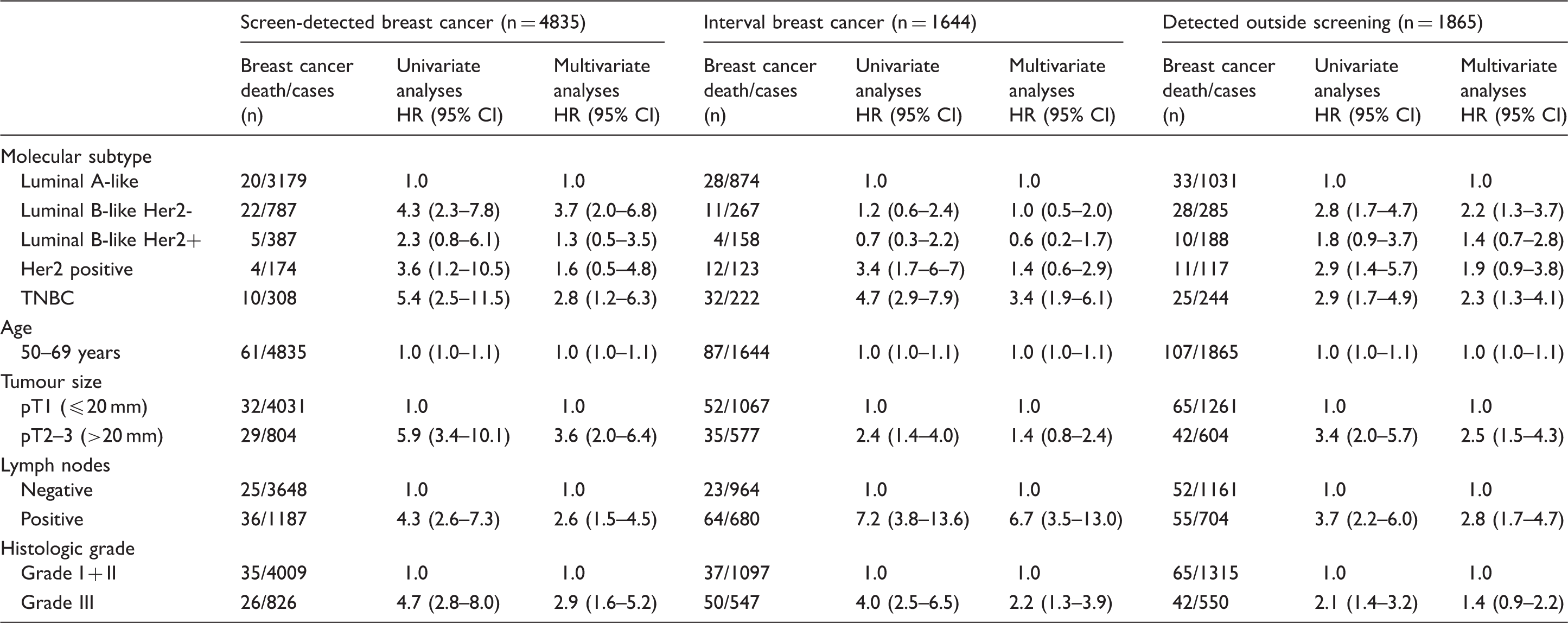
Adjusted analyses showed that ever screened women had an HR of breast cancer death of 1.3 (95% CI: 1.0–1.7), using ever-screened as the reference group (data not shown). No significant interaction was found between the study covariates (age at diagnosis, tumour size, histologic grade, and lymph node involvement). The results remained stable when county was included in the model (results not shown).
Discussion
Our study identified prognostic and predictive favourable tumour characteristics, a better disease-specific survival, and lower risk of breast cancer death among women with screen-detected compared with symptomatic cancer. The results remained stable after adjusting for factors related to lead-time and length bias. The findings support results from other studies.5,9,10,15,25,26
Our results support the view that lower rates of breast cancer death are due to screening, through detecting the tumours before they progress to a higher malignancy grade, and to a more aggressive phase in the natural history of the disease.1–3,25 Our study, which includes breast cancer cases diagnosed from 2005 and later, verifies the strongly predictive effect of tumour size and lymph node status on breast cancer survival in univariate and multivariate analyses, regardless of detection mode. This in turn indicates the continuing relevance of early diagnosis and treatment of the disease.
Luminal A-like tumours were more frequent among women with screen-detected compared with symptomatic cancer. In contrast, Her2 positive and TNBC were less frequently screen-detected. Favourable histopathologic tumour characteristics were observed among screen-detected versus symptomatic breast cancer in all subtypes. This is also described in other studies.3,8,12,16 Screen detected cancers are detected in an earlier stage, have a lower histologic grade and Ki67 index, and are more frequently hormone receptor positive and HER2 negative tumours. A Finnish study from 2004 followed around 3000 women for breast cancer specific death for 15 years, and found detection mode to be an independent prognostic factor. 11 The estimates were adjusted for age, tumour size, axillary lymph node status, histologic grade, and hormone receptor status. Sihto et al. 13 included information on subtypes when comparing the same study groups, and concluded that the distribution of molecular subtype differed by detection mode and that the better outcome for women with screen-detected cancer was partly explained by the subtype. In the Norwegian study by Collett et al. 4 negative estrogen receptor status and higher p53 expression were more common in interval cancer compared with screen-detected breast cancer, despite analysing tumours with similar size. However, whether the improved prognosis for screen-detected cancers is fully explained by earlier detection is not yet fully understood. Previous criticism regarding within-stage shift is eliminated in our study by including tumour size, histologic grade, lymph node status, subtypes, and age at diagnosis in the regression model.
Lead-time and length bias represent a concern when comparing survival among women with different detection modes. Adjusting for tumour size and lymph node involvement has been shown to decrease lead-time bias, 8 while including histologic grade and subtype in adjusted survival analysis has been shown to decrease the influence of length bias.10,11,26 We also used a method described by Duffy et al. 24 to adjust for lead-time. Our results remained stable, both when we adjusted for prognostic factors (tumour size, histologic grade, and lymph node involvement) and predictive tumour characteristics (subtypes), and also when we used the method described by Duffy et al. 24 Our results therefore suggest that detection mode is an independent prognostic factor, as indicated in other studies.5,12
The HRs did not differ significantly between IC and OS. This is as expected, according to the tumour characteristics given in Table 2. Studies have shown inconsistent findings, but might be hampered by small study populations and different definitions of IC. 27 Detection of small aggressive tumours at screening will reduce the burden of large aggressive ICs, and thereby improve survival among screened women. HRs for women with IC may be biased in favour of the ICs in our study, because some women have mammography at private clinics between two invitations to the programme, and their IC is therefore detected as asymptomatic cancer detected outside the screening programme, and their IC is therefore detected as asymptomatic cancer at private clinics within the two-year period after the prior screening exam. 28 In our sensitivity analysis, never screened women showed a borderline higher HR of breast cancer death when ever screened was used as the reference group. The HR for ever versus never screened is less biased by the “healthy screen effect” compared with the estimate given for the detection mode (SDC, IC, and OS). Women in the target group of the screening programme should therefore be requested to attend mammographic screening to increase survival and decrease mortality from the disease, and to minimize the healthy screen bias.
A substantial limitation of this study is the dichotomous variables used for axillary status and tumour size, and lack of information about multi-focality. Information about isolated tumour cells on sentinel lymph node biopsy and whether the positive axilla refers to micro or macro-metastasis was not available from the Cancer Registry, or from the pathology notes alone. Reviewing the medical reports for each of the cases included in this study is the next step to understand these issues. We grouped tumour size according to the pT system (20 mm or less and above 20 mm), because exact tumour size was available only for screen-detected and interval breast cancer. Again, access to individual medical reports is the next step to understand the value of exact versus a dichotomous value of tumour size on breast cancer survival.
Lack of information about treatment represents another limitation of our study, however, in Norway women are offered the same treatment regardless of mode of detection. There are 16 breast centres in Norway, all established as a part of the staggered implementation of the NBCSP from 1996–2005. Our study period ran from 2005 to 2011. There is no reason to assume geographical differences, and including county in the survival model did not influence the HR. However, women attending screening are often considered to be more aware of health issues compared with non-attenders, and this healthy screen effect or selection bias might influence the survival among screen-detected women.
The concordance between molecular subtypes based on microarrays and IHC analyses is 75–90% 29 , and the value of the high number of cases is shown to be a strength in the present study. However, no access to information on the proliferation marker Ki67 in this registry-based study is a limitation related to the sub-classification of luminal tumours. Such limitations, when using large population based databases, are in contrast to analyses based on data from pathologists dedicated for particular studies, and often including complete reclassification of cases. Another missing factor in this study was information about mammographic density. High mammographic density is shown to be associated with tumour growth factors. 30 Women with interval cancer are shown to have denser breasts than women with screen-detected tumours. The less favourable prognosis might be related to delayed detection due to masking.
The large number of breast cancer cases included in the study and the high compliance of the data represent strengths of the study. However, there were only 255 breast cancer deaths, which is mirrored in the confidence intervals of the HR’s. The study followed the women for breast cancer death for six years only, and a repeated study with longer follow-up might add further knowledge to this topic.
Our findings support previous conclusions that tumour size and node status remain strong prognostic factors for survival, both in univariate and multivariate analyses, 3 and additional analyses showed that the HR for breast cancer death remained stable also when including only tumour size, node status, and age in the adjusted analyses. Detection mode was shown to be an independent prognostic factor in which it represents an important research and clinical implication. Information about detection mode should therefore be taken into account when planning treatment, in order to avoid overtreatment. Our study supports previous studies suggesting that case report forms and research databases should include the method of detection to improve the accuracy of the study analyses.
Conclusion
Women with screen-detected breast cancer have better survival compared with women diagnosed with interval breast cancer or breast cancer detected outside the screening programme, independent of age, tumour size, lymph node involvement, histological grade, and subtype. The benefits and harms of including detection mode as a marker of prognosis and as a guide in therapy decision-making should be further investigated.
