Abstract
Objective
Participation, an indicator of screening programme acceptance and effectiveness, varies widely in clinical trials and population-based colorectal cancer (CRC) screening programmes. We aimed to assess whether CRC screening participation rates can be compared across organized guaiac fecal occult blood test (G-FOBT)/fecal immunochemical test (FIT)-based programmes, and what factors influence these rates.
Methods
Programme representatives from countries participating in the International Cancer Screening Network were surveyed to describe their G-FOBT/FIT-based CRC screening programmes, how screening participation is defined and measured, and to provide participation data for their most recent completed screening round.
Results
Information was obtained from 15 programmes in 12 countries. Programmes varied in size, reach, maturity, target age groups, exclusions, type of test kit, method of providing test kits and use, and frequency of reminders. Coverage by invitation ranged from 30–100%, coverage by the screening programme from 7–67.7%, overall uptake/participation rate from 7–67.7%, and first invitation participation from 7–64.3%. Participation rates generally increased with age and were higher among women than men and for subsequent compared with first invitation participation.
Conclusion
Comparisons among CRC screening programmes should be made cautiously, given differences in organization, target populations, and interpretation of indicators. More meaningful comparisons are possible if rates are calculated across a uniform age range, by gender, and separately for people invited for the first time vs. previously.
Introduction
Participation is a key indicator of a cancer screening programme’s acceptance and effectiveness. High uptake in the target population is necessary for a screening programme to achieve the mortality reductions demonstrated in clinical trials.1,2 Clinical trials and population-based programmes of colorectal cancer (CRC) screening have shown that participation varies widely and is relatively low. 3 A recent review found that few organized CRC screening programmes and pilot studies met the European recommendation for minimum uptake of 45% and a desirable participation rate of 65–70%.3,4 This may be partly due to the relatively recent implementation of CRC screening programmes compared with those for breast and cervical cancer.5,6
Population characteristics and programme design and implementation can affect participation rate.7–9 Inconsistencies in how CRC screening participation is defined, measured, and reported4,10 make direct comparison of participation rates challenging. Multiple indicators of screening participation have been developed, including coverage by invitation, coverage by examination, the participation rate, and long-term adherence over two or more screening rounds.3,11 To investigate how organized CRC screening programmes in different countries define and measure participation in screening, a study was initiated through the International Cancer Screening Network (ICSN), a consortium of countries that have population-based cancer screening programmes and active efforts to evaluate and improve cancer screening (http://appliedresearch.cancer.gov/icsn). Study aims were to assess the extent to which CRC screening participation rates can be compared across programmes, factors that may influence participation rates, and the feasibility of using questionnaires to collect data on CRC screening participation to improve the validity of direct comparisons across programmes and jurisdictions.
Methods
A Screening Participation Rates Work Group, formed following the ICSN-meeting in Denmark in June 2008, designed a two-part study to assess measurement of CRC screening participation rates among ICSN countries with organized CRC screening programmes:
In May 2011, 29 ICSN country representatives were emailed a questionnaire to ascertain whether they had an organized CRC screening programme (defined as targeting a specific population group, having a mechanism for identifying people eligible for screening, and sending letters inviting eligible people in the target population to participate in screening), characteristics of the programme (eg. when launched, targeted age groups, number of rounds completed, test modality used, screening interval), and how screening participation was defined and measured. In part II of the study, a follow-up questionnaire was e-mailed in February 2012 to 19 programme representatives in 12 countries, who had indicated that their country had an organized CRC screening programme using guaiac-based fecal occult blood test (G-FOBT) or fecal immunochemical test (FIT) for primary screening. One programme participating in Part I of the study reported flexible sigmoidoscopy as their primary screening modality, and we excluded this programme from Part II because of difficulties in making comparisons with G-FOBT/FIT-based programmes. The follow-up questionnaire requested data for the programme’s most recent, completed screening round, broken out by age at invitation in five-year groupings (50–54, 55–59, 60–64, 65–69, and 70–74) and by gender. Requested data were: number of people in the target population, number of these eligible for screening, number of eligible people invited to screening, number of eligible people invited for the first time, number of people screened of those invited, number of people screened of those invited for the first time, and the number of people screened of those invited for whom there was a final test result. We also asked about programme organizational and operational features, eg. type of G-FOBT or FIT kit used, number of samples collected and recommended test frequency, whether dietary restrictions are required for test kit completion, how invited people obtain test kits, and criteria for exclusion from screening.
For each programme participating in part II, we calculated three measures of screening participation derived from the European Guidelines for Quality Assurance in Colorectal Cancer Screening:
3
i) coverage by invitation (number of people invited to screening divided by number of people eligible for screening in the target population), ii) coverage by the screening programme (number of people screened [of those invited] divided by the number of people eligible for screening in the target population), and iii) uptake or participation overall (number of people screened [of those invited] divided by the number of eligible people invited). We also calculated a fourth measure adapted from the European Network for Indicators in Cancer project:
12
uptake or participation at first invitation (number of people invited for the first time who were screened divided by the number of eligible people who were invited for the first time).
Statistical analysis
Participation rate (in percent) by programme was calculated as the number of participants divided by the number of invited individuals, and stratified by gender and age group. Proportions with 95% confidence intervals were calculated to evaluate whether participation rates differed significantly between genders, and among age groups, and initial versus subsequent invitation round. Because information from Italy summarized 79 regional and local FIT-based programmes, we treated Italy as a single programme.
Results
Characteristics of organized G-FOBT/FIT-based CRC screening programmes
Characteristics of Organized G-FOBT/FIT-Based Colorectal Cancer Screening Programmes Participating in the ICSN Survey.
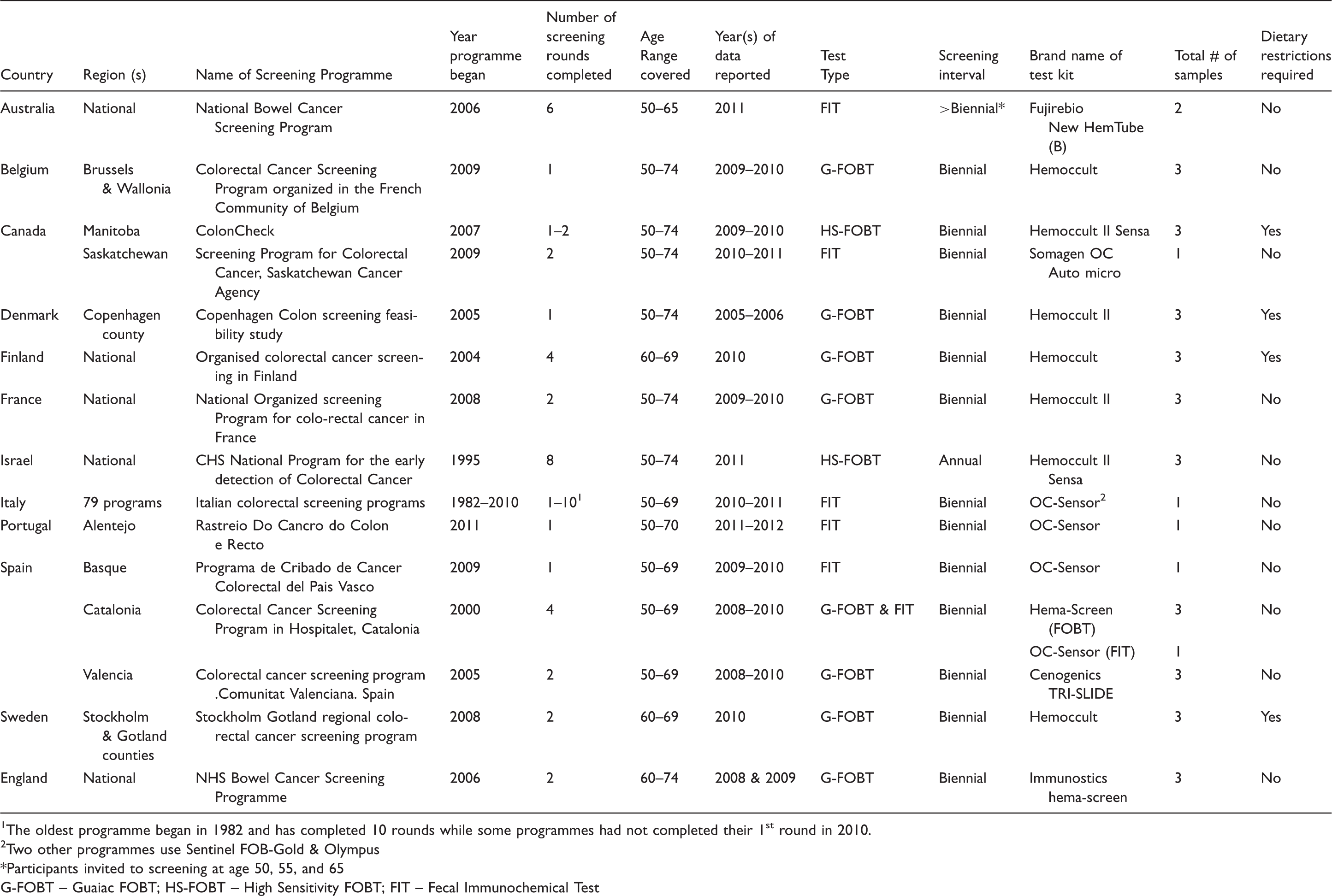
The oldest programme began in 1982 and has completed 10 rounds while some programmes had not completed their 1st round in 2010.
Two other programmes use Sentinel FOB-Gold & Olympus
Participants invited to screening at age 50, 55, and 65
G-FOBT – Guaiac FOBT; HS-FOBT – High Sensitivity FOBT; FIT – Fecal Immunochemical Test
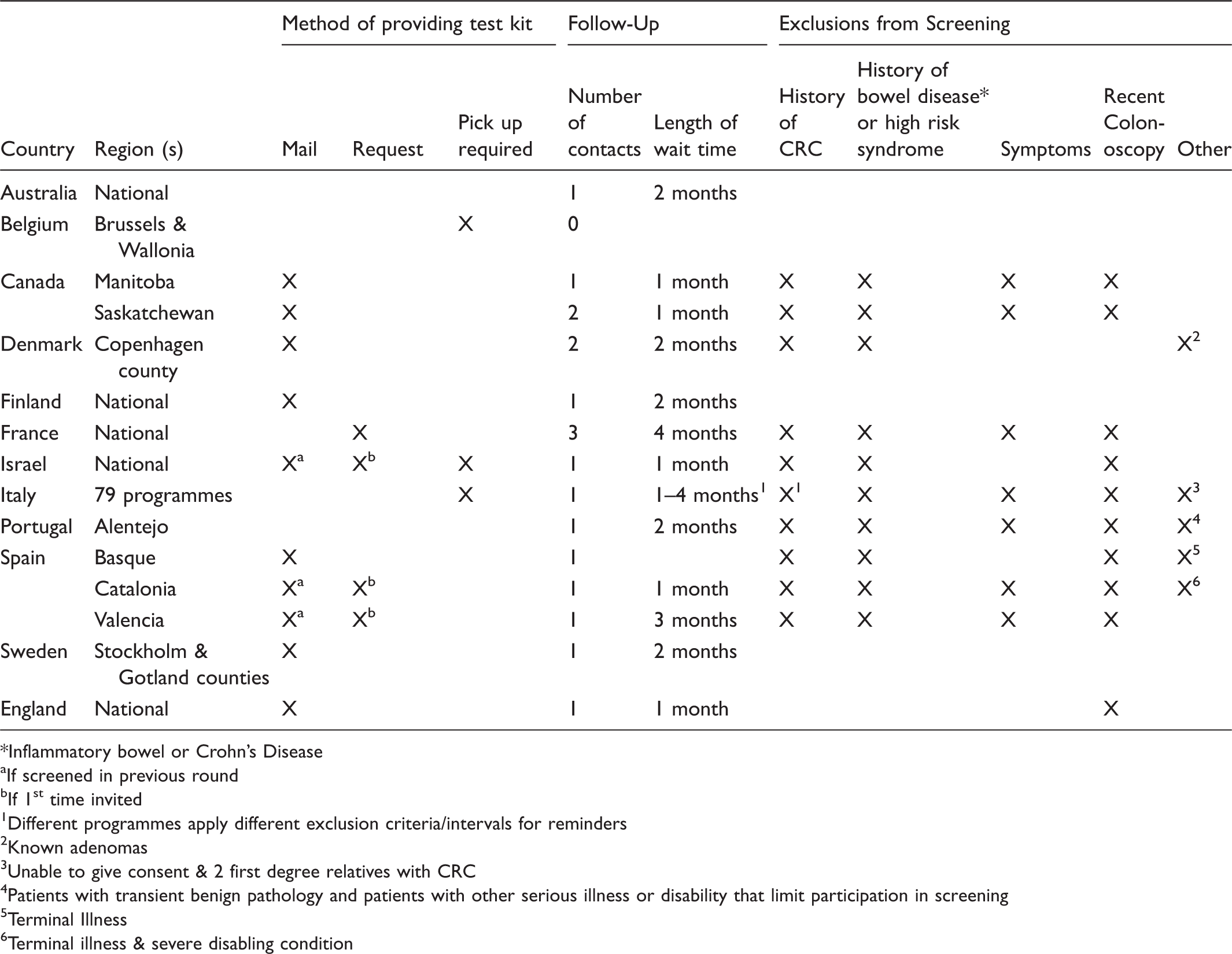
Organized G-FOBT/FIT-Based Colorectal Cancer Screening Programmes: Means of Providing Test Kits, Follow-Up, and Exclusions from the Programme.
Inflammatory bowel or Crohn’s Disease
If screened in previous round
If 1st time invited
Different programmes apply different exclusion criteria/intervals for reminders
Known adenomas
Unable to give consent & 2 first degree relatives with CRC
Patients with transient benign pathology and patients with other serious illness or disability that limit participation in screening
Terminal Illness
Terminal illness & severe disabling condition
All programmes except Brussels & Wallonia (Belgium) sent reminders to individuals who had not returned a completed test kit within a specified time period (Table 2). Ten programmes sent one reminder, two sent up to two reminders, and one sent up to three. Reminders were sent between one and four months after the initial invitation (mode two months). None of the programmes used reminder telephone calls.
Eligibility criteria also varied between programmes (Table 2). Four programmes (Australia, Brussels & Wallonia [Belgium], Finland, Stockholm & Gotland [Sweden]) had no exclusions. The other programmes excluded people based on one or more of the following factors: personal history of CRC, adenomas, or inflammatory bowel disease; family history of CRC or high-risk genetic syndrome; recent colonoscopy; and symptoms of CRC or other bowel disease or life-limiting chronic illness.
Participation indicators in organized G-FOBT/FIT-based CRC screening programmes
Participation Indicators for 15 G-FOBT/FIT-Based Colorectal Cancer Screening Programmes.
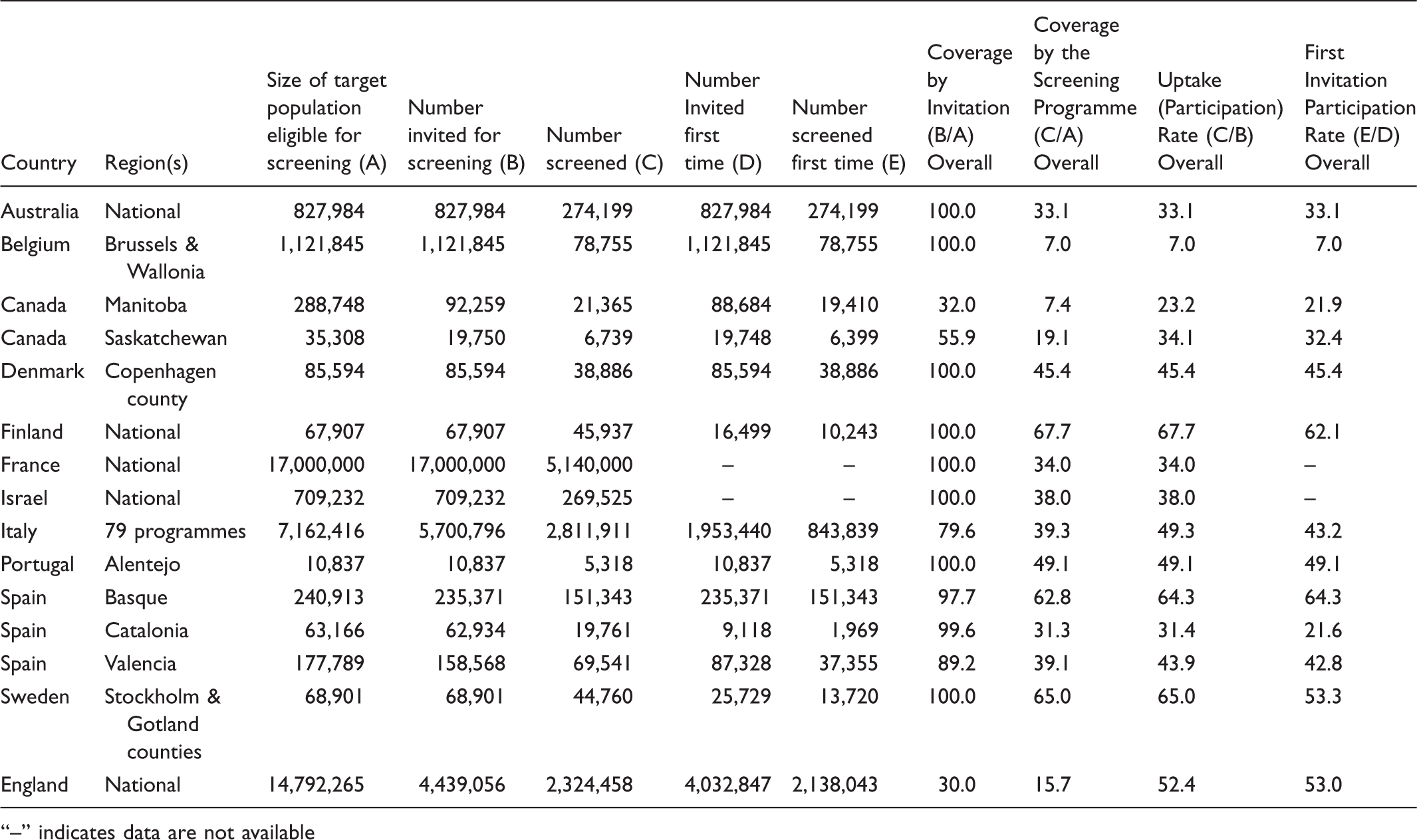
“–” indicates data are not available
Figure 1 shows that participation rates were statistically significantly higher among women than men for all programmes except Brussels & Wallonia (Belgium), where they were equivalent. The gender difference was most evident in Finland, with 75% participation rate among women compared with 60% among men.
Comparison of participation rates among 15 G-FOBT-based colorectal cancer screening programmes by gender*.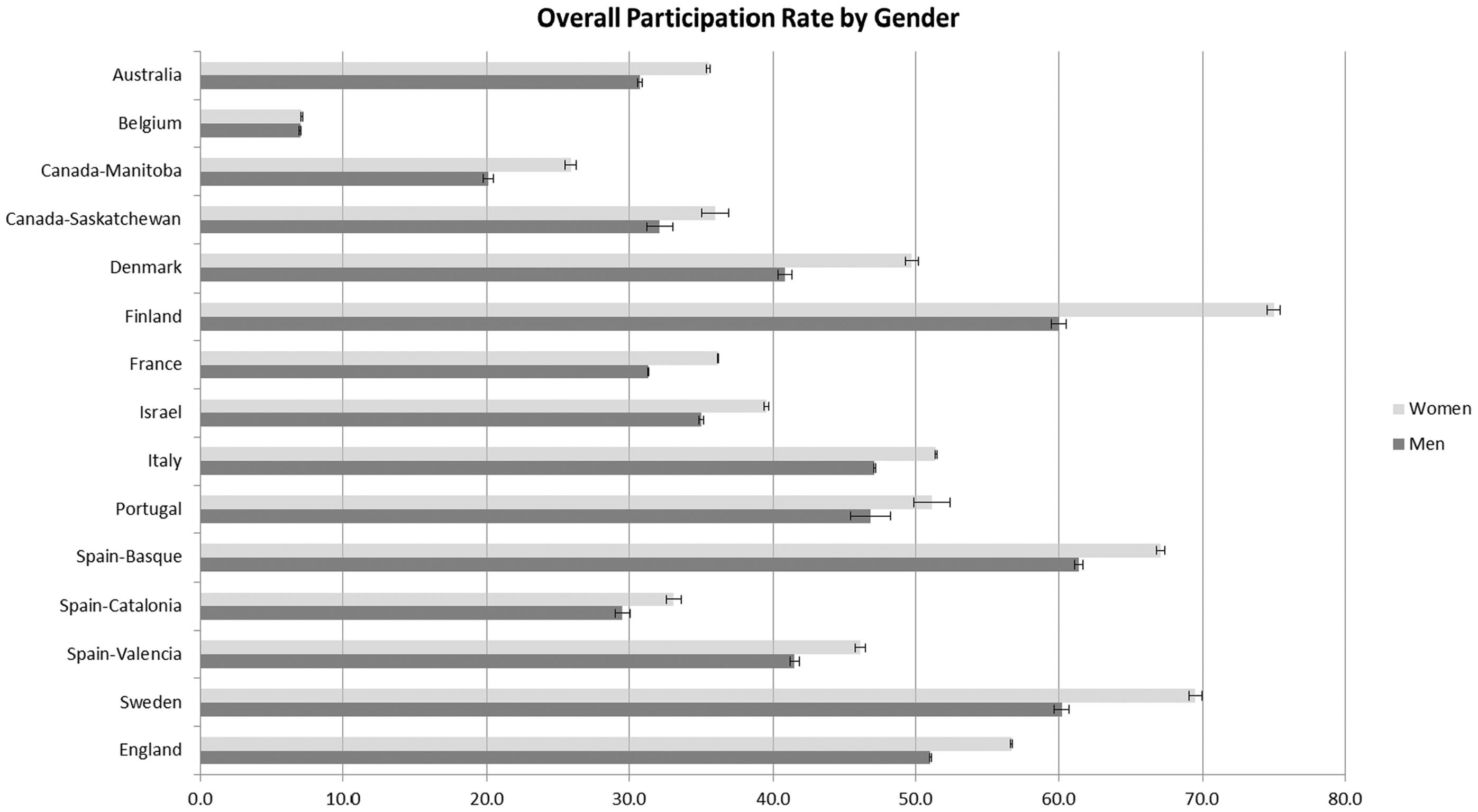
Comparison of Uptake/Participation Rates among 13 G-FOBT/FIT-Based Colorectal Cancer Screening Programmes, by Age Group and Initial versus Subsequent Invitation.
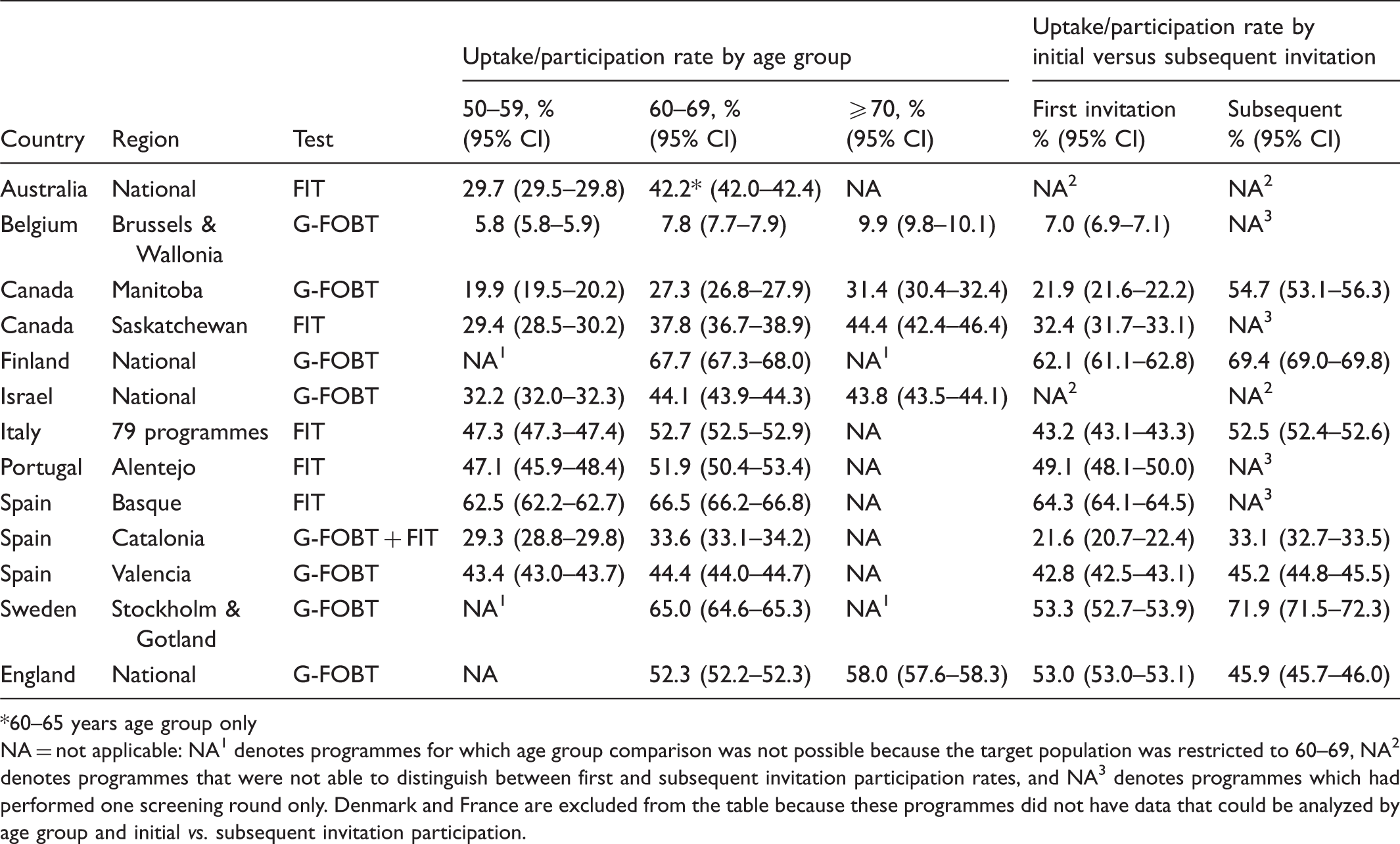
60–65 years age group only NA = not applicable: NA1 denotes programmes for which age group comparison was not possible because the target population was restricted to 60–69, NA2 denotes programmes that were not able to distinguish between first and subsequent invitation participation rates, and NA3 denotes programmes which had performed one screening round only. Denmark and France are excluded from the table because these programmes did not have data that could be analyzed by age group and initial vs. subsequent invitation participation.
In seven programmes able to evaluate differences in uptake between first and subsequent invitation (Table 4), subsequent invitation participation rates were statistically significantly higher than first invitation rates in Manitoba (Canada), Finland, Italy, Catalonia (Spain), Valencia (Spain), and Sweden. In England, subsequent invitation participation was lower than first invitation participation by about seven percentage points. Manitoba (Canada) showed the largest difference between first and subsequent invitation participation (21.9% v 54.7%). Averaged across the seven programmes, subsequent invitation participation rates were approximately nine percentage points higher than first invitation participation rates.
Discussion
Comparisons of CRC screening participation rates are hampered by diverse methods of defining, measuring, and reporting participation.4,10 Even though we used a standardized set of definitions to compare four participation indicators (coverage by invitation, coverage by the screening programme, overall uptake or participation, and first invitation participation) among 15 G-FOBT/FIT-based CRC screening programmes, and attempted to collect data in a uniform way, there was considerable variability in the indicators across programmes. Coverage by invitation ranged from 30–100%, coverage by the screening programme and overall uptake or participation both ranged from 7–68%, and first invitation participation from 7–64%. Only six programmes met the European Guidelines’ minimum participation target of 45%. 3 We also showed the influence of three factors on participation rates: gender (participation was higher among women than men), age (higher participation among those aged 60–69 and 70+ than younger age groups), and subsequent v first invitation participation (those invited in a previous screening round had higher participation than initial invitees).
Our study demonstrates that it is possible to collect and report participation data in a more standardized way to facilitate comparisons, but also illustrates the considerable diversity in screening programme policies, organization, and implementation that complicate the evaluation and comparison of participation indicators.
Screening participation tends to increase as organized programmes mature and make adjustments to better meet the needs of their populations.13–15 Some programmes in our study (Israel, Italy, and Catalonia [Spain]) had been in place for more than a decade, but others, established more recently (eg. Australia, England, and regional programmes in Canada and Spain) were still in the roll out phase. As our study encompassed programmes with only one completed screening round (as well as those with multiple rounds), participation in subsequent invitations could not be assessed in all programmes. In our study, the participation rate was increased and the variability across countries and regions was reduced, when comparing initial and subsequent screening in all programmes, apart from England (Table 4). The paradoxical decrease in participation in the subsequent invitation round in the expanding English programme could be related to a large proportion of invitations addressing individuals at an initial phase despite being invitations in the subsequent programme round. We believe that our finding of an increased participation in the subsequent round compared with the first invitation, also documented in other reports from ongoing national and regional programmes, represents an established and generalizable result.
Programmes also differed in size, from a few thousand to several million, and in the age groups targeted. Our study confirmed that younger age groups are less likely to participate in CRC screening,15–20 and so the ability to examine participation rates by discrete age groups is important, but two of the programmes in our study could not provide data by narrower age groups to permit comparisons with other programmes. Reporting programme data for five year (ie. 50–54, 50–59) or ten year (ie. 50–59) age groupings, instead of the entire age range of the target population (ie. 50–74), and calculating age-specific participation rates would make cross-programme comparisons more meaningful.
Exclusion criteria present another cross-programme comparison challenge. Programmes with exclusion criteria produce a reduced denominator, and generate a seemingly higher compliance rate than programmes that invite the entire targeted age group. 4 Four programmes in our study had no exclusion criteria; others excluded people based on one or more aspects of their colorectal cancer risk or health status.
Most programmes in our study used G-FOBT test kits requiring three stool samples, as opposed to one for the FIT-based programmes. Four of the G-FOBT-based programmes also imposed dietary restrictions for successful test completion. Due to lack of randomization of the test used, we did not assess whether participation rates varied by test type, number of stool samples required, or use of dietary restrictions. Even if the same test (G-FOBT) and dietary restrictions were used, programmes could differ in ways of providing the test. In 10 programmes in our study, the test kit was mailed together with the invitation, but in other programmes participants had to ask for the kit, or collect it at a clinic/pharmacy, which might lower participation21,22 and make cross-programme comparisons difficult. Programmes also differed in their use of reminders to individuals who failed to return a completed test kit. Although all but one programme reported mailing reminders, the number of reminders varied from one to three, and the timing of the reminders ranged from one to four months following the initial invitation to screening.
Our study has limitations. Only countries that participate in the ICSN could be included, somewhat hampering the generalizability of the results, although several ICSN countries (the Netherlands, Luxembourg, New Zealand) have recently launched organized CRC screening programmes, and a larger, more comprehensive study of participation indicators may be feasible in the future. In addition, the observational study design limited our ability to evaluate the influence of various programme features on participation indicators. Although the gender and screening history trends seem common across countries, the baseline differences could be due to cultural, health-systems’ characteristics, and organizational differences rather than the actual screening strategy.
Conclusion
Establishing common measures is an important initial step in the ability to compare participation across screening programmes. Our results show that more meaningful comparisons of CRC screening participation indicators across programmes are possible if participation indicators are calculated using uniform definitions, and differences in programme organization and population characteristics are taken into account. The majority of organized screening programmes underperform in uptake, with a general trend toward lower uptake among men and younger age groups, and the reasons for such trends need to be addressed in future studies. Comparability of measures of cumulative participation is of particular importance, given ongoing efforts to implement and evaluate CRC screening programmes.
Footnotes
Acknowledgements
We thank Kathy Sedgwick of NOVA Research Company for data collection assistance.
