Abstract
Objectives
The absolute annual risk of patients with Barrett's oesophagus (BO) developing oesophageal adenocarcinoma (OAC) is ≤0.5%. Screening BO patients for malignant progression using endoscopic surveillance is widely practised. To assess the efficacy and cost-effectiveness of this, we developed a protocol for a randomized controlled trial of surveillance versus ‘at need’ endoscopy.
Methods
In a multicentre trial, 3400 BO patients randomized to either 2-yearly endoscopic surveillance or ‘at need’ endoscopy will be followed up for 10 years. Urgent endoscopy will be offered to all patients who develop symptoms of dysphagia, unexplained weight loss >7lb (3.2kg), iron deficiency anaemia, recurrent vomiting, or worsening upper gastrointestinal symptoms. Participants must have endoscopically and histologically confirmed BO, with circumferential BO ≥1cm or maximal tongue/island length ≥2 cm. Candidates with existing oesophageal high-grade dysplasia or cancer, or previous upper gastrointestinal cancer will be excluded. Primary outcome will be overall survival. Secondary outcomes will be cost effectiveness (cost per life year saved and quality adjusted life years); cancer-specific survival; time to OAC diagnosis and stage at diagnosis; morbidity and mortality related to any interventions; and frequency of endoscopy.
Conclusions
This randomized trial will provide data to evaluate the efficacy and cost-effectiveness of screening BO patients for OAC.
Keywords
Introduction
The incidence of oesophageal adenocarcinoma (OAC) rose dramatically in Western populations in the latter 20th century,1–4 with over 5,000 cases each year in the UK. Only 20–30% of oesophageal cancers are potentially curable at presentation, and overall 5-year survival rate in the UK for 2005–9 was 13%. 5
Guidelines for surveillance of Barrett's oesophagus.
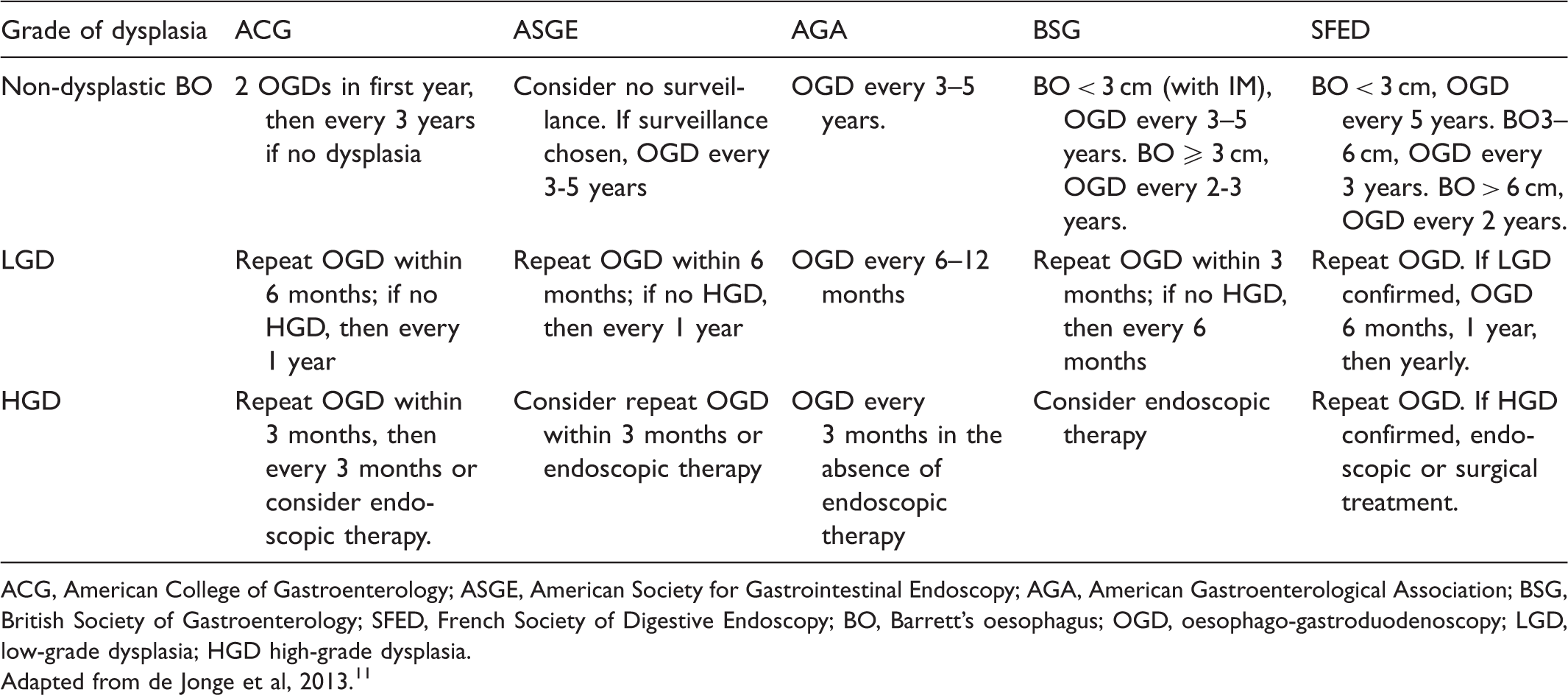
ACG, American College of Gastroenterology; ASGE, American Society for Gastrointestinal Endoscopy; AGA, American Gastroenterological Association; BSG, British Society of Gastroenterology; SFED, French Society of Digestive Endoscopy; BO, Barrett's oesophagus; OGD, oesophago-gastroduodenoscopy; LGD, low-grade dysplasia; HGD high-grade dysplasia.
Adapted from de Jonge et al, 2013. 11
Current evidence is derived from retrospective cohort and comparative studies, some showing improved outcomes and/or earlier stage at diagnosis in patients in surveillance programmes.12–19 A recent population wide study in Northern Ireland found that diagnosis of BO was associated with improved survival from oesophageal adenocarcinoma, and this effect persisted after adjustment for lead and length time bias. 20 A study in the Netherlands with a similar design found that, for patients who were subsequently diagnosed with oesophageal adenocarcinoma, a diagnosis of BO was only correlated with an improved outcome if the patient participated in an adequate surveillance programme. 21 However, several large retrospective studies have found no benefit from surveillance, and reached the opposite conclusion.22,23 These retrospective studies are limited by a number of potential sources of bias and confounders. Lead and length time bias may affect the results, and those patients electing to enter surveillance programmes may be systematically different from those who decide not to undergo surveillance (eg. Barrett's length, co-morbidities etc). A randomized-controlled trial is the only study design capable of accounting for these sources of bias.
Without hard data on efficacy, there is concern that surveillance is not cost-effective in its current forms.11,24,25 The observational data available have been modelled to provide an incremental cost-effectiveness ratio of Barrett's surveillance under ideal assumptions. However, these models have reached varying conclusions,26–29 with all models finding cost-effectiveness highly sensitive to the interval of surveillance, the risk of developing OAC, and the efficacy of surveillance in preventing mortality from OAC. A randomized trial could provide robust evidence for the efficacy of surveillance in reducing OAC mortality.
We aim to undertake a large, multicentre randomized controlled trial, the “BOSS” trial, to determine (i) efficacy and (ii) cost-effectiveness of two-yearly endoscopic surveillance compared with no routine surveillance in patients with BO.
Methods
Trial design
The BOSS trial is a multi-centre randomized 2-arm pragmatic parallel group trial of 3400 BO patients, followed up for 10 years. As both patients and clinicians will be aware of the intervention, a blinded trial is precluded.
Inclusion criteria
Patients must be aged over 18, able to give written consent, and fit for endoscopy. They must have had confirmatory endoscopy within two years, with histology reported as ‘diagnostic’, ‘in keeping with’ or ‘corroborative of’ Barrett's metaplasia, with circumferential BO of at least 1 cm, or a 2 cm non-circumferential tongue or island.
Exclusion criteria
Patients with high-grade dysplasia or carcinoma at enrolment will be excluded. Patients with low grade dysplasia (LGD) may join at their clinician's discretion (this aims to increase recruitment of low risk patients, but because only a subset of LGD patients are recruited separate results will not be presented). Patients with a history of upper gastrointestinal or other cancers, where the investigator considers the research to be an added burden to the participant, will be excluded. Participants in the ongoing AspECT trial are also ineligible. 30
Recruitment
Participants will be identified at local trial sites (see Appendix 1), either following endoscopy with a new diagnosis of BO, or from existing disease registers. Participants must have been informed of the risk of BO developing into oesophageal cancer, either at the visit when the invitation letter is issued, or on a documented previous occasion. Patients who meet the eligibility criteria and provide informed consent will be randomized to the study.
Interventions
The experimental intervention will be endoscopy every two years +/− 3 months with quadrantic biopsies taken every 2 cm. Final scheduled endoscopy will be no later than the 10th anniversary of recruitment date. The control intervention will be no surveillance endoscopy (or ‘at need’ endoscopy only). Patients in both arms will be offered urgent endoscopy if they develop dysphagia, unexplained weight loss of more than 7lb (3.2kg), iron deficiency anaemia, recurrent vomiting, or worsening upper gastrointestinal symptoms. All other care for patients (including decisions about treatment of oesophageal dysplasia/carcinoma, and altered endoscopy frequency following this) should follow standard practice for the treating hospital. Patients who develop LGD should remain in the study.
Outcomes
The primary outcome is overall survival, defined as the time from randomization to death from any cause. The secondary outcome of cost-effectiveness will be assessed through cost per life year saved, and cost per quality adjusted life year (QALY) saved from a health service perspective comparing surveillance every two years with ‘at need’ endoscopy.
The following secondary outcomes will be compared between the two arms: cancer-specific survival; time to diagnosis of OAC; stage of OAC at diagnosis; morbidity and mortality related to endoscopy, oesophageal surgery or other endoscopy-related interventions; and frequency of endoscopy. Cancer-specific survival is defined as the time from randomization to death from: oesophageal cancer; gastric or oesophageal cancer; and all cancers.
Sample size
For the superiority analysis of the primary outcome of overall survival, 3400 BO patients will allow us to detect a hazard ratio of 1.3 at 93% power (2-sided test at the 5% significance level). This assumes all cause mortality has an exponential time to conversion with a constant all cause mortality rate of at least 1.25% per year; recruitment for two years, follow-up for 10 years, and a 10% loss to follow-up from national flagging. For the non-inferiority analysis of overall survival with 3400 patients there is 87% power to conclude non-inferiority of ‘at need’ endoscopy if there is no underlying difference between the arms, assuming a non-inferiority margin of 5% absolute difference in 10 year survival rate, ie. if there is a difference in 10 year survival of less than 5% between the groups then we will conclude that ‘at need’ is non-inferior to surveillance.
Randomization
Randomization codes will be computer-generated by the Centre for Statistics in Medicine, Oxford, and administered by the Gloucestershire trials office. Block randomization will use varying block size, stratified on three factors: age at BO diagnosis (<65, ≥65); maximum length of Barrett's metaplasia segment (<2 cm, ≥2 cm and ≤3 cm, >3 cm and ≤8 cm, >8 cm); and Barrett's newly diagnosed (yes, no) (defined as date of endoscopic diagnosis of BO <4 months before the date of consent to trial entry).
Statistical analyses for primary objective
The primary objective will be assessed on the intention to treat (ITT) population since this is a trial of policy rather than simply efficacy. For the primary outcome of overall survival, the primary analysis will be a stratified log-rank test comparing the two groups, stratified for all variables used as randomization strata. A Kaplan-Meier plot will also be presented. A multivariate Cox model will also be fitted to the data, if the proportional hazards assumption is appropriate, to estimate hazard ratios with 95% confidence intervals (CIs). This model will include all stratification variables, and other prognostic factors (including gender, obesity, use of proton pump inhibitors, previous indefinite or low grade dysplasia, time from Barrett's to randomization). Results of an unadjusted (univariate) Cox model will also be presented. If neither group is superior, the assay sensitivity to investigate non-inferiority will be assessed and if possible the non-inferiority will be tested on the ITT and per protocol sample, with both analyses given equal weight. Non-inferiority of the ‘at need’ arm will be concluded if the two-sided CI for absolute difference in 10 years event rates between the two arms excludes 5%. This will be based on event rates estimated from the multivariate Cox model.
Statistical analyses for secondary objectives
Methods for analysing cancer specific survival and the time to OAC diagnosis will be identical to those used for the primary outcome of superiority for overall survival. For the analysis of stage of OAC at diagnosis, tumour/node/metastasis stage and randomization arm will be cross-tabulated, and a chi-squared test for trend will be used to compare the two arms. If the proportional odds assumption is valid, ordinal logistic regression will be used to estimate effect size and to adjust for stratification and other prognostic variables. Alternatively, stages 1–2 versus stages 3–4 will be compared in logistic regression. To analyse morbidity and mortality, the number of participants experiencing at least one Serious Adverse Event (SAE) at any time during the trial will be tabulated by arm and tested using a chi-squared test. Odds ratios will be presented for: experiencing any SAE, and separately by type; and from probit regression, adjusting for stratification variables. Prognostic factors will also be adjusted for in the same model, provided that there are enough events to avoid over-fitting the model. Generalized linear regression models will be used to analyse frequency of endoscopy, assuming a Poisson distribution for number of endoscopies, and using the log link function. A multivariate analysis will be carried out adjusting for prognostic variables, as well as a univariate analysis to assess the effect of covariate adjustment.
Cost-effectiveness analysis
A cost-effectiveness analysis will compare surveillance with ‘at need’ endoscopy from a UK health service perspective. Data will be presented as the extra cost per extra health benefit of surveillance every two years compared with ‘at need’ endoscopy, or “incremental cost effectiveness ratio” (ICER). Costs per life year saved and per QALY saved will be presented for both the within trial period, based on the observed data, and for patient lifetimes, based on an extrapolation model. Health Service resource use will be collected from healthcare records, and a biennial questionnaire will collect information about BO medications taken in the preceding three months. Quality of life data will be collected using the EQ-5D instrument from the biennial questionnaires and following endoscopy events (scheduled and unplanned). The ICERs calculated from the within trial data will have a range of uncertainty due to the statistical uncertainty around the estimates of costs and effects. Bootstrap sampling techniques will be used to assess the uncertainty, which will be presented using CIs for cost-effectiveness, where appropriate, and through cost-effectiveness acceptability curves, that graphically displays the probability of a given ICER. A Markov model will be constructed to extrapolate the data beyond the 10 years of the trial and to explore other issues such as variations in OAC incidence rates in different centres and the most cost-effective interval for endoscopic surveillance. Data from BOSS will be supplemented with data from other sources as appropriate. Extensive sensitivity analysis will explore the importance of modelling assumptions for the lifetime cost-effectiveness results.
Ethics
All participants will give written informed consent. Ethical approval for BOSS was granted by UCLH Research Ethics Committee Alpha in September 2008 (subsequent amendments also approved). Approval will be needed from each site host NHS organization before the trial commences.
Trial committees and interim analyses
An independent Data and Safety Monitoring Committee (DSMC) will oversee trial conduct. If an interim analysis of the primary aim four years after the last patient recruitment suggests that superiority might be demonstrated prior to the end of the trial, a second analysis may be performed. The decision to stop the trial rests with the Trial Steering Committee (TSC) of independent clinicians and statisticians, and a BO patient representative. The BOSS Chief Investigator will lead the Trial Management Group, who will implement TSC decisions.
Discussion
Surveillance programmes to screen BO patients for malignant progression are advocated by gastroenterological societies worldwide, and consume substantial resources. These programmes have been based on observational data that are subject to bias, and a desire to ‘do something’ for patients with a known risk factor for oesophageal cancer, but their efficacy has never been assessed in a randomized trial. The BOSS trial is the first to evaluate the efficacy of endoscopy surveillance compared with ‘at need’ endoscopy in BO.
Efficacious surveillance requires accurate endoscopic and pathological recognition of early dysplastic and malignant changes, and a sufficiently short interval between endoscopies to monitor progression and enable effective treatment. These factors must be balanced against the acceptability and risks to the patient of multiple endoscopies, and the costs of such a programme, particularly as the risk of progression to cancer may be below 0.5% per year.31–33 The secondary end-points in this trial will provide information on many of these variables to inform future decision making.
With a minimum study period of 12 years, there will be advances in detection and intervention that could pose ethical questions about ensuring optimal care for all patients in the trial. Since the trial's inception, the British Society of Gastroenterology (BSG) has revised guidelines on who should receive surveillance, and optimum intervals, and evidence has emerged about the risks of progression in LGD.10,34 Our study follows the 2005 BSG guidelines, using 2-year intervals, though subsequent guidelines have increased the interval for some patients considered to be at low risk of progression. The secondary end-points of the trial will allow us to model strategies to estimate how effective screening might have been if new technologies and practices had been instituted at baseline.
The trial will also face other challenges, including the acceptability of randomizing to an ‘at need’ arm, particularly in centres where surveillance has long been the standard treatment, and patients and clinicians may have preferences for continued surveillance. The long-term commitment required from patients and clinicians increases the risk of withdrawal and loss to follow up. While the study design and scale is feasible within the context of a nationally funded healthcare system, the applicability of the findings to other settings will require interpretation. There is a risk of contamination of the allocated treatment groups, if patients in the ‘at need’ arm present with factitious symptoms to ensure regular endoscopy. Conversely, poor adherence to scheduled endoscopies for those in the surveillance arm could affect outcomes for this group. The unavoidable lack of blinding is a potential source of bias, particularly in subjective outcome measures such as self-reported quality of life, although it is unlikely to have a major impact on the primary outcome of all cause mortality.
Key strengths of the trial design include its long follow-up period, pragmatic design to ensure safe care for patients in the ‘at need’ arm, and recognition that non-inferiority testing is an important outcome, as this would be sufficient to prefer a policy of ‘at need’ endoscopy.
Conclusions
The BOSS Trial represents an opportunity to answer categorically the key questions of efficacy and cost-effectiveness necessary to recommend or refute the value of endoscopy screening for patients with BO. It will assess the benefits of current surveillance in the UK, and may also enable risk stratification to identify those who may benefit most from targeted surveillance at an appropriate interval. The trial results will enable more effective and cost-effective BO management.
Footnotes
Trial Registration: ISRCTN54190466
The authors have no conflicts of interest.
Acknowledgements
The study is funded by the National Institute for Health Research (NIHR) Health Technology Assessment Programme (ref 05/12/01). We acknowledge the contributions of the National Cancer Research Network Consumer Liaison team and Mr Charles Brownhill, a patient with Barrett's oesophagus who is a member of the trial steering committee.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Trial Status
BOSS began recruiting in March 2009. The recruitment target of 3400 patients randomized was reached ahead of schedule, in October 2011.
