Abstract
Objectives
The sensitivity of screening mammography is much lower among women who have dense breast tissue, compared with women who have largely fatty breasts, and they are also at much higher risk of developing the disease. Increasing mammography screening frequency from biennially to annually has been suggested as a policy option to address the elevated risk in this population. The purpose of this study was to assess the cost-effectiveness of annual versus biennial screening mammography among women aged 50–79 with dense breast tissue.
Methods
A Markov model was constructed based on screening, diagnostic, and treatment pathways for the population-based screening and cancer care programme in British Columbia, Canada. Model probabilities and screening costs were calculated from screening programme data. Costs for breast cancer treatment were calculated from treatment data, and utility values were obtained from the literature. Incremental cost-effectiveness was expressed as cost per quality adjusted life year (QALY), and probabilistic sensitivity analysis was conducted.
Results
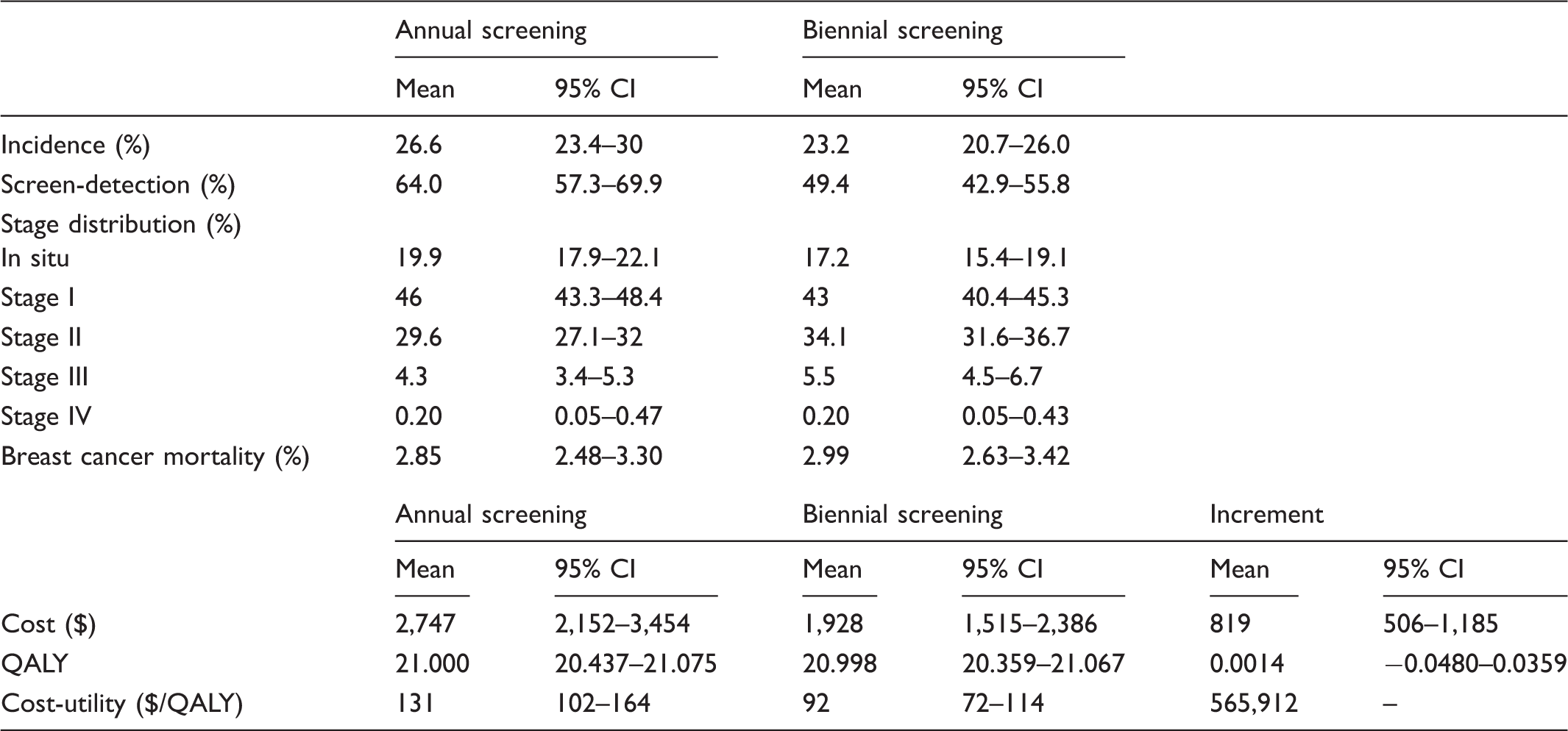
Compared with biennial screening, annual screening generated an additional 0.0014 QALYs (95% CI: −0.0480–0.0359) at a cost of $819 ($ = Canadian dollars) per patient (95% CI: 506–1185), resulting in an incremental cost effectiveness ratio of $565,912/QALY. Annual screening had a 37.5% probability of being cost-effective at a willingness-to-pay threshold of $100,000/QALY.
Conclusion
There is considerable uncertainty about the incremental cost-effectiveness of annual mammography. Further research on the comparative effectiveness of screening strategies for women with high mammographic breast density is warranted, particularly as digital mammography and density measurement become more widespread, before cost-effectiveness can be reevaluated.
Introduction
Breast density – a measure of the relative amount of epithelial and connective tissue to fat – is a strong risk factor for breast cancer,1,2 and dense breast tissue may mask tumours on mammograms, reducing the effectiveness of regular screening. 3 Women with breast density of over 75% as measured on mammograms are at least four times more likely to develop breast cancer than women with very low (<10%) breast density.4–7 The sensitivity of mammography screening has been reported to be as low as 62% in women with extremely dense breasts, compared with 88% in women with almost entirely fatty breasts. 8 Consequently, women with dense breasts are significantly more likely to be diagnosed with interval cancers (vs. screen-detected cancers) than women with non-dense breasts.3,9,10
There is continued debate in the literature regarding the effect of breast density on breast cancer outcomes. Survival for interval cancer is worse than for screen-detected cancer.11,12 Women with dense breasts are reportedly more likely to be diagnosed with larger tumours,13–16 but findings for nodal involvement and other prognostic factors are mixed, with some studies reporting an association between breast density and higher risk disease,13,14 and others reporting no effect.15,16 Breast density alone does not appear to be an independent predictor of survival; controlling for patient and disease characteristics, including stage and method of detection, breast cancer survival among women with dense breasts is no different from that among women without dense breasts. 17 In one study, the case fatality ratio among women with dense breasts was found to be lower than that among women with fatty breasts; 18 however, women with dense breasts remained at significantly higher risk of breast cancer mortality, due to their elevated risk of developing the disease. 18
More frequent mammography screening has been proposed as a way to improve the early detection of cancers in women with dense breasts. Studies comparing the outcomes at different breast cancer screening intervals for women with dense breasts have reported mixed results. A model by van Gils et al. found that screening women with ≥75% breast density annually instead of biennially could result in a 36% reduction in interval cancers in that subpopulation.
19
Similarly, by simulating mean sojourn time for cancers in dense and non-dense breasts, Chiu
With the high risk of developing breast cancer, and a potential reduction in interval cancers, increasing the frequency of mammography screening from every two years to annually for women with dense breasts may be an attractive policy option, but there is significant uncertainty around the cost-effectiveness of this intervention. Screening women over age 50 with dense breast tissue every two years has been found to be cost-effective at a US$50,000/QALY threshold, compared with screening every 3–4 years. 22 For annual screening, however, the incremental cost-effectiveness is over US$340,000/QALY. 22
Until a recent policy change, 23 women in British Columbia (BC) with dense breasts were offered annual screening from ages 40 to 49, and biennial screening from ages 50–79. The purpose of this study was to assess the incremental cost-effectiveness of annual screening mammography from ages 50–79 for women with ≥75% mammographic breast density.
Methods
Model
We constructed a Markov model to estimate the benefits and costs, from the health system perspective, associated with annual versus biennial screening mammography for women with high breast density. The model structure was based on screening, diagnosis, and care pathways for the Screening Mammography Program of BC (SMPBC), a population-based screening programme, and the BC Cancer Agency (BCCA). An advisory panel of clinicians and decision-makers, including a radiologist, oncologists, epidemiologists, and the director of screening operations, led the development of the model structure (Figure 1). The model simulates a hypothetical population of 50 year-old women with dense breasts participating in the SMPBC, with screening annually or biennially to age 79, assuming full participation. The model was constructed with a six month cycle length and a lifetime time horizon. The model structure, costs and utilities were the same in the annual and biennial arms of the model; only the transition probabilities differed.
Model Structure. In the annual screening arm, women alternate between ‘screening’ and ‘no screening’ states every cycle (6 months); in the biennial arm, women spend three cycles in the ‘no screening’ state (indicated by dashed line).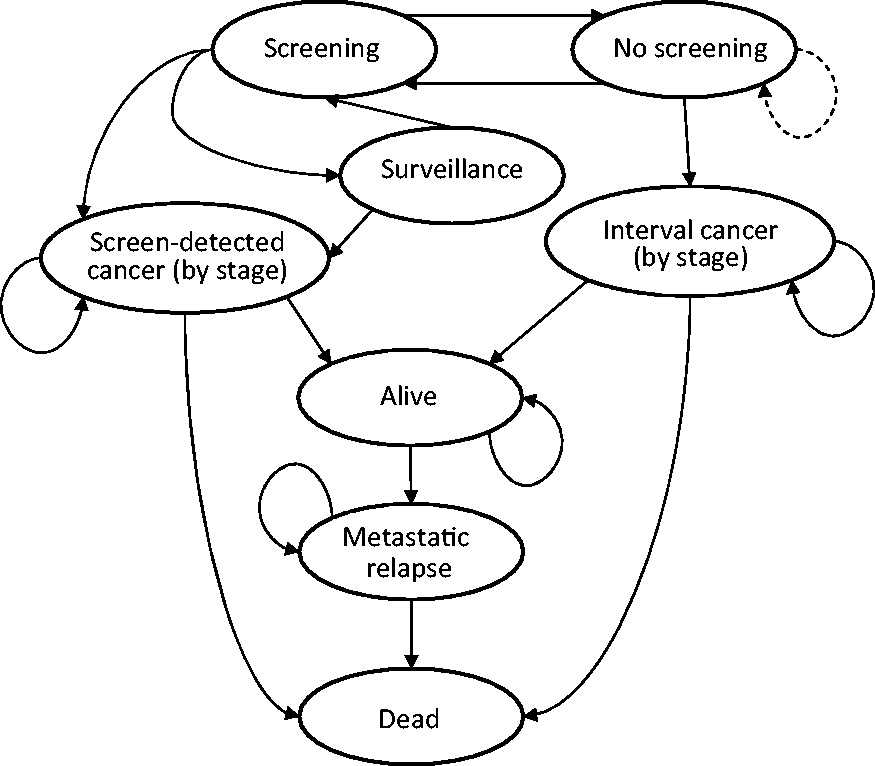
Probabilities
Transition probabilities for model.

transition probabilities for abnormal screen and cancer diagnosis are specific to women with extremely dense breasts (≥75% density); beta distributions were fit to probability estimates for probabilistic sensitivity analysis.
interval cancer risk expressed per 6-month model cycle.
Stage distributions by detection method were fit to Dirichlet distributions for probabilitistic sensitivity analysis.
Survival by stage and detection method was modeled with Weibull functions; parameters were fit to multinormal distribtuions for probabilistic sensitivity analysis.
The probability of a woman having an abnormal screen and undergoing diagnostic workup was calculated by analyzing SMPBC screening results from 2005 to 2007. The subset of screening centres reporting Breast Imaging, Reporting and Data System (BI-RADS) density was used to calculate the distribution of breast density ≥75% (BI-RADS category 4) and screen positivity rates. Cancer incidence for women with dense breasts was estimated by applying the density distribution and relative rates by density category 6 to rates calculated for all SMPBC participants from 2005 to 2007. Age-specific incidence for screen-detected and interval cancers were calculated separately for participants who had been screened annually (defined as <16 months since preceding screen) or biennially (18–30 months since preceding screen). Observed screen positivity rates and incidence by screening frequency were used directly; no assumptions regarding sensitivity and specificity were required. SMPBC data from 2003 were analyzed to determine the probability of a woman undergoing additional surveillance after receiving a false positive screening result.
Stage distribution of screen-detected and interval cancers and survival by stage were assumed to be no different for women with dense breasts than for the general breast cancer population, and were calculated from the BC Cancer Registry. Breast cancer survival rates by stage were calculated by fitting a series of Weibull models to Registry data. All-cause mortality was from Statistics Canada life tables. 24
Utility
Utility weights and cost inputs for model.
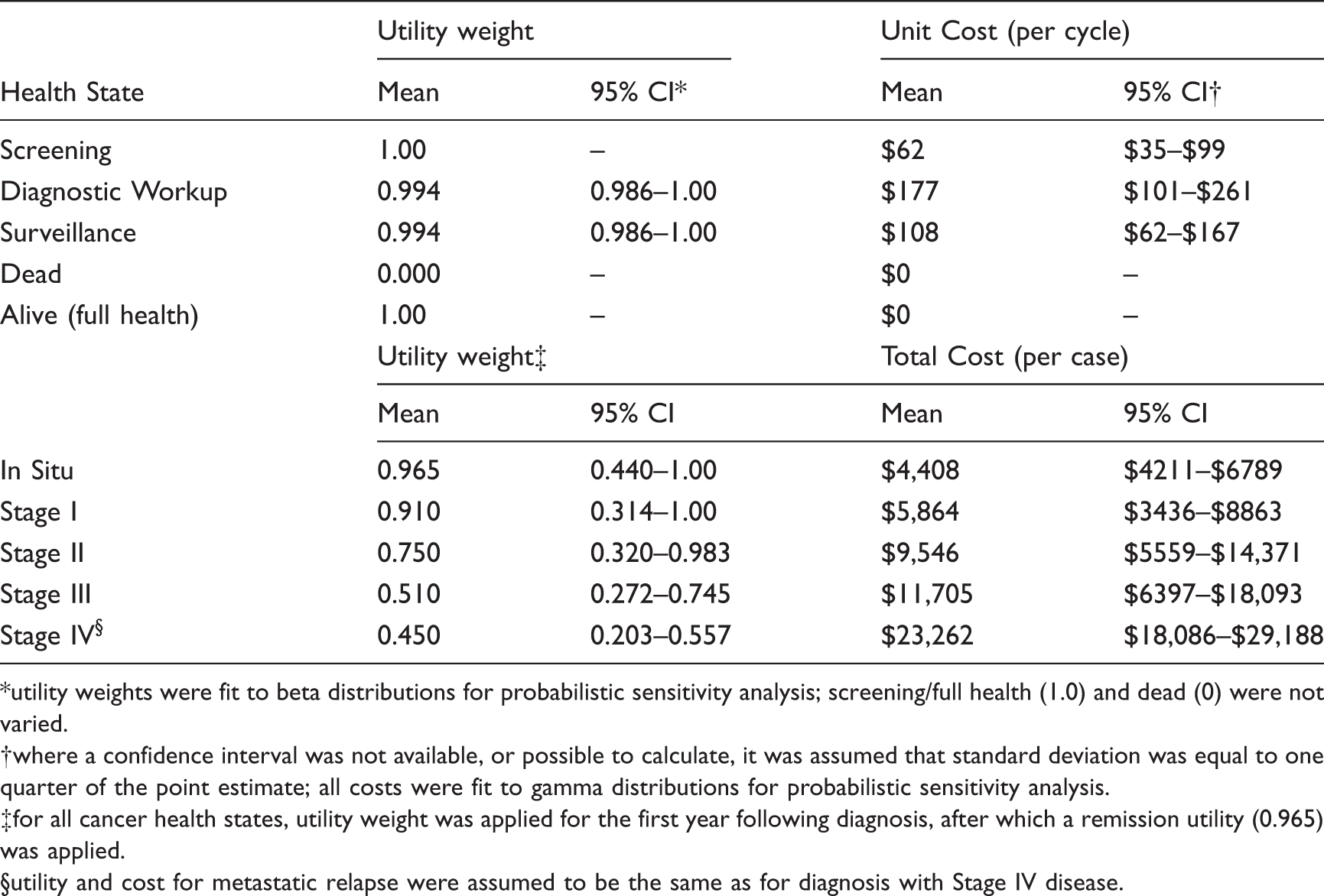
utility weights were fit to beta distributions for probabilistic sensitivity analysis; screening/full health (1.0) and dead (0) were not varied.
where a confidence interval was not available, or possible to calculate, it was assumed that standard deviation was equal to one quarter of the point estimate; all costs were fit to gamma distributions for probabilistic sensitivity analysis.
for all cancer health states, utility weight was applied for the first year following diagnosis, after which a remission utility (0.965) was applied.
utility and cost for metastatic relapse were assumed to be the same as for diagnosis with Stage IV disease.
Costs
Costs included: the mean cost of a mammogram, from operating costs of the SMPBC; 29 mean costs associated with diagnosis or surveillance following an abnormal screen, including diagnostic mammogram, ultrasound, consultations, core biopsy, or fine needle aspiration procedures; and the cost of treating breast cancer (Table 2).
The mean costs of diagnostic work-up and surveillance were calculated by combining patient-level resource use from the SMPBC data from 2003, and procedure costs estimated from the BC Medical Services Fee Schedule. 30 The cost of treating breast cancer was calculated using treatment records from breast cancer patients in their first year after diagnosis. For in situ disease and stage I–III breast cancer, the frequency of systemic therapy, radiation therapy, and surgical procedures were calculated from BCCA administrative data from 2002. The BCCA pharmacy provided the mean drug and delivery cost for systemic therapy protocols. The BCCA radiation therapy programme provided the average number of radiation fractions received by patients one year post diagnosis according to stage, and a mean cost per fraction estimated using the radiation therapy operating budget. The cost of surgery (mastectomy and lumpectomy) was estimated by combining professional costs from the BC Medical Services Fee Schedule, with local case cost and hospitalization costs. The cost of treating metastatic breast cancer was based on a study of BC women in 1995–1996. 31 The cost of chemotherapy for metastatic breast cancer was updated using BCCA systemic therapy utilization data from 2002–2007, and the cost of hospitalization and radiotherapy was inflated to 2007 values. 32 All costs in the model are expressed in 2007 Canadian dollars.
Analysis
The model was built and analyzed using TreeAge Pro 2013. 33 The cohort accumulates quality-adjusted life years (QALYs) and costs associated with each health state in every cycle. Costs and QALYs were discounted at 3% per annum. The changes in costs and quality-adjusted life expectancy for annual screening, relative to biennial screening, were used to calculate an incremental cost-effectiveness ratio (ICER) expressed as dollars per QALY gained.
Probabilistic sensitivity analysis was conducted using a Monte Carlo simulation with 1000 trials sampling from distributions for all parameters – probabilities, costs, and utilities. Beta distributions were used for utilities and probabilities, gamma distributions were used for costs, Dirichlet distributions were used for stage distributions, and multinormal distributions were used for parameters for survival curves. One-way sensitivity analysis was carried out on the cost of a screening mammogram, the cost of diagnostic work-up, utility adjustment, and discount rate.
Ethics approval was obtained from the University of British Columbia/British Columbia Cancer Agency Research Ethics Board.
Results
Model results and results of cost-effectiveness analysis for annual vs. biennial screening in women with dense breasts.
A scatter plot of the incremental cost and QALYs from 1000 replications (Figure 2) shows the simulations clustered around the y-axis. In 43.4% of simulations, annual screening resulted in a loss of QALYs relative to biennial screening. The cost-effectiveness acceptability curve (Figure 3) indicates that annual screening mammography has only a 23.5% probability of being cost-effective at a willingness to pay of $50,000/QALY, and a 37.5% probability of being cost-effective at a willingness to pay of $100,000/QALY.
Scatter plot of incremental cost and effectiveness for 1000 simulations.Ellipse indicates 95% of confidence limit. Cost-effectiveness acceptability curve for annual vs. biennial screening.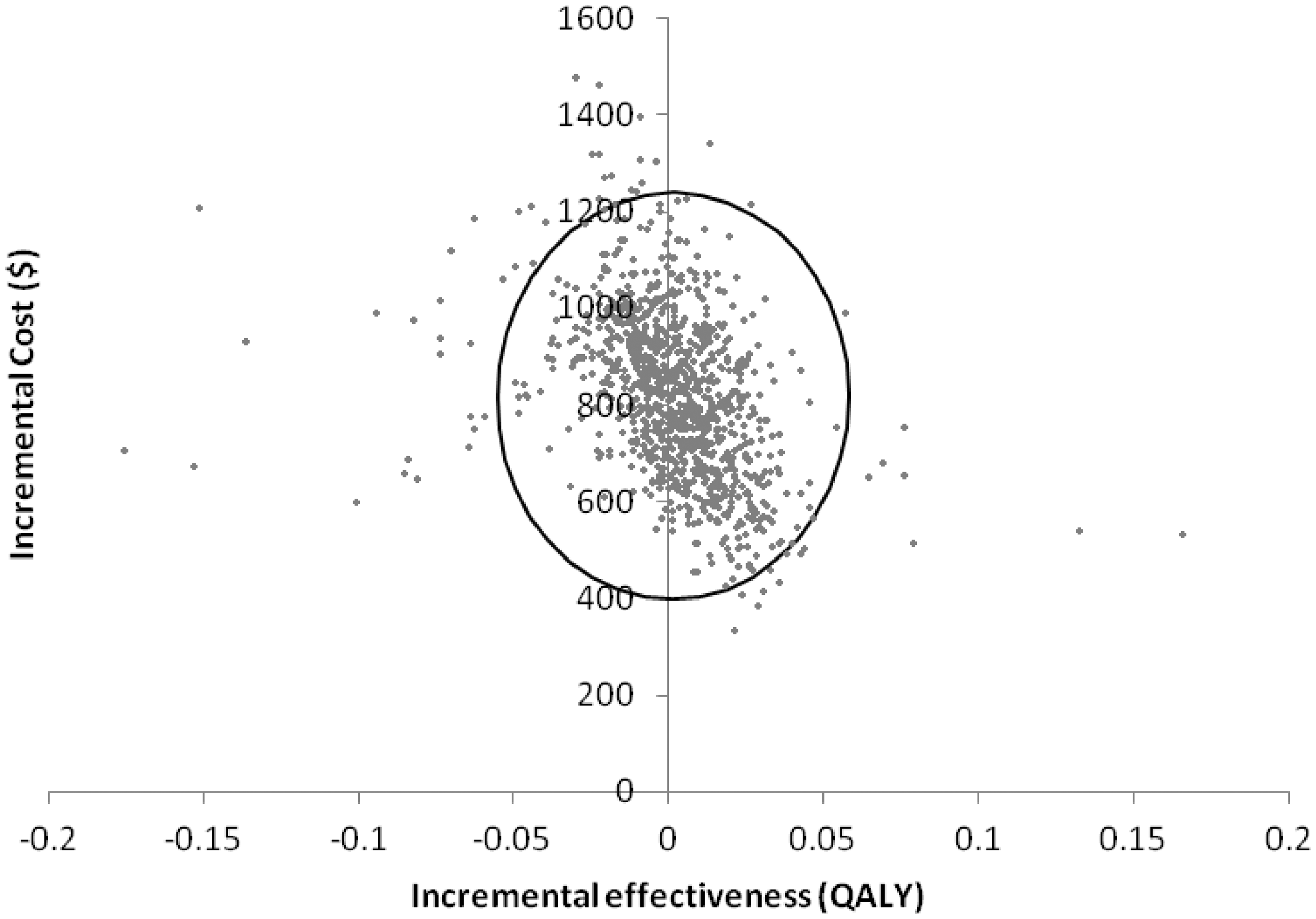
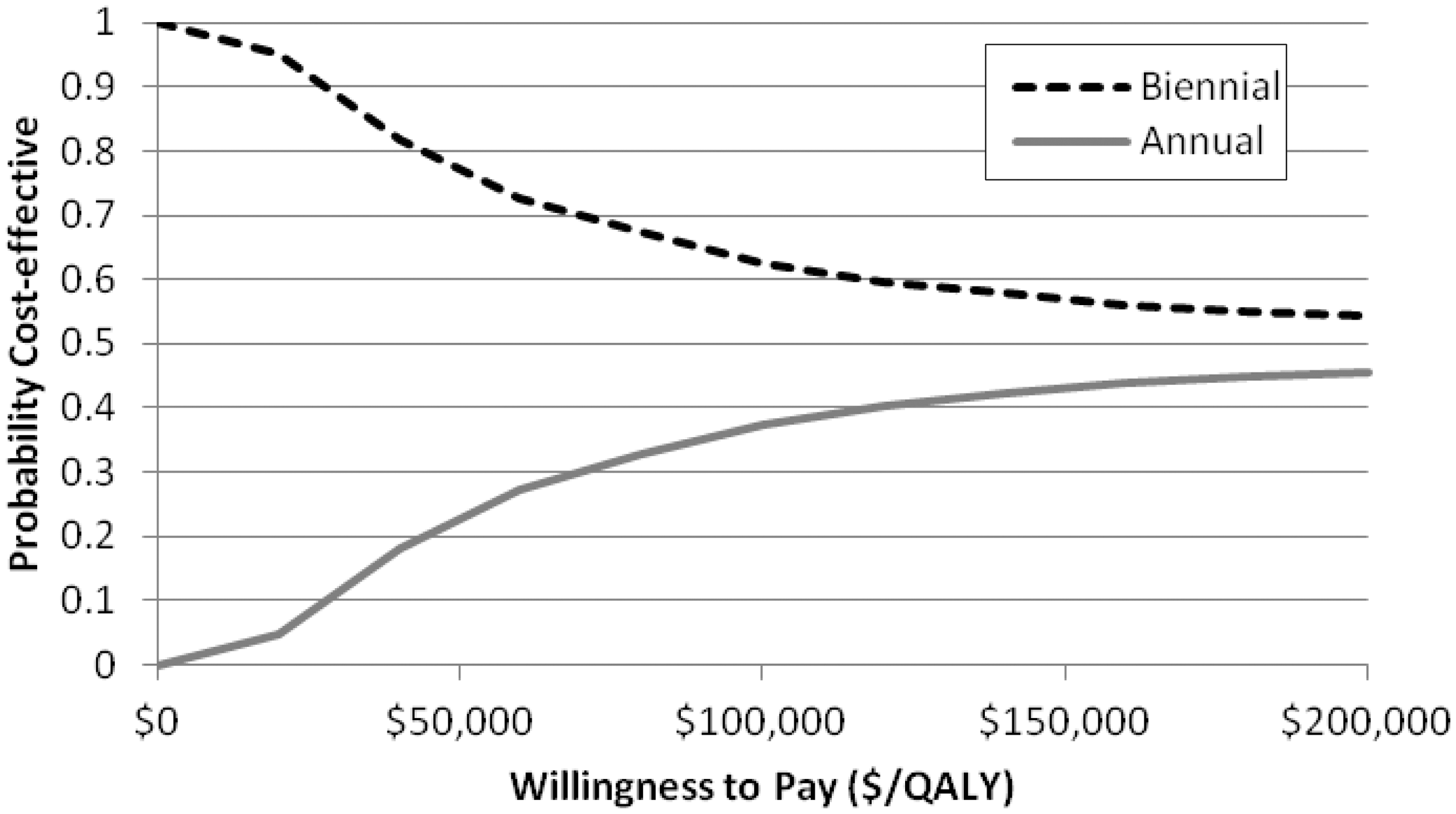
One-way sensitivity analysis indicates that the results were sensitive to the cost of a screening mammogram, with an ICER range of $412,423/QALY-$776,748/QALY as cost was varied from $35 to $99; results were far less sensitive to the cost of diagnostic work-up, which resulted in an ICER range of $546,448/QALY-$588,817/QALY as cost was varied from $101 to $261. Adjusting for utility had a large impact on cost effectiveness estimates: without applying utility weights, annual screening resulted in a mean gain in life expectancy of 0.0042 years (95% CI: −0.0201–0.0287), for an ICER of $194,281/life-year gained. Discounting assumptions also had a large effect. Without discounting, annual screening cost $82,387/QALY, while at a discount rate of 5%, annual screening was dominated by biennial screening, with a loss of 0.0013 QALYs at an additional cost of $666.
Discussion
Our model predicted that there is a small mean incremental benefit to women with dense breasts who undergo annual screening mammography from ages 50 to 79, compared with biennial screening; however, there is considerable uncertainty around that result, with 43% of simulations resulting in a loss of QALYs. The model also predicted a positive incremental cost in all simulations.
Annual screening mammography for women with dense breasts has only a 38% probability of being cost-effective at a willingness-to-pay threshold of $100,000/QALY, with a mean ICER of over $500,000/QALY. This value far exceeds the commonly cited cost-effectiveness thresholds of £20,000–£30,000/QALY (approximately $30,000–$45,000/QALY) 34 or $50,000/QALY. 35 A retrospective analysis of the cost-effectiveness on various screening mammography strategies found that the cost-effectiveness varied widely depending on screening interval and age at first and last screen. 36 The study did not evaluate the cost-effectiveness of annual screening mammography for women with high mammographic breast density, but the results show that frequency of screening mammography has a large impact of the cost-effectiveness of the intervention. For women with dense breast tissue, Schousboe et al. reported that biennial screening, relative to screening every 3–4 years, is cost-effective at a threshold of US$50,000/QALY in women over the age of 50. 22 The authors also found that targeting higher frequency screening at women with other risk factors – a family history of breast cancer, or a prior breast biopsy – improved cost-effectiveness. 22 However, they report that the ICERs for annual screening vs. biennial screening, for all age groups and categories of breast density, were over US$340,000/QALY. Our results indicate that there is little additional benefit from annual screening in all women with dense breasts, but further personalization of screening frequency may improve these outcomes.
The uncertainty around the benefit of annual screening mammography for women with dense breasts is consistent with the literature. The clinical impact of biennial versus annual screening has not been clearly demonstrated. While models have predicted a reduction in interval cancers between 36% and 50%,9,19 there is little trial or observational data on the potential stage shift and mortality reduction associated with higher frequency screening in this population. We found a trend towards reduced breast cancer mortality with annual screening, and a small but significant increase in breast cancer incidence. This difference in incidence probably represents overdiagnosis attributable to annual screening. Overdiagnosis was not explicitly built into the model; this difference arose from the observed cancer incidence in women screened annually versus biennially in the SMPBC. Estimated overdiagnosis rates vary widely 21 and there is little information on the effect of screening frequency. Compared with annual screening, biennial 37 or triennial 38 screening strategies reportedly decrease overdiagnosis by less than half. Screening women aged 70–79 in the model was also a contributor, as overdiagnosis increases with age. 39
Effectiveness estimates in the model are very finely balanced, with many simulations resulting in a net loss of QALYs. The sensitivity to utility weights also illustrates this result. Reductions in utility from overdiagnosis of breast cancer and false positive screening results largely offset the small gains in life expectancy from reduced breast cancer mortality. Discounting highlights the differential timing of these effects. The potential harms of screening (false positives and overdiagnosis) occur well before the potential benefits (reduced breast cancer mortality and extended life expectancy), and when these future benefits are discounted at 5%/year, annual screening results in a net loss of QALYs.
Transition probabilities for screening were calculated using SMPBC data from 2005–2007, and a limitation of the study is that during those years screening was conducted largely with film mammography. As reported by the Digital Mammography Imaging Screening Trial, digital mammography is more accurate than film in younger women and in women with dense breasts. 40 In BC, screening sites are currently being converted to digital mammography. The film mammography data probably underestimates the effectiveness of current digital screening practice, but because both the intervention and comparator arms of the current model would be affected, it is difficult to infer the impact on cost-effectiveness. Using data for film mammography also underestimates screening cost; however, digital mammography is potentially cost-effective in this population of high-risk women. 41 Screening with tomosynthesis, ultrasound, or magnetic resonance imaging has also been proposed in this population. Digital breast tomosynthesis, which uses a series of images to produce three-dimensional data, has been found to have significantly higher sensitivity and specificity than digital mammography. 42 Breast MRI has been recommended for women with a lifetime risk of breast cancer over 20–25%, 43 but there is little data on its effectiveness as a screening tool in women for whom breast density is their only risk factor. The addition of ultrasound to mammography for women with high breast density has been found to significantly increase the sensitivity of screening, but with an increase in the risk of false positive results. 44 Recently-adopted legislation in some American jurisdictions now requires radiologists to inform women of their breast density and recommend additional screening with ultrasound; however, more long-term clinical effectiveness and cost-effectiveness evidence is required to better understand the impact of these recommendations 45 and to inform future screening policy.
A limitation of this model is that it considered costs and outcomes in a defined population of women with dense breasts. The cost of assessing breast density among all screening participants, the accuracy of density assessment, or changes in women’s breast density over time were not considered.
Furthermore, current definitions of breast density, measured as the relative area of the breast made up of fibroglandular tissue, do not reflect the three-dimensional distribution of breast tissue. Methods to model volumetric breast density from two-dimensional mammography images or from three-dimensional tomosynthesis are being validated against area-based measurements. 46 The clinical significance of volumetric breast density as a predictor for breast cancer risk or screening effectiveness is not yet known.
The model relied heavily on routinely collected observational data from the SMPBC and the BC Cancer Registry. The data are representative of local experience, but also present some limitations, as observational data may suffer from unadjusted confounding in a way that screening effectiveness estimates from randomized controlled trials do not.
This study is the first to evaluate the cost-effectiveness of annual screening mammography for women who have high mammographic breast density. The results indicate that there is considerable uncertainty about the incremental effectiveness of the intervention, but also suggest that annual screening for women aged 50–79 is not cost-effective, with a mean ICER of over $500,000/QALY. Further research on the comparative effectiveness of screening strategies for women with high mammographic breast density is warranted, as are subsequent economic evaluations exploring the value-for-money implications of these strategies.
Footnotes
Acknowledgements
The authors thank Dr. Paula Gordon, Medical Director of the Sadie Diamond Breast Program at BC Women’s Hospital, for her contributions to the study and comments on the manuscript. The authors also thank members of the Screening Mammography Program of British Columbia, the British Columbia Breast Tumour Group, and participants in the provincial screening mammography programme.
Funding
This study was funded by Canadian Institutes of Health Research grant no. 162964 as part of a programme of research into setting priorities for cancer control. The Canadian Centre for Applied Research in Cancer Control is funded by the Canadian Cancer Society.
Conflict of interest
The authors have no conflicts of interest to declare.
