Abstract
Objectives
We here present the results of the first eight years of the newborn hearing screening programme in Iran, with a view to establishing the prevalence of hearing impairment among infants, and the efficacy of the programme.
Methods
A total of 3,350,995 infants were screened using the series method of transient evoked otoacoustic emissions (TEOAEs)/automated auditory brainstem responses (AABRs), between 2005 and 2012. The infants were first tested for TEOAEs (three times). Based on the results of this test, the positive cases were referred to the next stage, where they were tested for AABRs. If they also tested positive on AABRs, they were referred to the diagnostic and rehabilitation stages.
Results
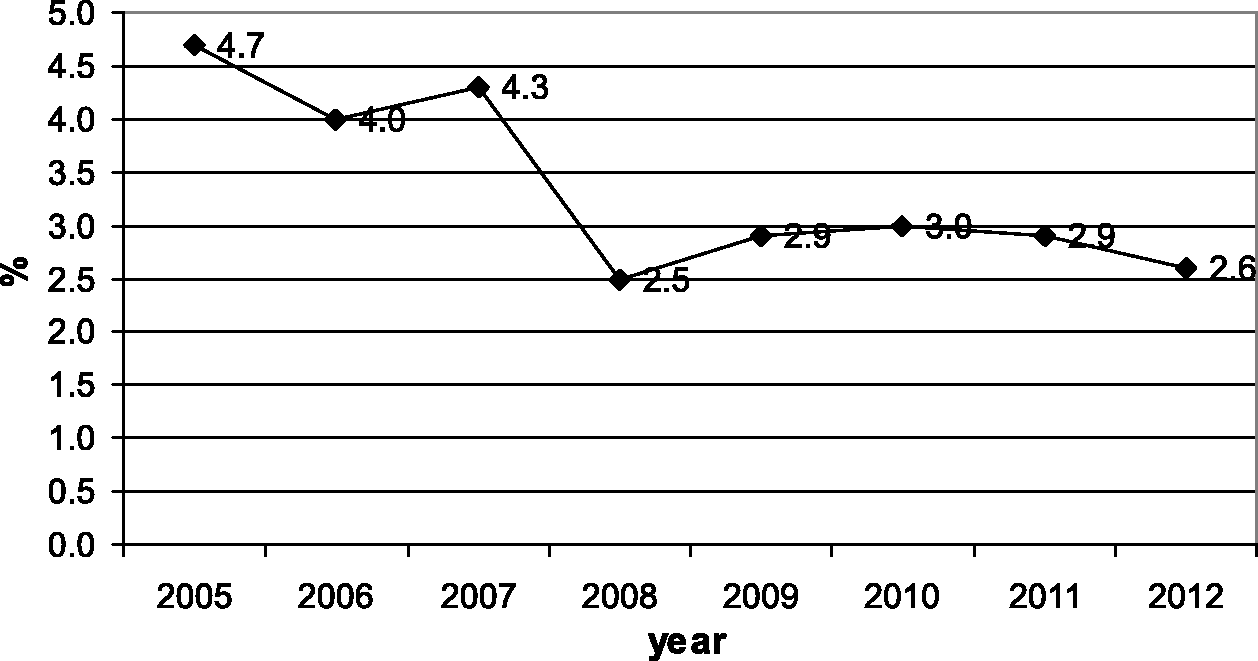
Results of this study indicated an infant hearing impairment prevalence of 3 per 1000. Although this rate was as high as 5 per 1000 in the early years of the programme, it decreased to 2.6 per 1000 in the last year. The absolute referral rate was 14.5% in the first stage, which decreased to 0.9% and 0.2% in the second and the third stages, respectively. The follow-up rate was 70% in the first stage, which increased up to 73% and 85% in the second and the third stages, respectively.
Conclusion
The study results suggest that the prevalence of hearing impairment in infants in Iran is comparable with that in developed and developing countries, and that the series TEOAEs/AABRs method used in the screening programme in Iran is efficient.
Introduction
Hearing impairment is one of the leading causes of infant disability throughout the world, with both short- and long-term adverse effects on infant development, including verbal, physical, cognitive, and social skills, which can negatively influence their education and occupational skills.1–4 At least one-third of infants with hearing impairment will have neuro-deficiencies later in life. 5 Risk factors for hearing impairment include family marriage, infectious diseases during the pregnancy, some ototoxic drugs, birth deficiencies, neonatal jaundice, and ear infection. 6 As hearing impairment may occur without apparent symptoms, lack of knowledge among family members, lack of knowledge about available care centres, and lack of knowledge about the prevalence and serious side effects of this disability among specialists, mean that cases may be detected so late that the child suffers permanent severe disability. This may have been avoided with earlier intervention. 5
Research indicates that screening programmes are the most effective way for early detection of hearing impairment among infants, and can improve their development. 7 Studies suggest that infants whose hearing impairment has been detected before the age of six months have more capabilities than those with later (8-10 months) diagnosis. Early and effective intervention improves verbal and cognitive skills, 8 thus demonstrating the importance of effective screening programmes.9–12
The World Health Organization encourages the early detection of infant hearing impairment, 13 and in the US and UK, for example, 99% of the newborns are screened for this disability before hospital discharge.14,15,16 Poland has now also introduced a similar programme. 17 In developing countries, such as Brazil and Oman, hearing screening programmes are just being developed. 18
Although studies have indicated that the burden of disease due to hearing impairment is much higher in Asia and Africa compared with other regions of the world, 19 hearing impairment in Iran was diagnosed at 2–2.5 years. From 2005, the State Welfare Organization of Iran introduced a new screening programme for early hearing detection and intervention (EHDI) (between 1 and 6 months). Implementation and coverage of the programme varies greatly from city to city, and region to region. The EHDI programme is not legislatively mandated. This paper presents the results of the first eight years of the screening programme in Iran.
Methods
The hearing screening programme in Iran was introduced in 2005, and utilizes the series method of OAEs/AABR to detect permanent bilateral or unilateral hearing loss of 30 dB or worse in the range 0.5–4 KHz. The programme is currently conducted in 381 maternity centres in 323 cities, with plans to extend this coverage. For healthy babies, screening is recommended by the 3rd day of life and before discharge from the maternity ward in hospital, and must occur before the 14th day of life. For severely ill babies and those who have failed to complete the procedures, screening is performed as soon as is practical considering the baby’s medical condition, and before the end of the 1st month of life.
Figure 1 is a flow-diagram of the series method used in the study. All infants are first tested for transient evoked otoacoustic emissions(TEOAEs) (three times for each case), and are then categorized into the following groups based on their test results:
Infants with pass response: in this group, the infants were divided into two groups:
Low-risk infants: educational brochures containing information on hearing, speech, language, and cognitive skills developmental milestones were given to the parents of these infants before discharge. The parents were informed if re-screening was necessary after discharge. High-risk infants: these infants were tested for automated auditory brainstem responses (AABRs) within one month of the TEOAEs test. Infants with at least one risk factor fell under this category. Infants with referral response: all infants with this kind of response were tested for AABRs as soon as possible, and all before the age of 1 month. They were then categorized into two groups based on their AABRs results:
Infants with pass response: the infants were further categorized into two groups:
a1 low-risk infants: educational brochures containing information on hearing, speech, and language development milestones were given to the parents of these infants before discharge. a2 high-risk infants: Although these infants passed the AABRs test, they were followed up regularly (twice before the age of 6 months, and once before the age of 9 months in the first year, once before the age of 18 months in the second year, and once before the age of 30 months in the third year). During these follow-ups, a diagnostic AABR was carried out and the stages of hearing development were checked in each session. Infants with referral response: these infants had some level of hearing impairment and were referred to the closest diagnostic centre, where the type and level of hearing impairment was diagnosed before the age of 3 months. Infants with incomplete response: for some infants, it was not possible to perform the TEOAEs test three times, mainly because of the existence of any mass in their ears, environmental noise, uncalibrated instruments, parents’ refusal for their child to take the test, or unsatisfactory conditions of the infants. This group was tested again after one week. They were then categorized into the above groups, based on the test results. If the response was again incomplete, they were tested for AABRs. Flow-diagram of the series method used in the study.
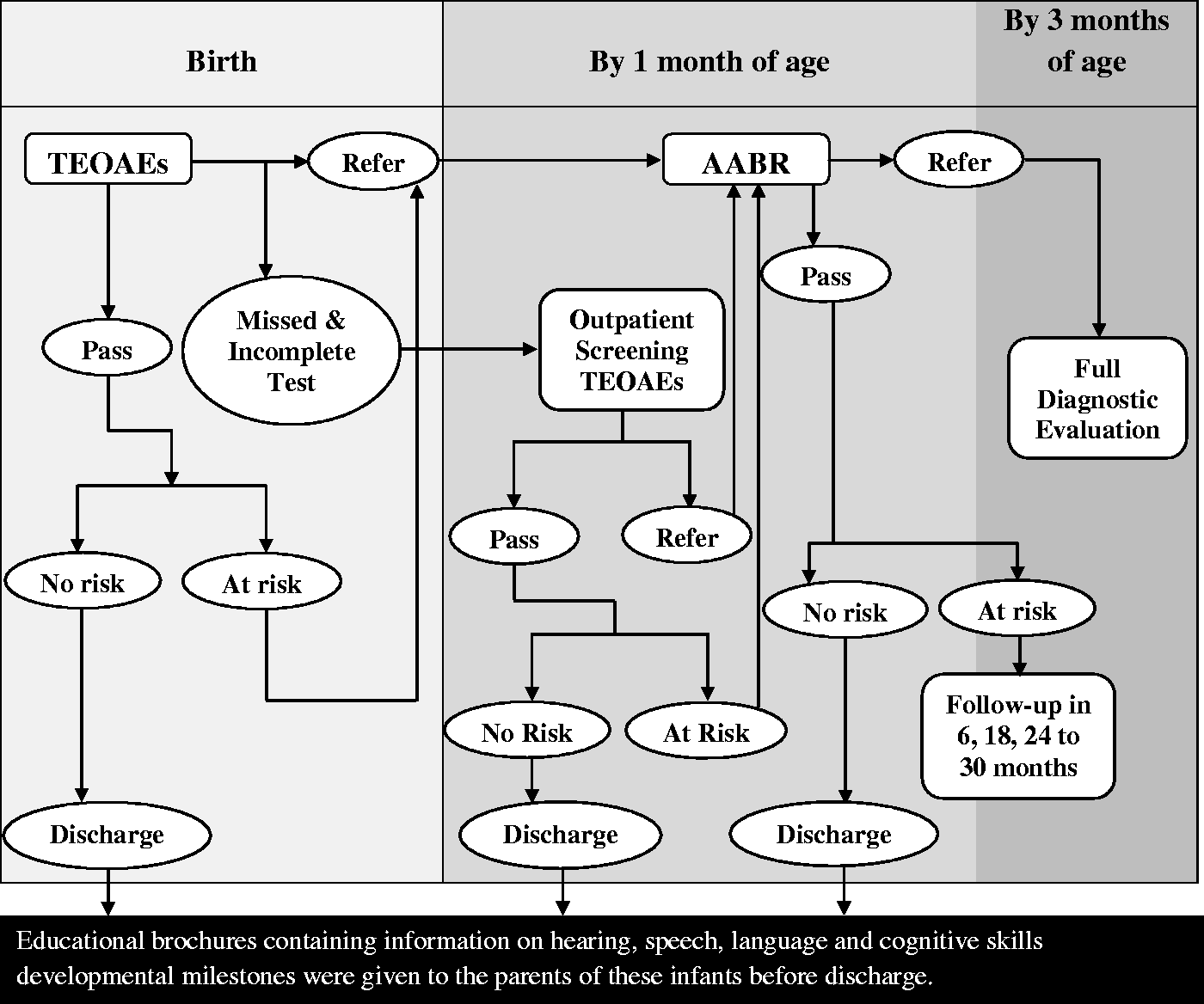
The newborn hearing screening is mostly performed by audiologists, nurses, midwives and trained health technicians. The programme is variously paid for by parents (financially supported by the state welfare organization of Iran), the government (for public hospitals), or hospitals (partially funded in Iran).
Results
The total number of infants screened, referred, and diagnosed with hearing impairment during the screening programme.
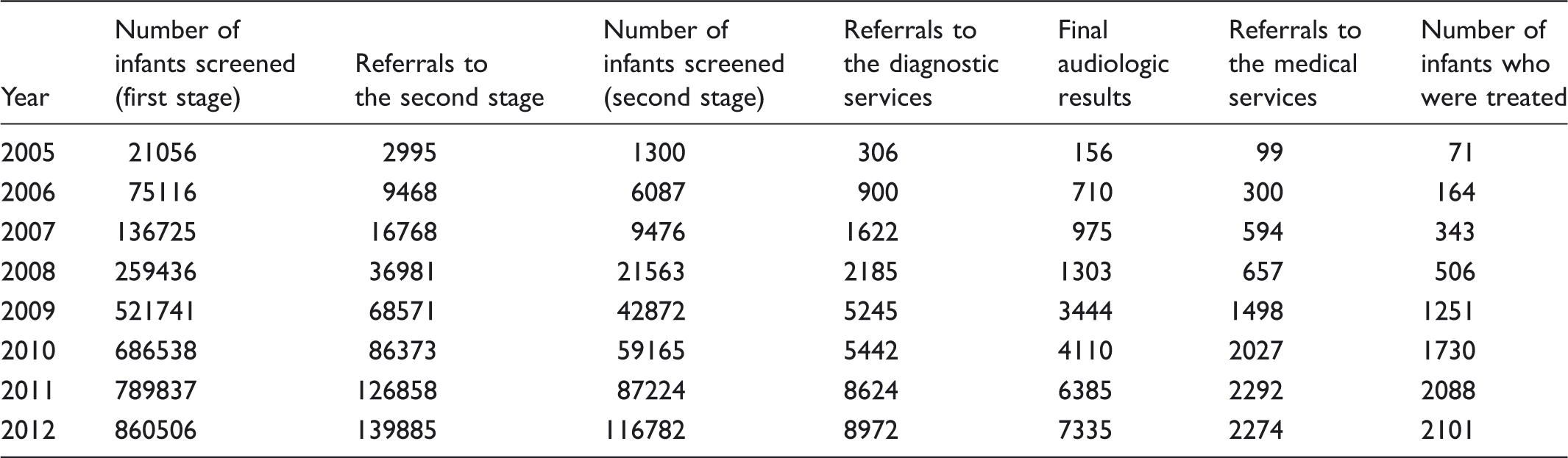
Figure 2 shows coverage rates of early hearing detection and intervention (EHDI) programmes in separate provinces across the country from 2005 to 2012. The screening rates varied from 22% in two provinces, up to 100 %. Screening rates among provinces that implemented the Universal Newborn Hearing Screening protocol significantly increased from 2005 through 2012. The overall screening rates increased from about 10% in 2005 to greater than 60% in 2012. Coverage is currently estimated as higher than 65%.
Coverage rates of early hearing detection and intervention (EHDI) programmes in separate provinces in Iran from 2005 to 2012.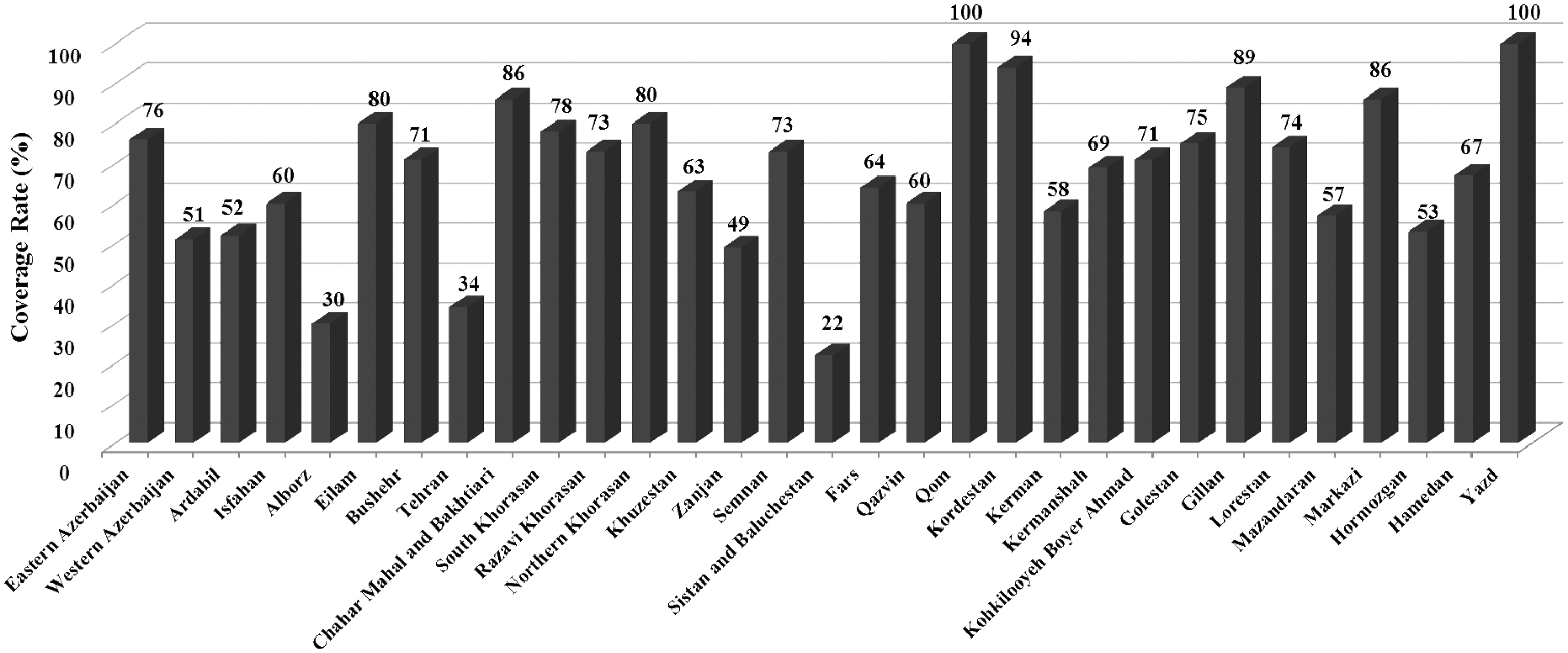
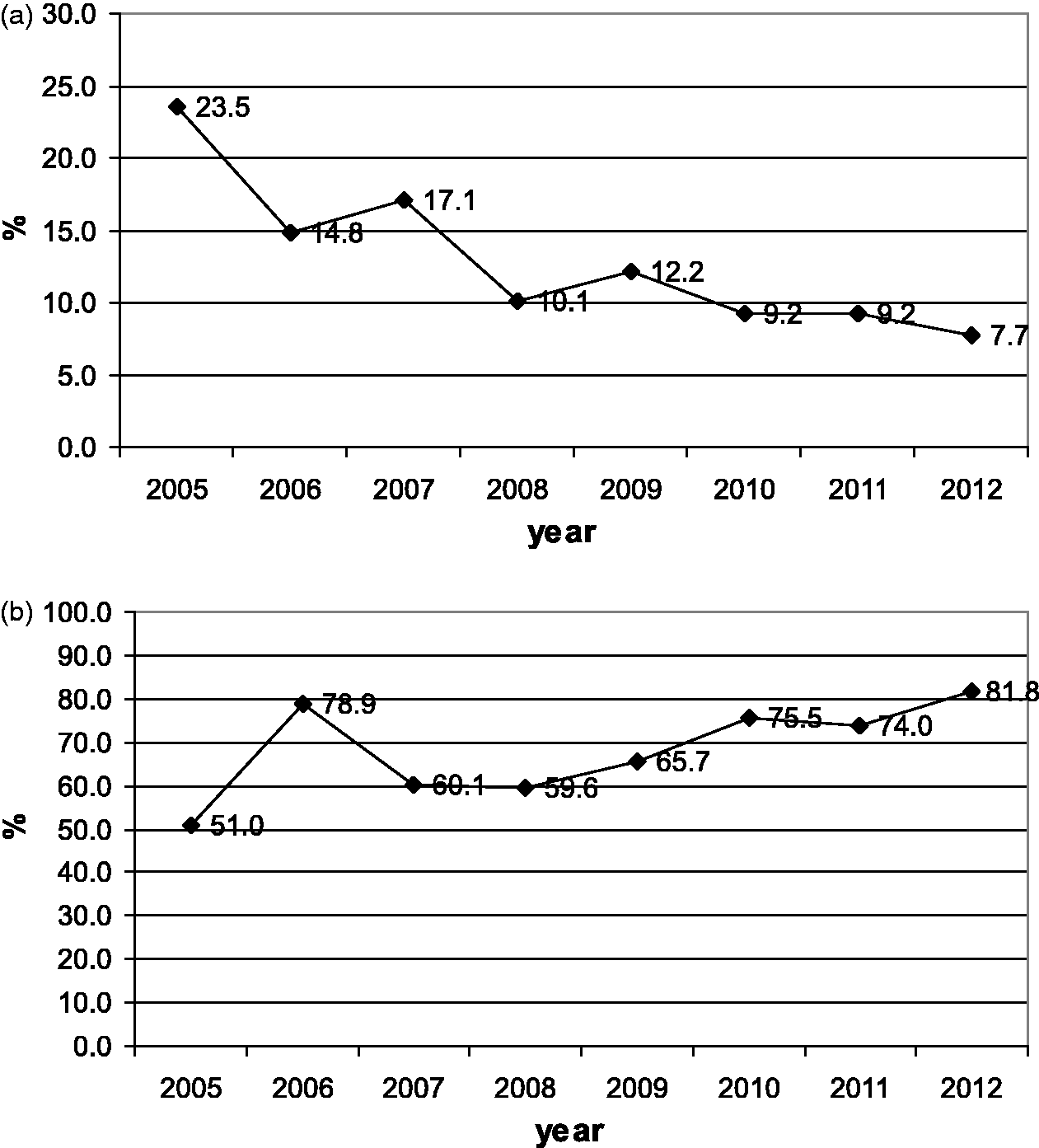
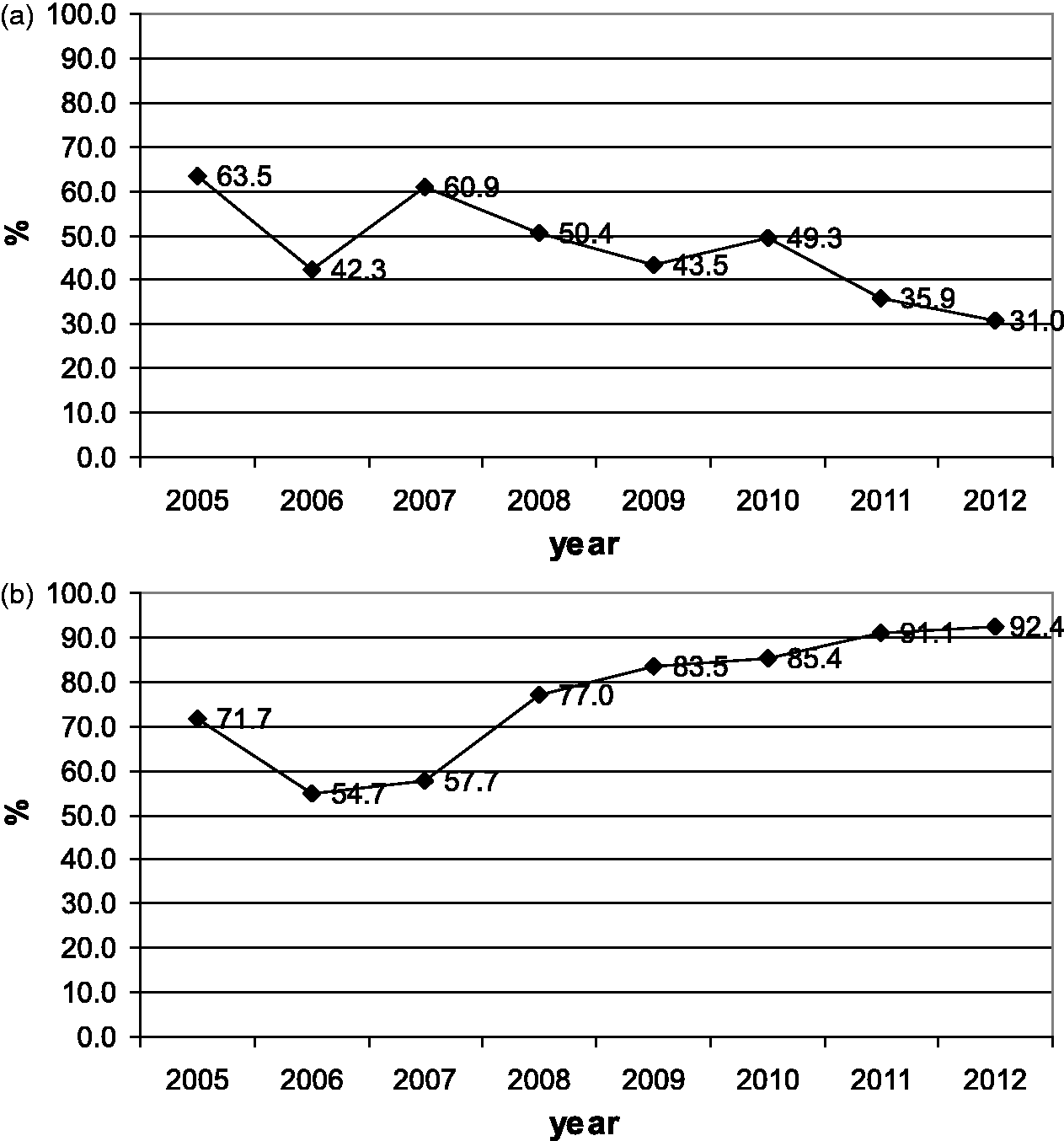
Figure 3a and b show the percentages of infants referred to and tested in the second stage for each year of the study period. During the study period, the percentage of infants referred to the second stage increased from 14% to 16%, and the percentage of infants tested in the second stage increased from 43% to 83%. Figure 4a and b display the same percentages for third stage referral and testing. The percentage referred to the third stage decreased from 23% to 8%, while the percentage tested increased from 51% to 81%. Figure 5a and b give referral and intervention rates for the intervention stage, with referral decreasing from 63% to 31%, and intervention increasing from 71% to 92%. Figure 6 indicates the prevalence of hearing impairment among Iranian infants during the study period. Prevalence decreased from 4.7% in the first year to 2.6% in the eighth year of the study.
The percentage of infants referred to the second stage (a) and the percentage of infants tested in that stage during each year of the study period (b). The percentage of the infants referred to the third stage (a) and the percentage of infants tested in that stage during each year of the study period (b). The percentage of the infants referred to the intervention stage (a) and the percentage of infants in whom intervention occurred in that stage during each year of the study period (b). The prevalence of hearing impairment among Iranian infants during the study period.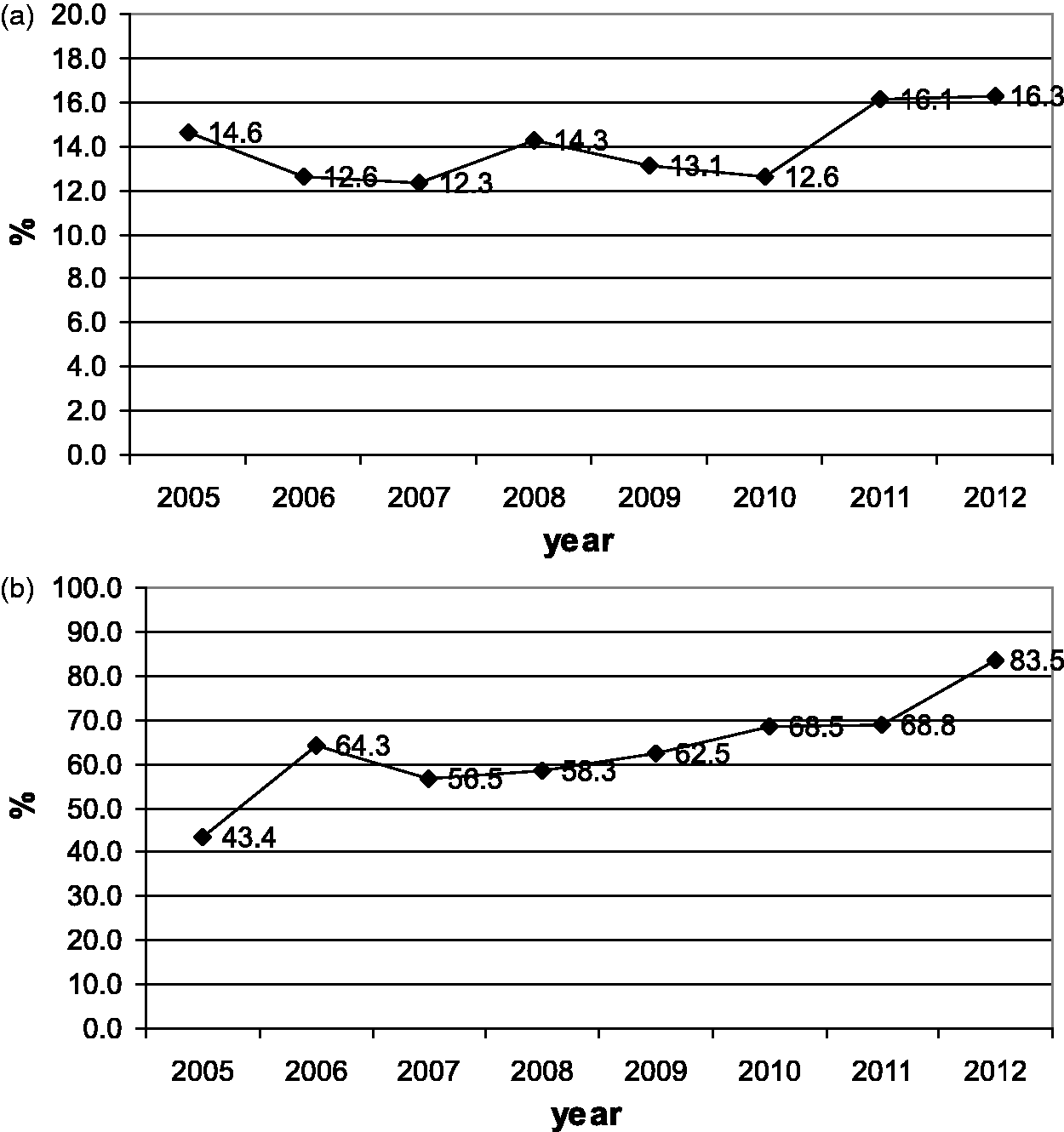
Discussion
This is the first report of statewide comprehensive newborn hearing screening outcomes in Iran from the first eight years of the programme. There are about 1.4 million births in Iran each year, over 90% delivered at a hospital or maternity centre. There are more than a thousand private OB-GYN clinics across the country.
Two different models are currently used to identify hearing loss in infants and young children in Iran: the hospital-based universal newborn hearing screening programme, recommended by the State Welfare Organization of Iran and by health professionals, and outpatient-based newborn screening which is carried out in some provinces. The results from the screening programme are of great importance in providing the evidence for the burden of hearing impairment in Iran, and in estimating the effectiveness of such programmes in the country. 11 In addition, it should be noted that the series method used in the present study is effective; previous studies have indicated that using of a single-step method or the parallel method can significantly underestimate the prevalence of hearing impairment. 11
Results from this study indicate that the mean prevalence of severe to profound hearing impairment in Iran is 3 per 1000. This figure was 5 per 1000 in the first year, but decreased to 2.6 per 1000 in the last year of the study. Comparison of this rate with those reported by previous studies revealed that the prevalence of hearing impairment in Iran is similar to that of developed as well as developing countries. For example, a 2007 pilot study in South Africa reported a prevalence of 3 per 1000; 11 another study in Western Cape province, South Africa, indicated a hearing impairment prevalence of 4.5 per 1000. 20 This difference might have been due to the fact that the second study was conducted in one part of South Africa, while the first one was a national study but only in the private sector. 20 A screening programme on 19700 newborns in Italy found a prevalence of 1.7 per 1000, 21 and similar findings have also been reported by other studies conducted in developed countries. 22
This study considered both unilateral and bilateral fails on newborn hearing screening as referrals. All newborns with unilateral referrals were considered in the same way as infants with bilateral referrals and followed similar diagnostic work-up protocols. According to Chang et al 2009, unilateral referrals on neonatal hearing screening should be considered a risk factor for significant hearing loss in the opposite ear that initially passes the screen. 23
The absolute referral rate in different stages of the screening programme is an indicator of the efficacy of the programme; 11 a decreased absolute referral rate is translated into increased efficacy. In the present study, the average absolute referral rate was 14.5% in the first stage, decreasing to 0.9% and 0.2% in the second and the third stages, respectively. This is an acceptable range of referral rates, similar to those observed by studies in both developed and developing countries.17,24 For example, in the Swanepoel (South Africa) study, the overall referral rate was 11%, 11 the study by Friderichs et al reported a referral rate of 9.5% in the first stage (in clinics), decreasing to 3% in the second stage, 20 and Wang et al. in China, reported a referral rate of 14.7%. 25 Even lower referral rates have been observed in studies conducted in other countries.21,26
The follow-up rate is also critical to evaluate the efficacy of the screening programme. The average follow-up rate in the Iranian programme was 70% in the first stage, increasing to 73% and 85% in the second (diagnostic) and the third (intervention) stages. These referral rates are quite high, but comparable with those observed by previous studies. For example, Friderichs et al reported follow-up rates of 85% in clinics and 91.8% in hospitals, 20 slightly higher than those observed in the present study. In the early years of the Iranian programme the follow-up rates were low, but could be significantly increased by taking necessary steps. The average follow-up rate in the first three years of the programme were 57%, 65%, and 58% for the first, second, and third year stages, respectively. However, the corresponding values for the last five years were 71%, 74%, and 88%.
One of the strengths of the programme was the series method, in which both TEOAEs and AABRs tests were used. Results from the previous studies have indicated that this method leads to more accurate estimates of the prevalence of hearing impairment, mainly because in the screening programmes relying solely on TEOAEs, auditory neuropathy/auditory dyssynchrony (AN/AD) is not accounted for, which in turn leads to underestimation of the prevalence. In addition, use of either TEOAEs or AABRs as the sole stage in the screening programme increases the referral rate, which in turn decreases the efficacy of the screening programme. 27 The cost of the series method is higher than that of single-step methods.
The biggest challenges in the programme were the lack of human resources (especially audiological professionals), difficulty with follow-up of those who did not pass hearing screening (tracking system), lack of national and provincial tracking software for universal newborn hearing screening or EHDI across the country, and the difficulties of centralized programme implementation in remote locations and/or rural areas. Although the results were lower than expected, the newborn hearing screening programme had only been implemented for a limited time and follow-up and data tracking were not satisfactory. Data-collection efforts are now ongoing, and more accurate figures are expected to become available.
Conclusions
The results from this study suggest that the prevalence of hearing impairment in infants in Iran is comparable with that of both developed and developing countries. The referral and follow-up rates at different stages during the programme also suggest that the series TEOAEs/AABRs method is efficient in Iran. Continuation and improvement of this screening programme provide a great opportunity for early detection of hearing impairment in Iranian infants, which in turn can significantly decrease the burden of disease due to hearing impairment.
Conflict of interest
The authors have no relevant financial interest in this article.
Footnotes
Funding
The State Welfare Organization of Iran.
Acknowledgements
The study for this paper was financially supported by the State Welfare Organization of Iran. We gratefully acknowledge the support and generosity of the ENT and Head & Neck Research Center of Iran University of Medical Sciences (IUMS) for Arts Research, without which the present study could not have been completed.
