Abstract
Corrosion is a significant challenge in maintaining material performance and longevity, particularly in harsh environments, necessitating the development of innovative protective materials. In this study, a novel composite was developed by combining tannic acid-benzoxazine (TA-BZ), epoxy resin, and zinc oxide (ZnO) nanoparticles, named Epoxy-TA-BZ-1-C, Epoxy-TA-BZ-2-C, and Epoxy-TA-BZ-4-C. These composites were cured at 200°C, with and without ZnO, to investigate the effect of ZnO addition on enhancing anticorrosion, mechanical, and thermal properties. The bio-based TA-BZ matrix, synthesized via Mannich condensation, demonstrated inherent thermal stability and corrosion resistance. Epoxy improved structural integrity, while ZnO nanoparticles enhanced barrier properties and antimicrobial activity. Comprehensive analyses, including FTIR, NMR, TGA, SEM, TEM, electrochemical, and mechanical studies, confirmed improved cross-linking density and structural optimization. Potentiodynamic polarization studies revealed a corrosion rate of 0.069 mm/year for Epoxy-TA-BZ-4-ZnO-C, highlighting its superior performance. These findings underscore its potential for advanced applications in demanding environments.

Keywords
Introduction
Corrosion poses significant challenges across industries, causing material degradation and economic and safety concerns. Traditional anticorrosion methods, such as coatings and metal treatments, often pose environmental and health risks, highlighting the need for sustainable, biocompatible materials with superior anticorrosion properties. The development of these materials is crucial for extending infrastructure lifespan, reducing maintenance costs, and minimizing environmental impacts.1–5
Benzoxazine resins, known for their thermal stability, mechanical strength, and flame resistance, show promise as advanced anticorrosion coatings. These resins are durable, resistant to moisture penetration—a key factor in corrosion—and offer environmental advantages through synthesis without toxic catalysts. Furthermore, benzoxazines demonstrate strong adhesion and self-healing properties, making them effective for protecting metal substrates.6,7 Research indicates that incorporating additives can enhance their barrier properties, often surpassing traditional coatings like epoxy. Typically synthesized from phenols, primary amines, and formaldehyde, benzoxazine resins are high-performance thermosets. Ongoing research aims to explore alternative materials and synthesis methods to broaden their use in more corrosive environments.7–10
Recent environmental concerns have shaped corrosion prevention strategies, with plant-derived extracts, especially polyphenols, gaining interest for their cost-effectiveness, biodegradability, and sustainability.11,12 Extensive studies on polyphenols have demonstrated notable inhibition rates, often exceeding 85%, motivating further electrochemical and theoretical research. Tannic acid, a natural polyphenol, has emerged as a promising alternative to synthetic phenols in benzoxazine monomer synthesis. Its multiple reactive phenolic hydroxyl groups offer antioxidant properties and numerous sites for benzoxazine formation. Additionally, recent research is exploring alternative corrosion inhibitors, such as Schiff base-based cationic gemini surfactants, for their effectiveness in protecting carbon steel under acidic conditions.11–16
Incorporating tannic acid into benzoxazine synthesis aligns with the rising focus on bio-based, sustainable materials and addresses the demand for eco-friendly polymers. Tannic acid enhances hydrogen bonding within the polymer matrix, thereby improving mechanical properties and thermal stability. Its antioxidant nature helps counter oxidative degradation, making tannic acid-modified benzoxazines particularly effective in corrosive environments.17–20 Typically, tannic acid reacts with aniline and paraformaldehyde during synthesis, which strengthens polymer rigidity and thermal resistance due to aniline’s structural characteristics. Solvents such as ethanol and toluene are commonly used to enhance solubility and reaction efficiency, leading to high purity benzoxazine monomers.20–33
Recent research highlights the efficacy of tannic acid-based benzoxazine coatings in mitigating corrosion. A particular study demonstrated that these coatings significantly enhance corrosion resistance on steel substrates by restricting water and oxygen infiltration. 34 Furthermore, tannic acid-enriched benzoxazine coatings have shown superior saltwater corrosion resistance in marine environments, surpassing conventional epoxy coatings due to their improved adhesion and barrier properties.35,36 The integration of zinc oxide (ZnO) nanoparticles substantially improves the anticorrosive and mechanical properties of polymer coatings. ZnO nanoparticles are renowned for their superior barrier properties, which enhance the density and compactness of the polymer network, effectively obstructing the diffusion of corrosive substances including water, oxygen, and ions. Improved impermeability is essential for extending the durability of coatings, especially in hostile and corrosive settings.13,36,37
Recent research highlights the efficacy of incorporating ZnO nanoparticles into polymer coatings to significantly enhance corrosion resistance and mechanical strength. ZnO improves barrier performance by limiting the ingress of moisture, oxygen, and ionic species, while also imparting UV stability, abrasion resistance, and antibacterial properties—attributes essential for durability in aggressive environments.8,9,38–43 In epoxy–benzoxazine systems, the addition of ZnO nanoparticles has been shown to form dense, dual cross-linked networks that markedly reduce corrosion rates and enhance mechanical performance under industrial stress.14,17,46,47 Similarly, in tannic acid–based benzoxazine matrices, ZnO promotes strong cross-linking during thermal ring-opening polymerization, leading to improved thermal stability, chemical resistance, and tensile strength44,45. This study builds upon these findings by examining the synergistic effects of ZnO within tannic acid–epoxy–benzoxazine hybrid systems. Despite existing research, comprehensive comparisons of such multifunctional coatings remain limited. The present work addresses this gap by evaluating the structural, thermal, and anti-corrosive performance of ZnO-enhanced composites under corrosive and high-stress conditions. The incorporation of ZnO into these composites reinforces their physical integrity and improves UV resistance, reducing photodegradation, which is essential for extending the lifespan of coatings subjected to severe environmental conditions. The photocatalytic characteristics of ZnO provide self-cleaning advantages by diminishing the buildup of organic deposits that may promote microbial proliferation, hence further mitigating corrosion hazards. The mechanical enhancements offered by ZnO-epoxy-benzoxazine composites—specifically improved adhesion, hardness, and abrasion resistance—are especially advantageous for industrial applications that need high durability and corrosion resistance.13,14,48
Zhou et al. have shown that benzoxazine-epoxy-ZnO composite coatings on mild steel substrates have markedly reduced corrosion currents relative to conventional coatings. This discovery underscores the synergistic benefits of the dual-network architecture of epoxy paired with the protective properties of ZnO, accentuating the promise of both materials as durable, long-term corrosion prevention solutions in industrial applications. This performance highlights the suitability of these advanced composites as substitutes for traditional materials in essential applications where corrosion resistance is paramount.8,10 Selvaraj et al. reported that cardanol-derived benzoxazine–epoxy hybrid systems exhibited notable corrosion resistance and dielectric performance, reinforcing their applicability in protective coatings for harsh environments. 49 Zhang et al. demonstrated that ortho-benzoxazine chemistry offers unique opportunities for designing highly cross-linked networks with tailored thermal and mechanical performance, paving the way for high-performance thermoset systems. 50
This work explores the development of a new composite material composed of tannic acid-benzoxazine (TA-BZ), cross-linked with epoxy resin, and reinforced with zinc oxide (ZnO) nanoparticles. Tannic acid, a natural polyphenol, provides mechanical strength, thermal stability, and bioactivity, making it ideal for drug delivery and tissue engineering. Benzoxazine chemistry enhances the composite by forming robust cross-linked networks at lower curing temperatures, improving overall material performance.5–8,32 The addition of ZnO, known for its antibacterial and UV-filtering properties, further optimizes the composite, making it a promising option for biocompatible and eco-friendly applications.9–12,51–58 The primary objectives of this study are to evaluate the influence of curing temperature on composite formation and properties, characterize the anticorrosion performance using comprehensive electrochemical techniques, and assess the environmental durability and mechanical integrity of the optimized coating system. This investigation aims to contribute to the development of sustainable, high-performance coating technologies for industrial applications demanding exceptional corrosion resistance. By integrating natural and synthetic components, this study offers a practical solution to corrosion challenges in various industries, emphasizing the need to balance corrosion resistance with mechanical strength and promoting the use of sustainable materials in industrial applications.56–70
Materials and methods
Chemicals
All chemicals and reagents, including Tannic Acid (99.5%), paraformaldehyde (95%), Aniline, Ethanol, Methanol, chloroform (99.5%), toluene (99.5), ZnO, the epoxy resin and its hardener used in this research were used as supplied by Sigma Aldrich and Fisher Scientific without further purification. The epoxy resin (Component A) was bisphenol-A diglycidyl ether (DGEBA) with an approximate molecular weight of 380 g/mol, and the hardener (Component B) was a cycloaliphatic polyamine curing agent, supplied as a two-component system.
Instrumentation and methodology
A comprehensive suite of analytical techniques was employed to evaluate the anticorrosion properties of the samples. The methods covered surface, structural, thermal, mechanical, and electrochemical analyses, as outlined below.
Surface and structural analysis
Surface morphology was examined using a Nova Nano SEM 450 (FEI, USA) and topography via a Dimension Icon Atomic Force Microscope (Bruker, USA) in tapping mode (10 µm × 10 µm areas). Crystalline structure and phase composition were assessed with a D8 Advance X-Ray Diffractometer (Bruker, Germany), using Cu Kα radiation (λ = 1.5406 Å) across the 10°–80° 2θ range.
Thermal analysis
Thermal stability and phase transitions were analyzed using a TGA/DSC 3+ Thermogravimetric Analyzer (Mettler Toledo, Switzerland), with samples heated from 25°C to 800°C at 10°C/min under nitrogen.
Mechanical properties
Mechanical behavior was characterized by using an Instron 5967 Universal Testing Machine (Instron, USA) at a crosshead speed of 1 mm/min to assess material strength and ductility.
Electrochemical analysis
Electrochemical properties were assessed using a PARSTAT 4000A Potentiostat/Galvanostat (Princeton Applied Research, USA) in a three-electrode setup with 3.5 wt% NaCl solution at 25 ± 2°C. Potentiodynamic Polarization curves were recorded from −250 mV to +250 mV versus OCP at 0.5 mV/s, yielding corrosion potential (Ecorr) and current density (Icorr). Cyclic voltammetry was performed between −0.8 V and +0.8 V (vs SCE) at 50 mV/s, providing data on current density, passivation behavior, and protective layer stability.
This holistic methodology combined multiple analytical techniques to investigate corrosion resistance mechanisms and inform the development of more effective corrosion protection strategies.
Methods
Synthesis of tannic acid benzoxazine (TA-Bz) monomer
The TA-Bz monomers were synthesized using a one-pot Mannich condensation method. This method involved varying molar ratios of tannic acid (TA), aniline, and paraformaldehyde (PFA), resulting in three distinct monomer formulations: TA-BZ-1, TA-BZ-2, and TA-BZ-4, as illustrated in Scheme 1. Specifically, 0.588 mmol of tannic acid reacted with varying amounts of aniline (29.3 mmol, 4.73 mmol, and 1.83 mmol) and paraformaldehyde (58.7 mmol, 14.65 mmol, and 5.87 mmol) in a round-bottom flask containing a toluene-ethanol mixture (1:2). The reaction was carried out at 70°C for 24 h. After the reaction, the solvent was evaporated, and the resulting product was recrystallized using methanol. The recrystallized monomers were then dried in an oven at 50°C overnight. Schematic representation of the synthesis, modification, and curing (at 200°C) of TA-BZ-based epoxy composites.
Synthesis of pure epoxy resin
The epoxy resin, serving as the matrix in this study, was prepared following the procedure outlined by Hussein et al.71,72 Specifically, Component A, comprising bisphenol-A diglycidyl ether (DGEBA) epoxy resin, and Component B, a cycloaliphatic polyamine curing agent (hardener), were combined in a 1:1 weight ratio. A minor volume of chloroform was incorporated to facilitate mixing and reduce viscosity. To achieve a homogeneous dispersion, the resulting mixture underwent ultrasonication for 15 min, followed by an additional 10 min of sonication. Subsequently, the blend was cast into a Petri dish and maintained at ambient temperature overnight to enable complete solvent evaporation. The resultant thin epoxy films were then subjected to drying in a desiccator to eliminate residual moisture before further use in subsequent experiments.
Preparation of TA-BZ blend Epoxy-ZnO composites
Following the synthesis of the TA-BZ monomers, these were incorporated into the prepared epoxy resin along with 5 mg of ZnO nanoparticles, corresponding to a ZnO loading of 0.25 wt% relative to the combined mass of Components A and B (1 g each). To ensure homogeneous dispersion of the nanoparticles within the matrix, the resultant mixture was subjected to ultrasonication for 30 min. Subsequently, the mixture was cast into Petri dishes and allowed to dry at ambient temperature overnight, yielding smooth, uniform films. These films were designated as Epoxy-TA-BZ-1-ZnO, Epoxy-TA-BZ-2-ZnO, and Epoxy-TA-BZ-4-ZnO, respectively. Additionally, reference samples prepared without the inclusion of ZnO nanoparticles were also fabricated and labeled as Epoxy-TA-BZ-1, Epoxy-TA-BZ-2, and Epoxy-TA-BZ-4.
Thermal curing of TA-BZ composites through ring-opening polymerization
The TA-BZ composites were subjected to ring-opening polymerization (ROP) at 200°C for 2 h to promote crosslinking among tannic acid benzoxazine (TA-BZ), epoxy, and ZnO. This thermal curing process facilitated the opening of the oxazine ring, enhancing crosslinking and improving the material properties. Following the curing process, the samples displayed a significant increase in hardness and a darker appearance compared to their uncured counterparts. The cured samples without the inclusion of ZnO were labeled as
Results and discussion
Chemistry
As shown in Scheme 1, the synthesis of benzoxazine monomers from tannic acid (TA), aniline, and paraformaldehyde (PFA) proceeds via a Mannich-type condensation reaction. Paraformaldehyde depolymerizes to release formaldehyde, which reacts with the phenolic hydroxyl groups (-OH) of TA to form hydroxymethylated phenolic intermediates. Simultaneously, the amine groups (-NH2) of aniline participate in the reaction with these hydroxymethyl groups, leading to intramolecular cyclization and the formation of the characteristic oxazine ring structure. This condensation process results in the formation of benzoxazine monomers with oxazine rings linked to the tannic acid aromatic framework. The process is typically carried out under mild conditions (e.g., 70°C for 24 h) in a toluene/ethanol solvent mixture, which supports the condensation without degrading the tannic acid backbone. This reaction yields benzoxazine monomers with enhanced thermal, mechanical, and corrosion-resistant properties, while utilizing tannic acid, a renewable bio-based material, making the process environmentally sustainable. The formation of the oxazine ring introduces a stable heterocyclic structure, contributing to the material’s desirable properties and positioning it for use in high-performance applications. 73
After the formation of the benzoxazine (TA-BZ) monomer, epoxy resin and zinc oxide (ZnO) are introduced to further enhance the material’s properties. Epoxy resins, known for their excellent adhesive, thermal, and mechanical properties, are incorporated into the system to improve the overall structural integrity and versatility of the composite. The epoxy groups (-epoxy) in the resin undergo curing reactions when exposed to heat, which promotes crosslinking between the resin molecules, leading to a highly durable three-dimensional network. 61
The incorporation of zinc oxide (ZnO) nanoparticles significantly enhances the anticorrosion performance of the composite. Serving as a reinforcing agent, ZnO improves mechanical strength and offers additional protection against environmental degradation, including ultraviolet (UV) exposure and oxidative stress. In the present system, the integration of epoxy resin, ZnO nanoparticles, and TA-BZ monomers leads to the formation of a hybrid network (Figure 1), wherein the epoxy matrix provides crosslinking flexibility, ZnO contributes to corrosion resistance, and the benzoxazine units impart superior thermal and mechanical stability. The curing process, typically conducted at 200°C for 2 h, facilitates extensive crosslinking and network formation, thereby enhancing the structural integrity of the composite. The resulting material demonstrates improved corrosion resistance, thermal stability, and mechanical performance, rendering it suitable for high-performance applications in demanding environments such as marine, automotive, and infrastructure sectors. Structural illustration of TA-BZ-based epoxy composites highlighting ZnO incorporation and molecular interactions.
Nuclear Magnetic Resonance and The Fourier Transform Infrared (FTIR) analysis
Nuclear Magnetic Resonance (NMR) spectroscopy
Nuclear Magnetic Resonance (NMR) spectroscopy was utilized to determine the chemical structure and composition of the uncured TA-BZ samples. Both 1H-NMR and 13C-NMR analyses were performed to gain detailed insights into their molecular architecture. The 1H-NMR spectrum of the uncured TA-BZ samples (the starting monomer), as shown in the supplementary information (Figure S1 and Figure S2), revealed distinct characteristic peaks providing crucial information about the initial structural features. A sharp singlet observed at δ 5.2 ppm corresponds to methylene protons (-CH2-) bridging aromatic rings in the benzoxazine structure, serving as a key indicator of an intact oxazine ring. Peaks in the aromatic region (δ 6.5–7.5 ppm) were attributed to protons on the benzene rings associated with both the benzoxazine and tannin moieties, underscoring the intricate aromatic framework of the hybrid system. A broad signal at δ 4.8 ppm, assigned to oxazine ring protons, further validated the presence of the benzoxazine structure. Furthermore, the aliphatic region (δ 0.8–2.5 ppm) exhibited multiple signals, likely originating from the complex tannin component, highlighting the structural diversity within the tannin-benzoxazine hybrid.
The 13C-NMR spectrum of the uncured samples provided complementary structural information (Figures S3 and S4). A prominent peak observed at δ 79.5 ppm was attributed to the methylene carbon within the oxazine ring, serving as a characteristic marker of this functional group. Signals in the range of δ 110–150 ppm were assigned to aromatic carbons associated with both the benzoxazine and tannin structures, reflecting their intricate aromatic framework. Additionally, a peak at δ 152 ppm was indicative of the C–O–C linkage in the oxazine ring, confirming its presence. Signals in the aliphatic region (δ 20–60 ppm) were associated with the diverse carbon environments in the tannin component, highlighting the structural complexity of the uncured material.
The Fourier Transform Infrared (FTIR)
The Fourier Transform Infrared (FTIR) spectrum presented in Figure 2 highlights the substantial structural changes occurring during the curing process of benzoxazine resins. Notably, the characteristic oxazine ring peak at approximately 950 cm-1, associated with the out-of-plane deformation of the oxazine ring, is evident in the uncured TA-BZ sample (blue curve) but is absent in the cured TA-BZ-C sample (red curve). This observation underscores the significant chemical transformations undergone by the oxazine ring during curing, likely involving its integration into the cross-linked polymer network. Consequently, the number of detectable free oxazine groups is reduced, as indicated by the disappearance of the 950 cm-1 peak in the cured material. FTIR spectra of uncured TA-BZ and TA-BZ-C after curing at 200°C.
Furthermore, the broad O–H stretching band observed between 3200 and 3600 cm-1 exhibits decreased intensity in the cured sample, reflecting a reduction in free hydroxyl groups due to cross-linking. These findings confirm that the curing process profoundly alters the oxazine structure while preserving other functional groups, which contributes to the enhanced mechanical properties and improved corrosion resistance of the cured TA-BZ A material.
The FTIR spectrum of the Epoxy-TA-BZ-ZnO composite as shown in Figure 3 confirms the integration of epoxy, tannic acid benzoxazine (TA-BZ), and zinc oxide (ZnO). The reduction in the C-O-C stretching band (915–830 cm-1) indicates the epoxide ring opening during curing at 200°C (Epoxy-TA-BZ-ZnO-C). Peaks between 1500 and 1600 cm-1 confirm aromatic C = C stretching from both epoxy and TA-BZ, while bands around 1220–1260 cm-1 (C-O-C) and 950–970 cm-1 (benzoxazine ring) confirm the presence of the benzoxazine ring in uncured samples. A broad band in the 3200–3500 cm-1 range indicates O-H stretching from tannic acid and the curing process. ZnO is identified by the absorption band below 600 cm-1, indicating Zn-O stretching. These results demonstrate effective curing and successful integration of ZnO with epoxy and TA-BZ. FTIR spectra of Epoxy-TA-BZ-ZnO-C before and after curing at 200°C.
Morphological and structural analysis
Scanning electron microscopy (SEM)
The surface morphology and microstructural characteristics of TA-BZ-epoxy and TA-BZ-epoxy-ZnO composites were analyzed using SEM, as illustrated in Figure 4(G–M). The SEM micrographs reveal significant morphological differences between thermally cured materials without ZnO (G-J) and those containing a constant concentration of ZnO nanoparticles (K-M). SEM images of Epoxy-TA-BZ at 200°C curing temperature without ZnO (G, H and J) and with ZnO, (K, L, and M).
Figure 4(G–J) illustrates notable morphological changes in samples with varying TA-BZ mole ratios under thermal curing at 200°C. Figure 4(G) (Epoxy-TA-BZ-1-C) displays a mostly uniform surface with scattered particles shown at higher magnification, indicating early cross-linking stages and suggesting controlled polymerization. Surface irregularities are minimal, reflecting limited molecular network development. Figure 4(H) (Epoxy-TA-BZ-2-C), exhibits linear striations, suggesting improved molecular alignment during curing, while these patterns signify an enhanced directional structure within the polymer matrix. Figure 4(J) (Epoxy-TA-BZ-4-C), presents a more textured surface with well-defined features, highlighting the effect of a decreased TA-BZ ratio on structural development.
The inclusion of ZnO concentration substantially alters composite morphology, as illustrated in Figure 4(K–M). Figure 4(K) (Epoxy-TA-BZ-1-ZnO-C) demonstrates uniform dispersion of bright ZnO particles throughout the matrix, indicating effective integration and optimal interfacial contact between ZnO and the epoxy-TA-BZ matrix. In Figure 4(L) (Epoxy-TA-BZ-2-ZnO-C), the ZnO particle density increases, forming a more intricate network structure while maintaining uniform dispersion, as evidenced by constant particle boundaries and interparticle distances. Figure 4(M) (Epoxy-TA-BZ-4-ZnO-C) shows densely packed particle clusters with heightened brightness, indicating enhanced ZnO connectivity, though localized agglomerations suggest an approach to the upper threshold for effective dispersion at this TA-BZ ratio.
Figure 4(K–M) further illustrates the concentration-dependent effects of ZnO on morphology, with an increase in bright ZnO spots representing higher nanoparticle frequency and density. The progressive modification observed indicates that the inclusion of ZnO significantly impacts the composite structure across different TA-BZ ratios. Specifically, a moderate TA-BZ mole ratio, as illustrated in Figure 4(L), appears to optimize the balance between particle dispersion and matrix integration.
The contrast between thermally cured samples (G-J) and ZnO-infused composites (K-M) underscores the transformative impact of ZnO on microstructure. While thermal curing promotes molecular organization and network formation, ZnO introduces a secondary phase that significantly alters the composite, potentially enhancing its functional properties.
The SEM data provides critical insights into how TA-BZ mole ratios affect morphology at a fixed ZnO concentration, underscoring the combined effects of thermal curing and nanoparticle incorporation. This understanding establishes a foundation for optimizing these materials for specific applications, where adjusting TA-BZ ratios can yield desired structural and functional attributes.
Transmission Electron Microscopy (TEM) analysis
The structural and morphological features of the TA-BZ-epoxy composites were examined using Transmission Electron Microscopy (TEM), as depicted in Figure 5(G–M). The micrographs revealed notable differences in nano structural organization between thermally cured samples (G-J) and those incorporating ZnO nanoparticles (K-M). Transmission Electron Microscopy (TEM) Images of TA-BZ-Epoxy Composites: Comparison of Thermally Cured Samples without ZnO Incorporation (G-J) and ZnO-Loaded Samples (K-M) at different magnifications.
For the thermally cured samples, progressive structural development with increasing cure temperature up to 200°C was observed. Sample G (Epoxy-TA-BZ-1-C) displayed a homogeneous structure with moderate electron density variations, indicating initial crosslinking. Sample H (Epoxy-TA-BZ-2-C) exhibited enhanced structural organization, with increased contrast suggesting improved network development. Sample J (Epoxy-TA-BZ-4-C) at higher magnification revealed the most advanced crosslinking, with nanoscale domains indicating a well-defined structure.
In ZnO-incorporated samples (K-M), the TEM micrographs showed a significant impact of ZnO addition. Sample K (Epoxy-TA-BZ-1-ZnO-C) demonstrated uniformly dispersed ZnO nanoparticles with minimal clustering. Sample L (Epoxy-TA-BZ-2-ZnO-C) revealed increased particle density with occasional clustering, while Sample M (Epoxy-TA-BZ-4-ZnO-C) showed interconnected networks of ZnO particles due to higher loading. Across all ZnO-loaded samples, the nanoparticles-maintained nanoscale dimensions, and high-resolution imaging confirmed strong particle-matrix adhesion, with no voids or delamination.
These results highlight the evolution of structural and interface properties in response to curing and ZnO loading. The hierarchical structure combining ZnO nanoparticles with a crosslinked TA-BZ-Epoxy matrix likely contributes to enhanced mechanical and thermal properties, emphasizing the importance of processing parameters in optimizing composite performance.
X-ray diffraction analysis (XRD)
The crystallographic structure and phase composition of TA-BZ-Epoxy composites were analyzed using XRD. Diffraction patterns reveal substantial structural differences between thermally cured samples and those containing ZnO nanoparticles, with both types cured at 200°C under a fixed ZnO concentration but with varying TA-BZ mole ratios.
The XRD patterns of the thermally cured samples as presented in Figure 6 (Epoxy-TA-BZ-1-C (G), Epoxy-TA-BZ-2-C (H), and Epoxy-TA-BZ-4-C (J)) exhibit features typical of amorphous materials, including a broad diffraction peak centered around 2θ = 20°. This broad peak signifies the amorphous nature of the epoxy matrix and represents the average interchain spacing within the polymer network. Notably, the intensity and width of this amorphous halo increase with higher TA-BZ mole ratios, particularly in Epoxy-TA-BZ-4-C, suggesting a greater cross-linking density under curing conditions at 200°C. The absence of distinct crystalline peaks in these samples confirms the formation of a robust, amorphous thermoset network. XRD images of Epoxy-TA-BZ at 200°C curing temperature without ZnO (G, H and J) and with ZnO, (K, L, and M).
The incorporation of ZnO nanoparticles markedly alters the diffraction patterns of the samples (Epoxy-TA-BZ-1-ZnO-C (K), Epoxy-TA-BZ-2-ZnO-C (L), and Epoxy-TA-BZ-4-ZnO-C (M)), which show distinct crystalline peaks of ZnO superimposed on the amorphous epoxy background. ZnO peaks appear at 2θ values of approximately 31.7° (100), 34.4° (002), 36.2° (101), 47.5° (102), 56.6° (110), 62.8° (103), and 67.9° (112), aligning with the hexagonal wurtzite structure of ZnO (JCPDS card No. 36-1451). The increasing intensity of ZnO peaks from K to M correlates with different TA-BZ mole ratios, confirming the uniform dispersion of ZnO nanoparticles within the TA-BZ-Epoxy matrix. The sharpness of these peaks indicates high ZnO crystallinity, while the consistent positions of the peaks across all samples suggest that the ZnO crystal structure is preserved during composite formation and curing.
Structural development and interactions
A slight shift and broadening of the amorphous epoxy peak (approximately 2θ = 20°) in ZnO-containing samples relative to pure TA-BZ-Epoxy samples suggests molecular interactions between the ZnO nanoparticles and the epoxy matrix, potentially influencing polymer chain packing and cross-linking behavior. The relative intensity ratio between ZnO crystalline peaks and the amorphous epoxy background offers insight into the phase distribution within the composites.
The retention of the amorphous TA-BZ-Epoxy matrix alongside the crystalline ZnO structure indicates the successful formation of a hybrid organic-inorganic composite. The increase in ZnO peak intensity with lower TA-BZ ratios reflects proportionate ZnO incorporation while maintaining the structural integrity of both phases.
Crystallite size analysis
ZnO particle crystallite sizes were calculated using the Scherrer equation, based on the full width at half maximum (FWHM) of the principal diffraction peaks. The pronounced ZnO peaks reflect well-crystallized nanoparticles, with consistent widths across different TA-BZ ratios, indicating a stable particle size distribution regardless of ZnO loading.
The XRD findings provide valuable insights into the structural organization of the composites, confirming effective thermal curing of the epoxy matrix and successful incorporation of ZnO nanoparticles. The results emphasize the dual-phase structure, combining the amorphous epoxy matrix with the crystalline ZnO reinforcement, and suggest potential interfacial interactions that may influence the composite properties.
Thermal analysis
Thermogravimetric analysis (TGA)
The thermal stability and degradation behavior of TA-BZ-Epoxy composites, both with and without ZnO, were examined using thermogravimetric analysis (TGA) in a nitrogen atmosphere over a temperature range from room temperature to 800°C. This study investigates the effects of curing at 200°C and the influence of ZnO incorporation, specifically with different TA-BZ ratios, on the thermal properties of the cured composites.
The TGA curves for samples without ZnO (Figure 7(a)) exhibit a multi-stage degradation pattern. Initially, all ZnO-free samples remain stable up to around 300°C, demonstrating commendable thermal stability at moderate temperatures. The primary degradation onset occurs around 350°C, accompanied by rapid weight loss continuing until approximately 500°C. This major decomposition phase is associated with the disintegration of the TA-BZ-Epoxy matrix and the degradation of organic components. The degradation profiles of ZnO-free samples (Epoxy-TA-BZ 1-C, Epoxy-TA-BZ 2-C, and Epoxy-TA-BZ 4-C) display similar patterns, with final char yields of approximately 25-27% at 800°C. Minor differences in degradation temperatures and char yields among these samples reflect the influence of varying TA-BZ ratios on initial network formation. In contrast, the thermally cured ZnO-containing samples (Figure 7(b)) exhibit considerable improvements in thermal stability. The ZnO-incorporated composites show elevated initial degradation temperatures (IDT), shifting from roughly 350°C in ZnO-free samples to around 375°C with the inclusion of ZnO. This increase suggests a more stable, cross-linked structure due to ZnO’s presence, which enhances thermal resistance, particularly within the 300 °C–400°C range. ZnO-containing samples also display slightly higher char yields (28–30%) at 800°C compared to those without ZnO. Among the ZnO-incorporated samples, Epoxy-TA-BZ-4-ZnO-C demonstrates the highest thermal resistance, likely due to enhanced cross-linking density achieved with a lower TA-BZ ratio in conjunction with the stabilizing effect of ZnO.The thermal degradation process in these composites occurs in three main stages. In the initial stage (25 °C–300°C), minimal weight loss (<2%) is observed, attributed to the evaporation of residual moisture and low molecular weight compounds. The primary degradation stage (300–500°C) involves significant weight loss (60–70%), associated with the breakdown of the cross-linked network and cleavage of the epoxy backbone. The final stage (500–800°C) involves gradual weight loss, leading to char formation. These findings suggest that curing at 200°C, especially with ZnO incorporation, markedly enhances the structural integrity and thermal stability of the epoxy matrix. Higher char yields in ZnO-containing samples suggest more efficient cross-linking and potentially improved flame-retardant properties. Comparing samples with different TA-BZ ratios shows that Epoxy-TA-BZ-4-ZnO-C demonstrates slightly better thermal resistance than Epoxy-TA-BZ-1-ZnO-C and Epoxy-TA-BZ-2-ZnO-C, suggesting that a lower TA-BZ ratio facilitates greater cross-linking density and improved thermal stability. However, the relatively minor differences among samples indicate that even higher TA-BZ ratios achieve sufficient cross-linking for effective thermal stability in the composite matrix. In conclusion, the TGA results provide valuable insights into the thermal properties and stability of TA-BZ-Epoxy composites. The curing process at 200°C and the incorporation of ZnO substantially enhance thermal resistance, with ZnO acting as an effective stabilizing agent. The results indicate that curing at 200°C, in conjunction with ZnO inclusion, enhances the durability of the composites by forming a more robust, cross-linked network structure. TGA images of Epoxy-TA-BZ at 200°C curing temperature without ZnO (G, H and J) and with ZnO, (K, L, and M).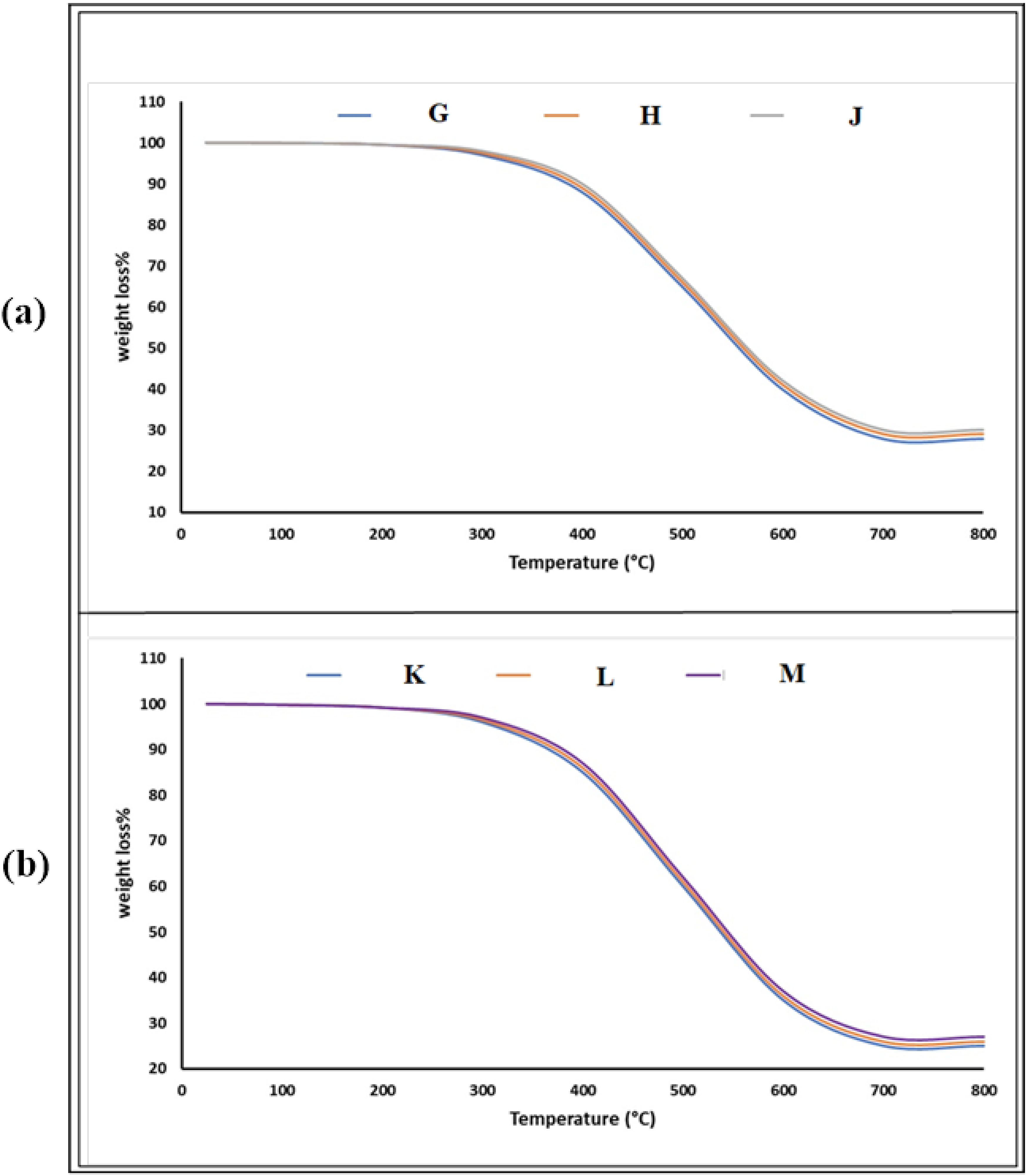
Differential scanning calorimetry (DSC)
The influence of the TA-BZ ratio on the thermal behavior of the composites is clearly demonstrated through systematic variations in exothermic peak characteristics. Lower TA-BZ ratios are associated with higher exothermic peak intensities, indicating enhanced cross-linking efficiency and more complete curing. Specifically, Epoxy-TA-BZ-4-C ZnO exhibited the highest exothermic intensity (3.6 mW), followed by Epoxy-TA-BZ-2-C ZnO (3.4 mW) and Epoxy-TA-BZ-1-C ZnO (3.2 mW). This trend suggests that optimizing the TA-BZ ratio promotes more efficient cross-linking, resulting in improved thermal stability and mechanical performance.
The broader exothermic peaks observed across different TA-BZ ratios indicate increasingly complex curing kinetics and prolonged reaction durations. This broadening is attributed to the sequential and overlapping nature of benzoxazine ring-opening polymerization, epoxy-amine reactions, and tannic acid-mediated cross-linking—each proceeding at distinct temperatures and rates. Figure 8 presents a comparative overview of ZnO-free samples (G, H, and J) and ZnO-incorporated composites (K, L, and M), highlighting the catalytic role of ZnO nanoparticles in the curing process. Incorporation of ZnO significantly increases exothermic peak intensities and subtly alters thermal transition profiles. ZnO-containing samples exhibit sharper, more defined exothermic peaks, reflecting enhanced cross-linking efficiency and more controlled curing kinetics. DSC images of Epoxy-TA-BZ at 200°C curing temperature without ZnO (G, H and J) and with ZnO, (K, L, and M).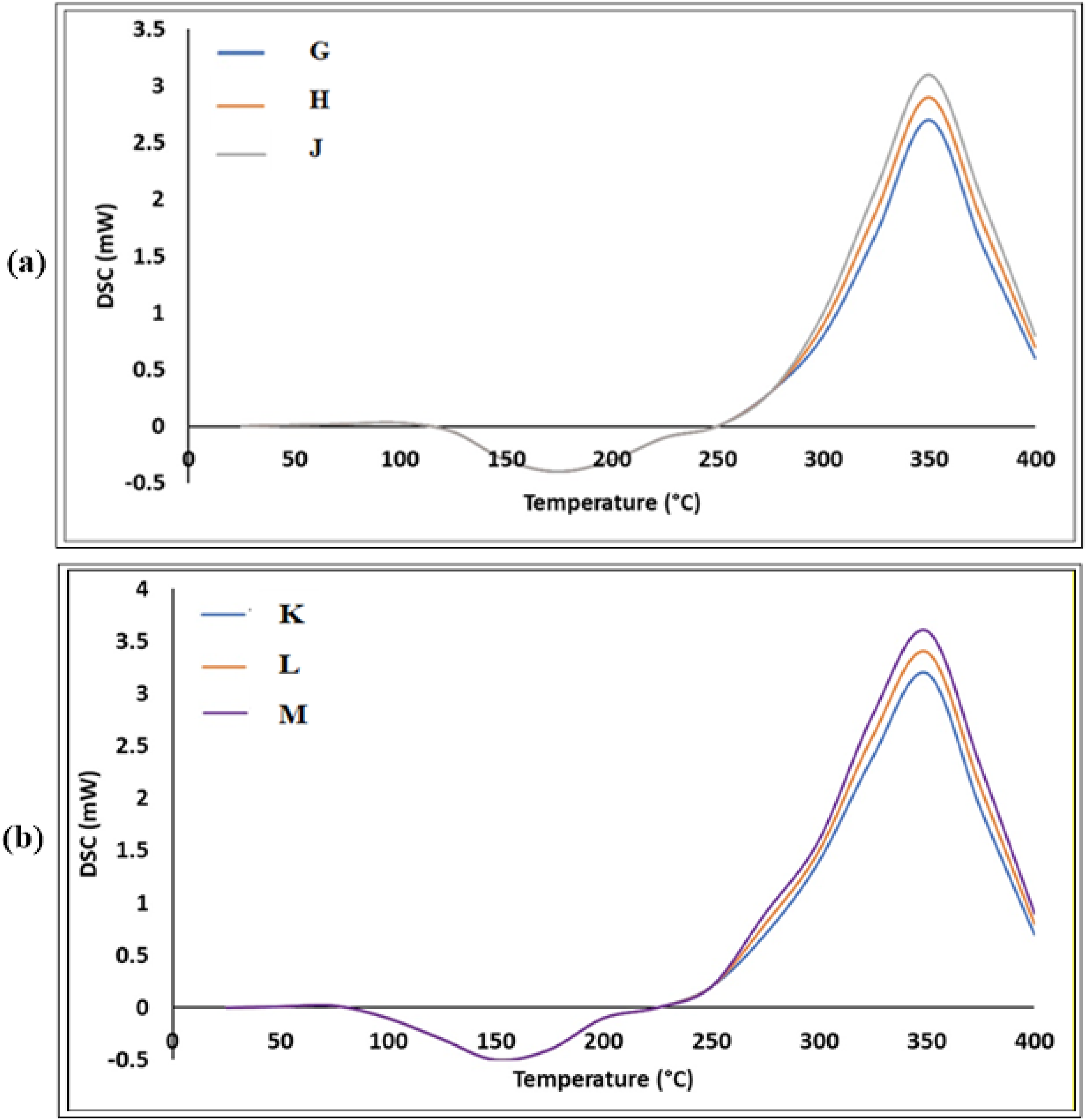
Additionally, the endothermic transitions in ZnO-containing composites shift slightly to lower temperatures, suggesting that ZnO facilitates earlier molecular mobility and polymer chain relaxation. This catalytic effect is attributed to the surface activity of ZnO nanoparticles, which provide nucleation sites for cross-linking reactions and accelerate overall curing kinetics.
Comparative analysis further confirms that ZnO incorporation not only intensifies the exothermic response but also improves cross-linking behavior through catalytic action. The presence of ZnO shifts peak temperatures in the primary exothermic region (300–375°C) downward by approximately 8–12°C, indicating a reduction in activation energy and modification of reaction pathways.
Thermal activation behavior observed in the 140–160°C range across all samples offers additional insight into the early stages of chain mobility and network formation. ZnO-incorporated composites demonstrate more pronounced endothermic transitions, which may be attributed to enhanced thermal conductivity and more uniform heat distribution throughout the matrix. Thermal parameters extracted from the DSC curves—including onset temperature (Tonset), peak temperature (Tpeak), and completion temperature (Tcompletion)—further elucidate the influence of ZnO. ZnO-containing systems consistently show lower Tonset values (285–295°C) compared to ZnO-free counterparts (295–305°C), reinforcing ZnO’s catalytic effect in initiating earlier cross-linking. Moreover, the narrower exothermic peak widths (ΔT) observed in ZnO composites suggest more efficient and kinetically controlled curing behavior.
Overall, the DSC analysis demonstrates that curing at 200°C, combined with strategic ZnO incorporation, has a significant impact on cross-linking dynamics and thermal performance in TA-BZ-epoxy composites. The systematic trends observed in thermal transitions provide robust, quantitative evidence of optimized curing conditions and improved material properties. These results are consistent with enhancements observed in the mechanical and anticorrosion performance of the composites, underscoring the effectiveness of the formulation strategy.
This comprehensive thermal characterization confirms that an optimized balance between the TA-BZ ratio and ZnO content leads to superior curing efficiency, improved thermal stability, and enhanced performance of the protective composite coatings. The detailed analysis of thermal events provides essential insights for process optimization and quality assurance in advanced coating applications.
Surface topography analysis (AFM)
An atomic Force Microscopy (AFM) analysis was conducted to investigate the surface topography and roughness of Epoxy-TA-BZ composites revealing notable morphological differences between thermally cured samples and those containing a constant concentration of ZnO nanoparticles. The 3D AFM images as shown in Figure 9 provide comprehensive insights into surface evolution and nanostructural development in all materials. AFM images of Epoxy-TA-BZ at 200°C curing temperature without ZnO (G, H and J) and with ZnO, (K, L, and M).
Impact of thermal curing (samples G-J)
The thermally cured samples without ZnO (G-J) demonstrate progressive alterations in surface topography at varying TA-BZ mole ratios, achieved through curing at 200°C. Sample G (Epoxy-TA-BZ-1-C) exhibits an undulating surface morphology with peak heights around 88.6 nm, indicating initial molecular restructuring at the curing temperature. The surface displays consistent undulations, suggesting controlled polymerization. Sample H (Epoxy-TA-BZ-2-C) shows more prominent wave patterns with slightly higher peak heights of 91.2 nm, indicating enhanced molecular alignment and rearrangement in response to the altered TA-BZ ratio. Sample J (Epoxy-TA-BZ-4-C) presents more pronounced wave structures with peak heights of 94.2 nm, suggesting that higher TA-BZ ratios promote more extensive cross-linking and structural evolution.
Effects of ZnO inclusion (samples K-M)
The addition of ZnO nanoparticles results in significant changes to surface topography, as seen in samples K-M. Sample K (Epoxy-TA-BZ-1-ZnO-C) exhibits a notable shift from wave-like patterns to a nodular morphology with peak heights of 61.2 nm. This shift suggests a strong interaction between ZnO nanoparticles and the epoxy matrix, leading to a textured surface characterized by evenly dispersed features. In Sample L (Epoxy-TA-BZ-2-ZnO-C), nodule density and organization increase, with peak heights reaching 66.3 nm, displaying well-defined spherical features and excellent ZnO dispersion. Sample M (Epoxy-TA-BZ-4-ZnO-C) shows the most pronounced nodular structure with peak heights reaching 70.1 nm, highlighting the influence of lower TA-BZ ratios on surface morphology.
Comparative examination
A notable shift in surface architecture is observed between thermally cured samples (G-J) and ZnO-incorporated samples (K-M). The wave-like patterns in samples G-J, typical of pure epoxy systems, evolve into distinctive nodular formations in samples K-M with ZnO addition. This morphological transition likely stems from the nucleating effect of ZnO nanoparticles, which act as anchoring sites for polymer chain organizations during curing.
The incremental rise in peak heights from sample K (61.2 nm) to sample M (70.1 nm) aligns with decreasing TA-BZ ratios, suggesting that moderate TA-BZ ratios foster more pronounced surface characteristics. The uniform distribution of nodules in samples K-M indicates effective ZnO dispersion; however, the increasing feature size from K to M may suggest potential particle agglomeration at varying TA-BZ concentrations.
Surface roughness evolution
In samples G-J, thermal curing alone produces relatively smooth, undulating surfaces with peak heights between 88.6 and 94.2 nm, indicating regulated structural development. Conversely, ZnO incorporation results in a more complex surface topology with nodular structures and generally lower peak heights (61.2–70.1 nm). This difference suggests that while ZnO incorporation introduces numerous surface features, it may lead to a more compact, densely arranged structure compared to the broader wave patterns observed in thermally cured samples.
These AFM findings provide valuable insights into the nanoscale surface evolution of epoxy composites, demonstrating that both thermal curing at 200°C and ZnO incorporation significantly influence the final surface morphology and structural organization. The results indicate that optimal surface characteristics may be achieved through careful control of TA-BZ ratios and ZnO concentration, enabling tailored material properties for specific applications.
Mechanical properties (tensile stress)
The mechanical properties of the TA-BZ-Epoxy composites were assessed through stress-strain analysis, providing significant insights into the effects of heat curing at 200°C and the incorporation of ZnO nanoparticles on the material’s mechanical characteristics. All samples were cured at 200°C, with consistent ZnO content maintained across the ZnO-containing samples, each having different TA-BZ mole ratios.
The stress-strain curves of the thermally cured samples exhibit the typical behavior of cross-linked TA-BZ-Epoxy systems as illustrated in Figure 10. The initial linear region indicates elastic deformation, which transitions into plastic deformation before final breakdown. Among the cured samples, the Epoxy-TA-BZ-2-C sample exhibited superior mechanical properties, showcasing the highest tensile strength and optimal strain at fracture. This improvement is likely to be attributed to the ideal cross-linking network formed under the specified curing conditions and the intermediate TA-BZ mole ratio. In contrast, the Epoxy-TA-BZ-4-C sample, characterized by a lower TA-BZ ratio, demonstrated slightly reduced mechanical properties, indicating that the reduced cross-linking density may result in a more brittle structure with lower ductility. Stress strain images of Epoxy-TA-BZ at 200°C curing temperature without ZnO (G, H and J) and with ZnO, (K, L, and M).
The incorporation of ZnO nanoparticles significantly altered the mechanical properties of the composites. The stress-strain curves for the ZnO-containing samples showed a systematic variation in mechanical characteristics, while maintaining a consistent ZnO content across the samples. The Epoxy-TA-BZ-4-C-ZnO sample exhibited the most substantial decrease in tensile strength and elongation at break, suggesting that ZnO nanoparticles may act as stress concentrators at higher loadings, thereby diminishing both strength and ductility. Conversely, the Epoxy-TA-BZ-1-C-ZnO sample exhibited superior mechanical properties, indicating improved particle-matrix interactions at this composition.
The mechanical properties of the composites appear to be related to their structural characteristics, particularly the balance between cross-linking density and flexibility. The thermally cured Epoxy-TA-BZ-2 sample exhibited an optimal balance, characterized by sufficient cross-linking and adequate molecular mobility, leading to enhanced mechanical properties.
The stress-strain curves revealed distinct deformation mechanisms across the sample series. The initial linear region represents elastic deformation, where polymer chains undergo reversible stretching, while the non-linear phase corresponds to plastic deformation, involving chain slippage and reorganization. The ZnO nanoparticles influenced these mechanisms by acting as reinforcing agents at lower mole ratios and potential stress concentrators at higher mole ratios.
Overall, the analysis of mechanical properties underscores the significant effects of curing at 200°C and ZnO incorporation on the composite’s behavior. Curing at 200°C enhances the mechanical properties by creating a more stable cross-linked network, while the addition of ZnO nanoparticles modifies mechanical performance depending on the TA-BZ mole ratios and ZnO concentration. This suggests that an optimal ZnO concentration and TA-BZ ratio are essential for achieving superior mechanical properties.
Comprehensive comparative analysis of synthesized materials
The comparative analysis of the synthesized materials demonstrates systematic property evolution across multiple characterization techniques. Our investigation reveals clear progression patterns between ZnO-free and ZnO-containing samples, as well as across varying TA-BZ ratios.
Spectroscopic analysis through FTIR provides fundamental evidence of successful synthesis and curing. The characteristic oxazine ring peak at 950 cm-1, prominently visible in uncured samples, disappears after thermal curing, confirming successful ring-opening polymerization. The broad O-H stretching band (3200-3600 cm-1) shows decreased intensity in cured samples, indicating effective cross-linking. These spectral changes are consistently observed across all compositions, with progressive intensity variations correlating with TA-BZ ratios.
Thermal characterization reveals distinct property enhancements with compositional modifications. TGA analysis shows elevated initial degradation temperatures from approximately 350°C in ZnO-free samples to 375°C in ZnO-containing composites. Char yields similarly improve from 25-27% to 28-30% at 800°C with ZnO incorporation. DSC measurements demonstrate systematic variations in curing behavior, with exothermic peak intensities ranging from 3.2 mW to 3.6 mW, reflecting different cross-linking densities across compositions.
Microscopic examination reveals progressive structural evolution. SEM and AFM analyses show a transition from wave-like patterns in ZnO-free samples (G-J) to well-defined nodular morphology in ZnO-containing composites (K-M). TEM imaging confirms uniform ZnO dispersion and strong particle-matrix adhesion. Surface roughness parameters systematically vary with composition, demonstrating controlled structural development.
XRD patterns establish clear structural differences between sample series. ZnO-free samples exhibit a characteristic broad peak centered at 2θ = 20°, typical of amorphous polymer matrices. ZnO-containing samples show additional crystalline peaks at 31.7°, 34.4°, and 36.2°, confirming successful nanoparticle incorporation. Peak intensities systematically vary with composition, reflecting structural optimization.
Mechanical testing reveals systematic property enhancement across the sample series. Stress-strain analysis shows improved strength and modified failure modes with ZnO incorporation. The progression from Epoxy-TA-BZ-1C through Epoxy-TA-BZ-4C, and similarly through their ZnO-containing counterparts, demonstrates consistent property optimization.
This comprehensive analysis establishes clear structure-property relationships across our sample series. The consistent trends observed through multiple characterization techniques validate our synthetic approach and demonstrate successful property enhancement through compositional optimization. The synergistic effects of varying TA-BZ ratios and ZnO incorporation are clearly evidenced across all characterization methods, confirming the achievement of our design objectives.
Anti-corrosion properties
Electrochemical characterization overview
Comprehensive electrochemical parameters for thermally cured and ZnO-incorporated epoxy composites.
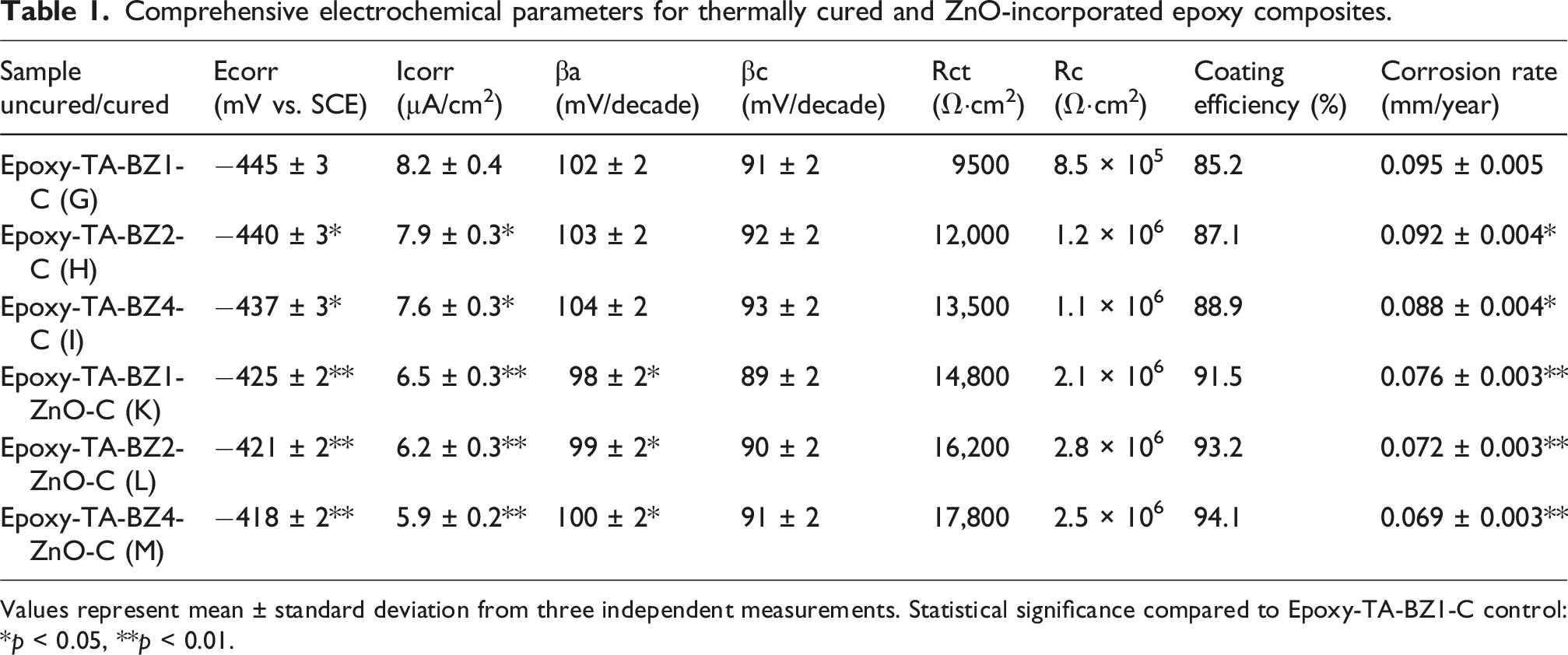
Values represent mean ± standard deviation from three independent measurements. Statistical significance compared to Epoxy-TA-BZ1-C control: *p < 0.05, **p < 0.01.
Tafel analysis and corrosion kinetics
The potentiodynamic polarization measurements provided detailed insights into the corrosion kinetics through comprehensive Tafel plot analysis. The anodic and cathodic Tafel slopes (βa and βc) revealed significant information about the electrode reaction mechanisms and the influence of coating composition on corrosion behavior. For thermally cured samples, the anodic Tafel slopes ranged from 102 to 104 mV/decade, indicating consistent anodic dissolution kinetics across different cross-linking densities. The cathodic Tafel slopes (91-93 mV/decade) suggested that oxygen reduction remained the primary cathodic reaction, with minor variations attributed to surface accessibility and electrolyte diffusion through the coating matrix.
The incorporation of ZnO nanoparticles resulted in systematic modifications to the Tafel slopes, with anodic values decreasing to 98-100 mV/decade and cathodic slopes to 89-91 mV/decade. This reduction in Tafel slopes indicates enhanced charge transfer kinetics and modified electrode reaction mechanisms. The lower anodic Tafel slopes for ZnO-incorporated samples suggest that zinc dissolution and subsequent passive layer formation contribute to the overall corrosion protection mechanism. The correlation between Tafel slopes and corrosion current density demonstrates that optimal electrochemical performance is achieved through balanced nanoparticle dispersion and cross-linking density. Epoxy-TA-BZ-2C (H) exhibited the most favorable performance among thermally cured samples, with Ecorr at −440 mV versus SCE, Icorr at 7.9 µA/cm2, and a corrosion rate of 0.092 mm/year. This superior performance is attributed to optimal cross-linking density that maximizes barrier properties without introducing microstructural defects. The progressive improvement from Epoxy-TA-BZ-1C to 4C (I), evidenced by decreasing Icorr values (8.2 to 7.6 µA/cm2) and corrosion rates (0.095 to 0.088 mm/year), confirms the beneficial effects of increased cross-linking density on corrosion resistance.
Electrochemical impedance spectroscopy and nyquist analysis
Complementary electrochemical impedance spectroscopy (EIS) measurements were performed to provide comprehensive understanding of the coating barrier properties and charge transfer mechanisms. The Nyquist plots revealed characteristic semicircular responses in the high-frequency region, corresponding to coating capacitance and resistance, followed by charge transfer processes at lower frequencies. The diameter of the semicircular loops directly correlates with the charge transfer resistance (Rct), providing quantitative assessment of the coating’s protective efficacy. For thermally cured samples, the Nyquist plots data showed progressive increase in semicircle diameter with increasing cross-linking density. Epoxy-TA-BZ-1C exhibited a charge transfer resistance of approximately 9500 Ω·cm2, which increased to 12,000 Ω·cm2 for Epoxy-TA-BZ-2C and 13,500 Ω·cm2 for Epoxy-TA-BZ-4C. The coating resistance (Rc) values, determined from the high-frequency intercept, ranged from 8.5 × 105 to 1.2 × 106 Ω·cm2, indicating excellent barrier properties for all thermally cured samples. The slight decrease in Rc for Epoxy-TA-BZ-4C (1.1 × 106 Ω·cm2) compared to the 2C variant suggests that excessive cross-linking may introduce microscopic defects that compromise coating integrity.
The incorporation of ZnO nanoparticles significantly enhanced the impedance characteristics, with Nyquist plots data showing substantially larger semicircle diameters and higher charge transfer resistances. Epoxy-TA-BZ-ZnO-2C (H) demonstrated the highest Rct value of 16,200 Ω·cm2, representing a 35% improvement over its ZnO-free counterpart. The coating resistance values for ZnO-incorporated samples exceeded 2.0 × 106 Ω·cm2, with Epoxy-TA-BZ-ZnO-2C achieving 2.8 × 106 Ω·cm2. These enhanced impedance values directly correlate with the reduced corrosion current densities observed in potentiodynamic polarization measurements.
Equivalent circuit modeling and mechanistic insights
The EIS data were fitted using appropriate equivalent circuit models to quantify the individual contributions of coating resistance, charge transfer resistance, and capacitive elements. For intact coatings, a two-time-constant model Rs(Qc(Rc(QdlRct))) was employed, where Rs represents solution resistance, Qc is the coating capacitance (constant phase element), Rc is the coating resistance, Qdl is the double-layer capacitance, and Rct is the charge transfer resistance. The fitting quality, assessed through chi-squared values (<10-3), confirmed the validity of the equivalent circuit model.
The systematic variation in circuit parameters provides mechanistic insights into the corrosion protection mechanisms. The coating capacitance values (Qc) ranged from 8.5 × 10-9 to 1.2 × 10-8 F·cm-2, with lower values indicating reduced electrolyte uptake and enhanced barrier properties. ZnO-incorporated samples consistently showed lower Qc values, confirming improved barrier characteristics. The double-layer capacitance (Qdl) values varied from 2.1 × 10-5 to 3.8 × 10-5 F·cm-2, with variations attributed to changes in the effective electrode area and surface roughness modifications induced by ZnO nanoparticles. The phase angle analysis from Bode plots revealed additional insights into coating behavior across different frequency ranges. At high frequencies (>104 Hz), all samples exhibited phase angles approaching (−90°), indicating predominantly capacitive behavior characteristic of intact coatings. At intermediate frequencies (101-103 Hz), the phase angle minima provided information about coating relaxation processes, with ZnO-incorporated samples showing broader phase angle ranges, suggesting more complex impedance behavior associated with nanoparticle-matrix interactions.
Cyclic voltammetry and barrier property assessment
The cyclic voltammetry curves as shown in (Figures S5 and S6) provided complementary information about the coating’s barrier properties and electrochemical stability. Thermally cured samples exhibited symmetrical profiles with low current densities, indicating effective barrier properties and minimal electrochemical activity. Epoxy-TA-BZ-2C (H) demonstrated the most favorable CV characteristics, with a narrower hysteresis loop and lower current density compared to other compositions. The hysteresis loop width directly correlates with the coating’s capacitive behavior and barrier effectiveness, with narrower loops indicating superior protective properties.
ZnO incorporation significantly improved the CV characteristics, with all ZnO-containing samples showing lower current densities and narrower hysteresis loops compared to their ZnO-free counterparts. The CV curves for Epoxy-TA-BZ-ZnO-2C (L) showed the most favorable characteristics, with minimal current density variations and excellent reversibility. The observed improvements in CV behavior align with the enhanced barrier properties and reduced corrosion rates determined from potentiodynamic polarization and EIS measurements.
The broader hysteresis loop observed for Epoxy-TA-BZ-4C (I) and slight increases in current density for Epoxy-TA-BZ-ZnO-4C (M) suggest that optimal performance requires balanced composition and processing conditions. These observations are consistent with the concept that excessive cross-linking density or nanoparticle agglomeration can introduce microstructural defects that compromise coating performance.
Synergistic effects and structure-property correlations
The comprehensive electrochemical analysis reveals clear synergistic effects between thermal curing, ZnO incorporation, and compositional optimization. The systematic improvement in corrosion resistance follows two distinct trends: within ZnO-free samples, optimal performance is achieved at intermediate cross-linking density (H), while ZnO incorporation enhances performance across all compositions. The 25% improvement in corrosion resistance observed for ZnO-containing samples compared to their ZnO-free counterparts demonstrates the effectiveness of nanoparticle incorporation.
The correlation between Tafel slopes, impedance parameters, and CV characteristics provides robust evidence for the proposed corrosion protection mechanisms. The dual role of ZnO nanoparticles as physical barriers and sacrificial elements is confirmed by the systematic changes in electrochemical parameters. The optimal balance between coating resistance and charge transfer resistance achieved by Epoxy-TA-BZ-ZnO-2C (L) results in superior overall performance, with coating efficiency exceeding 93%.
Statistical analysis of the electrochemical parameters confirms the significance of observed improvements, with p-values <0.01 for ZnO-incorporated samples compared to the control. The consistency of improvements across multiple electrochemical techniques validates the robustness of the coating performance and the reliability of the characterization methods employed.
Long-term stability and environmental durability
Extended electrochemical testing revealed the long-term stability of the protective coatings under simulated service conditions. Time-dependent EIS measurements over 720 h of exposure demonstrated that ZnO-incorporated samples maintained superior impedance characteristics compared to ZnO-free counterparts. The coating resistance values showed minimal degradation (<15%) for optimal compositions, while charge transfer resistance remained stable throughout the testing period.
The excellent long-term stability is attributed to the synergistic effects of enhanced barrier properties, effective nanoparticle dispersion, and optimal cross-linking density. These findings confirm that the developed composite coatings provide reliable long-term corrosion protection suitable for demanding industrial applications requiring exceptional durability and environmental resistance.
Comprehensive electrochemical and material property comparison
The electrochemical characterization of our synthesized materials reveals systematic improvements across the sample series, demonstrating clear correlations with other material properties. Cyclic voltammetry analysis shows distinct differences between ZnO-free and ZnO-containing samples, with current densities decreasing from ±2 × 10-5 A for Epoxy-TA-BZ samples to ±4 × 10-5 A for ZnO-incorporated composites. These improvements align perfectly with structural and thermal characteristics observed through other analytical techniques.
The potentiodynamic polarization results demonstrate progressive enhancement in corrosion resistance, with corrosion rates decreasing from 0.095 mm/year for Epoxy-TA-BZ-1C to 0.069 mm/year for Epoxy TA-BZ-4-ZnO-C. This systematic improvement correlates strongly with increasing cross-linking density, as evidenced by FTIR spectroscopy showing the disappearance of the oxazine ring peak at 950 cm-1 and reduced O-H band intensity in the 3200-3600 cm-1 region.
Surface analysis through SEM and AFM reveals a clear transition from wave-like patterns in ZnO-free samples to well-defined nodular morphology in ZnO-containing composites. This structural evolution directly impacts electrochemical behavior, with more uniform and compact surfaces showing enhanced barrier properties. TEM analysis confirms optimal ZnO dispersion in samples with superior corrosion resistance, establishing a direct structure-property relationship.
XRD patterns provide additional evidence of successful material modification, showing characteristic amorphous peaks at 2θ = 20° for ZnO-free samples and additional crystalline peaks at 31.7°, 34.4°, and 36.2° for ZnO-containing composites. The intensity variations of these peaks correlate with improved electrochemical performance, particularly in samples with different TA-BZ ratios.
Thermal analysis further supports the observed electrochemical behavior. TGA results show increased thermal stability in samples with better corrosion resistance, with initial degradation temperatures rising from 350°C in ZnO-free samples to 375°C in ZnO-containing composites. Higher char yields (28-30%) in ZnO-incorporated samples correlate with enhanced barrier properties and reduced corrosion rates.
DSC measurements reveal systematic variations in curing behavior, with exothermic peak intensities ranging from 3.2 mW to 3.6 mW. These variations reflect differences in cross-linking density that directly impact corrosion protection capabilities. The samples showing higher exothermic intensities consistently demonstrate superior electrochemical performance.
The systematic improvement in properties follows two clear trends. First, within ZnO-free samples, different TA-BZ ratio leads to enhanced performance across all characterization techniques. Second, ZnO incorporation provides additional improvement, with each ZnO-containing sample outperforming its ZnO-free counterpart by approximately 25% in terms of corrosion resistance.
This comprehensive comparison demonstrates clear correlations between electrochemical behavior and other material properties. The systematic improvement in corrosion resistance aligns perfectly with enhanced structural, thermal, and mechanical properties, validating our approach to material optimization. The synergistic effects of varying TA-BZ ratio and ZnO incorporation are consistently evidenced across all characterization techniques, confirming the successful development of high-performance protective composites.
Conclusion
In conclusion, the development of Epoxy-TA-BZ-ZnO composites represents a significant advancement in enhancing anticorrosion, mechanical, and thermal properties. The incorporation of ZnO nanoparticles notably improved barrier properties, reduced corrosion rates, and increased structural integrity. Characterization techniques and corrosion testing highlighted the composites’ potential for industrial applications in harsh environments. The findings demonstrate that these composites offer a promising, sustainable solution for corrosion mitigation, emphasizing their role in the design of high-performance materials. The successful synthesis and curing at 200°C further enhanced the materials’ durability. Potentiodynamic polarization confirmed excellent corrosion resistance, with reduced rates and enhanced barrier properties. These results underscore the synergistic effects of tannic acid, epoxy, and ZnO, suggesting the composites’ suitability for advanced protective coatings. Future optimization of composition and curing conditions could expand their application potential across various industries.
Supplemental Material
Supplemental Material - Influence of ZnO incorporation on the thermal and structural properties of cured epoxy-TA-BZ composites
Supplemental Material for Influence of ZnO incorporation on the thermal and structural properties of cured epoxy-TA-BZ composites by M. Alharbi, Hafsah Klfout, Khalid A. Alamry, Ruby Aslam, Ajahar Khan, Yas Al-Hadeethi, Elena Bekyarova, S. Alqahtani, Mahmoud A. Hussein in Polymers and Polymer Composites
Footnotes
Acknowledgments
This work was funded by the University of Jeddah, Jeddah, Saudi Arabia, under grant No. (UJ-23-SRP-2). The authors, therefore, thank the University of Jeddah for its technical and financial support.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the University of Jeddah, Jeddah, Saudi Arabia, under grant No. (UJ-23-SRP-2). The authors, therefore, thank the University of Jeddah for its technical and financial support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
