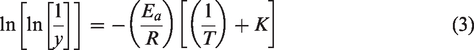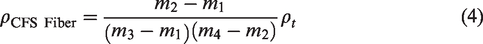Abstract
In this research article, a leftover of Cymbopogon flexuosus stem (CFS) collected from the oil extraction industry was examined for its ability as a reinforcing agent in a polymer composite. Anatomical, morphological, physical, chemical, mechanical, and thermal characteristics of the CFS fiber were examined. Chemical analysis revealed the presence of higher amount of cellulose (68.13%), which offers better bonding properties and higher tensile strength (431.19 ± 23.96 MPa). Moreover, the density of the fiber (1270 kg/m3) found using physical analysis was less than that of synthetic fibers, which paves a path in replacing hazardous synthetic fiber. Solid-state nuclear magnetic resonance and Fourier transform infrared spectrum spectroscopy analyses were conducted to study the functional groups of the extracted CFS fiber. The thermal stability (253.17°C), activation energy (73.01 kJ/mol), and maximum degradation temperature (345.08°C) were investigated by thermogravimetric analysis. X-ray diffraction analysis confirmed the semi-crystalline nature of the fiber with crystallinity index (46.02%) and crystallite size (13.96 nm). The CFS had a smooth surface, as conformed by an atomic force microscopy and scanning electron microscope analysis. Altogether, this study highlights the feasibility of leftover CFS fiber residue as reinforcement in biopolymer matrices replacing synthetic fiber.
Introduction
Cymbopogon flexuosus (CF) is commonly known as lemon grass, Cochin or Malabar grass, barbed wire grass, east Indian grass, citronella grass, amongst others, and grows about 2 m high [1]. CF are a perennial plant that is native of South India and Sri Lanka, but now it is spread across Africa, America, Australia, and Europe. CF plant belongs to the family of Poaceae grasses. In India, it grows in all the regions, especially more abundantly in the regions 4200 m above the sea level with an average rainfall of 200–250 cm annually [2]. CF can be cultivated in any wasteland, and their well-branched root aids the conservation of water and soil. It flowers frequently, and leaves are linear and lanceolate. There are two types of species of C. flexuosus, which are identified by the stem color such as red and white. The red stem species were named as C. flexuosus var. flexuosus and white stem species were named as C. flexuosus var. albescens. The red stem species were cultivated commercially and considered as a true lemongrass [1,3]. Aromatic oils were extracted from the shoot of CF, which smells like lemon. CF leaves and oils act as a stress and anxiety reducer, has antifungal detoxifying abilities, reduces fever, helps in digestive problems, lowers cholesterol, acts as a natural bug repellant, fights the flu and colds, reduces inflammation, exerts antioxidant effects. Kerala in India is number in the annual production and exporter of lemongrass oil. Nearly 90% of world trade is shipped from the Cochin Port, and hence it is properly named as Cochin oil. The oils are extracted by the steam distillation process. After distillation, stem residues are thrown out as waste that is dried and fired, which creates environmental issues [4]. Every year, in a 1-ha CF field, nearly 12–15 tons of residues are available for disposal as waste.
Environmental concerns have led researchers to use renewable and biodegradable resources as substitute materials for reinforcement in the polymer matrix because of their unique features compared to conventional synthetic fibers. Several health problems such as allergy, skin infection, cancer are associated with the long-term usage of synthetic fibers [5]. Moreover, synthetic fibers are non-biodegradable and non-recyclable, which create environmental issues [6]. These issues motivated researchers to search viable natural fibers extracted from root, stalk, stem, fruit, leaf, and bark of various new plants for high-quality fibrous applications especially in automotive, textile, marine, construction, and packaging industries. Recently, various fibers such as Phoenix dactylifera L [7], Dracaena reflexa [8], Chrysopogonzizanioides [9], Saccharum spontaneum [10], Thespesia populnea barks [11], guaruman fiber [12], Acacia tortilis [13], corn stalk waste [14], Phoenix pusilla leaves [15], Cocciniagrandis L [16], Tamarindus indica L [5], Juncus [17], and Pennisetum purpureum stem [18] were examined to study their feasibility as a reinforcing agent in the polymer composite. The discarded Cymbopogon flexuosus stem (CFS) from a distillation plant for oil extraction was investigated in this study. To the best of our knowledge, research report is not available on the characterization of CFS fibers as potential polymer reinforcement. In this investigation, leftover residues of C. flexuosus stem were used to determine their feasibility in polymer composite and textile industry by examining their chemical composition, physical properties, microstructure, crystalline properties, thermal stability, chemical functional groups, tensile strength, and surface roughness.
Materials and methods
Extraction of CFS fiber
Leftover CFS were collected from residue of lemongrass oil extraction plant in Aromatic and Medicinal Plants Research Station (formerly known as Lemongrass Breeding Station), Ernakulam, Kerala. The collected CFS were cleaned to remove dirt and submerged into water for 15 days to enhance the microbial degradation process. After 15 days, the stems were washed out scrupulously in freshwater. Then, the stems were placed in direct sunlight for a week to remove the wetness present in it. The stems were mined using a metal-teeth brush to remove the outer layer and soils present in them (Figure 1).

(a) Cymbopogon flexuosus (Lemon grasses) plant and flower, (b) stem residue and cross section of stem, (c) stem under microbial degradation process, (d) extracted stem fiber.
Cf stem anatomy
The extracted stem was used for anatomical studies to analyze the microstructure. The stem fibers were sliced in 0.1 × 0.1 cm size and soaked in the formalin-aceto-alcohol solution. The above steps were followed for 24 h to conserve the tissues. Then, the tissues were dried out through a graded (CH3)3COH and enclosed in a paraffin block [19]. Thereafter, these blocks were sliced in size of 10–12 μm and affixed to a glass slide. To make a clear distinction between different kinds of tissue, they were counterstained with a mixture of safranin and toluidine blue [20]. The fiber anatomy was analyzed with the help of a polarized light microscope by examining the slides.
13C Nuclei solid-state nuclear magnetic resonance
Solid-state nuclear magnetic resonance (NMR) spectra were analyzed to determine the chemical composition of CFS using JEOL ECX 400 MHz spectrometer. The sample is placed in a strong magnetic field and analysis is conducted by calculating the interaction of nuclear spins. This investigation was conducted in cross polarization mode at a sample spinning rate of 10 kHz. The experiment was done at 25°C with 13C nuclei operating frequency of 75.46 MHz.
Fourier transform infrared spectrum
The nature of the chemical functional group of CFS fiber was identified by obtaining an infrared absorption spectrum by Fourier-transform infrared spectroscopy (FTIR) spectroscopy. The extracted CFS fibers were dried and crushed to a fine powder. A spectrometer (model FTIR-8400S; Shimadzu, Korea) was used to attain the spectra in the range of 500–4000 cm−1, in a resolution of 2 cm−1 and scanning range of 32 scans per min.
X-ray diffraction
Physical characteristics and crystallographic structure of the CFS fiber were studied using X-ray diffraction (XRD) analysis. The powdered CFS fiber prepared by standard protocols was tested (Bruker-D8 XRD model). XRD operating conditions were set as wavelength 0.154 nm, scans of scattering angle (2θ) against intensity from 10 to 80 θ (step size and scan of 0.002, 10.16 s at room temperature) with a setting of 30 mA and 40 kV. The following equation [21] was used to calculate the crystallinity index (CI) of the CFS
Thermogravimetric analysis
A thermal study of powdered CFS fiber was carried out using a thermogravimetric analyzer (SIINT 6300; Japan). Sample weighing 5.856 mg was treated at 10°C/min from 28°C to 600°C at airflow of 50 ml/min. The variation in the mass was recorded for every 5°C to evaluate the thermal stability of the CFS fiber.
Broido’s equation [22] was used to calculate the kinetic activation energy (Ea) of the CFS fiber to authenticate the CFS fiber as reinforcement in a composite application
Chemical compositions
Standard test procedures were used for calculating cellulose, hemicelluloses, moisture, lignin, ash, and wax contents in the CFS fiber. The cellulose weight fraction was evaluated based on Kushner and Hoffer method, and hemicellulose percentage was calculated by NFT 12-008 standard [23]. Moisture content in the CFS fiber was determined with the use of an electronic moisture analyzer (Mettler Toledo HS153). The Klason method was used to assess CFS fiber’s lignin content according to APPITA P11s-78 standard [24]. Conrad method [25] was used to quantify the wax content. The ash content was determined using the procedure stated by Technical Association of the Pulp and Paper Industry [26].
Physical composition
Density measurement
The density of the CFS fiber was measured by a pycnometer [27] using toluene (known as Toulon) using the following equation
Diameter measurement
Measurement of fiber diameter is complicated due to uneven fiber shape with artifacts. However this can be resolved by measuring 25 samples of CFS fiber kept under a Carl Zeiss optical microscope (resolution of 1 micrometer) in a longitudinal direction [28]. Diameter at four places in each sample was recorded, and the average value was computed to determine the fiber diameter.
Single CFS fiber tensile test
A Zwick/Roell Universal Testing Machine was used to determine the tensile properties of the CFS fiber. The test was performed according to the ASTM D3822-07 standard. Owing to the uneven nature of the fiber such source, maturity, mechanism of fiber extraction, 30 samples for each gauge lengths (50, 40, 30, 20, and 10 mm) were tested with a stable crosshead speed of 0.1 mm per minute with 65% relative humidity; room temperature was maintained to determine the average tensile value of the fiber [5]. The microfibril (α) angles of the CFS fiber were calculated [29] using the equation given below
Where α is the microfibril angle (in degree), ε is the strain, ΔL is elongation at break (in mm), and Lo is gauge length (in mm).
Mechanical properties of natural fiber are difficult to determine as they depend on plant maturity, extraction methods, defects in fiber surfaces, difference in diameter, climate, and soil conditions of the plant. Therefore, is essential to use statistical analysis techniques to assess the mean values of mechanical properties. All 30 samples results were analyzed by Weibull distribution in Minitab 17 software.
Scanning electron microscope
The morphology study of the CFS fiber was ascertained by a scanning electron microscope (SEM) using VEGA 3TESCAN. Before coating the fibers were sliced to a height of 10 mm for easy access. Then the thin gold film coating was done on the fiber surface to enhance the image resolution as well as to avoid the accumulation of electron beam charging effects during analysis. Then they were dried in an vacuum for half an hour at 100 °C and surface structure of CFS were recorded with different magnifications performed under 10–30 kV accelerating voltage.
Edx spectroscopy analysis
Elemental composition in the surface of the CFS fiber is determined by Energy Dispersive X- Ray Spectroscopy (INCAPentaFETx3), which is equipped with SEM machine. Using a special tape samples were mounted on SEM holder and measurements were made on surface of CFS fiber at five different locations and average value was measured. Existence of elements present on the fiber external surface was determined in weight and atomic percentage.
Atomic force microscopy
The surface roughness parameters were analyzed with the help of XE-70 AFM (Park Systems Ltd, Korea) equipped with a piezoelectric scanner with a tip radius of curvature less than 10 nm. This atomic force microscope is capable of providing resolution up to nano-scale accuracy. The images were collected in non-contact mode. Initially the sample of 0.01 g of CFS fiber was mixed in 50 mL of acetone and sonicated for 20 min. Then the solution was dried in room temperature and surface topography analysis was performed under atmospheric condition.
Results and discussion
Analysis of CFS stem anatomy
The transverse section (TS) of a well-developed matured CF stem is demonstrated in the Figure 2(a) to (d) at different magnifications. The stem is thin and circular in the transactional view, which consists of a typical atactostele that is a feature of monocot stem. The surface of the stem is smooth and even. A thin epidermal layer of small, circular, thick walled cells and thick continuous cortical cylinder of fibers with very thick, lignified secondary walls are observed in the Figure 2(a).These cortical fibers (CFi) are circular, less compact and holds small, circular central cell lumen. Figure 2(b) shows a clear view of outer cortical vascular bundles and cortical fibers at 40× magnification. The cells towards the interior of cortical zone are also fibers, which are slightly longer and less compact with wide and circular lumen at the centre. These fibers have thin primary wall and thick secondary wall. Along with the cortical fiber cylinder, there are several radial layers are present, those are radially elliptical, thick walled cells. These radial lines of cells separate the cortical vascular bundles at regular intervals. Throughout the stem, ground parenchyma (GPa) cells are evenly distributed (Figure 1a). Such ground parenchyma cells are polygonal in outline and thin walled. The compactly arranged cells in ground parenchyma tissue form uniform circular vascular bundles (VB). These vascular strands are collateral with vessels and arranged in ‘Y’ shaped outline where the protoxylem elements appeared in endarch orientation. There are two extensive circular metaxylem elements located side by side and a circular unit of phloem is seen in between metaxylem elements. Besides that, one protoxylem element is present in the inner vascular bundle (INVB). The inner vascular bundles are 200 µm in thick and which are surrounded by a thick bundle sheath of fibers (Figure 2c). Such bundle sheath fibers are narrow, angular, thick walled and compact.

Optical microscopic transverse section of CFS – (a) 10×, (b) 20× (c) 40× and (d) 80×.
Moreover, the anatomical study clearly revealed the position and features of the fibers present in the CFS. Fibers are present in the outer cylinder where cortical bundles are seen. The presence of fibers in the outer cortical and inner cortical portions shows the plants has potential of yielding natural fibers for futuristic applications. It is also observed that the size of fibers increases centripetally. The inner optical fibers are angular where wide circular cell lumen is present. The inner fiber cells possess a diameter of 6 µm, cell wall thickness of 4 µm and a cell lumen is up to 8 µm wide, while the outer optical fibers are more or less wide and angular in outline. They are 15 µm wide with a cell wall thickness of 5 µm, in which the primary walls are 2 µm thick and the lumen is 3 µm wide (Figure 2d).
Analysis of X-ray diffractogram
X-ray diffractogram of CFS in Figure 3(a) shows two enlarged diffraction peaks of 15.72° (1 1 0) and 21.96° (2 0 0), which are clearly observed in most of the plant fibers belonging to crystallographic plane. As reported by various researcher [6,16,24,30,44,71] the two major peaks present around 2θ = 15.72° and 21.96° are the relevant peaks of cellulose I and IV of a monoclinic structure. The peak observed at 2θ = 15.72° shows the existence of amorphous constituent in the CFS fiber. Using equation (1), the CI of the CFS was calculated to be 46.02 nm. The higher CI values indicate better orderly nature of cellulose crystals [37]. The resulted CI value is greater than Artisdita hystrix (44.85 nm) [52], Juncus (43 nm) [17], Cyperus pangorei (41 nm) [47], Tridax procumbens (34.6 nm) [24], and Juncus effuses L (33.4 nm) [55]. During polymer reinforcement, a higher value of crystallite size suggests a decrease in chemical reactivity and water absorption capacity of the fiber [44].Using equation (2), crystallite size (L) of CFS fibers was found to be 65.49 nm, which was higher than that of other natural fibers such as Raffia textilis (32 nm) [72], T. procumbens (25.04 nm) [24], red banana peduncle (12.1 nm) [23], Cissus quadrangularis root (7.04 nm) [29], aerial roots of banyan tree (6.28 nm) [27], T. populnea (3.576 nm) [11], Prosopis juliflora bark (2.8 nm) [40], C. grandis stem (1.91 nm) [16], and chakshir (1.6 nm) [54]. Crystallinity index and Crystallite size of CFS fiber in comparison with other natural fiber is represented in the Table 1.
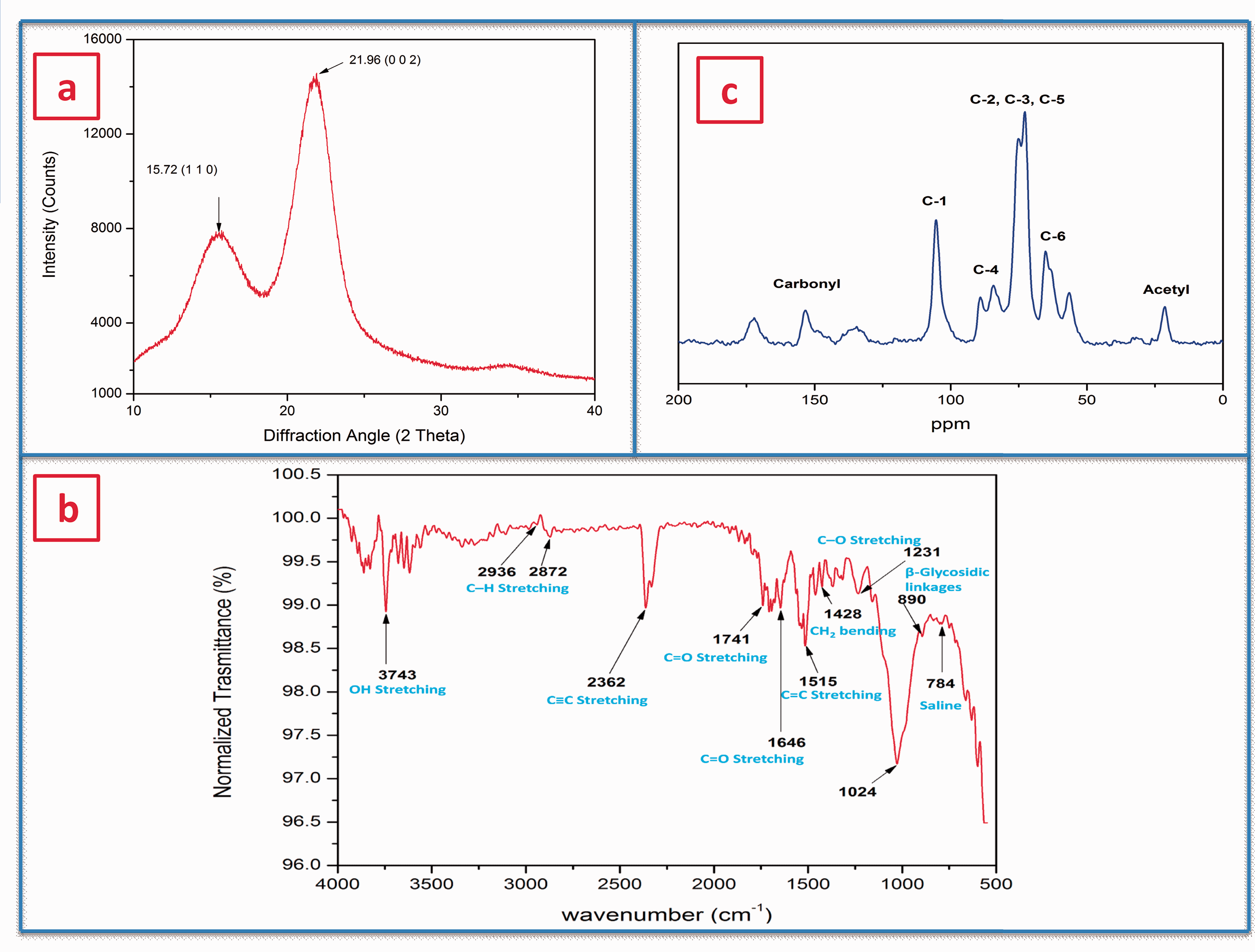
(a) XRD peak of the CFS fiber, (b) FTIR vibrational spectra of CFS fiber, and (c) NMR spectrum of CFS fiber.
Listing the properties of CFS fiber in comparison with other fiber.

Analysis of FTIR vibrational spectra
Figure 3(b) shows the FTIR vibrational spectra of the raw CFS fiber from 500 to 4000 cm−1, which exhibits the existence of cellulose, hemicelluloses, and lignin in the form of different absorption bands. Table 2 lists different band positions of the observed CFS fiber in comparison with other reports. The higher intensity around 3100–3800 cm−1 (3743 cm−1) is attributed to the presence of hydrogen-bonded OH stretching. The peaks at 2936 and 2872 cm−1 indicate C–H stretching vibration from CH and CH2 in cellulose and hemicellulose [14,58].
FTIR peak observed for CFS fiber.

The peak at 2362 cm−1 indicates the presence of wax or a substance like wax, which denotes an alkyl chain band [16,59]. The peaks at 1741 and 1646 cm−1 confirm the existence of carbonyl C = O acetyl group of hemicelluloses [73]. The sharp peak at 1515 cm−1 indicates the presence of water in the CFS fiber(C = C groups) of lignin, and the peak at 1428 cm−1 is due to (CH2) methylene bending frequency of cellulose. The band at 1231 cm−1 is attributed to the availability of the C–O stretching vibration of the acetyl group in lignin. The appearance of a wideband around 1024 cm−1 is assigned to the C–O and O–H stretching of hydroxyl and ether groups in cellulose. The band seen at 890 cm−1 confirms the β-glycosidic linkages between the monosaccharides. The peak at 784 cm−1 shows the existence of saline in the CFS [28]. FTIR peak observation and the functional groups present in the CFS is represented in the Table 1.
Analysis of 13C (CPMAS) NMR spectrum
The existence of cellulose, hemicellulose, and lignin contents in the CFS fiber was again confirmed by experimenting with high-resolution 13C (CPMAS) NMR spectrum, as shown in Figure 3(c). The peak observed at 65.125 ppm corresponds to the amorphous cellulose contents (C-6) [58]. The peak between 84.26 and 89.15 ppm confirms the presence of C-4 carbon in the crystalline domain. Among the peaks observed, the maximum intensity in the peaks is seen from 75.2 to 72.71 ppm (assigned as C-2, C-3, and C-5), which indicates a higher amount of carbon rings of cellulose present in the CFS fiber [58]. The peak at 105.45 ppm is related to C-1 (anomeric carbon) contents present in the fiber. The peak at 21.24 ppm is allocated to acetyl groups of hemicelluloses [46]. The small peak occurred between the spectra 134.48 and 172.26 ppm is related to lignin constituent. Similar peaks were noticed in various biofibers of plants such as Furcraea foetida [34], Ficus religiosa [58], and Napier grass [18].
Analysis of TGA and DTG
The determination of CFS fiber thermal stability is another important criterion as it determines the correct range of composite processing temperature. Figure 4(a) shows the combined profile of thermogravimetric analysis (TGA) and derivative thermogravimetry (DTG) of the powdered CFS fiber. The initial degradation of 8.41% occurred at 95.37°C, which indicates evaporation of moisture and wax contents present in the CFS fiber. Degradation peak occurred at 253.17°C with a mass loss of 4.42%, which is relevant to the reduction of hemicelluloses, lignin, and glycosidic links of cellulose [28]. Hence, the CFS fiber is thermally stable up to 253.17°C. A major decline in mass (around 59.04%, noted by the steep curve) occurred from 253.17 to 352.60°C due to complete degradation of the cellulose, as reported for various other fibers such as the root of the banyan tree (370°C) [27], F. foetida (355°C) [34], C. grandis L (351°C) [16], D. reflexa (348°C) [8], C. quadrangularis stem (342°C) [28], and root of Ficus religiosa tree (325°C) [58]. Further, less prominent drops in mass (26.69%) at 477.74°C occurred due to omission of lignin, and remaining mass of 1.44% above 477.74°C was considered as a charred residue of the fiber [27]. The major peak observed in DTG at 345.08 °C indicates thermal degradation of cellulose I and the entire decomposition of α-cellulose. A similar drop in the peak was also observed in Artisdita hystrix leaf fiber (298.2°C) [52], F. foetida (320°C) [34], C. grandis stem (320°C) [16], Ficus religiosa tree root (325°C) [58], C. quadrangularis root (328.9°C) [29], and Lygeum spartum (338.7°C) [53].
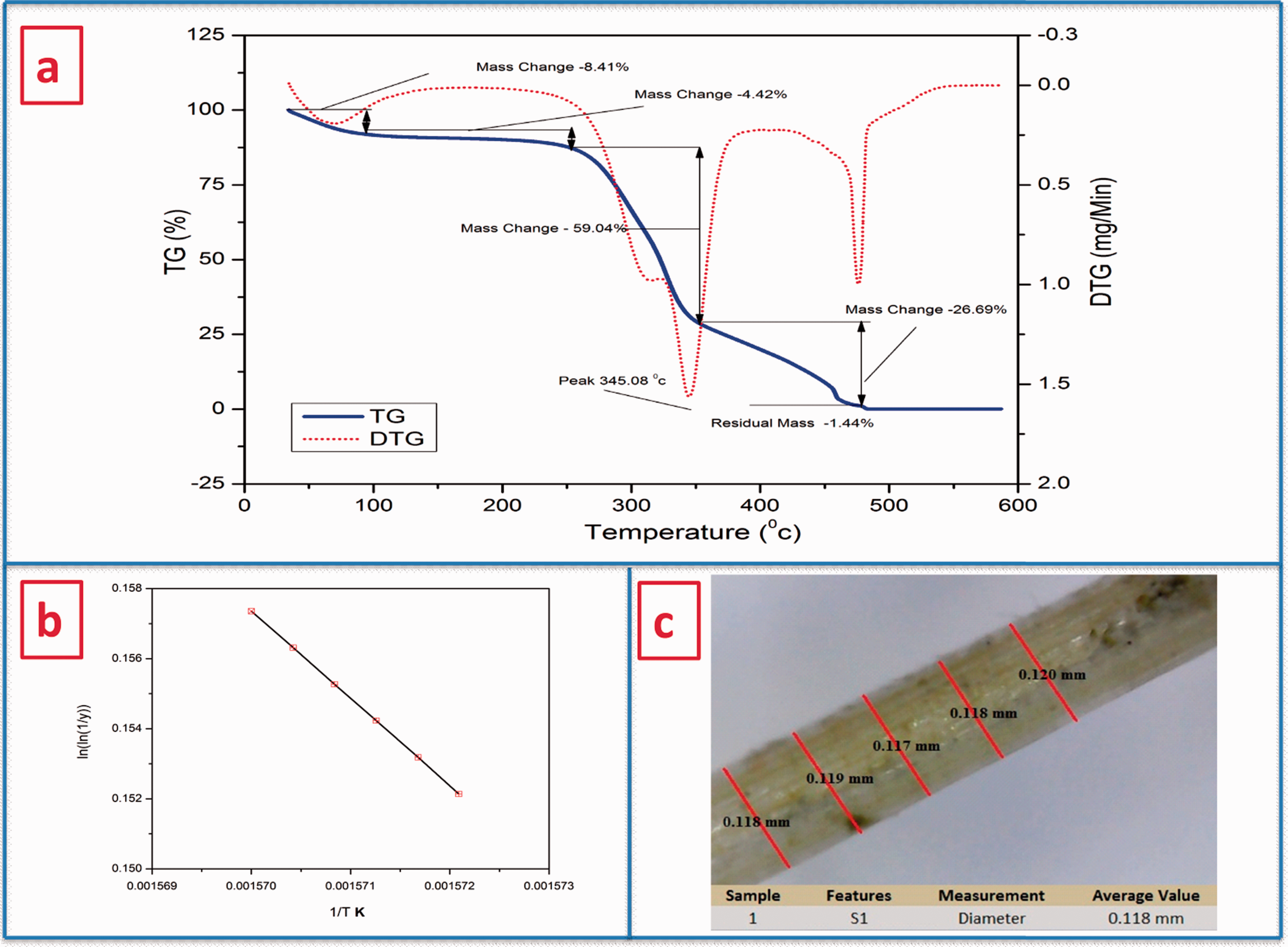
(a) TGA/DTG, and (b) Broidio’s plot of CFS fiber, and (c) Optical microscopic view of CFS fiber for diameter confirmation.
The thermal stability of the CFS fiber is one of the important factors that were measured by calculating the kinetic activation energy (Ea). Broidio’s plot (Figure 4b) was used to interpolate kinetic activation energy and Ea was found to be 73.01 kJ/mol, which is within the specified range for natural materials (60–170 kJ/mol) [34]. This shows that the CFS fiber enhances thermal stability to hold up the polymerization process temperature while manufacturing fiber-reinforced polymer composite. The above result confirms that the CFS fiber–reinforced composite can withstand temperature up to 345.08°C.
Analysis of physical composition
Evaluation of fiber density is vital as it differentiates natural fibers from synthetic fibers. The average density of the CFS fiber was found to be 1270 kg/m3, which is significantly lower than that of synthetic fibers such as E-glass fiber (2500 kg/m3), carbon (1400 kg/m3), aramid fibers (1400 kg/m3) [28], and some natural fibers such as cotton (1600 kg/m3), C. grandis stem (1517.5 kg/m3), C. quadrangularis root (1510 kg/m3), hemp (1500 kg/m3), seagrass (1500 kg/m3), flax (1500 kg/m3), ramie (1500 kg/m3), sisal (1500 kg/m3), L. partum L (1499 kg/m3), T. populnea (1412 kg/m3), kenaf (1400 kg/m3), piassava (1400 kg/m3), and Kydia calycina (1400 kg/m3) [29,67]. This property makes the CFS fiber suitable for the fabrication of lightweight composite materials. The average diameter from the 25 samples was found to be 0.118 mm using optical microscope and shown in Figure 4(c). CFSF density and its diameter were compared with those obtained for other natural and synthetic fibers (Table 1).
Analysis of chemical composition
Mechanical characterization of natural fiber can be done based on the weight percentage of each chemical composition. The higher cellulose content in the fiber guarantees higher tensile strength and modulus of elasticity [60]. The CFS fiber showed high cellulose content of 68.13%, which is higher than that of G. tilifolia (62.8%) [42], C. grandis (62.35%) [45], P. juliflora bark (61.65%) [40], Saharan aloe vera (60.2%) [22], C. dichotoma (59.7%) [34], A. hystrix (59.54%) [52], areca palm leaf stalk (57.49%) [68], seagrass (57%) [28], Ficus religiosa tree root (55.58%) [58], Napier grass strands (47.12%) [49], alfa (45.4%) [74], Althaea officinalis L (44.6%) [50], Arundo donax (43.2%) [51], and Pergularia tomentosa L seed fiber (43.8%) [56]. The CFS fiber cellulose content was near to that of other natural fibers such as Borassus fruit (68.94%) [8], Cyperus pangorei (68.5%) [47], Perotis indica (68.4%) [44], aerial roots of banyan tree (67.32%) [27], Rhectophyllum camerunense (68.2%) [8], Acacia arabica (68.10%) [8], and Acacia leucophloea (68.09%) [69]. When hemicellulose content in the fiber was increased, an adverse effect on the strength of the fiber was observed, which was due to disintegration of cellulose microfibrils [11]. The CFS fiber had 11.56% hemicelluloses content, which was comparatively lower than alfa (38.5%) [28], seagrass (38%) [28], mulberry barks (37.38%) [70], Napier grass strands (31.27%) [49], bamboo (30%) [76], C. dichotoma (23.6%) [34], G. tilifolia (21.2%) [42], A. donax (20.5%) [51], mendong grass (20.2%) [8], Heteropogon contortus (19.34%) [64], areca palm leaf stalk (18.34%) [68], Sida cordifolia (17.63%) [46], P. juliflora bark (16.14%) [40], P. tomentosa L seed fiber (16%) [56], S. rhombifolia (15.43%) [60], Borassus fruit (14.03%) [8], Saharan aloe vera (14.2%) [22], root of Ficus religiosa tree (13.86%) [58], Salago fiber (13.6%) [65], A. leucophloea (13.60%) [69], A. officinalis L (13.5%) [50], aerial roots of banyan tree (13.46%) [27], C. grandis (13.42%) [16], Dichrostachys cinerea (13.08%) [59], T. populnea (12.64%) [11], Manicaria saccifera palm (12%) [57], and A. hystrix (11.35%) [52]. Higher lignin content prevents microbial attack in the fiber, but at the same time, it shows a negative influence on fiber structure and its morphology [59]. The CFS fiber contains 23.9% lignin, which is moderate. While manufacturing polymer composite structure, fiber with low wax content will establish a good bonding capacity [30]. The wax content of sisal (2%) [28], alfa (2%) [28], P. tomentosa L seed fiber (1.88%) [56], aerial roots of banyan tree (0.81%) [27], hemp (0.8%) [28], kenaf (0.8%) [28], C. grandis (0.79%) [16], T. populnea (0.76%) [11], Ficus religiosa tree root (0.72%) [58], Areca palm leaf stalk (0.71%) [68], T. procumbens (0.71%) [24], P. juliflora bark (0.61%) [40], Acacia planifrons (0.57%) [43], and D. cinerea (0.57%) [77], which is comparatively higher than the CFS fiber wax content (0.53%). A similar bonding effect was also observed with moisture content present in the fiber. The CFS fiber had a low moisture content (9.17%), which was lower than Salago fiber (50.21%) [65], M. micrantha Kunth ex H.B.K. (34.30%) [71], S. rhombifolia (12.02%) [60], T. procumbens (11.2%) [24], T. populnea (10.83%) [11], Sansevieria ehrenbergii (10.55%) [38], aerial roots of banyan tree (10.21%) [27], D. cinerea (9.82%) [59], P. juliflora bark (9.48%) [40], red banana peduncle (9.36%) [23], and Areca palm leaf stalk (9.35%) [68]. Amorphous nature was reduced due to ash content in the fiber [45]. The CFS fiber had 4.81% ash content, which was higher than C. grandis (4.38%) [16], P. indica (4.32%) [44], mendong grass (4.2%) [48], S. rhombifolia (4.07%) [60], A. planifrons (4.06%) [43], aerial roots of banyan tree (3.96%) [27], red banana peduncle (2.79%) [23], P. tomentosa L seed fiber (2.74%) [56], A. officinalis L (2.3%) [50], A. donax (1.9%) [51], T. populnea (1.80%) [11], and Areca palm leaf stalk (1.43%) [68]. CFS fiber chemical weight % were compared with those obtained for other natural fibers (Table 1).
Analysis of single CFS fiber tensile test
For any natural fiber, chemical composition (especially cellulose content) significantly influences its mechanical properties such as tensile strength [46]. Table 3 shows the results of tensile characteristics, Young’s modulus, strain to failure % of the CFS fiber tested under different gauge lengths (GLs) where a minor deviation exists with respect to GL. Higher GL shows a sudden failure due to a larger raise of flaw as they are interlinked with each other. Increase in the GL will increase Young’s modulus and decrease the strain to failure % of the CFS fiber [16]. The tensile strength of the CFS fiber was found to be 431.19 ± 23.96 MPa, which was higher than other natural fibers such as cotton (400 MPa) [28], Alfa (350 MPa) [28], L. spartum L (280 MPa) [53], G. tilifolia (273 MPa) [42], A. donax (248 MPa) [51], C.pangorei (196 MPa) [47], Borassus fruit (175.52 MPa) [8], P. purpureum (73 MPa) [78], M. sacciferapalm (72.09 MPa) [57], C. grandis (65.2 MPa) [16], C. dichotoma (65.2 MPa) [42], coconut tree leaf sheath (46.4 MPa) [58], T. procumbens (25.75 MPa) [24], Napier grass strands (13.15 MPa) [49], and ramie (8.08 MPa) [58]. The microfibrillar angle was determined to find the angle in which cellulose microfibrils embed in the lignin matrix. Smaller microfibril angle (MFA) will result in higher fiber rigidity [60]. The MFA of the CFS fiber was calculated using equation (5) and found to be 6.4°, which was lower than that of mendong grass (22.9°) [48], T. populnea (13.94°) [11], T. procumbens (13.4°) [24], C. grandis (13.25°) [16], A. hystrix (12.64°) [52], L. spartum L (12.65°) [53], red banana peduncle (12.64°) [23],Saharan aloe vera (11.1°) [22], and P. juliflora bark (10.64°) [40]. CFS fiber tensile characteristics, Young’s modulus, strain to failure %, and microfibril angle were compared with those obtained for other natural fibers (Table 1).
Mechanical properties of C. flexuosus stem fiber.

The obtained experimental results of cross-sectional area, tensile strength, Young’s modulus, and elongation of the CFS fiber were analyzed using Weibull statistical distribution fit plot for 20 trials, shown in Figure 5(a) to (d). All experimental data recorded are found inside the line and fit rightly to the distribution, showing conformity to correlation factor R2 = 0.95, which is nearer to 1. Calculated P value from the distribution plots is less than 0.05, suggesting a confidence level of 95% between the specimens. The four parameter Weibull distribution provides the close value of mechanical properties to the average experimental value obtained.

Weibull distribution plot for (a) cross-sectional area, (b) Young’s modulus, (c) tensile strength, (d) elongation at break of CFS fiber.
Surface morphology
The surface morphology analysis of the CFS fiber, shown in Figure 6, was carried out using SEM with varying magnifications. This study was carried out to determine whether CFS fiber is an effective reinforcing agent in a composite and remains firm when fiber is pulled out. Partially smooth surface was observed in the fiber, which might be due to the presence of hemicellulose and wax contents seen as white layers in the image [58]. Precasting fiber-cleaning treatments can be processed to remove the starch grains and impurities present. Although the presence of pores and debris seen in the fiber affects the tensile property, it aids better interfacial bonding with the matrix polymer by increasing the surface roughness.

Scanning electron micrographs of CFS Fiber – (a) 100×, (b) 300×, and (c) 500×.
Edx spectroscopy analysis
Figure 7(a) represents composition of chemical elements distributed on the surface of CFS fiber in relation with weight and atomic percent. The major peaks of carbon, oxygen and silica was observed. The CFS fiber also contain a small amount of antimony, zirconium and also with minor traces of elements such as niobium and silica (less than 1 wt%). The weight percentage of carbon found to be 52.32 wt%, which is greater than other natural fibers such as Acacia tortilis (42.591 wt%) [13], Coccinia grandis L (44.591 wt%) [16], Cotton fibers (46.1 wt%) [58] and less compared to sugar palm fiber (87.93 wt%) [79], Ficus religiosa (67.48 wt%) [58], Furcraea foetida (66.43 wt%) [34], Dracaena reflexa (65.13 wt%) [8], Calotropis procera (61 wt%) [80], Jute fiber (55.68 wt%) [58] and aerial root of banyan tree (52.28) [27]. This might be due to higher non-cellulosic material content in CFS. However, the concentration of oxygen was found to be (43.23 wt%) which is less than Coccinia grandis L (54.38 wt%) [16], Acacia tortilis (40.509 wt %) [13], Dracaena reflexa (34.20 wt%) [8], Furcraea foetida (33.57 wt%) [34], Ficus religiosa (25.32 wt%) [58], and sugar palm fiber (5.86 wt%) [79]. The Carbon and oxygen were found to be the predominant elements, which represents the fiber is organic in nature. Table 4 presents the elemental weight and atomic percentage of CFS fiber along with other natural fibers.

(a) EDX analysis,(b) 2D roughness surface texture, (c) 3D roughness surface texture, (d) 2D line diagram for roughness measurement of FF, and (e) roughness parameters of CFS Fiber.
EDX elemental comparison of CFS fiber with other few natural fibers [8,58].

Atomic force microscopy analysis
AFM 2D and 3D surface topographies of the CFS fiber are shown in Figure 7(b), (c), (d) and (e). The average roughness value (Ra) of the CFS fiber was reflected in the lower value of Ra as 5.311 nm, which shows that surface is smooth due to the impurity present and lignin coverage in the fiber. The surface skewness (Rsk) value of the fiber was calculated to be −0.788 nm. A negative value in skewness shows that surface of the fiber is porous [58]. Roughness kurtosis (Rku) magnitude is an important parameter to decide if a surface is rough or spiky. Its magnitude was observed as 4.761 nm, which was greater than 3 nm, confirming the spiky surface, which might be due to wax content. Average absolute height roughness (Rz), maximum peak valley height (Rt), and root mean square surface roughness (Rq) of the CFS fiber showed low values of 35.005 nm, 45.424 nm, and 7.130 nm due to smoothness of the surface. Moreover, the Rq/Ra ratio (Gaussian distribution) was 1.34, which is slightly greater than the acceptable limit of 1.31 for the most tribological surfaces [43]. Hence, the above result confirms that a slight surface treatment is needed to improve roughness before reinforcing in a composite.
Conclusion
In this study, fibers were extracted from the stem of CF plants that were discarded from the oil extraction industry. The fibers extracted were examined for their potentiality as a reinforcement in the polymer composite. They compete with hazardous synthetic fiber with its excellent properties. The following outcomes were drawn from the test results: CFS fiber density was determined as 1270 kg/m3, which is significantly lower than artificial fibers and some potential natural fibers. The chemical composition analysis and FTIR results showed that the CFS fiber has greater cellulose content (68.13%). The SEM image analysis of fiber revealed the presence of shallow pore fractions, which enhance the fiber bonding with the matrix while reinforcing. The TGA revealed that the CFS fiber was thermally stable up to 253.17°C, which is well enough to withstand the polymerization process temperature. Using Broido’s plot, kinetic activation energy was calculated to be 73.01 kJ/mol. From the XRD report, the CI and CS of the CFS fiber was calculated to be 46.02% and 13.96 nm. The AFM micrographs revealed that the CFS fiber has significantly smooth surfaces and likely need surface treatment for improvements in the surface roughness. The monotonic tensile test revealed that the CFS fiber has higher tensile strength and Young’s modulus (431.1.9±52.56 MPa and 59.881±11.19 GPa, respectively) compare to other natural fibers.
Thus, the characterization of discarded CFS fibers proves that they not only act as a substitute for harmful synthetic fibers in the composite and textile industry but also lead to zero waste management in the industry by eliminating resources that are sent as a disposal.
Supplemental Material
sj-jpg-1-jit-10.1177_15280837211007507 - Supplemental material for Characterization of industrial discarded novel Cymbopogon flexuosus stem fiber: A potential replacement for synthetic fiber
Supplemental material, sj-jpg-1-jit-10.1177_15280837211007507 for Characterization of industrial discarded novel Cymbopogon flexuosus stem fiber: A potential replacement for synthetic fiber by S Raja, R Rajesh, S Indran, D Divya and G Suganya Priyadharshini in Journal of Industrial Textiles
Footnotes
Acknowledgements
SR acknowledges the Rohini College of Engineering and Technology, Shiv Kumar - copyeditor, and Pinnacle Bio-Sciences for providing research lab facilities to carry out his research work. The authors also acknowledge the Department of Nanotechnology, Noorul Islam University, Kumarakoil, Tamilnadu, India, for sharing their facilities to complete this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.