Abstract
Polyaniline zinc oxide (PANI-ZnO) composites were synthesized via a chemical route, varying zinc oxide content from 20 wt.% to 25 wt.%. The structural integrity of the PANI-ZnO composites was confirmed through X-ray diffraction (XRD) analysis. Two-probe measurements were conducted to evaluate DC conductivity as a function of temperature in the range (
Keywords
Introduction
Conducting polymers have garnered significant interest for applications ranging from organic electronics and energy storage devices to anticorrosion coatings. Their unique properties arise from a chemical polymerization process in which small monomer units react to form long, interconnected chains or three‐dimensional networks. The degree of polymerization (DOP), which indicates the number of monomers that join during the reaction, is a key factor in determining the structural and functional properties of the resulting polymer. 1
Among these materials, polyaniline (PANI) is particularly noteworthy due to its ease of synthesis, environmental stability, and adjustable electrical conductivity. Aniline (C6H5NH2) the building block for PANI has diverse applications in industries such as dye manufacturing, biomedical research, and even aerospace, highlighting the versatile nature of its derived polymer. 2 Conductive polymers like PANI, often described as “synthetic metals,” exhibit remarkable magnetic, electrical, and optical properties, making them suitable for advanced devices including organic solar cells, light-emitting diodes, and biosensors.3,4
A promising strategy to further enhance the performance of PANI is the incorporation of nanomaterials such as zinc oxide (ZnO) nanoparticles.5–10 The formation of PANI–ZnO composites via chemical oxidative polymerization combined with sol–gel synthesis of ZnO has demonstrated notable improvements in properties such as crystallinity and conductivity.11,12 ZnO nanorods, when integrated with the PANI matrix, not only influence the microstructure but also interact with the polymer chains to tailor the conduction and thermal behaviour of the composites.13,14
Furthermore, optimizing the ZnO content and doping levels has proven critical in enhancing the DC conductivity and semiconducting behavior of these composites. For instance, an appropriate ZnO concentration has been shown to improve device performance in applications like supercapacitors, electrochromic batteries, and even in metal–insulator–semiconductor devices such as Schottky diodes.15–17 These observations underscore the importance of nanoscale engineering in modulating the intrinsic properties of conductive polymers.
In this work, we present a comprehensive investigation into the synthesis, characterization, and electrical performance of PANI–ZnO nanocomposites. By systematically varying the ZnO content and employing advanced fabrication techniques, we aim to elucidate the interplay between the composite’s microstructure and its conductive properties. Our study not only deepens the understanding of doping and conduction mechanisms in PANI-based systems but also highlights their potential for practical applications in corrosion protection, thermoelectric devices, and energy storage systems.18–23 This integrated approach to material design and characterization emphasizes the importance of tailoring composite materials to meet the demands of next-generation electronic and energy technologies.
Recent studies have continued to push the boundaries of PANI–ZnO nanocomposite research. For instance, Patel et al. 24 introduced a novel one‐pot synthesis technique that enhances the interfacial bonding between ZnO nanostructures and the PANI matrix. Their method simplifies the fabrication process while delivering composites with superior thermal stability and improved electrical conductivity. Similarly, Lee et al. 25 demonstrated that surface functionalization of ZnO nanoparticles can fine‐tune the optoelectronic properties of PANI–ZnO composites, which is critical for applications in flexible electronics and energy storage devices.
Experimental
Materials
Synthesis of polyaniline and PANI-ZnO composite.

Chemicals used in the synthesis of polyaniline/polyaniline-ZnO composite
Polyaniline was synthesized by a chemical polymerization of aniline monomer. A solution of ammonium persulfate (APS) was prepared by dissolving 10.00 g (0.05 M) of APS in 44 ml of distilled water. This mixture, which acts as an oxidizing agent, was stirred continuously using a magnetic stirrer for 30 min to form the APS solution. Additionally, 400 ml of distilled water and 5.00 ml of hydrochloric acid (0.13 M) were also used in the experiment. Highly dangerous fumes were emitted by the heating process, and the pH was adjusted up to 1. The APS solution was added drop by drop to the aniline HCl solution while it was being stirred continuously at 5°C. This process gives off heat, and it needs to be stirred for up to 4 h to complete polymerization.
After completed the polymerization process the precipitate was filtered and washed with distilled water multiple times to reduce its acidity and adjust its pH to neutral the residue was filtered using Whatman filter paper with the help of a vacuum pump and then left in open air to remove surface moisture. It was subsequently dried in a vacuum oven at 60°C for 24 h until a constant mass was achieved. The resulting dried sample of pure polyaniline was then ground using an agate mortar to obtain fine powder. The final yield of pure polyaniline was 4.00 g. ZnO composites with pure polyaniline were prepared by adding 20 wt.% and 25 wt.% ZnO to the filtered polyaniline solution, followed by stirring for 2 h.
Measurements
The X-Ray diffraction studies were performed using X-Ray diffractometer with Cu-K 
Results and discussion
X-ray diffraction
X-Ray diffraction has been achieved from Institute Advanced Material (IAM) from BZU, Multan. The X-Ray diffraction pattern of PANI and PANI-ZnO composites also studied in this chapter. These tools have a wavelength of 1.54 A° by using Cu-K X-Ray diffraction pattern of pure polyaniline.
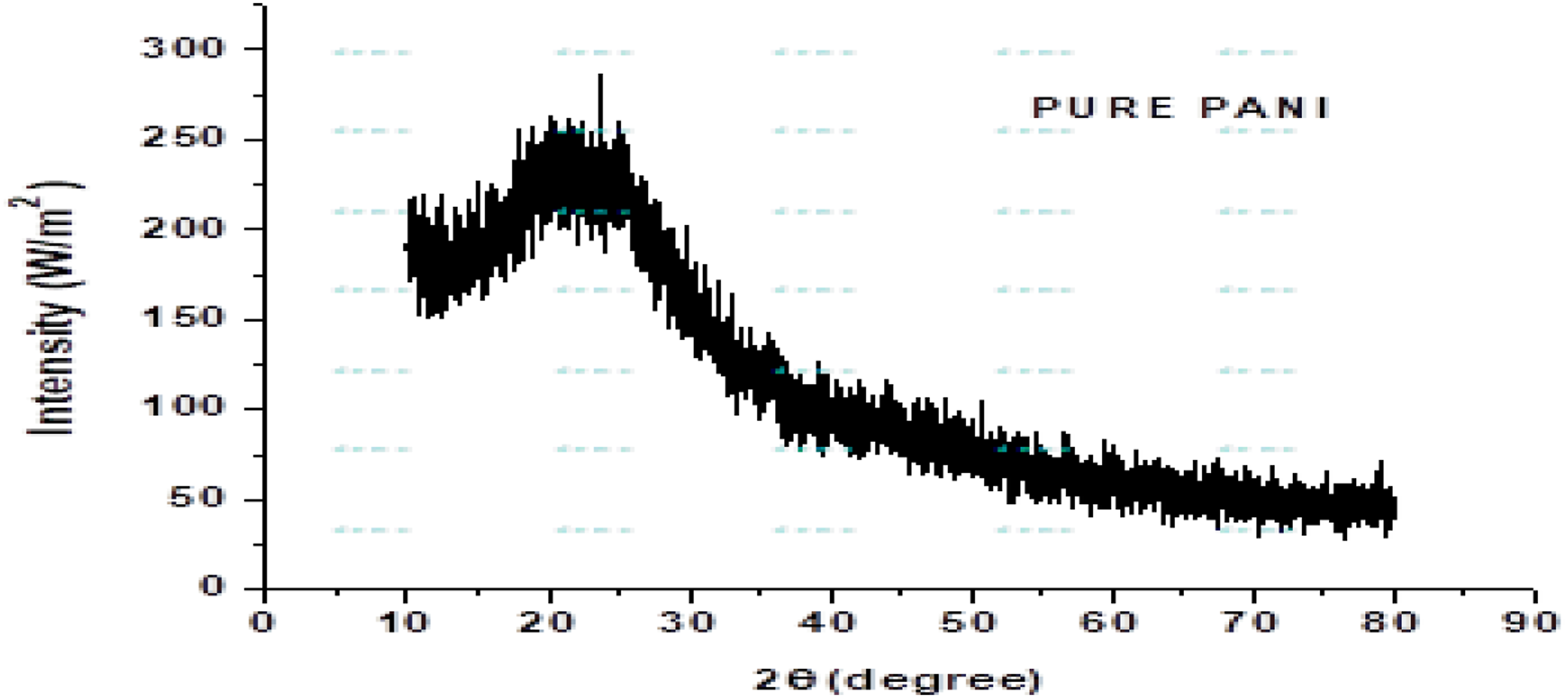
X-ray diffraction of polyaniline
The X-Ray diffraction analysis mechanisms is used to determine the different crystallinity structure which is present in Polyaniline. The structural behaviour of PANI and PANI-ZnO composites were studied by X-Ray diffraction. The X-Ray diffraction studied of Polyaniline reveal a broad peak at angle (degree) 2θ = 25.30°. This broad peak which is appear in the X-Ray diffraction pattern reveal the amorphous nature.
X-ray diffraction of zinc oxide (ZnO)
Figure 2 shows the X-Ray diffraction pattern of pure Zinc oxide (ZnO). X-Ray diffraction results declares that Zinc Oxide are well crystalline where highest intense peak is observed. The existence of sharp peaks indicates that the powder was crystalline and the single phase of ZnO. PANI-ZnO composites shows ZnO peaks as well as PANI, indicated that ZnO crystallite have been uniformly mixed with PANI.
26
X-ray diffraction of Zinc oxide.
X-ray diffraction analysis of ZnO
X-ray diffraction analysis of zinc oxide (ZnO).


X-ray diffraction of polyaniline-zinc oxide (PANI-ZnO composite)
Figure 3 shows the X-Ray diffraction pattern of PANI-ZnO composites by increase the concentration 20 wt.% and 25 wt.% of ZnO in Polyaniline chain. PANI-ZnO composites gives the highest sharp peaks of ZnO with Polyaniline which confirmed that the crystallinity of ZnO have been consistently mixed within the Polyaniline chain. It is confirmed that PANI interrelate with ZnO particles and the PANI molecular chain are stressed and leading to lower crystallinity. The peaks position of PANI-ZnO composites were shifted toward the small angle, the intensity of peaks increasing by increase the wt% of ZnO content in Polyaniline chain. X-ray diffraction of (a) PANI 20 wt.% ZnO (b) PANI 25 wt.% ZnO in stacking form.
D.C conductivity of polyaniline and PANI-ZnO composites
The D.C conductivity of Polyaniline is deliberate by temperature dependent by using 2-probe method and followings Figure 4 shows the variation of D.C conductivity at different temperature of pure PANI and PANI-ZnO composites. In both cases observed that conductivity was going to increase by increasing the temperature and wt.% of composite. Conductivity (σ) versus Temperature dependent for (a) polyaniline (b) PANI 20 wt.% ZnO (c) PANI 25 wt.% ZnO.
Conductivity was increased by increasing the wt.% of ZnO, the reason is that carrier concentration is proportional to the conductivity. ZnO is the n-type semi-conductor which having large band gap about 3.4
Conductivity was also increased by increasing the temperature, the reason is that the efficiency of charge transfer increase and the covalent bonds was broken easily which was present between PANI and PANI-ZnO Composites. Its resistance decreased so; conductivity increased by increasing the temperature.
Here the conductivity of the Polyaniline is increased by increasing the temperature
Conductivity(σ) versus temperature dependent for (a) Polyaniline-25 wt.% ZnO (b) PANI-20 wt.% ZnO (c) pure PANI.

Temperature dependent D.C conductivity
The relation between temperature and dc conductivity in polymer can provide essential information about the nature of phenomenon correlated with charge transport mechanism into a polymer
Now exponent n in the above equation is the effective dimensionally for charge transport mechanism that may depend upon inter chain coupling, “n” is the positive integer 1, 2, 3…for 1-dimension, 2-dimension or 3-dimension respectively variable range hopping model which is proposed by Mott charge transport mechanism.
The nearest neighbour Hopping process with a distribution of Activation energies can give the same type of exponential temperature dependence for conductivity.
The Activation energy is defining as
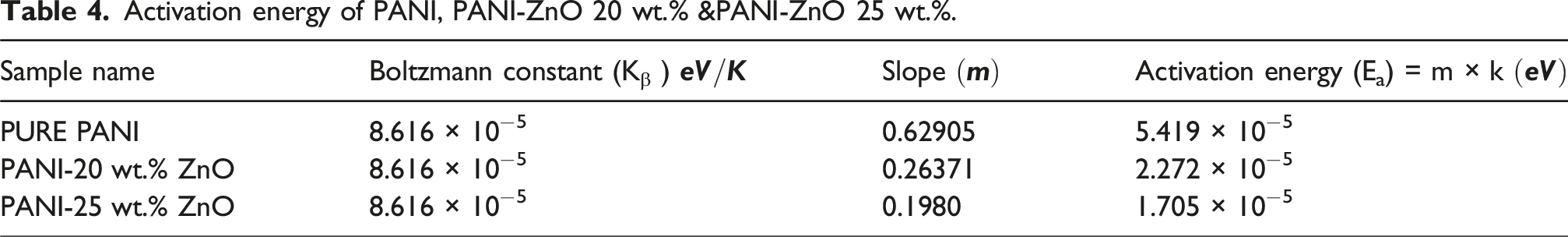
Activation energy of PANI and PANI-ZnO composite
When Polyaniline was doped with Zinc Oxide
Here we observed that activation energy about at 0 wt.% of composite is 5.419
In Figure 5 by using linear fitting on the calculated data the slope Conductivity (σ) versus 1/temperature (K −1) dependent for (a) PANI-25 wt.% ZnO (b) PANI-20 wt.% ZnO (c) polyaniline.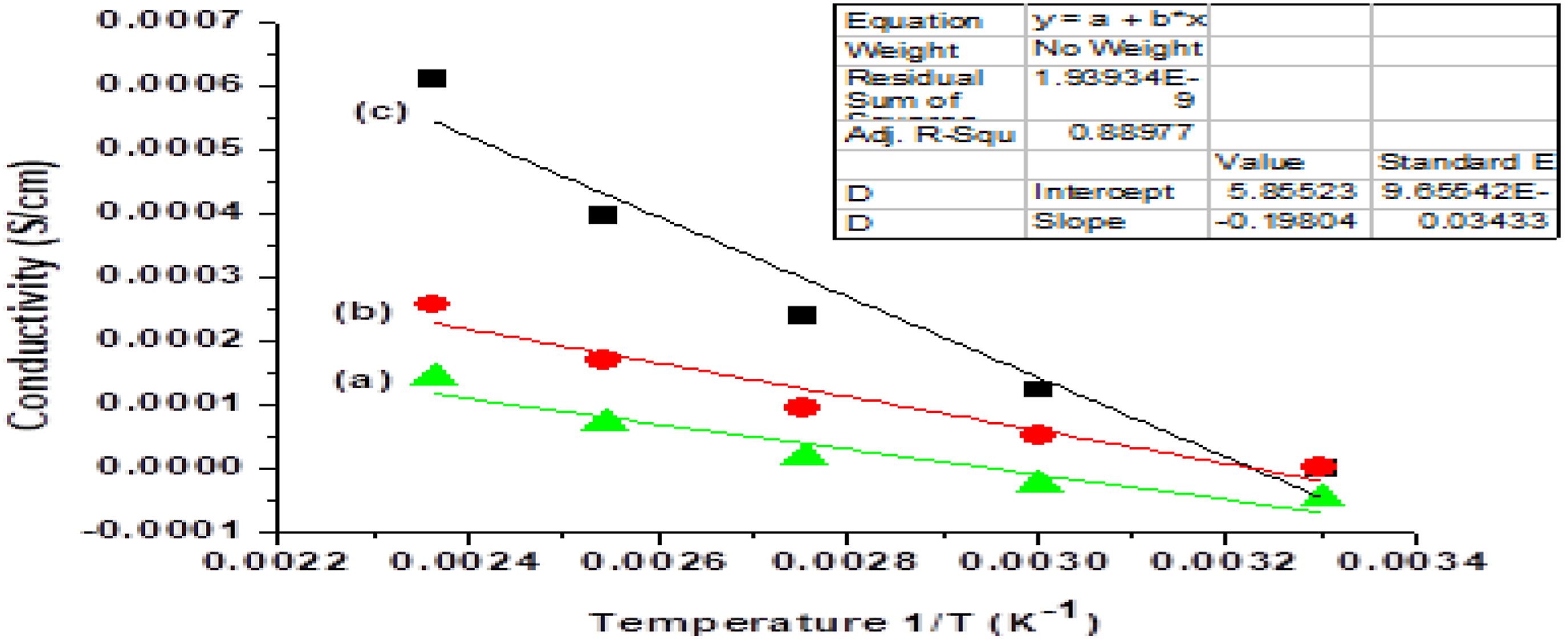
Activation energy

Activation energy of PANI, PANI-ZnO 20 wt.% &PANI-ZnO 25 wt.%.
taking log on both side above equation

Compare equations (1.4) and (1.5)
Slope is calculated by m
Activation energy is obtained by Ea = m
Activation energy versus content of ZnO (wt.%)
In Figure 6 shows that activation energy decreases rapidly by increases the wt.% of ZnO in Polyaniline chain. Activation energy is the minimum amount of energy is required to initiate the chemical reaction in a system. Plot of activation energy versus wt.% of ZnO in PANI.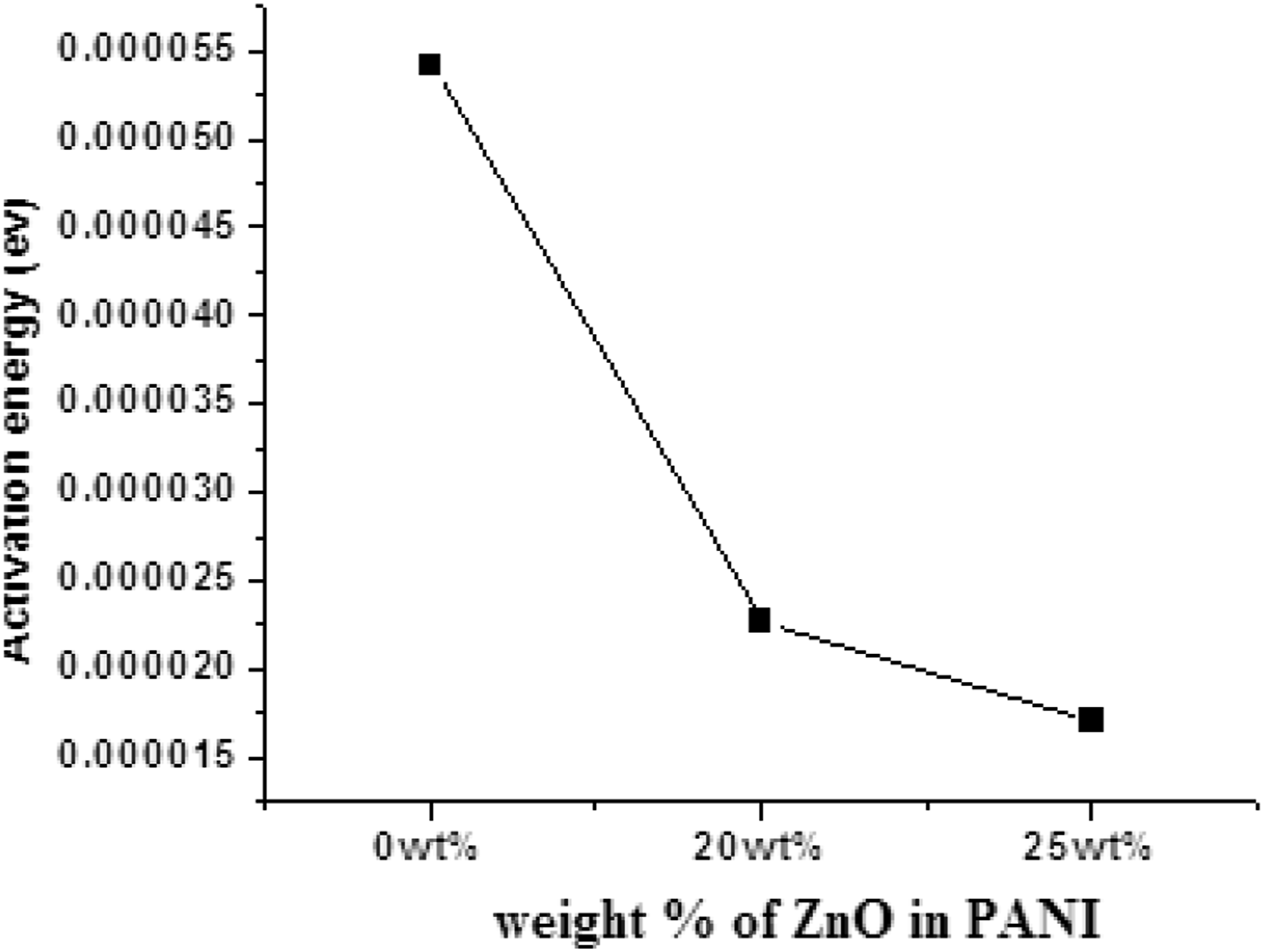
The activation energy is calculated from the above equation the slope is m
Device fabrication for seebeck measurement
Seebeck coefficient of PANI&PANI-ZnO.

Seebeck coefficient of polyaniline and PANI-ZnO composite
Figure 7 shows the Seebeck coefficient graph of Polyaniline and Polyaniline-composites. The graph shows that Seebeck coefficient (S) of PANI-composites rapidly increase than pure Polyaniline. By increasing the temperature and wt.% of ZnO Seebeck coefficient increased, the reason is that the efficiency of charge carriers was increased so, more Seebeck voltage is created so, Seebeck coefficient was increased. Seebeck coefficient range of Polyaniline is Seebeck coefficient of (a) PANI- 20 wt.% ZnO and (b) PANI- 25 wt.% ZnO.
Scanning electron microscopy (SEM)
The SEM image of pure PANI exhibits complete amorphous regions as shown in (Figure 8(a)). This image clearly reveals that surface of PANI is not smooth. Uneven lumps and holes visible in PANI are suitable for adsorption. It is found that the doping of ZnO has a strong effect on morphology of the resulting PANI-ZnO composites (Figure 8(b) and (c)). In case of PANI-ZnO composites, the SEM micrographs revealed a dual phase, the platelet as well as the flaky structure with an interlocking arrangement of granular particles. This suggests that most of ZnO nanoparticles coated with PANI and have formed a network during the polymerization process. Scanning electron micrographs (SEM) of (a) pure ZnO (b) pure PANI (c) PANI-ZnO.
Computational analysis

Conclusion
➢ Polyaniline and PANI-ZnO composites have been successfully synthesised by in-situ chemical polymerization methods and in-situ methods. ➢ The X-ray diffraction pattern confirmed that zinc-oxide nanoparticles are crystals, while pure polyaniline exhibits an amorphous nature. However, the PANI-ZnO composites pattern indicates a crystalline nature, suggesting a polyaniline interact with zinc oxide. The literature has reported on all the planes of zinc oxide. ➢ DC conductivity as a function of temperature ➢ Seebeck co-efficient measurement of PANI-ZnO composites also task this project which was not reported in literature. Nano-voltmeter ➢ The scanning electron micrographs (SEM) show dual phase, the platelet as well as the flaky structure, in PANI-ZnO.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
