Abstract
This study investigated the potential of bis(2-hydroxyethyl) terephthalate (BHET), derived from the glycolysis of polyethylene terephthalate (PET) waste, as an additive in the synthesis of flexible polyurethane foam (FPUF), addressing the escalating environmental concerns associated with PET waste accumulation and the demand for sustainable material solutions. The influence of varying BHET content on the synthesis process, cellular architecture, and the intricate interplay between compositional variations and the resultant physical, mechanical, and thermal properties of FPUFs has been investigated. Our examination approach included analyses of foam density and cell morphology, comprehensive Fourier transform infrared (FTIR) spectroscopy for chemical structure elucidation, thermogravimetric analysis (TGA) to assess thermal stability, and differential scanning calorimetry (DSC) alongside mechanical property evaluations to discern the impact of BHET on foam performance metrics. Results emphasized the pivotal role of BHET in refining foam characteristics, where its inclusion facilitated the formation of foams with improvement in cell uniformity, subsequently affecting the foam’s apparent density. FTIR spectra analysis provided insight into the hydrogen bonding dynamics within polyurethane segments, revealing how BHET integration influenced microphase separation and the structural coherence of the material. The thermal and mechanical property assessments through TGA and mechanical testing demonstrated that the addition of BHET substantially augmented the thermal stability and mechanical performance of FPUFs.
Keywords
Introduction
Polyethylene terephthalate (PET), a thermoplastic polymer, is extensively used across various industries, including the manufacture of a wide range of consumer goods, including packaging, textiles, and automotive components. Its prevalence in these applications is due to its exceptional mechanical properties, chemical resistance, and transparency.1,2 The global production of PET has been rapidly increasing over the past decades, reaching approximately 30 million tons per year. 3 This growth in PET production has led to a significant increase in PET waste generation, posing serious environmental challenges. Waste PET contributes to the accumulation of non-biodegradable waste in landfills and the pollution of ecosystems.2,4 In response, sustainable and eco-friendly PET recycling and valorization strategies have emerged, including mechanical and chemical recycling, as well as energy recovery.2,5 Chemical recycling, in particular, is gaining traction for its ability to decompose PET into monomers or oligomers, which are subsequently repurposed for new polymer synthesis. 6
The growing concern for environmental sustainability and the need to reduce dependence on fossil fuels have driven extensive research efforts in polymer science. In particular, the development of eco-friendly and renewable materials has become a crucial aspect of modern materials research. One area that has garnered significant interest is the incorporation of recycled monomers in the synthesis of polymeric materials, aiming to create sustainable and versatile products. One such monomer, bis(2-hydroxyethyl) terephthalate (BHET), derived from glycolysis of PET litter, has shown promising potential in the production of polyurethane (PU). Mendiburu-Valor et al. 7 showcase an innovative approach where sustainable thermoplastic polyurethanes (TPU) are synthesized using a recycled BHET monomer obtained from marine PET litter and a renewable castor oil-derived macrodiol. This eco-friendly method involves a closed reactor process at a relatively low temperature and short reaction time. The resulting TPUs with varying BHET contents exhibit favorable thermal, thermomechanical, and mechanical properties, making them attractive candidates for sustainable material applications. In addition, another study 8 focuses on thermoset pUs synthesized using a recycled BHET monomer derived from marine PET litter and a biobased polyol from castor oil. The study demonstrates that increasing the recycled BHET content in pUs enhances thermomechanical stability, resulting in materials with higher stiffness and improved mechanical properties. The pUs synthesized from recycled BHET exhibit comparable performance to those from fossil-derived commercial BHET, emphasizing the potential of recycled monomers in sustainable polyurethane synthesis. Li et al. 9 explore a different aspect of recycling PET waste, focusing on the glycolytic degradation of waste PET textiles to produce BHET for flame-retardant rigid polyurethane foams. The incorporation of BHET and dimethyl methylphosphonate as a flame-retardants yields foam with improved flame resistance, making them suitable for potential fire safety applications.
Flexible polyurethane foam (FPUF), which was first commercialized in the early 1950s, has emerged as a pivotal material in a multitude of industrial applications.10,11 It offers a unique combination of properties, such as low density, excellent cushioning, insulation, and sound absorption, which make it suitable for numerous applications.10,12,13 The synthesis of FPUF involves the chemical reaction between toluene diisocyanate (TDI) and a polyol, a process that yields a material with unique and adaptable characteristics. 14 However, the production of PU foam relies heavily on petrochemical feedstocks, raising concerns about resource depletion and environmental impact. 12 Incorporating waste PET-derived additives in PU foam can potentially alleviate these concerns while providing additional benefits, such as improved foam properties and reduced reliance on petroleum-based raw materials. 15 One promising approach is to use BHET as an additive in FPUF. BHET, a product obtained from the glycolysis of PET waste, has been reported to enhance the properties of various polymers, including polyurethanes, when added in small amounts. 7
Despite the significant progress in using recycled BHET for producing thermoplastic and thermoset polyurethanes,7,8,16 limited research has focused on the incorporation of BHET as an additive in FPUFs. This study investigates the potential of using BHET as an additive in FPUF to valorize waste PET and enhance foam properties. The research explores the effects of incorporating varying concentrations of BHET on the physical, mechanical, and thermal properties of FPUFs, providing a new approach to improving the sustainability and performance of flexible polyurethane foams.
Experimental
Materials and equipment
The chemicals used in this study were obtained as follows: Ethylene glycol (EG) and zinc acetate, which are essential for the synthesis of BHET, were purchased from Sigma-Aldrich. For the experimental production of FPUF, the study used VORANOL 3322, a polyol product from Dow Chemical. This polyol is characterized by a hydroxyl number between 45.5 and 49.5 mg KOH/g and has a molecular weight of approximately 3400 g/mol. The foam formulation also utilized 2.4-2,6-toluene diisocyanate (TDI 80:20), commonly referred to as 2.4-2.6 TDI. The process also incorporated distilled water as a chemical blowing agent, in addition to catalysts such as amine (DABCO 33-LV) and stannous octoate (KOSMOS 29), which were essential for the reaction kinetics. To improve the stability of the foam, Niax Silicone L-618, a silicone-based surfactant, was introduced. It is important to note that all chemical reagents used in this investigation were used in their original, unaltered form to ensure the integrity of the experimental results.
Glycolysis of polyethylene terephthalate
Obtaining bis(2-hydroxyethyl) terephthalate (BHET),17,18 illustrated in Figure 1, involved mixing flakes of PET bottles and ethylene glycol in a ratio of 40:60 w/w (PET to glycol), incorporating zinc acetate at a ratio of 0.5% w/w relative to PET. This mixture was transferred to a round-bottom flask, attached to a condenser, and positioned on a magnetic stirrer. The reaction was conducted at 190°C for 8 hours. Upon completion, the mixture was cooled to room temperature, at which point the introduction of water facilitated the precipitation process. The precipitate was collected and purified by recrystallization, resulting in a white crystalline product. PET depolymerization by glycolysis for BHET preparation.
The NMR spectra of the synthesized BHET were recorded using a 300 MHz Bruker NMR spectrometer with CDCl3 as the solvent. The NMR signals (δH and δC) observed in the spectra are described below: • δH (300 MHz, CDCl3): δ 8.15 (s, 4H, aromatic CH), 4.60-4.40 (m, 4H, CO(O)CH2), 3.10-3.90 (m, 4H, (OH)CH2), 2.20-1.90 (s, 2H, OH). • δC (300 MHz, CDCl3): δ 61.29 (CH2), 67.05 (CH2), 129.71 (aromatic CH), 133.87 (quaternary aromatic C), 166.05 (C(O)).
The NMR spectra in Figures 2 and 3 show the successful synthesis of BHET. 1H-NMR spectra of BHET in CDCl3. 13C-NMR spectra of BHET in CDCl3.

Synthesis of flexible polyurethane foam
Formulation of FPUFs with various quantities of BHET.

*pphp: parts per hundred polyol.
Characterization
Apparent density
The measurement of apparent density, in accordance with ISO 845:2006 standards, involved evaluating five foam cubes, each with dimensions of 50 mm × 50 mm × 50 mm. The samples were accurately weighed using an analytical balance, and their dimensions were ascertained with a digital vernier caliper. The apparent density figures were calculated based on the average mass-to-volume ratio.
Scanning electron microscopy
Foam samples were characterized using various techniques. The cellular structure was examined through SEM imaging (SEM IT500 HR, JEOL, Japan) at 5 kV with 50x magnification, and cell diameter was quantified using ImageJ software.
Fourier transform infrared spectroscopy
The structural properties of various FPUFs were characterized using the (Bruker Alfa II) equipped with Attenuated Total Reflectance (ATR) mode. The analysis was conducted over a spectral range from 400 to 4000 cm−1.
Differential scanning calorimetry
DSC analysis using a Q20 TA instrument was performed to determine the glass transition temperature of the foams. The procedure involved a temperature scan from −80 to 100°C using two distinct heating cycles. First, the sample was equilibrated at −80°C, followed by a heating ramp to 100°C at a rate of 20°C/min, and then cooled back to −80°C at 5°C/min. This step was essential to eliminate any prior thermal history. This was followed by a reheating phase at 20°C/min to 100°C, specifically to determine the glass transition temperature.
Thermogravimetric analysis-thermal desorption analysis
The thermogravimetric analysis of the foams was executed using a Perkin Elmer Diamond TGA/DTA over a temperature span of 30°C to 750°C. This procedure was carried out at a heating pace of 10°C per minute, aimed at identifying the rate of weight loss experienced by the foams as the temperature reached 750°C. The analysis was conducted in the presence of nitrogen (N₂).
Mechanical properties
Mechanical parameters of foam samples were measured using a universal testing machine from IDM Instruments, Australia. Compressive stress analysis at 65% strain was performed in accordance with ASTM D 3574 B using foam specimens measuring 200 mm × 200 mm x 50 mm compressed at a rate of 50 mm/min. Tensile properties were determined at a tensile rate of 300 mm/min in accordance with ASTM D 3574 E, and tear strength was determined at a rate of 250 mm/min in accordance with ASTM D 3574 F.
Results and discussions
Density and cell morphology
The density of polyurethane foams (PUFs) is a critical parameter that significantly impacts their functional characteristics.19–21 The desired density can be fine-tuned through various methods, including selecting blowing agents, optimizing processing conditions, and choosing specific types of polyols or isocyanates to meet precise property and application requirements.22,23 This study investigates the effect of BHET incorporation into FPUFs. As shown in Figure 4, an increase in BHET within the foam formulation initially results in a decrease in density, with the most significant drop observed as BHET content reaches two pphp. Beyond this concentration, the trend in density flattens out, suggesting that the impact of BHET on foam expansion and stabilization reaches an equilibrium. Further insight into this phenomenon is provided by the cell size distribution data in Figure 5, which is supported by the SEM micrographs in Figure 6. These results offer a detailed representation of the microscopic structural changes, deepening our understanding of the BHET effect. The findings show that BHET addition results in a more homogeneous cell size distribution. An increase in BHET content is correlated with a decrease in average cell size and an improvement in cell uniformity. In particular, the FPUF2 sample exhibits a smaller average cell diameter and more uniform cells compared to the FPUF0 sample. The homogenization of cell size with BHET addition may be attributed to a more controlled cell nucleation and growth process, wherein BHET moderates cell formation and stabilization, thus leading to a more even cell size distribution. Moreover, the initial viscosity of the reactive mixture, which could be affected by BHET, might also play a role in determining the morphology and size of the foam cells.
24
The introduction of BHET seems to adjust the foam formation process by influencing both the blowing and gel reactions. This effect is reflected in the viscosity changes of the reactive mixture, which in turn affects the chemical kinetics, promoting the development of a foam structure with uniform cell distribution. Impact of BHET content on the density of FPUFs. Distribution of cell diameter of FPUFs. Visualization of FPUFs with different content of BHET through SEM micrographs: (a) FPUF0, (b) FPUF1, (c) FPUF2, (d) FPUF3, (e) FPUF4, (f) FPUF5.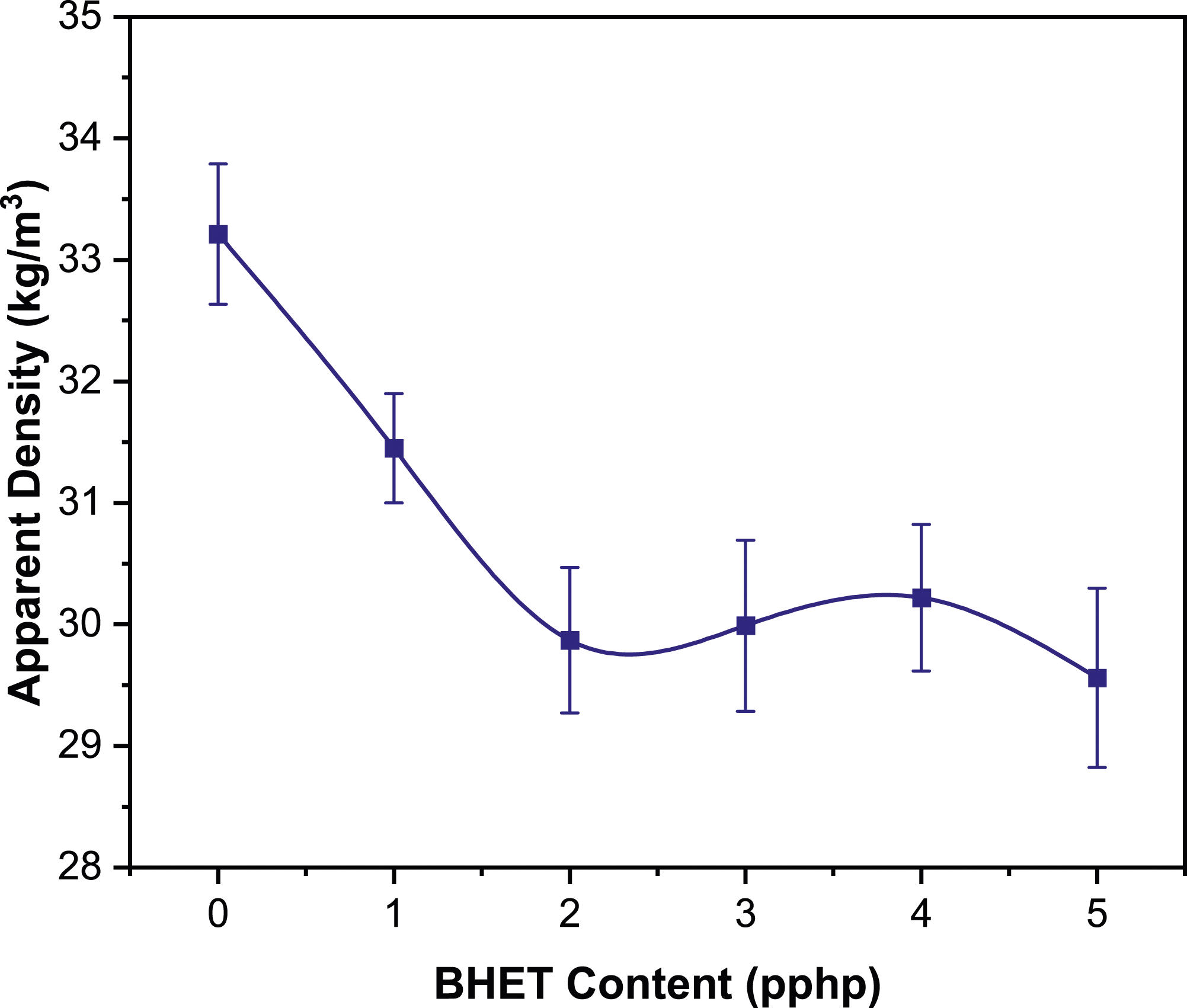


Fourier transform infrared analysis
FTIR spectroscopy is extremely useful in revealing the complex chemical composition of FPUFs. The carbonyl region of the FTIR spectrum is highly responsive to hydrogen bonds urea, which includes both monodentate and bidentate urea, as well as hydrogen bonds urethane. Urea groups, known for their stronger specific interactions and greater structural rigidity compared to urethane groups. Therefore, the common practice of using hydrogen-bonded urea, particularly in its bidentate form, indirectly indicates the degree of microphase separation.14,25–30
Figure 7 presents the FTIR spectra for a series of FPUFs (FPUF0 through FPUF5). The broad band at 3290 cm−1 is attributed to the stretching vibration of hydrogen-bonded N-H groups, suggesting the incorporation of urea and/or urethane in hydrogen bonding. The notable bands at 1532 cm−1, 1222 cm−1, and 1090 cm-1 correspond to the N-H bending, C-N stretching, and C-O stretching of urethane linkages, respectively. These bands indicate that chemical reactions are taking place between hydroxyl groups and isocyanate, a hallmark of polyurethane formation.
31
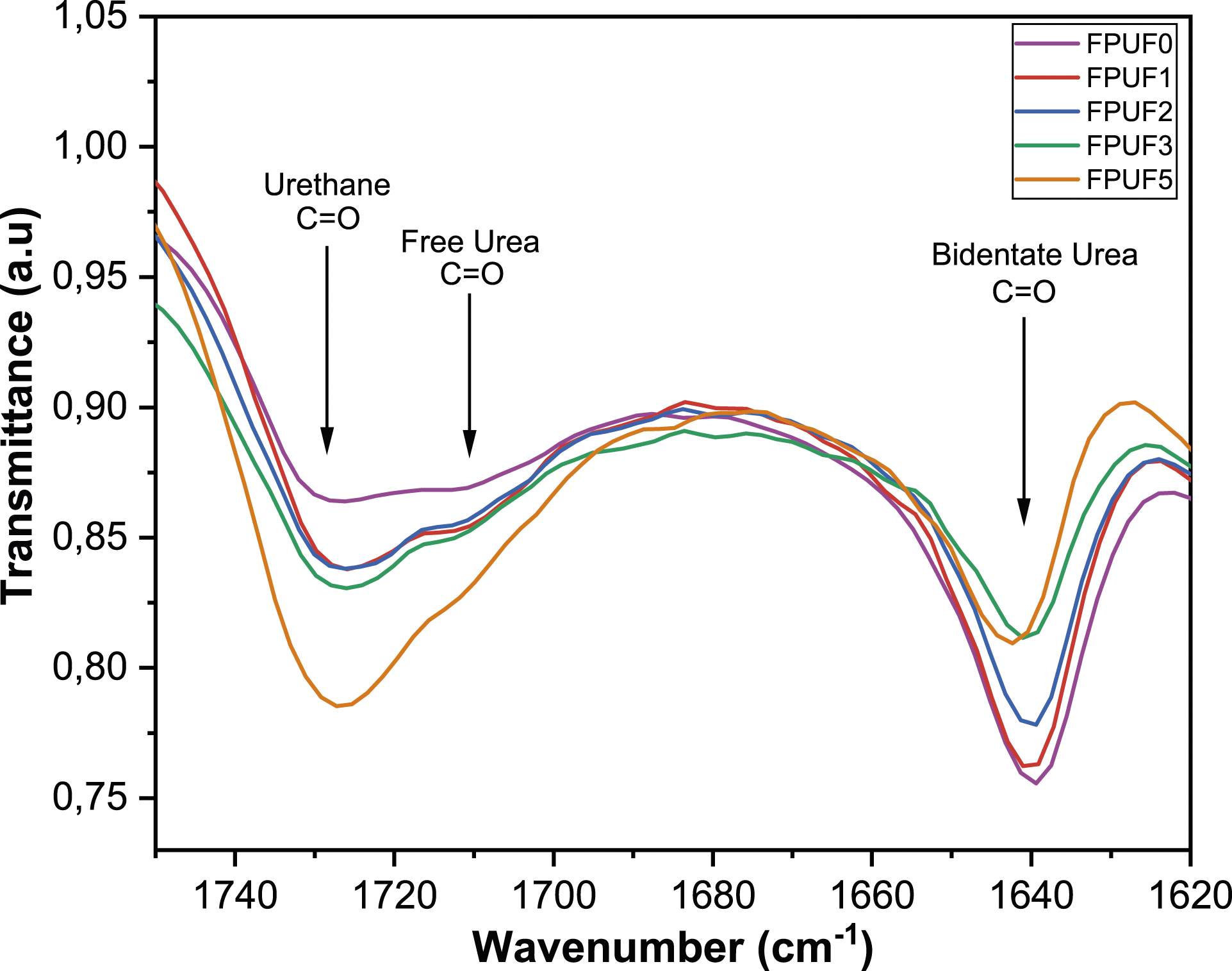
The carbonyl band ranging between 1800 cm−1 and 1600 cm−1 is particularly distinct, designed to investigate the hydrogen bonds formed between carbonyl groups and N-H groups, as shown in Figure 8. As BHET content varies, notable shifts are observed in the arrangement of polyurea segments, which play a crucial role in assessing the extent of microphase separation. A rise in BHET content correlates with a decrease in the intensity of bidentate urea groups, along with an increase in the intensity of free urea and urethane groups. These shifts imply a change in the molecular environment and intermolecular interactions within the material. The increase in the peak corresponding to free urea is particularly remarkable and could be the result of altered intermolecular interactions prompted by higher BHET content. The participation of BHET in reactions with isocyanate groups, leading to the formation of urethane groups, results in the release of additional free urea. This phenomenon, combined with the diminished intensity of bidentate urea groups, suggests a reduction in microphase separation. This reduction is posited to relate to the altered reaction kinetics, specifically the consumption of isocyanate groups by BHET, which favors the formation of urethane linkages. The complex interaction of these chemical processes highlights the sensitivity of the system to the BHET content, which affects the molecular structure and arrangement within the FPUFs. FTIR spectra for FPUF0, FPUF1, FPUF2, FPUF3, FPUF4 and FPUF5. The carbonyl region for FPUF0, FPUF1, FPUF2, FPUF3 and FPUF5.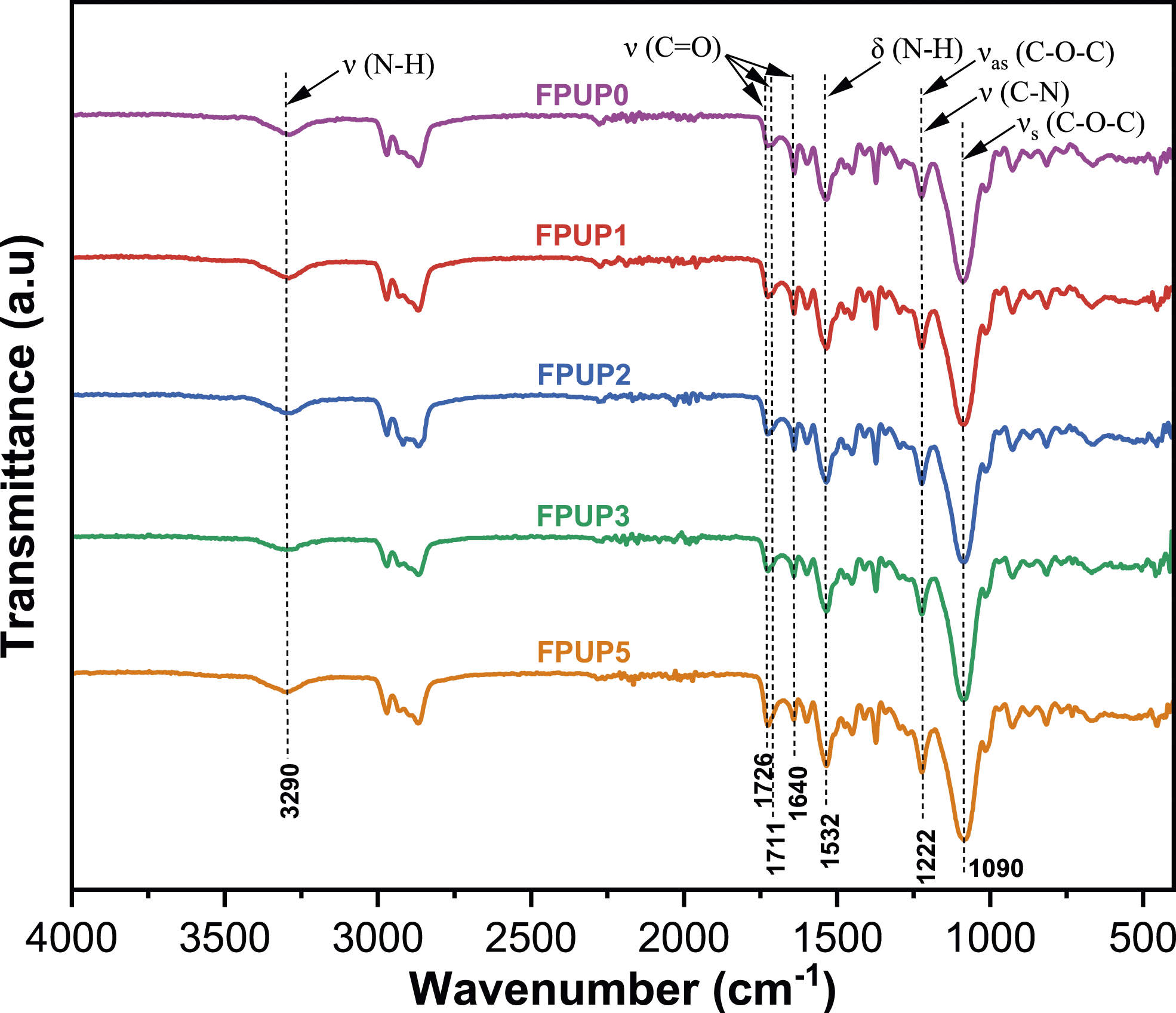
Thermal properties
Thermogravimetric analysis - thermal desorption analysis
The thermal properties of FPUFs containing varying contents of BHET have been systematically investigated through TGA-DTA analysis. Figure 9 illustrates the influence of BHET content on the thermal stability of the foams, revealing a distinct two-step degradation process corresponding to the degradation of the hard and soft segments. This observation aligns with previous studies,32,33 which confirm that the initial phase is mainly due to the degradation of the hard segments, while the subsequent phase is dominated by the degradation of the soft segments (SS). TGA-DTA thermograms for FPUF0, FPUF1, FPUF2, FPUF3 and FPUF5.
Further insight into the thermal decomposition process of FPUFs is provided by the DTA thermograms (Figure 9). These thermograms display two distinct weight loss regions: the first (200-300°C) is attributed to the decomposition of the hard segment (HS) with urethane bond cleavage. Critical observations indicate that an increased HS urethane content positively affects the thermal stability of the foams. The decomposition temperatures of the HS exhibit a significant increase, ranging from 274.35°C for FPUF0 to 288.03°C for FPUF5. In the second critical temperature range (300-400°C), the decomposition of the SS is observed. The introduction of BHET at different concentrations leads to a complex decomposition behavior that unfolds in several stages. These stages represent a cascade of degradation reactions, each with its own unique thermal signature, that progress incrementally across this temperature spectrum. As the BHET content increases, the complexity of thermal decomposition becomes more apparent. Not only is there a shift to higher temperatures, indicating greater thermal stability, but there is also a noticeable broadening and intensification of the DTA peaks. Particularly in the case of FPUF5, where the BHET content is highest, there is a specific change in the DTA peak characteristics. The peaks broaden, indicating that the degradation process covers a wider temperature range, possibly indicating simultaneous degradation of different components or a more gradual degradation process. At the same time, these peaks become more intense, indicating more exothermic degradation reactions. An intriguing third weight loss, observed above 500°C in FPUF3 and FPUF5 with higher BHET content, suggests secondary thermal decomposition events that reveal the presence of thermally stable components within the foam. These components, likely altered by the incorporation of BHET, decompose at higher temperatures, demonstrating the enhanced thermal stability of the material.
Differential scanning calorimetry analysis
DSC analysis was conducted to assess the impact of BHET on the soft segment’s structure, as shown in Figure 10. The analysis revealed that the DSC curves for all samples exhibited similar patterns, indicating that BHET’s presence did not significantly alter the fundamental thermal properties of the SS. In Table 2, the glass transition temperatures (Tg) of each FPUF are presented. While the Tg values showed only slight variations in the modified foams, this minimal change underscores a stability in the thermal response of the SS. This suggests that the structural integrity of the SS is maintained across all samples, irrespective of the differences in the structures of the urea and urethane phases. DSC thermograms for FPUF0, FPUF1, FPUF2, FPUF3 and FPUF5. Glass transition temperature of FPUFs with different BHET contents.

Mechanical property
The mechanical properties of FPUFs with varying BHET content is illustrated in Figures 11 and 12, demonstrating a synchronized trend in tensile strength, elongation at break, and tear strength. Each of these properties demonstrates an improvement with the incorporation of BHET up to a concentration of three pphp, beyond which there is a decline in performance. This initial increase suggests that small amounts of BHET enhance the polymer matrix, leading to a foam structure that can withstand greater tensile forces, elongate further before breaking, and resist tearing more effectively. These enhancements are likely due to improved phase adhesion or cross-linking density induced by the BHET. However, the subsequent decline in all three mechanical characteristics beyond the three pphp threshold indicates a threshold level for BHET integration. Beyond this point, the excess BHET probably reduces intermolecular forces, which compromises the foam’s ability to maintain structural integrity under stress. The similarity in the trend across these three measures underscores a common underlying mechanism influenced by BHET content that governs the foam’s mechanical behavior. This is further evidenced by the load at 65% strain data in Figure 12, where the material’s performance under significant deformation is assessed. The observed decrease in load capacity with higher BHET content aligns with the reduction in phase separation,
34
as seen in FTIR, reinforcing the concept that excess BHET induces softness into the foam. Impact of BHET content on the tensile strength and elongation at break of FPUFs. Impact of BHET content on the tear strength and compressive stress at 65 % strain of FPUFs.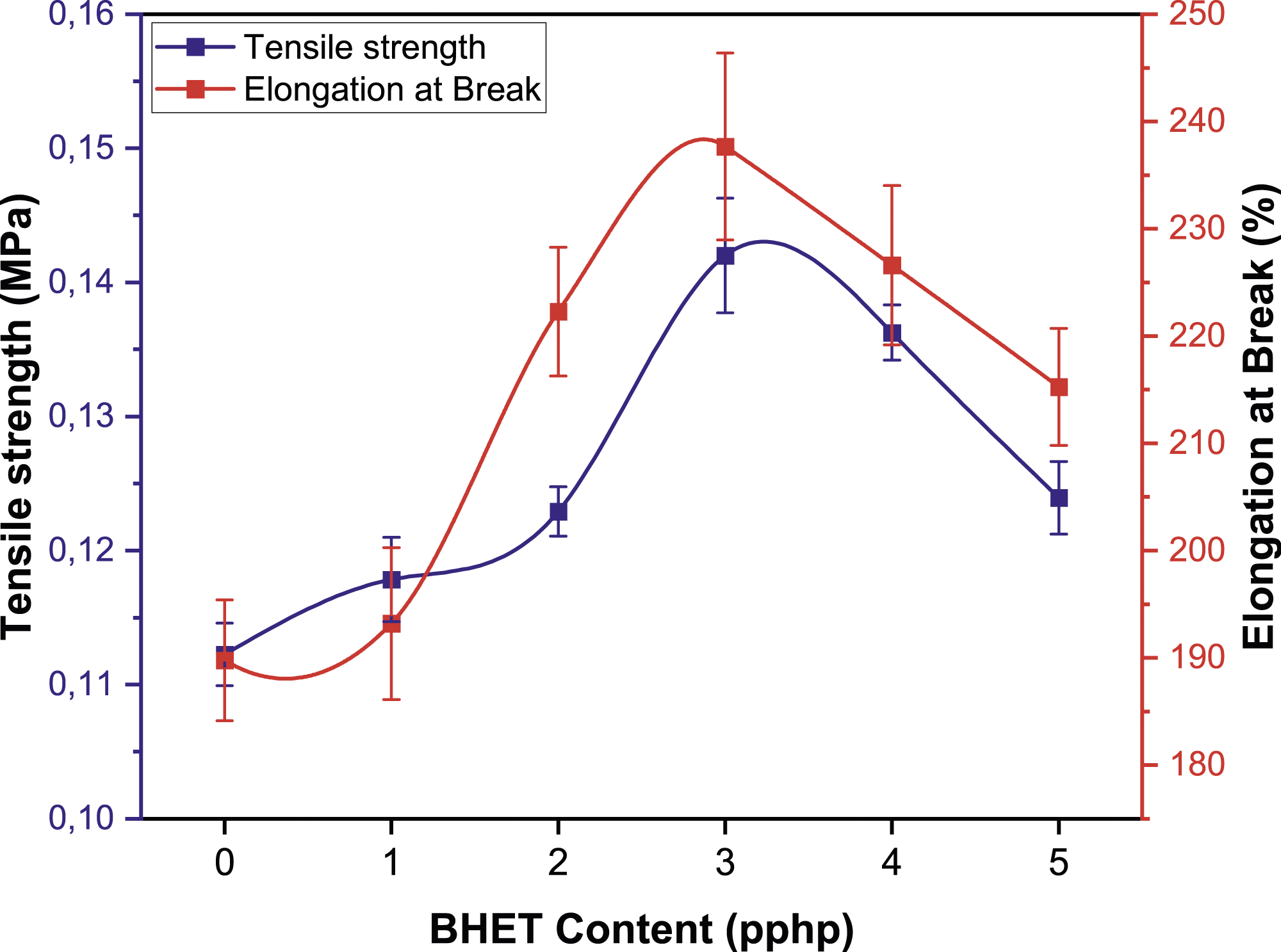

Conclusion
The incorporation of BHET in the synthesis of FPUF from PET waste revealed a clear relationship between BHET content and the foam’s morphological, mechanical, and thermal properties. Our results demonstrate that BHET plays a key role in modulating the foam formation process, affecting both the microstructure and performance characteristics of the resulting FPUFs. Specifically, the addition of BHET results in a noticeable improvement in foam cell uniformity and thermal stability. However, the improvement in mechanical properties is only observed up to an optimal BHET concentration, beyond which these mechanical benefits begin to diminish, highlighting the critical balance between BHET content and foam properties. This change in foam properties with varying BHET content is attributed to the adjustment of the dynamic balance between the chemical reactions involved in foam formation, specifically the gelation and blowing reactions. The incorporation of BHET significantly affects this equilibrium, allowing for a more refined cellular architecture within the FPUFs. In addition, the study highlights the role of BHET in influencing the microphase separation between HS and SS within the foam, a factor that critically affects the mechanical properties and thermal stability of the foam. Our research underscores that the improved properties of FPUFs with BHET are not simply a function of the concentration of the additive but are intricately linked to the degree of microphase separation and the dynamics of the chemical reactions during foam formation. The optimal mechanical strength observed at certain BHET concentrations is attributed to the optimal microphase separation achieved, which balances the structural integrity and flexibility of the foam. This balance is critical in developing FPUFs with desired properties for specific applications.
Footnotes
Acknowledgments
The authors extend their sincere appreciation to SALIDOR for their provision of the raw materials and the sample preparation equipment that were instrumental in the conduct of this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
