Abstract
In recent years, the production and consumption of plastics has increased. This growth in production has resulted in an increasing amount of plastic waste, which is a global problem. Among plastics, high density polyethylene (HDPE) is one of the most widely used polymers in the packaging industry, so its waste is used in the recycling process. One of the main disadvantages of accepting recycled HDPE in production processes is its odour, which makes it impossible to use in production lines. The aim of this work is to reduce and/or eliminate the odours present in the HDPE samples selected for recycling through the use of sepiolites. FTIR analysis of the HDPE samples sent for recycling showed the presence of small amounts of polypropylene (PP), (PET), flexible polyurethane, dirt and adhesives, which could be the cause of the bad odour. Washing the sample showed a decrease in odour, probably due to the removal of dirt and other products that can lead to the degradation of the plastics responsible for the odour. The characterisation of the mixture of sepiolites and HDPE aims to analyse the mechanical and thermal stability of the sample. Not only can pre-washing lead to odour reduction, but also extrusion and injection processes at elevated temperatures can lead to the removal of volatile or semi-volatile compounds. Sensory analysis showed a reduction in odour in HDPE samples with sepiolites, particularly with 15% Pensil sepiolite, to the detriment of mechanical stability. At lower concentrations of sepiolite, the results are good from a sensory, mechanical and thermal point of view, which could mean an industrial application in the manufacture of benches, flower pots, etc.
Introduction
According to Plastics Europe’s Plastics 2022 report, global plastics production reached 390.7 million tonnes (mt) in 2021, which is a 4.04% increase from 2020. Fossil-based plastics accounted for 90.2% (352.3 million mt) of this amount, while post-consumer recycled plastics accounted for 8.3% (32.5 million mt) and bioplastics accounted for 1.5% (5.9 million mt). In 2020, fossil-based plastics accounted for 87.1%, post-consumer recycled plastics accounted for 8.1%, and bioplastics accounted for 1.0%.1–3
In 2021, the largest percentage (19.3% of the total) of plastics produced was polypropylene (PP), followed by low-density polyethylene (14.4%), PVC (12.9%), and high and medium density polyethylene (12.5% of the total). Recycled plastics accounted for 8.3% of the production. In 2021, fossil-based thermosets excluding polyurethane and other fossil-based thermoplastics accounted for 7.1% of global production. PET accounted for 6.2%, polyurethane for 5.5%, polystyrene and EPS for 5.3%, and bioplastics for 1.5%. 1
The production of plastics has led to a rise in plastic waste, which is a global issue.3–7 Possible solutions for the plastic waste problem include treating and reusing the materials, which requires a shift from a linear to a circular economy.
The four treatment routes for plastic solid waste comprise primary (re-extrusion), secondary (mechanical), tertiary (chemical) and quaternary (energy recovery) technologies. Primary recycling involves the reintroduction of clean single-polymer waste into the extrusion cycle to produce similar products. 8
Mechanical recycling methods include sorting technologies, thermomechanical degradation and immiscibility of polymer blends, while chemical recycling techniques include chemolysis, pyrolysis, fluid catalytic cracking, hydrogen techniques and gasification. 9
Energy recovery is an achievable solution for plastic solid waste in general and municipal solid waste (MSW) in particular. The amount of energy produced in this way is sufficiently researched and is being integrated, little by little, in energy producing plants. 8
Recycled plastics can have an unpleasant odor, which makes them unsuitable for use in production lines.10–14 Research indicates that the unpleasant odor of recycled plastic materials may be due to different factors, such as: thermal and environmental degradation (humidity and temperature) of the polymer, additives, contact with other substances, improper storage of the container etc.15,16 Odor can be a limiting factor, which means that in some cases its most suitable use is for outdoor applications such as: benches, planters and litter garbage cans etc.
Various proposals can be found in the literature to reduce or eliminate odours, including chemical treatments and washing with water and detergent. However, treating all polymers with the same treatment is difficult due to the complexity of the recycled material samples. The washing and/or treatment system used depends on the type of plastic.17–19
HDPE, which is one of the most commonly used polymers in the packaging industry,20,21 presents a particular challenge. The fragment ends here.
Materials and methods
Starting material
The study utilised high-density polyethylene (HDPE) waste from Ecoembes (Figure 1) and sepiolites from Tolsa. HDPE sample supplied by ecoembes.
HDPE is a thermoplastic polymer that is easily processed through injection or extrusion and can be recovered through mechanical and thermal recycling. HDPE is identified with Plastic Identification Code 2.
High density polyethylene is a polymer with a linear structure and very few branches. It is obtained by polymerization of ethylene at relatively low pressures using catalysts. It is a high density polymer, colorless, odorless, non-toxic and resistant to both stress and chemical agents.
Sepiolite is a hydrated magnesium phyllosilicate with the molecular formula Mg4Si6O15(OH)2-6(H2O) and is chemically inert. Its crystalline structure creates zeolitic channels, which significantly increase its specific surface area and adsorptive capacity.22,23
Tolsa, the company, has provided adsorbents with a sepiolitic structure (Pansil and Pangel B20). Pansil is a hydrated magnesium silicate with a sepiolite purity of 85%. Pangel B20 is an organically modified magnesium silicate hydrate containing 15% of a quaternary ammonium salt.
Preparation of the starting material
12 plastic fragments of different shapes and colours were randomly selected from the HDPE starting material to obtain heterogeneous samples. The coloured and shaped fragments underwent a cleaning process involving chloroform and magnetic stirring for 5 min, followed by a solution of soap and water with magnetic stirring for a further 5 min, and finally rinsed with distilled water.
The HDPEw sample was then dried in an oven at 40°C for 10 h. This temperature was chosen to prevent oxidation of the HDPE, which can occur at 50°C or even at ordinary temperatures in the presence of light. For this reason, antioxidants are typically added.
The HDPEw samples were manually mixed with PANSIL and PANGEL B20 sepiolite at 5%, 10%, and 15% concentrations.
Processing of starting material
The HDPEw samples, both alone and mixed with sepiolites, were introduced into a twin-screw extruder L/D 24 of Dupra S.L. (Alicante, Spain) with a temperature profile of 185–190–195°C. The resulting mixture was then injected as standardised test specimens into a Sprinter 11t of Erinca S.L. (Barcelona, Spain), using a temperature profile of 185–190–195°C.
Nomenclature of samples
The samples prepared correspond to the following nomenclature:
Characterisation of samples
HDPEw chemical characterisation
The starting HDPEw sample was analysed using Fourier transform infrared spectroscopy with a Perkin Elmer Spectrum BX spectrometer (Perkin-Elmer España S.L., Madrid, Spain).
Infrared spectroscopy tests were conducted with 20 scans between 600 and 4000 cm−1 and a resolution of 16 cm−1, using an attenuated total reflectance (ATR) accessory, which is recommended for samples with low transparency.
Textural characterisation of pansil and pangel B20 sepiolites
To characterise the sepiolites, we determined their specific surface area (SBET) using a Quantachrome Autosorb 1 series equipment. This parameter is crucial for adsorbents because the greater the surface area, the higher the adsorption capacity due to increased interaction between the adsorbent and the adsorbate. The BET-Brunauer-Emmet-Teller isotherm was used to determine the specific surface areas of the adsorbents. This isotherm is based on the physical adsorption of a monolayer of inert gas (nitrogen) on the surface of the material being characterised. 24
Thermal characterisation of the samples
The HDPEw sample and those mixed with sepiolite were analysed using differential scanning calorimetry (DSC) with a Mettler Toledo 821 (Mettler Toledo, Schwerzenbach, Switzerland) and thermogravimetric analysis with a Linseis TGA PT1000 (Selb, Germany).
DSC tests were carried out using samples of 5–10 mg. The first test was conducted in a nitrogen atmosphere, with a heating and cooling rate of 10°C/min. The DSC test involved heating the sample from 30 to 170°C, followed by cooling to 80°C and then heating up to 270°C. This allowed for the removal of the thermal history of the sample and provided information about the material’s melting. The second test was conducted in an air atmosphere with a heating rate of 10°C/min from 30 to 190°C, followed by an isotherm at 190°C for 30 min. This test determined the onset of degradation by measuring the oxidation induction time (OIT).
Thermogravimetric analysis (TGA) is a thermal analysis method used to measure the mass of a sample over time as the temperature changes or at a constant temperature for a given time. It is employed to determine a material’s thermal stability, composition, purity, decomposition reactions, decomposition temperature, and absorbed moisture content of products.
The samples’ thermal stability was evaluated using TGA. The experiments were conducted using a sample weight of 15–20 mg in 70 ηl alumina crucibles. The samples were heated from 35°C to 700°C at a rate of 10°C/min in a nitrogen atmosphere (30 mL/min). The temperature at which degradation begins was measured at 5% degradation (T5%), and the maximum rate of decomposition (TMAX) was determined at the minimum peak of the first derivative of the TGA curves (DTG).
Mechanical characterisation of the samples
The mechanical characterization of the HDPEw sample and HDPEw samples with sepiolite was carried out using the Charpy impact testing machine. For the tensile tests, an Ibertest universal testing machine model ELIB 30 (Madrid, Spain) with a 5 kN load cell at a speed of 10 mm/min was used. Five specimens of each sample were tested, as indicated in the UNE-EN ISO 527 standard.
The Charpy impact test was conducted using a 1J pendulum and specimens notched with type A geometry according to UNE-EN ISO 179-1:2011, using a Charpy Pendulum from Metrotec S.A. (San Sebastian, Spain).
Morphological characterisation of the specimens
The specimens were morphologically characterised after the Charpy impact test using a Zeiss Ultra 55 model field emission scanning electron microscope (FESEM) (Oxford Instruments, Pleasanton, USA) operating at an accelerating voltage of 1 kV. To improve surface conductivity, the samples were coated with a gold-palladium layer using an Emitech SC7620 Sputter Mod Coater, Quorum Technologies (East Sussex, UK).
Odour test
For odour analysis, the samples were analysed according to the VDA 270 standard used for odour assessment in the automotive industry. For this purpose, 10 g of each sample was placed in a 1 L glass container and kept closed at 80°C for 2 h. After cooling to 60°C, ten volunteers (three men and seven women) aged between 21 and 56 years were asked to rate the overall odour of the samples on a scale of 1 - not perceptible, 2 - perceptible, not annoying, 3 - clearly perceptible, not annoying, 4 - annoying, 5 - very annoying and 6 - unbearable. The mean of these ratings was calculated and the degree was rounded down. All subjects had no known medical conditions at the time of the sensory evaluation. 25
Results and discussion
Sample preparation
From the HDPE sample supplied by Ecoembes, fragments of different shapes and colours were selected and washed to remove impurities and dirt from the HDPE sample, resulting in the HDPEw sample.
The HDPEw sample was mixed with sepiolite and introduced into an extruder and subsequently into an injector resulting in the HDPEw samples with sepiolites.
Nomenclature of the samples.

Characterisation of samples
The samples were characterised according to the scheme shown in Figure 2. Overall scheme of the process.
Characterisation of the raw material HDPEw, pansil and pangel B20
Chemical characterisation of the HDPEw sample
The chemical characterisation was carried out using FTIR spectra. These spectra allow us to determine the functional groups of the polymers, the degradation processes they have undergone or the presence of other materials. The following figures show the compounds present in the selected sample and the FT-IR spectra of HDPEw together with the compounds detected in the sample.
The spectra obtained show the different chemical compositions of the polymers present and show that some of the polymers studied are structurally composed of the same subunits and have peaks in common regions of the spectrum: C-C peaks around 1400–1600 cm−1, C-H peaks around 2800–3200 cm−1, aliphatic or aromatic -CH2 and -CH3 peaks between 500 and 1300 cm−1 (in polymers such as HDPE or PP, among others),
In the HDPE spectrum (Figure 3), three groups of bands are clearly observed corresponding to C-H bond stretching at 2919 cm−1, C-C stretching at 1462 cm−1 and -CH2 bending at 716 cm−1.
26
FT-IR spectrum: (A) Dark green fragment, high density polyethylene and (B) grey fragment, polyethylene terephthalate.
In the PET spectrum (Figure 3), an intense band corresponding to the C=O bond stress at 1700 and stress shifts between 1000 and 1100 cm−1 due to aromatic ring bond stresses are observed. The C-H bond stress bands at 2800 2900 cm−1 are weak, although still discernible. 27
In the PP spectrum (Figure 4), three groups of bands corresponding to C-H bond stretching at 2900 cm−1 and C-C stretching at 1450 1350 cm−1 and -CH3 bending between 1200 and 1000 cm−1 are clearly observed.
26
FT-IR spectrum: (A) Elongated white fragment, polypropylene and (B) brown fragment, flexible polyurethane foam.
The FTIR spectrum obtained for flexible polyurethane (PU) foam (Figure 4) shows the different PU bands, which can be easily identified using literature data 21 : the corresponding υ(N-H) band is observed between 3500 and 3300 cm−1, while the bands at 2919 cm−1 correspond to ν(-CH2) groups and the band at 1375 cm−1 to δ(NH) and the band at 1074 cm−1 to asymmetric ν(-CH2).
Material identification by FTIR of selected fragments (HDPEw sample).

Textural characterisation of the samples pansil and pangel B20
Characterisation of pansil and pangel B20 sepiolites.

However, it can be observed that the specific surface areas of Pansil and Pangel B20 are very different. Pansil is the solid with the highest value of this parameter (294 m2-g-1), while Pangel B20 is the adsorbent with the lowest surface area (95 m2-g-1). This difference does not allow us to analyse the influence of the specific surface area on the ability of these materials to retain volatile and semi-volatile compounds.
Thermal characterisation of HDPEw and HDPEw samples with sepiolites
Differential scanning calorimetry of samples
Differential Scanning Calorimetry (DSC) allows the characterisation of semi-crystalline polymers such as polyethylene (PE), polypropylene (PP), polyethylene terephthalate (PET), etc., which are systems containing a crystalline phase and an amorphous phase.28,29
Looking at Figure 5, the HDPEw sample shows a wide temperature gradient, the maximum temperature gives the actual melting temperature 137°C,
30
typical melting temperature of a high-density polyethylene, furthermore no other melting peaks are observed which represent other semi-crystalline plastic materials in significant proportions. Dynamic and N2 atmosphere calorimetric graph of the HDPEw material.
Another important parameter is the melting enthalpy, ΔHf. If the melting enthalpy of the fully crystalline polymer (ΔHf100 for HDPE 293 J/g) is known, the degree of crystallinity of an HDPEw polymer sample can be determined as follows (equation (1)):
Considering the calorimetric curves (Figure 5), we obtain a melting enthalpy of 96.70 J/g and therefore a crystallinity of 33.3%, calculated with equation (1).
Crystallinity is related to the mechanical strength of the thermoplastic. Semi-crystalline thermoplastics have a high mechanical strength, are more rigid and withstand creep and heat better than amorphous thermoplastics. On the other hand, they are less impact resistant and less flexible. Their macromolecules align in some areas and create small crystals within the amorphous matrix.
From the air DSC test, Figure 6, the oxidative induction time (OIT) of the HDPEw material is determined. The oxidative induction time (isothermal OIT) is a relative measure of the resistance of a (stabilised) material to oxidation. The sample is heated in a dynamic air atmosphere at a constant rate until the oxidation temperature is revealed by an exothermic deviation in the DSC heat flow curve. Dynamic-isothermal and air-atmosphere calorimetric graph of HDPEw material.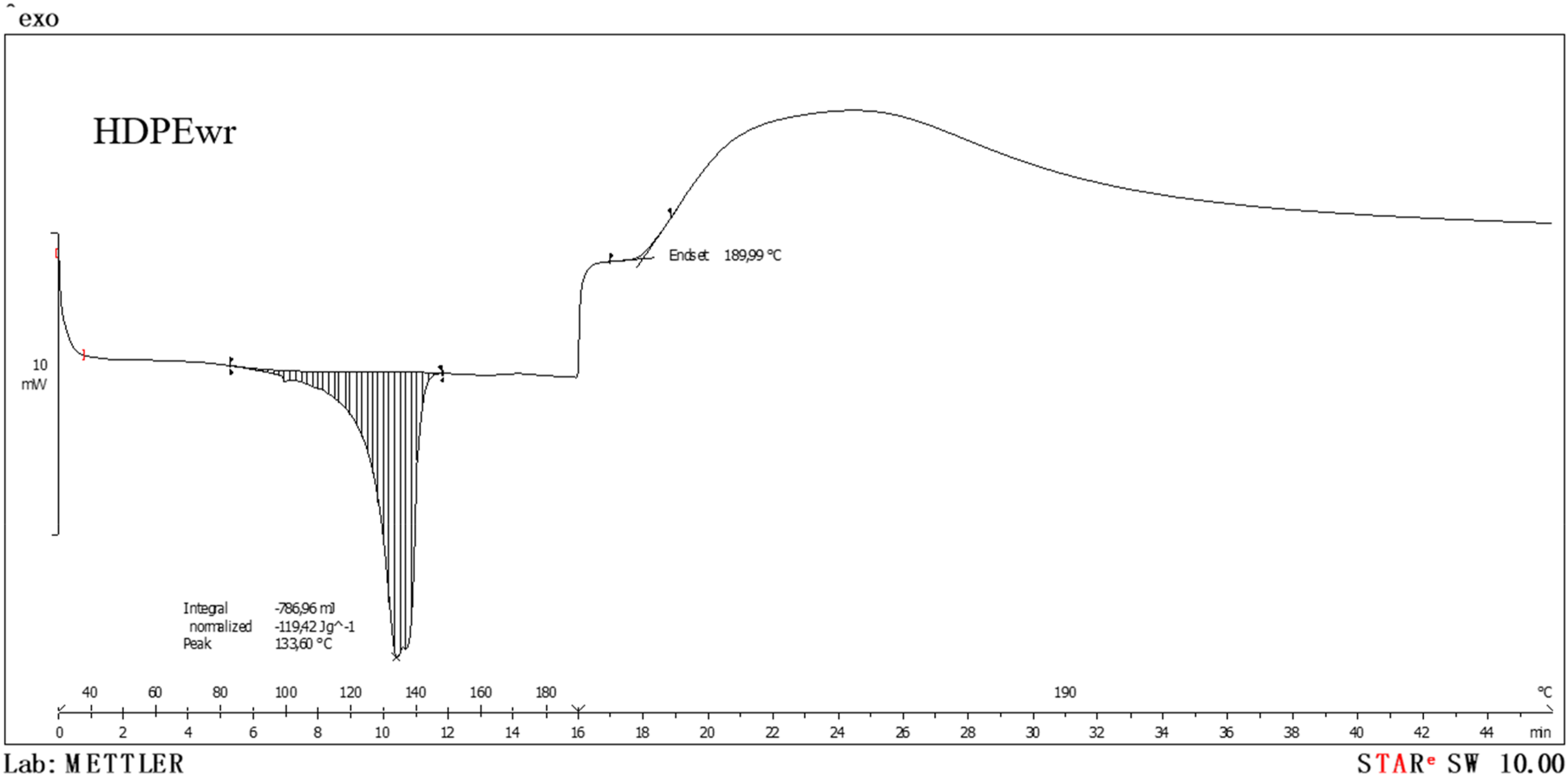
From Figure 6, which corresponds to the DSC in air atmosphere of the HDPEw sample, it can be seen that the OIT occurs at 2 min and 10 s at 190°C.
The degradation temperature of HDPEw, 190°C, is the maximum dwell temperature at which HDPEw can be processed without losing its physical and chemical properties. Degradation reduces the molecular weight of the polymer by breaking the covalent bonds of the chains formed during polymerisation.
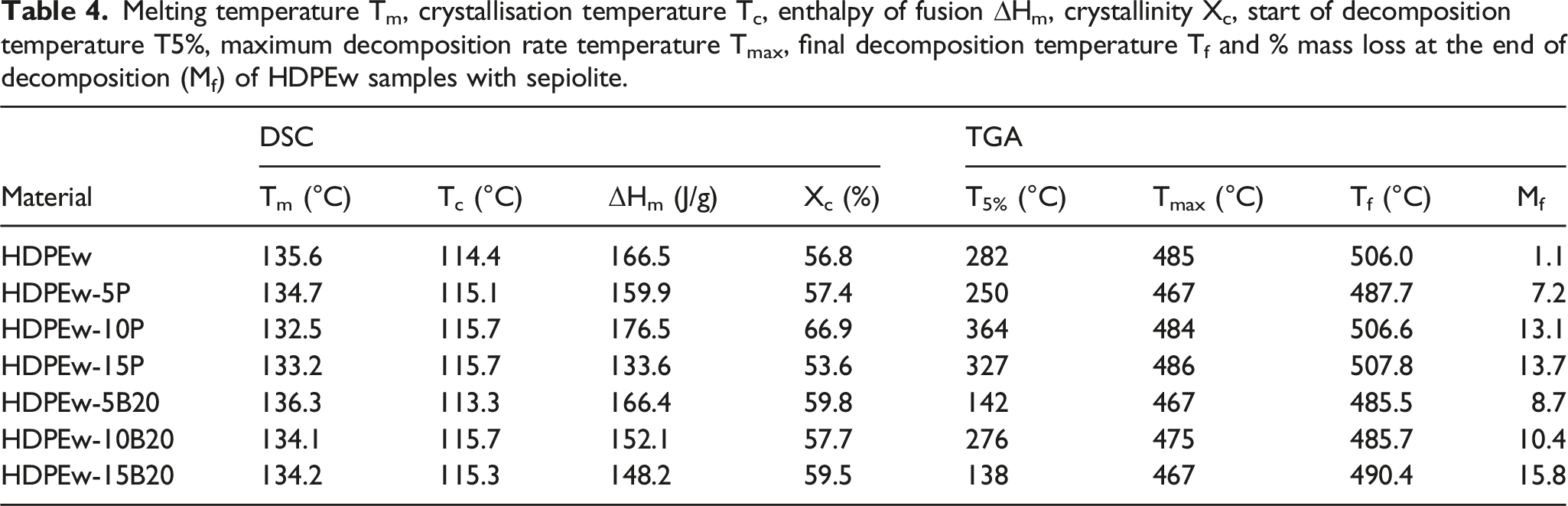
On the other hand, the thermal characterisation of the HDPEw samples with sepiolite by DSC can be seen in Figure 7 and the thermal parameters are presented in Table 4. It can be seen that the melting temperature (Tm) is around 135.6°C. The addition of PANSIL does not cause a significant change for contents of 5 and 10 wt.%, but an increase in Tm of 2°C is observed when the PANSIL content is increased up to 15 wt.%. On the contrary, the addition of B20 sepiolite does not produce a significant effect on the Tm of the HDPEw material, similar results were obtained by Singh et al. (2017)
31
using nanosepiolite in a polyethylene matrix.
17
Furthermore, it can also be observed that the degree of crystallinity increases with the incorporation of B20 sepiolite for all the contents analysed, suggesting that B20 sepiolite acts as a nucleating agent in HDPEw. However, PANSIL sepiolite causes no change in crystallinity at 5 wt.%, increases crystallinity at 10 wt.% and decreases the crystallinity value again when the content is increased to 15 wt.%. This behaviour suggests that concentrations above 10 wt.% of PANSIL result in poor dispersion of this sepiolite in the HDPEw matrix, which inhibits crystallisation. Calorimetric plots of HDPEw samples with sepiolite. (a) PANSIL at 5, 10 and 15 wt.% and (b) B20 at 5, 10 and 15 wt.%. Melting temperature Tm, crystallisation temperature Tc, enthalpy of fusion ∆Hm, crystallinity Xc, start of decomposition temperature T5%, maximum decomposition rate temperature Tmax, final decomposition temperature Tf and % mass loss at the end of decomposition (Mf) of HDPEw samples with sepiolite.
Thermogravimetric analysis HDPEw
Thermogravimetric analysis (TGA) is a thermal analysis method used to determine the thermal stability of a material, decomposition temperature and mass loss, among other parameters.
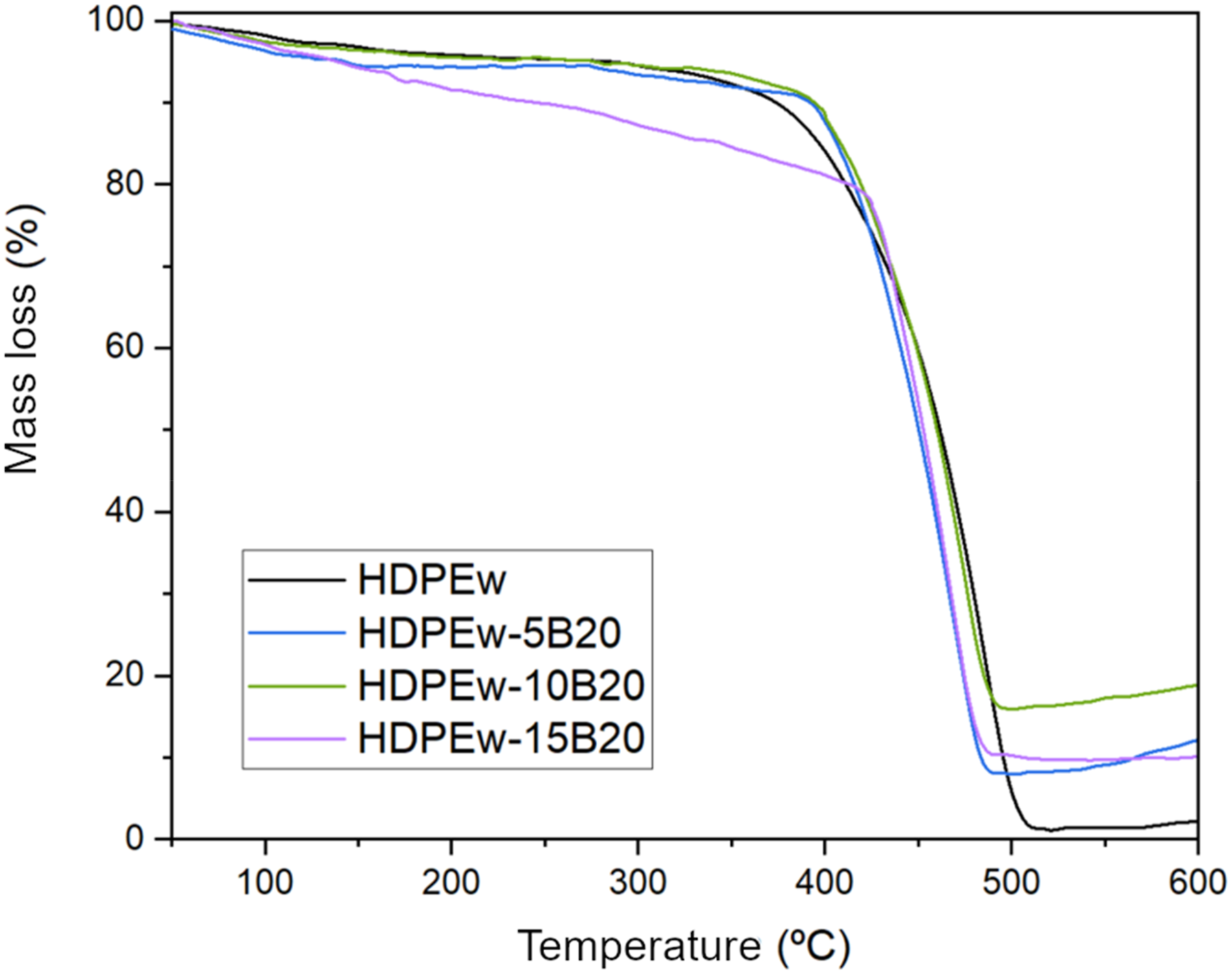
Figures 8 and 9 show the degradation profile of HDPEw. The final degradation of HDPEw is close to 100%, almost all of which is converted into volatile products. In the figure it can be seen how the thermogram shows a degradation from the beginning of the thermal treatment and ends around 506°C, as can be seen from the weight losses. TGA curves of HDPEw samples with PANSIL sepiolite at 5, 10 and 15 wt.%. TGA curves of HDPEw samples with sepiolite B20 at 5, 10 and 15 wt.%.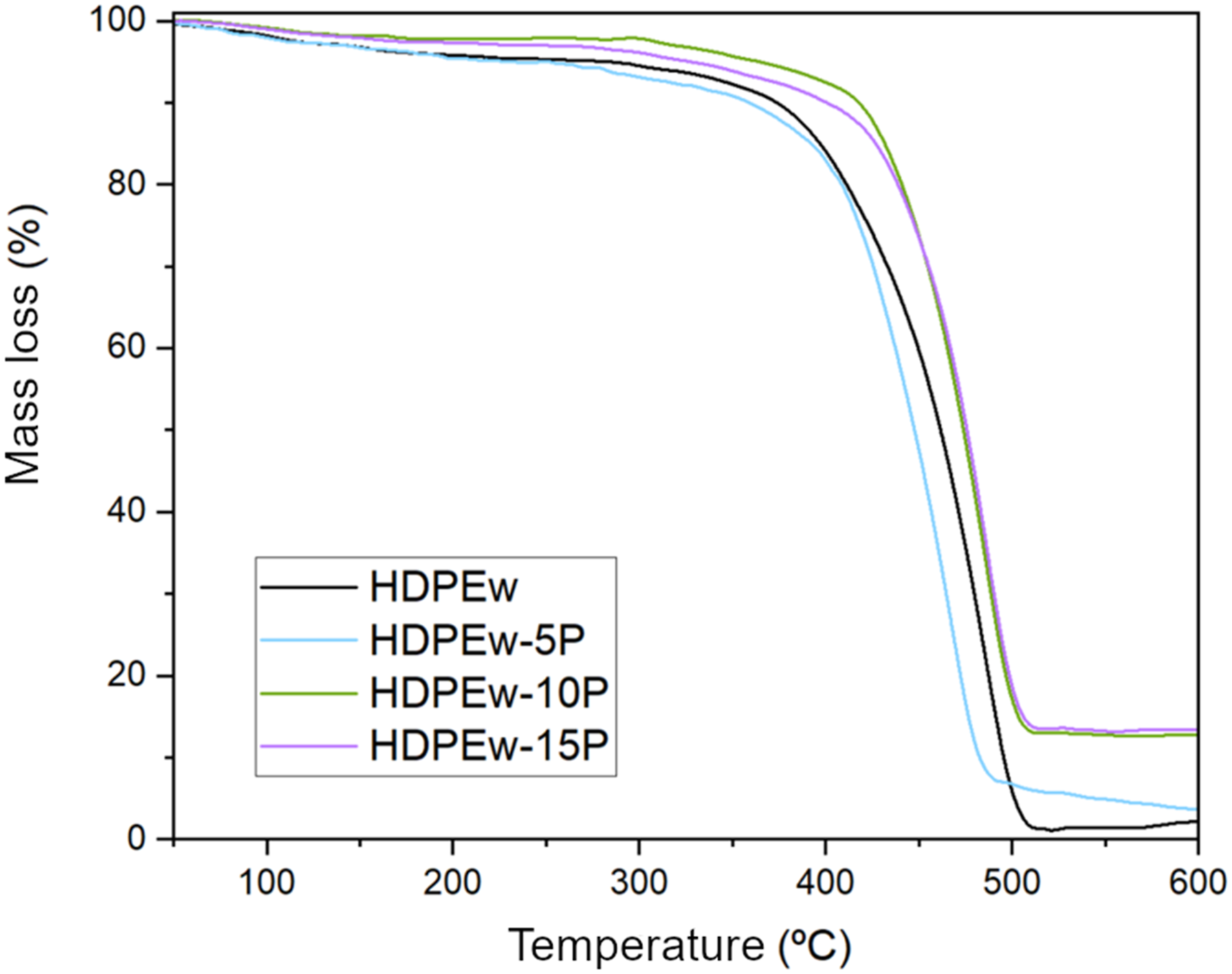
On the basis of the thermogram, we have determined the temperature at which 5% of the total T5% has been lost, which is 282°C, Tf, which indicates the end of the process and the temperature at which the maximum weight loss occurs, which is 506°C, and Tmax, the temperature at which the maximum rate of decomposition occurs, which is 485°C.
On the other hand, the thermogravimetric analyses carried out on the HDPEw samples with sepiolite can be seen in Figure 8 for the samples made with PANSIL sepiolite and in Figure 9 for the samples made with PANGEL B20 sepiolite. In addition, the main data obtained, such as the start and end temperature of degradation, the temperature of the maximum rate of degradation and the final mass of the different samples, can be found in Table 4. It can be observed that the incorporation of PANSIL in high percentages, 10 and 15 wt%, increases the thermal stability of HDPEw by 29 and 16%, respectively. On the contrary, Sepiolite B20 reduces the onset temperature of degradation of HDPEw. The temperature of the maximum degradation rate is reduced by 18°C when 5% by weight of PANSIL is added and it is also strongly reduced when B20 sepiolite is added, while it remains constant when 10 and 15% by weight of PANSIL sepiolite are added. As for the final degradation temperature, it can be seen that the incorporation of sepiolite B20 reduces it by more than 15°C and PANSIL sepiolite keeps it constant for values higher than 5% by weight. Furthermore, when analysing the residue resulting from the degradation, it is observed that it increases as the sepiolite content of the samples increases, due to the fact that it is an inorganic material.
Mechanical characterisation of samples
Mechanical performance values of HDPEw materials with sepiolite.

The tensile test shows that the HDPEw sample has an average strength of 21.2 MPa and an elongation of 125. These values are similar to other recycled HDPE. 32 On the other hand, the Charpy test allows us to determine the energy absorbed by the specimen during the Charpy impact, in our case the average impact energy absorbed by the HDPEw specimen is 33.7 kJ/m2. 33
On the other hand, the values of maximum tensile strength, elongation at break and energy absorbed by Charpy impact of HDPEw specimens with sepiolite, PANSIL and B20 can be found in Table 5. These results are also shown in Figure 10 for HDPEw specimens containing PANSIL sepiolite and in Figure 11 for HDPEw specimens containing PANGEL B20 sepiolite. Mechanical characterization, maximum resistance, elongation and Charpy impact energy absorbed of HDPEw samples with PANSIL sepiolite. Mechanical characterization, maximum resistance, elongation and Charpy impact energy absorbed of HDPEw samples with PANGEL B20 sepiolite.
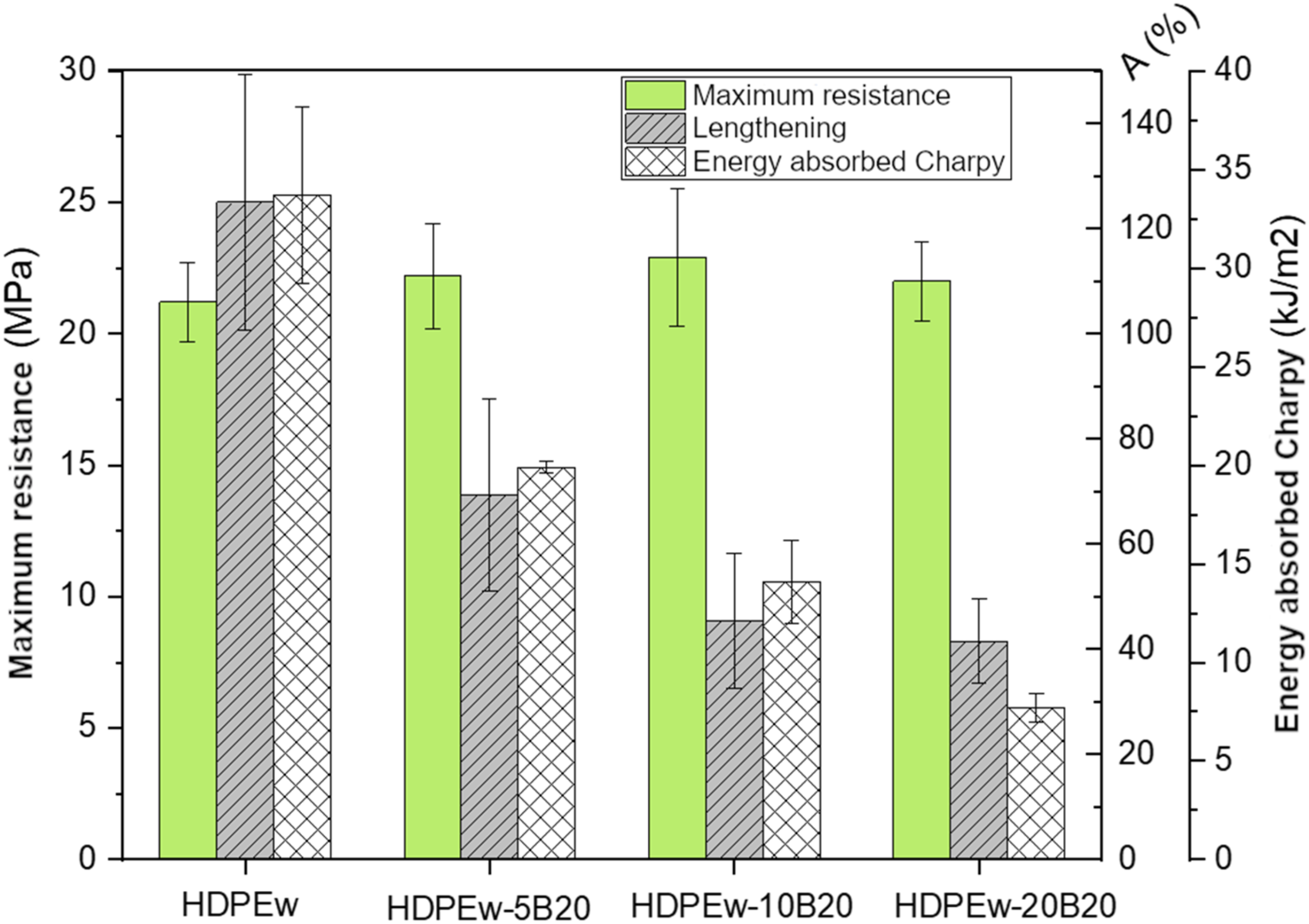
With regard to the characterisation carried out by tensile test, it can be seen that the maximum tensile strength remains practically constant with the incorporation of the different sepiolites, PANSIL and B20, and increases with respect to the HDPEw material by about 10% with PANSIL and 5% with B20. However, ductile properties such as elongation at break and Charpy impact energy absorption decrease significantly with increasing PANSIL and B20 content. The addition of 5 wt.% PANSIL reduces elongation by 44% and impact energy absorption by 33% and the addition of 5 wt.% B20 shows a similar behaviour, reducing elongation by 45% and impact energy absorption by 41%. The reduction in ductility increases with increasing sepiolite content, with the greatest loss in ductility being obtained with the addition of 15% sepiolite, i.e. 15% w/w PANSIL reduces the elongation at break by 73% and the impact energy absorption by 65%, and the addition of 15% w/w B20 reduces the elongation at break by 69% and the impact energy absorption by 76%. The reduction in impact energy is consistent with the reduction in toughness in the stress-strain curves (Figure 12) and is explained by the embrittlement effect of clays reported in the literature.34–39 Stress-strain curves for the prepared specimens.
Morphological characterisation of samples
Figure 13 shows FESEM images of the fracture surface of the HDPEw sample. FESEM images of the impact fracture surface at 250X of samples: (a) HDPEw, (b) HDPEw-5P, (d) HDPEw-10P, (f) HDPEw-15P, (c) HDPEw-5B20, (c) HDPEw-10B20 and (g) HDPEw-15B20.
The scanning electron microscope image for the HDPEw sample shows a homogeneous distribution with no heterogeneities corresponding to other plastics, which would indicate that they are present in a very small proportion.
Figure 13 shows FESEM images of the fracture surface of HDPEw samples with sepiolite (PANSIL and PANGEL B20) at 5, 10 and 15 wt.%. The images show that the distribution of sepiolite is similar for both types of sepiolite and an increase in the number of particles in the HDPEw matrix is observed with increasing sepiolite content. However, the addition of sepiolite, both PANSIL and B20, increases the number of micro-cracks and holes, which increases the roughness of the fracture surface. In addition, an increase in the heterogeneity of the impact surface is observed as the sepiolite content increases. A lack of interfacial adhesion between the sepiolite particles, both PANSIL and PANGEL B20, and the HDPEw matrix is also observed. All these peculiarities explain the embrittlement of the structure observed in the mechanical characterisation.
Odour analysis of samples
The odour of plastics can come from a variety of sources from the virgin starting material to additives or even odours that may be generated during the use of the plastic. 39
In the case of recycled plastics, odour can come from environmental or thermal degradation products, volatile and semi-volatile compounds from previous use, improper storage of containers, etc. 39 To determine the degree of bad odour, a sensory test was used with different volunteers previously selected according to their olfactory aptitude.32,40
Sepiolites have been used to retain liquids,40–42 but few tests have been carried out to retain this type of gas .
40
The following results (Figure 14) show the performance of sepiolites in retaining the gaseous compounds responsible for malodour in polymeric materials from plastic waste collection sites. Odour test values.
From the Figure 14 it can be seen that the washed HDPEw samples show a slight decrease in odour intensity compared to the unwashed samples, which is basically due to the carry-over of certain compounds by the chloroform, water and soap of the initial sample. Similarly, the extruded and injected samples lose some of this odour compared to the washed HDPEw, probably because the thermal treatments to which the sample is subjected during the extrusion and injection processes favour the elimination of the volatile compounds responsible for the bad odour. Similarly, the HDPEw Pansil samples reduce the odour much more than the HDPEw Pangel B20 samples, which could be due to the higher SBET specific surface area of the Pensil sepiolite 294 m2·g−1 compared to the Pangel B20 sepiolite 95 m2·g−1, as this would imply a higher contact surface area in the Pensil sepiolite samples.
Conclusions
The results described above show that the starting HDPEw material contains a number of impurities from other polymeric materials analysed by FTIR. However, these impurities are not very abundant as no melting peaks of other semi-crystalline polymeric materials are observed by DSC. On the other hand, mechanical characterisation shows that it has similar Charpy tensile strength, elongation and impact energy absorption characteristics to other recycled HDPE,
The sepiolites incorporated in the HDPE have a reinforcing effect, with PANSIL sepiolite increasing the tensile strength by 10% and B20 sepiolite by 5%. However, ductile properties such as elongation at break and Charpy impact energy absorption decrease, and this decrease increases with increasing amount of sepiolite. When the morphology of the fracture surface is analysed by FESEM, a homogeneous distribution of both sepiolites is observed, but both sepiolites increase the number of microcracks and the roughness of the fracture surface, and a lack of adhesion between the sepiolite and the HDPEw matrix is also observed. These features explain the embrittlement of the samples when sepiolite was added.
Regarding the thermal characterization, it is observed that the incorporation of sepiolites does not alter the melting temperature of HDPEw. However, it does affect the crystallinity depending on the type of sepiolite used. Thus, sepiolite B20 acts as a nucleating agent and increases the crystallinity while the addition of PANSIL sepiolite presents a different behavior depending on the percentage used, so the HDPEw-15P sample (53.6%) decreases the degree of crystallinity with respect to the HDPEw sample (56.8%) probably due to a poor dispersion that obstructs crystallization. On the other hand, PANSIL sepiolite at high percentages increases the thermal stability of HDPEw by delaying the onset of degradation at higher temperatures. On the other hand, B20 sepiolite reduces the onset temperature of HDPEw degradation.
The results obtained show that PANSIL sepiolite gives better results than B20 sepiolite, but it is necessary to improve the dispersion and adhesion of this inorganic filler in the HDPEw matrix.
Samples extruded and injected with sepiolites versus HDPEw lose some of this odour, probably due to heat treatments that favour the elimination of volatiles responsible for the bad odour. HDPEw Pansil samples reduce the odour much more than HDPEw Pangel B20 samples, probably due to their higher specific surface area, as this would imply a larger contact surface in the samples with the volatile compounds and a higher adsorption of these compounds.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the ERDF A way of making Europe and State Investigation Agency (PID2021-123753NA-C33 and PID2020-116496RB-C22) and European Union Next Generation EU/PRTR and MCIN/AEI.
