Abstract
In this study, diethoxydimethylsilane (DEDMS) was blended with chitosan (CS) to fabricate DEDMS/CS composite films, which were deposited on the surface of 316L stainless steel to assess their anticoagulant properties. A neat CS film exhibits a high concentration of bovine serum albumin (BSA) protein adhesion of 458.3 ± 6.2 μg/L in a bicinchoninic acid protein assay because the carbonyl and amide functional groups on the CS surface easily form hydrogen bonds with the carboxylic acid functional groups of the BSA protein. The DEDMS/CS composite films exhibited lower BSA adhesion concentration than neat CS films because some of the carbonyl and amide functional groups on the surface of CS were replaced by the –Si-O-Si and –Si-(CH3)2 functional groups. Increasing the DEDMS content in the DEDMS/CS composite films led to a higher concentration of –Si-O-Si and –Si-(CH3)2 functional groups on the surface and lower BSA adhesion concentration. Blending excess amounts of DEDMS caused an undesirable rougher surface morphology because of the hydrolysis and self-condensation reactions of DEDMS; this deteriorated the anticoagulant properties of the composite films. The study confirmed that a DEDMS/CS composite film with an appropriate DEDMS/CS content ratio of 8/92 mL/mL possesses hydrophobic characteristics and the lowest BSA protein adhesion of 155.5 ± 1.0 μg/L and has potential for biomedical coating applications.
Introduction
Metallic biomaterials such as stainless steel, cobalt–chromium alloys, pure titanium, Ti–6Al–4V alloys, Ti–6Al–7Nb alloys, and TiNi shape memory alloys have been used for surgical prosthetics and orthotics for decades. 1 These biomaterials typically possess superior mechanical properties and sufficient biocompatibility for use in medical and surgical devices. Nevertheless, when these metallic biomaterials are exposed to body fluids, their inadequate corrosion resistance and undesirable leaching of metallic ions may limit their practical applications. To address this issue, the surfaces of these biomaterials are usually protected by biomedical coatings, including ceramics, bioactive glass, and polymers.2–5
Chitosan (CS) is a bioactive coating that is widely used for biomedical applications because of its non-toxicity, biodegradability, biocompatibility, easy preparation, and relatively low cost.6–8 Numerous studies have reported that the addition of organic or inorganic additives to chitosan to form a composite film can effectively improve its physical and chemical properties.9–18 For instance, adding graphene oxide, graphene, carbon nanotubes, or montmorillonite to chitosan can enhance the mechanical properties of chitosan for implant applications,11–14 while blending chitosan with polycaprolactone, polylactide, polyglycolide, or poly (lactic-co-glycolic acid) is beneficial for its tissue engineering applications.15–18
Silanes, which are inorganic compounds containing one or many silicon–carbon bonds, are widely used as precursors to elemental silicon, polymer modifiers, and coupling agents to bond organic materials to inorganic materials. Various types of silane, such as tetraethyl orthosilicate (TEOS), (3-aminopropyl)triethoxysilane (APTES), octyltriethoxysilane (OTES), 3-glycidoxypropyl trimethoxysilane (GPTMS), 1,2-bis(triethoxysilyl)ethane (BTSE), and (3-aminopropyl)trimethoxysilane (APTMS), serve as protective coatings for metallic biomaterials because they improve protein adsorption, cell viability, bacterial reduction, and platelet adhesion of biomedical implant materials. 19
Recently, several studies have demonstrated that combining silane and chitosan as silane–chitosan composite films or hybrid scaffolds is promising for biomedical applications.20–23 Spirk et al. 20 proposed a fast and highly reproducible sol-gel process to prepare silane–chitosan composite films with controllable layer thicknesses and morphologies. Connell et al. 21 reported that chitosan–silica hybrid scaffolds using GPTMS in a sol–gel process were suitable for bone or cartilage regeneration. Palla-Rubio et al. 22 developed a silane–chitosan antibacterial coating using TEOS to avoid dental implant-related infections and promote the osseointegration of titanium implants. Réthoré et al. 23 synthesized a novel silanized chitosan, an injectable, self-setting, and cytocompatible hydrogel, to support the in vitro and in vivo viability and activity of human adipose stromal cells.
Except of the aforementioned silanes, diethoxydimethylsilane (DEDMS) is also a candidate material for silane–chitosan composite coatings to improve the physical and chemical properties of neat chitosan. DEDMS is an organically modified silicate with two methyl groups and is often used to incorporate the silicon-carbon bond into the organic-inorganic silica structure to prepare variety new materials. Nevertheless, the synthesis and characterization of diethoxydimethylsilane/chitosan (DEDMS/CS) composite films have not yet been investigated. Therefore, this study aimed to prepare DEDMS/CS composite films with various DEDMS/CS ratios and investigate the surface and protein adsorption properties of these DEDMS/CS composite films to assess their feasibility in biomedical coating applications.
Materials and methods
Preparation of DEDMS/CS composite films
The 316L stainless steel used in this study was purchased from Soonglee Metals Inc., Taiwan. The surface of the 361L stainless steel was progressively ground with abrasive paper, polished using 0.3 μm Al2O3 powder, and cut into specimens with dimensions of 20 × 30 × 1 mm with a low-speed diamond saw. The 361 L stainless steel samples were subsequently cleaned sequentially with isopropanol and deionized water to remove residual organic matter. The cleaned 361L stainless steel samples were immersed in piranha solution, a 3:1 (v/v) solution of concentrated sulfuric acid (95%), and hydrogen peroxide solution (35%) for 15 min to increase the amount of hydroxyl functional groups on the sample surface. The samples were then immersed in a solution of 2% v/v 3-aminopropyltriethoxysilane (APTES) in toluene at room temperature for 24 h to form siloxanes covalently bonded to the 361L stainless steel surface. After washing sequentially with toluene, ethanol, and deionized water, the siloxane bonded 361L stainless steel samples were immersed in a 25% glutaraldehyde solution and gently stirred at room temperature for 1 h. The glutaraldehyde solution served as a crosslinking agent, by which aldehyde groups can react with the amino group in APTES and with the primary amino group in the CS or DEDMS/CS composite.
The CS powder used in this study was purchased from C&B Co., Ltd., Taiwan. The CS powder was prepared from crab shells and had a molecular weight in the range of 10–50 kDa and a 90% degree of deacetylation. The DEDMS solution (97.0%) used in this study was purchased from Acros, US. The purchased CS powder (1.5 g) was first dissolved in a 100 mL acetic acid solution (99.7%) to form a CS solution, which was then mixed with various volumes of the DEDMS solution to form DEDMS/CS gels (DEDMS:CS = 0:100, 1:99, 2:98, 5:95, 8:92, and 10:90 mL/mL). Each DEDMS/CS gel was stirred at 50°C and 700 r/min for 3 h. The 361 L stainless steel samples were settled in stainless steel trays, and each DEDMS/CS gel was added to form a DEDMS/CS composite film on the surface of the 316L stainless steel sample, followed by drying in an oven at 45°C for 24 h. Then, the DEDMS/CS composite films were immersed in a 0.5 N NaOH solution for 2 h to neutralize excess acetic acid, and were washed repeatedly with deionized water until the pH value of the solution approached 7. Finally, each DEDMS/CS composite film was dried in ambient air at room temperature to obtain a dense film for subsequent characterization. The prepared DEDMS/CS composite films with DEDMS/CS volume ratios of 0:100, 1:99, 2:98, 5:95, 8:92, and 10:90 were designated as neat CS, DEDMS1, DEDMS2, DEDMS5, DEDMS8, and DEDMS10, respectively.
Characterizations
The functional groups of the DEDMS/CS composite films were determined using an attenuated total reflectance Fourier-transform infrared (ATR-FTIR) spectrometer (Spectrum 100, PerkinElmer). Each specimen was measured in the range of 4000–6500 cm−1 using 16 scans at a resolution of 4 cm−1. The wettability properties of the surface of the DEDMS/CS composite films were measured using the sessile drop method using a contact angle instrument (FTA125, First Ten Ångstroms, USA). Surface morphology observations and chemical composition analyses of the DEDMS/CS composite films were performed using a scanning electron microscope (SEM) (5136 MM, Tescan instruments) equipped with an energy-dispersive X-ray spectroscope (EDS) (X-Act, Oxford). The surface chemical compositions of the DEDMS/CS composite films were determined using X-ray photoelectron spectroscopy (XPS) (K-Alpha, Thermo Scientific) with a monochromatic Al Kα radiation source of 1468.6 eV. The survey spectrum of each specimen was determined in the range of 0–700 eV in 1 eV steps. The high-resolution Si2p spectra for each specimen were determined in 0.05 eV steps.
The anticoagulant properties of the DEDMS/CS composite films were assessed by determining the amount of bovine serum albumin (BSA) adsorbed on the film surface using a bicinchoninic acid (BCA) protein assay. The DEDMS/CS composite films were rinsed with phosphate buffer solution (PBS), immersed in 5 mL of BSA solution at 37°C for 24 h, and then rinsed again with PBS. The DEDMS/CS composite films were then immersed in 2 mL of sodium dodecyl sulfate (SDS) solution for another 24 h. A 0.1 mL sample of the solution was mixed with 1 mL of the BCA solution in a cuvette to determine its optical density (OD) value measured at 562 nm using a spectrophotometer (GENESYS 20, Thermo Scientific, USA). The concentrations of BSA adhered onto the films were calculated according to the determined OD values using a BSA concentration standard curve. The BSA and BCA used in this study were purchased from Bio Basic Inc, while PBS and SDS were purchased from UniRegion and Sigma-Aldrich, respectively.
Results and discussion
ATR- FTIR analysis
Figure 1 shows the ATR-FTIR spectra of the neat CS and the DEDMS/CS composite films with various DEDMS/CS volume ratios. As shown in Figure 1, the neat CS and DEDMS/CS composite films exhibit a broad characteristic peak at approximately 3000–3500 cm−1, which is attributed to the stretching vibrations of the –NH2 and –OH groups. The characteristic absorption band at approximately 2932 cm−1 corresponds to the –CH group. The characteristic absorption bands appearing at approximately 1642, 1586, and 1409 cm−1 were attributed to the –C = O carbonyl stretching of amide I, the –NH bending of amide II, and the –CN stretching of amide III, respectively. The absorption band appearing at approximately 1257 cm−1 is attributed to the –CO functional group. The characteristic absorption bands at approximately 1130 and 847 cm−1 were assigned to the –Si-O-Si and –Si–(CH3)2 groups, respectively, because of the presence of DEDMS. The signals of the –Si-O-Si and –Si-(CH3)2 groups became more significant with increasing DEDMS content in the DEDMS/CS composite films. ATR-FTIR spectra of neat CS and DEDMS/CS composite films with various DEDMS/CS ratios.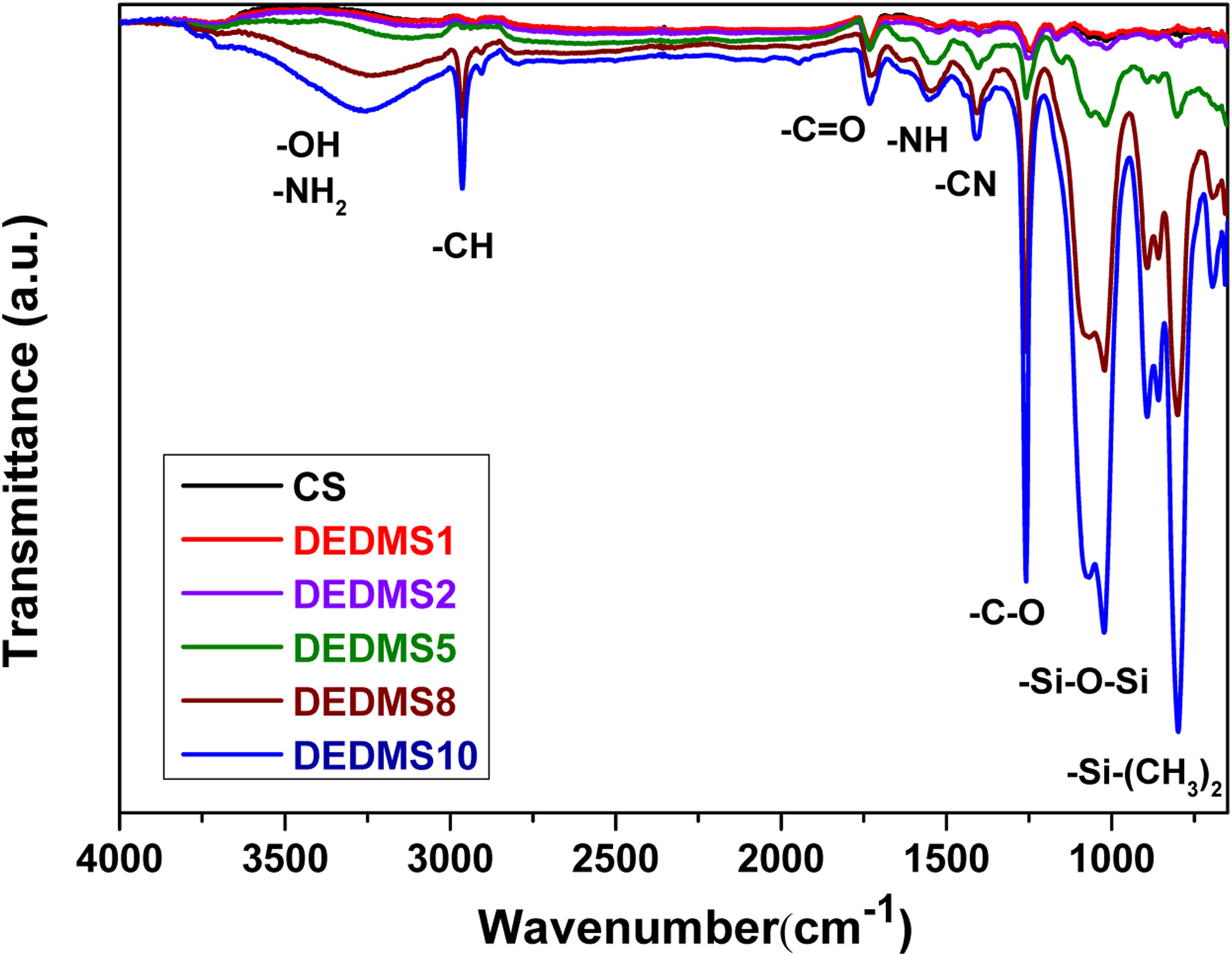
Wettability properties
Figure 2 shows the water contact angles of the neat CS and the DEDMS/CS composite films with various DEDMS/CS ratios. Figure 2 indicates that the neat CS film exhibited a relatively low water contact angle of approximately 75.4 ± 2.3°. The water contact angle values of the DEDMS/CS composite films all exceed 80°, indicating that the surface of the DEDMS/CS composite films became more hydrophobic than that of the neat CS film. The hydrophobic characteristic of the surface of the DEDMS/CS composite films was attributed to the abundant –Si-O-Si, and –Si–(CH3)2 functional groups on the surface, as shown in Figure 1. Among these DEDMS/CS composite films, DEDMS1 and DEDMS2 possessed a high water contact angle of above 95°. Nevertheless, the water contact angle of the DEDMS/CS composite films gradually decreased with increasing amount of DEDMS in DEDMS5, DEDMS8, and DEDMS10. Water contact angle of neat CS and DEDMS/CS composite films with various DEDMS/CS ratios.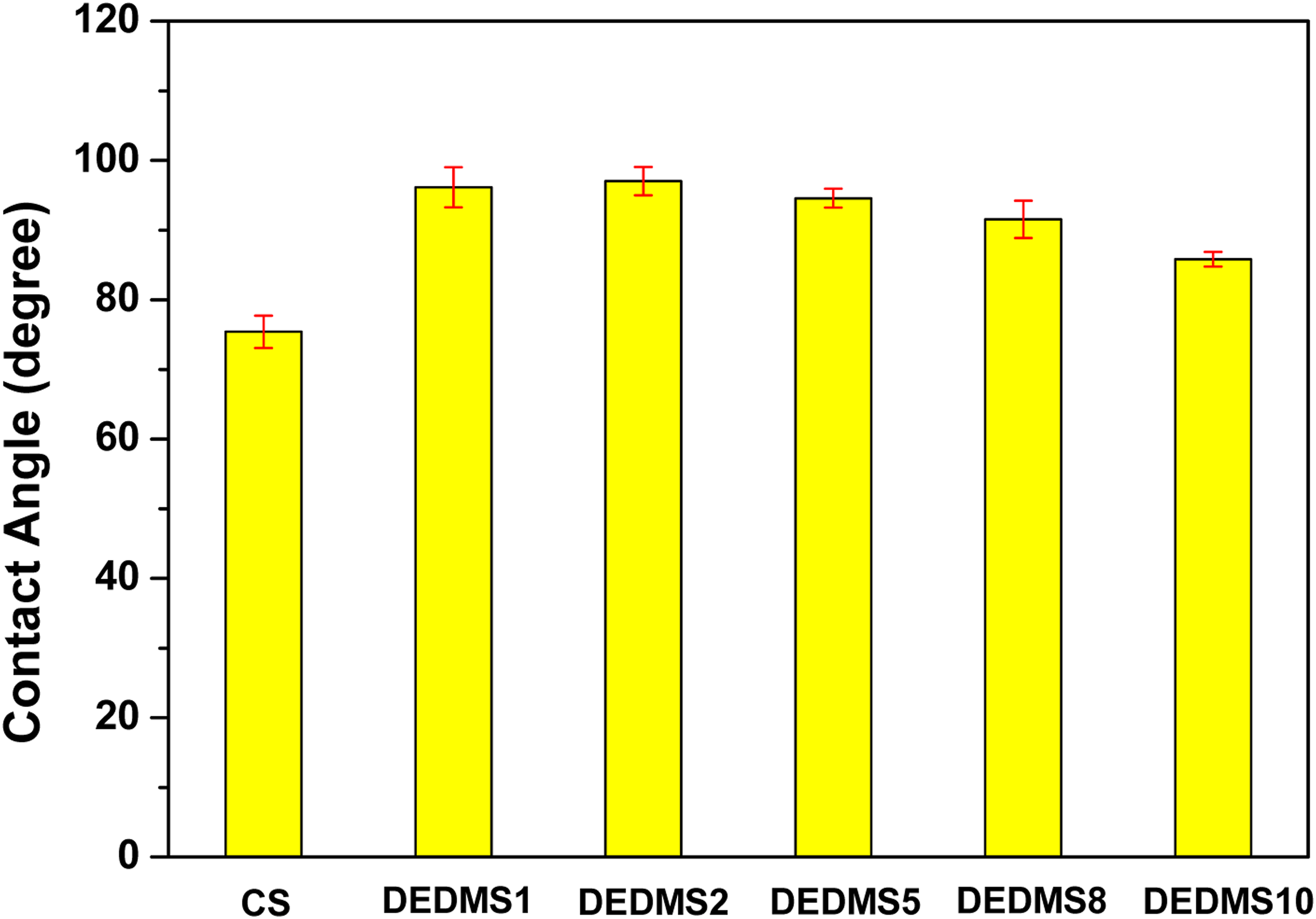
XPS measurements
Figures 3(a)–(f) show the XPS survey spectra of neat CS, DEDMS1, DEDMS2, DEDMS5, DEDMS8, and DEDMS10, respectively. Figure 3(a) demonstrates that the XPS spectrum of the neat CS film exhibits a significant C1s peak at approximately 284.4 eV, a N1s peak at approximately 398.5 eV, and a O1s peak at approximately 531.0 eV. Figure 3(b) shows that the XPS spectrum of the DEDMS1 composite film also shows C1s, N1s, and O1s peaks at approximately 284.4, 398.5, and 531.0 eV, respectively. The film also exhibits Si2s and Si2p characteristic peaks at approximately 150.7 and 101.7 eV, respectively. The inset in Figure 3(b) presents the high-resolution Si2p XPS spectra of the DEDMS1 composite film. The significant Si2p peak, which was not observed in the XPS result of the neat CS, originated from the blended DEDMS. In Figure 3(d)–(g), the survey and high-resolution Si2p XPS spectra of the DEDMS2, DEDMS5, DEDMS8, and DEDMS10 composite films are similar to those of DEDMS1. Figure 3 demonstrates abundant DEDMS on the surface of the DEDMS/CS composite films. XPS survey spectra for (a) neat CS, (b) DEDMS1, (c) DEDMS2, (d) DEDMS5, (e) DEDMS8, and (f) DEDMS10 composite films. The inset shows the high-resolution Si2p XPS spectra of each specimen.
SEM and EDS measurements
Figures 4(a)–(f) show the surface morphology images (500×) of the neat CS, DEDMS1, DEDMS2, DEDMS5, DEDMS8, and DEDMS10 composite films, respectively. Figure 4(a) shows that the neat CS film exhibits a smooth and homogeneous surface morphology, while Figure 4(b) shows that the DEDMS1 film exhibits small circular holes on its surface. As shown in Figures 4(c)–(f), both the number and radii of circular holes increase with an increase in the amount of blended DEDMS in the DEDMS/CS composite films. Figures 5(a)–(f) present the EDS mapping analysis of silicon for neat CS, DEDMS1, DEDMS2, DEDMS5, DEDMS8, and DEDMS10, respectively. Figure 5(a) reveals that no silicon atoms can be detected on the surface of the neat CS film. The number of silicon atoms on the surface of the DEDMS/CS composite films increases with increasing DEDMS content, as shown in Figures 5(b)–(f). These silicon atoms correspond to the –Si-CH3, –Si-O-Si, and –Si–(CH3)2 functional groups on the surface of the DEDMS/CS composite films. Figures 5(b)–5(f) also show that the silicon atoms are homogeneously distributed on the surface of these DEDMS/CS composite films. SEM images of the surface morphologies (500×) for (a) neat CS, (b) DEDMS1, (c) DEDMS2, (d) DEDMS5, (e) DEDMS8, and (f) DEDMS10 composite films. EDS silicon map results for (a) neat CS, (b) DEDMS1, (c) DEDMS2, (d) DEDMS5, (e) DEDMS8, and (f) DEDMS10 composite films.

Bicinchoninic acid (BCA) protein assay
Figure 6 presents the concentrations of adsorbed BSA on the surfaces of the neat CS and DEDMS/CS composite films in the BCA protein assay. As shown in Figure 6, the concentration of BSA adhered on the surface of the neat CS film is approximately 458.3 ± 6.2 μg/L. The concentrations of BSA on the surfaces of the DEDMS/CS composite films decrease significantly from 419.0 ± 7.2 μg/L (DEDMS1) to 155.5 ± 1.0 μg/L (DEDMS8) with increasing amounts of blended DEDMS; this indicates that adding DEDMS to the CS film effectively improves its anticoagulant properties. However, the concentration of adhered BSA on the surface of DEDMS/CS composite films slightly increases from 155.5 ± 1.0 to 167.6 ± 9.2 μg/L when the DEDMS content increases from 8 to 10%. The neat CS film exhibits high BSA protein adsorption because the carbonyl and amide functional groups on the surface of the CS film and the carboxylic acid functional groups of the BSA protein easily form hydrogen bonds. On the other hand, the BSA protein adsorption of all DEDMS/CS composite films is comparatively lower than that of the neat CS film, with the DEDMS8 demonstrating the lowest BSA protein adsorption of approximately 155 μg/L. This phenomenon can be attributed to the fact that part of the carbonyl and amide functional groups on the surface of the DEDMS/CS composite films are replaced by the –Si-O-Si and –Si-(CH3)2 functional groups, as demonstrated by the XPS and EDS results shown in Figures 3 and 5, respectively. BSA adsorption concentrations of neat CS and DEDMS/CS composite films with various DEDMS/CS ratios.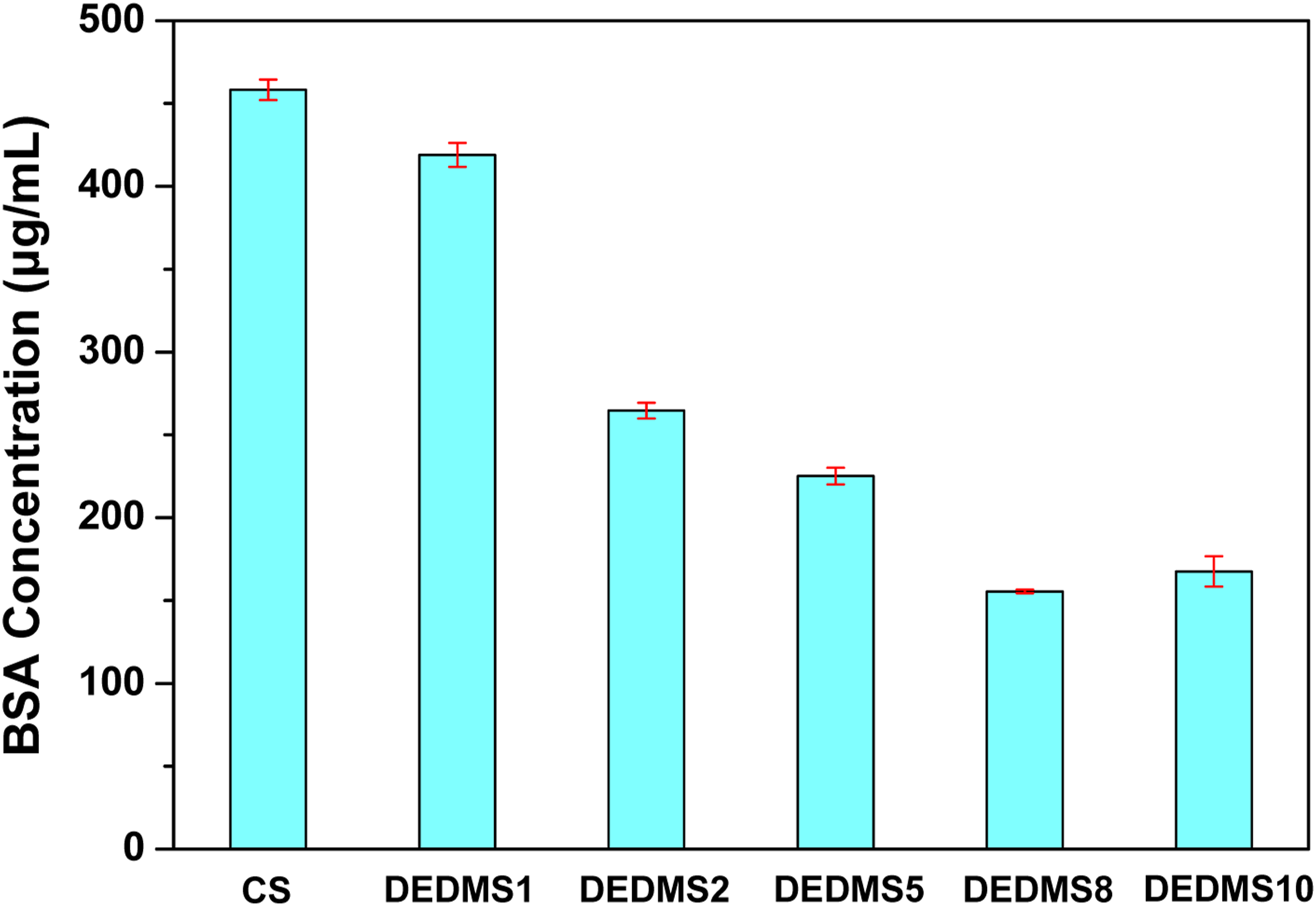
Figure 6 also shows that the concentration of BSA protein adsorption decreases significantly from DEDMS1 to DEDMS8. This is because there are more –Si-O-Si and –Si-(CH3)2 functional groups on the surface with increasing DEDMS content in the composite films, as shown in Figure 5. However, the DEDMS10 composite film exhibits a higher concentration of adhered BSA protein than DEDMS8, even though DEDMS10 possesses a higher DEDMS content than DEDMS8. This unexpected result stems from more small circular holes on the DEDMS10 surface than that of DEDMS8, as demonstrated in Figure 4. The small circular holes on the surface of the DEDMS/CS composite films originate from the hydrolysis and self-condensation reactions of DEDMS, which cause shrinkage of the composite films. Thus, the DEDMS/CS composite films containing higher amounts of DEDMS exhibit rougher surface morphologies, leading to a higher concentration of BSA protein. Therefore, the DEDMS/CS composite films possess better anticoagulant properties than neat CS films for in vivo bioactive material films, especially for highly hydrophobic surfaces. However, the amount of blended DEDMS in the composite films should be carefully controlled, as excessive blending with DEDMS typically causes a rough surface and deteriorates the anticoagulant properties of DEDMS/CS composite films.
Conclusions
In this study, DEDMS/CS composite films were fabricated and deposited on the surface of 316L stainless steel. The presence of abundant –Si-O-Si and –Si-(CH3)2 functional groups formed on the surface of the DEDMS/CS composite films caused a hydrophobic surface. The contents of –Si–O–Si and –Si–(CH3)2 functional groups on the film surface both increased with increasing DEDMS content in the DEDMS/CS composite films. The surface of the DEDMS/CS composite films became rougher with increasing DEDMS content owing to shrinkage of the composite films caused by hydrolysis and self-condensation reactions of DEDMS. BCA protein assay results showed that the DEDMS/CS composite films exhibited better protein adhesion properties than neat CS film because of the –Si–O–Si and –Si–(CH3)2 functional groups on the surface. Thus, it can be inferred that blending appropriate amounts of DEDMS with chitosan can effectively improve its protein adsorption properties; however, the surface flatness of DEDMS/CS composite films should be considered and improved to broaden their biomedical coating applications.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The authors gratefully acknowledge the financial support provided by the Ministry of Science and Technology (MOST), Taiwan, under Grant No. MOST 110-2221-E-197-013-MY2.
