Abstract
In this work, novel superabsorbent composites made of starch-grafted-polyacrylic acid/coal gangue were prepared via aqueous solution polymerization. The starch-g-polyacrylic acid/coal gangue (St-g-PAA/CG) composite with 10 wt.% CG showed the maximum water absorbency in distilled water (560 g·g−1) and a 0.9 wt.% NaCl solution (102 g·g−1). Moreover, this superabsorbent composite exhibited fine thermal stability and its swelling behavior was dependent on pH. In general, the swelling kinetics of the superabsorbent composites fitted with the Scott’s kinetic model. These composites not only showed excellent water absorption reversibility after five absorption/drying cycles, but also exhibited considerable environmental responsiveness after the swelling medium was changed from distilled water to the 0.9 wt.% NaCl solution. Based on these results, the prepared superabsorbent composites can be applied in several fields such as horticulture, agriculture, health care, and controlled drug delivery, while ensuring comprehensive utilization of solid wastes.
Introduction
Coal gangue (CG) is a complex solid waste from the coal industry whose main mineral phases are kaolinite, illite, and quartz. 1 Although China has already produced 3.8 billion tons of CG, stockpiles of CG are increasing at a rate of 0.2 billion tons per year. 2 Since existing technologies are not fully utilizing the CG reserves via high value-added approaches, CG has taken up plenty of space and led to various several critical environmental dilemmas, which have been perplexing the coal industry worldwide. 3 Accordingly, numerous techniques have been developed to increase CG utilization. For example, surplus CG has been sustainably utilized to generate electricity, 4 develop brick, stabilize soil, 5 and absorb toxic materials such as Hg (II) 6 and Cr(VI). 7 However, there has been insufficient research on the components and surface functional groups of CG, which has inhibited the development of novel CG transformation techniques aimed at resolving the inherent problems caused by waste CG.
A superabsorbent with a cross-linked network can easily absorb water, saline, or other solutions 8,9 by relying on its three-dimensional arrangement and different functional groups. Consequently, superabsorbent polymers have been employed in numerous fields, such as agriculture and adult incontinence products, 10 horticulture,11,12 absorbents, 13 and drug delivery systems. 14
However, superabsorbents derived from fossil commodities such as acrylic acid (AA) are extremely expensive, whereby limiting their application in many fields. Several studies have demonstrated that the absorption capacity of superabsorbent polymers can be enhanced by incorporating inorganic materials, which also reduces the cost of the product. Thus, the research and development of superabsorbent composites, especially those including inorganic materials have attracted increasing attention.
Wang et al. 15 synthesized a novel eco-friendly superabsorbent composite of chitosan-grafted-polyacrylic acid/poly-dimethylourea phosphate/calcined semicoke that exhibited a maximum water absorption capacity of 412.89 and 51.30 g·g−1 in deionized water and 0.9 wt% NaCl solution, respectively. A biodegradable superabsorbent polymer has been fabricated from zinc oxide and starch for use in plant growth. 16 Meanwhile, Le et al. 17 conducted a one-step synthesis of a super-absorbent nanocomposite hydrogel based on bentonite. Furthermore, novel superabsorbent hydrogels based on starch, gellan gum, citric acid, and nanoclays have been used for crop water management, which then are used as nutrients after decomposition. 18
The recent advent of “sustainable development” has enabled the rapid utilization of wastes as inorganic materials to fabricate superabsorbent composites. Industrial wastes19,20 and agriculture wastes 21 have been used to fabricate superabsorbent composites. Liu 22 synthesized a superabsorbent with waste hemicelluloses lye. An environmental-friendly superabsorbent composite 23 based on rice husk has also been observed to improve soil water retention and control urea release.
It is worth noting that CG components mainly compries inorganic minerals, 24 which include active functional groups such as the hydroxyl group or carboxyl group. These functional groups participate in the polymerization reaction by generating a positive interaction with the superabsorbent backbone. Furthermore, the introduction of inorganic minerals not only enhances the superabsorbent properties such as water absorption and stability, but also reduces incurring costs. Therefore, incorporating CG into superabsorbent composites is a practical approach. In this study, a novel superabsorbent composite was designed and fabricated via aqueous solution polymerization of starch and AA in the presence of CG. The factors influencing water absorbency, including starch content, initiator content, crosslinker content, CG content, neutralization degree, and pH value, were also studied. Moreover, the swelling kinetics and water absorption reversibility were tested. The environmental-responsiveness property was examined by studying their swelling and deswelling in distilled water and 0.9 wt.% NaCl solutions. Overall, this work not only provides a promising superabsorbent composite, but also facilitates fundamental investigation on the application of solid waste or minerals.
Materials and methods
Materials
Based on X-ray fluorescence (XRF) results, the CG employed in this study included 59.09% SiO2, 23.16% Al2O3, 7.26% Fe3O4, 3.25% CaO, and 3.03% K2O. Furthermore, it was acquired from the Yulin region in Shaanxi province, China. The CG was first calcined at 700°C under a heating rate of 5°C·min−1 for 8 h. Then, CG was alkali-modified using a solution of 2 wt.% sodium hydroxide to remove impurities. Meanwhile, soluble starch was purchased from Shen Gao Chemical Reagent Co., Ltd. (Tianjin, China). Potassium persulfate (KPS) was treated as the initiator and bought from the Ke Wei Chemical Reagent Company (Tianjin, China). Furthermore, AA and hydrochloric acid were obtained from the Da Mao Chemical Reagent Factory (Tianjin, China). The crosslinker N, N′-methylenebi-sacrylamide (MBA) and sodium hydroxide were sourced from Kermel Chemical Reagent Company (Tianjin, China).
Preparation of starch-g-poly (AA)/CG (St-g-PAA/CG) superabsorbent composites
Starch was dissolved in 50 mL of distilled water and stirred at 90°C for 30 min in a 500 mL four-neck flask equipped with a condenser, stirrer, thermometer, and nitrogen line. Thereafter, the solution temperature was reduced to 40°C and KPS was introduced and stirred. After 20 min, a solution containing 14.4 g of AA (neutralized with 5 mol·L−1 NaOH) and a specific amount of CG was added to the system. The resulting admixture was stirred for 20 min, and a certain amount of crosslinker MBA (5 g·L−1) was added. The reaction temperature was slowly raised to 70°C to initiate polymerization and this temperature was maintained for 3 h to complete the grate process. The reaction was subjected to mechanical stirring (500–700 rpm) during the total process. Next, by using varying amounts of CG, starch, crosslinker, and initiator as well as different degrees of neutralization, a set of superabsorbent composites was fabircated via the above-mentioned process. All samples were milled and sieved using a 24-mesh screen. The flowchart associated with the preparation of St-g-PAA/CG superabsorbent composites is shown in Scheme 1. Preparation flowchart of St-g-PAA/CG superabsorbent composites.
The polymerization process of St-g-PAA (without CG) was the same as that used for polymerizaing St-g-PAA/CG. That is, St-g-PAA/CG was fabricated using a free radical reaction implemented via solution polymerization, wherein starch was grafted with PAA chains and CG using KPS and MBA (Figure 1). When KPS is subjected to heating, SO4− radicals can be formed. Alternatively, carbon radicals can be produced via hydrogen abstraction in the starch backbone chain. Such active centers on the backbone chains can initiate the grafting of AA. Meanwhile, MBA facilitates the formation of a crosslinked structure during the reaction. Consequently, a new macroradical with four reactive sites was formed after KPS was added to the system. These sites can be linked with radicals on both AA and starch owing to the bifunctionality of MBA. Moreover, the CG grains were scattered throughout the polymeric network. The results of Fourier transform infrared (FTIR) spectroscopy and X-ray photoelectron spectroscopy (XPS) elucidated the grafting reaction between the OH groups of CG and the functional groups (COOH and –COO−) of St-g-PAA. These reactions lead to the grafting of PAA onto starch and CG. Mechanism governing the synthesis of the St-g-PAA/CG superabsorbent composite.
Water absorbency and swelling kinetics measurements
The water absorbency was tested at room temperature, and 0.1 g of the superabsorbent composite was immersed in 400 mL of distilled water for 24 h to attain swelling equilibrium. The water absorbency (W) was determined based on equation (1).
The water absorbency of the superabsorbent composite was also measured in 0.9 wt.% NaCl solution and tap water using the aboved-mentioned procedure.
The swelling kinetics of the sample in the distilled water was assessed follows: 0.1 g sample was immersed in 400 mL of distilled water for a set period of time. Then, the swollen gel was filtered using a 200-mesh screen, and the water absorbed (Wt) at a given time (t) was measured by weighing the swollen and dry samples (equation (1)). Three replicates were performed and the average value was recorded. The swelling kinetics of superabsorbent composites in 0.9 wt.% NaCl solution was measured using the same procedure.
Environmental-responsiveness of the superabsorbent composite
The environmental-responsiveness property of the compositeswas studied in terms of their swelling and deswelling characteristics in distilled water and 0.9 wt.% NaCl solution. That is, superabsorbent composites (0.1 g) were immersed in distilled water (400 mL) until equilibrium was attained. Then, the swollen samples were soaked in 0.9 wt.% NaCl solution for set time intervals. Finally, the swollen samples were filtered and weighed, after which the change in mass before and after swelling was calculated. The same procedure was repeated thrice, and a new batch of solution was used for every cycle.
Water absorption reversibility
The water absorption reversibility was studied by repeatedly swelling and drying the superabsorbent composites. The swollen samples were dried in an oven at 70°C and then immersed in distilled water until the gel reverted to its swollen state. The Wn value (where n represents the number of absorption/drying cycles) was calculated using equation (1). Three replicates were performed and the average was reported.
Characterization
The polymerization structure of superabsorbent composites was analyzed with a Fourier transform infrared spectrometer (Bruker Tensor27 FTIR instrument, Bruker analytical instruments, Karlsruhe, Germany) from 400 to 4000 cm−1 and by adopting the KBr pellet method. The morphologye of the samples was determined using field emission scanning electron microscope (FE-SEM) (Sigma 300, Carl Zeiss AG, Jena, Germany) after the samples were coated with a gold film. Energy-dispersive spectroscopy was combined with FE-SEM to analyze the elements in the samples. A composition analysis of GC was performed using XRF (ARLADVANT'X Intellipower 3600 scanning) by employing three crystal changers (PET, XS-55, and LiF200) for varying element analyses. The thermal behavior was analyzed using a thermogravimetric analyzer (STA 449 F5/F3 Jupiter, NETZSCH Scientific Instruments Trading Ltd., German) under a heating rate of 10°C/min, temperature range of 25°C to 700°C, and a flowing nitrogen atmosphere (flow rate was 50 mL/min). XPS was performed using the ESCALAB250 Xi spectrometer (Thermo Fisher, USA) equipped with an Al Kα X-ray source that produced an energy of 1486.6 eV.
Results and discussion
FTIR spectroscopy
Figure 2 shows the FTIR spectra of CG, St-g-PAA, St-g-PAA/CG, and the physical mixture of St-g-PAA and CG. For CG (Figure 2(a)), the peaks at 1150 cm−1, 469 cm−1, 589 cm−1, 800 cm−1, and 699 cm−1 were assigned to Si-O, Si-O-Si, and Si-O-Al bonds of SiO2 sourced from CG, respectively.
25
Besides the adsorption peaks at 2963 cm−1 and 2894 cm−1 were attributed to the -CH2 stretching vibration of carbonic matter sourced from CG. The peaks at 1650 cm−1and 1445 cm−1 were characteristic of the OH- or –COO− asymmetric stretching vibrations and –CH scissoring vibrations, respectively;
26
they were representative of the carbonic matter mixed with the silicate mineral in CG.
27
FTIR spectra of (a) CG, (b) St-g-PAA, (c) St-g-PAA/CG (10 wt.%), and (d) the physical mixture of St-g-PAA and CG (m/m = 10).
As shown in Figure 2(b), the peak at 1720 cm−1 was assigned to the C=O stretching vibration of un-neutralized St-g-PAA. The peaks at 1580 cm−1 and 1420 cm−1 were attributed to the asymmetric stretching and symmetric stretching vibrations of carboxylate groups, respectively. 28 Overall, the FT-IR analysis indicates that the polymerization grafting of PAA onto the starch backbone was successful.
In contrast to the FTIR spectra of CG and St-g-PAA, the characteristic peaks of St-g-PAA appeared in the FTIR spectra of St-g-PAA/CG (Figure 2(c)). The absorption bands at 1650 cm−1, 1580 cm−1, and 1420 cm−1 shifted to 1610 cm−1, 1568 cm−1, and 1410 cm−1, respectively. The peaks associated with the symmetric and asymmetric stretching vibrations of -CH2 at 2963 cm−1 and 2894 cm−1 became weak and were shifted to 2960 cm−1and 2883 cm−1, respectively. The peak assigned to Si-O stretching vibration at 1150 cm−1 was significantly weaker and shifted to 1100 cm−1. The peaks assigned to Si-O-Si and Si-O-Al at 800 cm−1, 699 cm−1, and 589 cm−1 became slightly weaker. Furthermore, compared to the characteristic peaks (Figure 2(d)) of St-g-PAA physically mixed with CG, the peaks of St-g-PAA/CG were observed to shift considerably. This may be because St-g-PAA can react with the –OH groups on the CG surface to form an ester, and then the doped CG leads to the formation of the 3D polymeric network.
XPS analysis
The XPS analysis was conducted to determine the interaction between CG and St-g-PAA. Figure 3(a) shows that after polymerization, the peaks assigned to Si 2p and Al 2p of CG appeared in the FTIR spectra of St-g-PAA/CG, which indicates that CG was present in the polymer matrix. Figures 3(b) and 3(c) display the C1s spectra of all the samples. The St-g-PAA peaks at 284.80, 286.37, and 288.58 were associated with the carbon atoms present in the form of C-C, C-O, and C=O, respectively.
29
The C-O and C=O peaks, which shifted to lower binding energies (BEs) after CG was incorporated, indicated a decrease in the electron density of the carbon atoms during polymerization. This result is a direct consequence of the Si content produced after CG bonds with C=O or C-O. XPS spectra: (a) CG, St-g-PAA, and St-g-PAA/CG; (b) C 1s of St-g-PAA; (c) C 1s of St-g-PAA/CG; (d) O 1s of St-g-PAA; (e) O 1s of St-g-PAA/CG.
As shown in Figures 3(d) and 3(e), the O1s spectra of St-g-PAA observed before and after incorporating CG include two peaks that are attributed to C-O (531 eV) and C=O (532 eV). More importantly, both peaks shifted to lower BEs (C=O: from 532.88 to 532.79; C-O: from 531.62 to 531.43), while the C-O peak intensity increased and the C=O peak intensity declined. This result also shows that the reaction between Si-OH (sourced from CG) and COO- (sourced from AA or starch) may decrease the electron density of the oxygen atoms. 30
These results established that CG was involved in copolymerization through the covalent bond formed between CG and the St-g-PAA chain, 31 which was in accordance with the FTIR analysis.
SEM analysis
The surface morphology and microstructure of St-g-PAA (Figure 4(a)) showed a compact and folded surface. Such a structure may make it hard for the water to permeate into the polymeric network. After incorporating CG into St-g-PAA (Figure 4(b)), the surface morphology of St-g-PAA/CG became coarse and fibrous, while comprising a few porous cavities, and exhibiting a gullies structure. Such a structure enables water permeation and leads to the formation of sites that interact with the hydrophilic groups of the graft copolymers,
32
thereby resulting in rapid permeation. Meanwhile, CG showed a sheet surface morphology (Figure 4(c)); these individual CGs were not spotted in St-g-PAA/CG, thereby demonstrating that CG particles can be scattered during polymeric reaction and can be integrated within the polymer matrix without aggregation. This result indicates that the superabsorbent composite is formed. SEM micrographs of (a) St-g-PAA, (b) St-g-PAA/CG (10 wt.%), and (c) CG; EDS spectra of (d) St-g-PAA, (e) St-g-PAA/CG (10 wt.%), and (f) CG.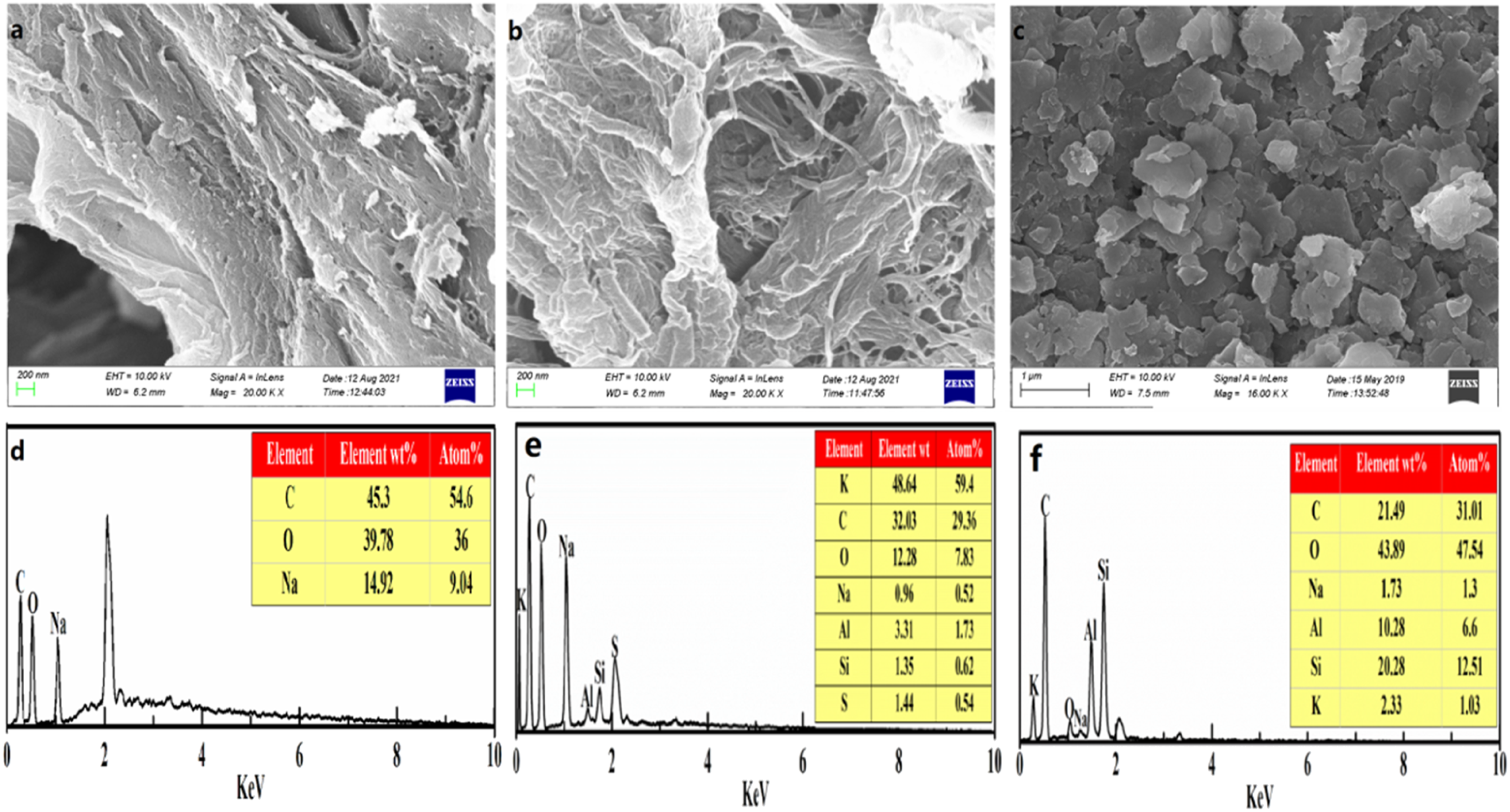
Additionally, EDS spectrograms of St-g-PAA, St-g-PAA/CG (10 wt.%), and CG are shown in Figures 4(d)–4(f), respectively. Since only the characteristic peaks corresponding to C, O, and Na elements were seen in the EDS spectrogram of St-g-PAA (Figure 4(d)), it can be concluded that its surface comprised only C, O, and Na. In St-g-PAA/CG (Figure 4(e)), Si, Al, K, and Na were also found (Figure 4(f)). This result shows that CG was magnificently scattered and embedded into the St-g-PAA network matrix. Overall, CG increased the absorption and holding capacities of the 3D network.
Thermogravimetric (TG) analysis
Figure 5 shows the thermal stabilities of CG, St-g-PAA, and St-g-PAA/CG, thereby reflecting the reciprocity of CG as well as the polymer matrix of St-g-PAA. The curve associated with CG (Figure 5(a)) showed that a significant thermal loss occurred around 377°C and a minor thermal loss was observed around 623°C. Overall, three distinct stages of thermal weight loss were observed: The first stage caused a 2.1% reduction in weight after the temperature was elevated from room temperature to 377°C, owing to the vaporization of water molecules and release of volatiles. The second stage to a 7.2% weight loss after the temperature was elevated from 377°C to 623°C, owing to the combustion of volatiles and fixed carbon. The third stage occurred after the temperature was elevated from 624°C to 800°C; only a slight weight loss was observed due to the minor decomposition of inorganic substances.
33
TG and DTG of (a) CG, (b) St-g-PAA, and (c) St-g-PAA/CG.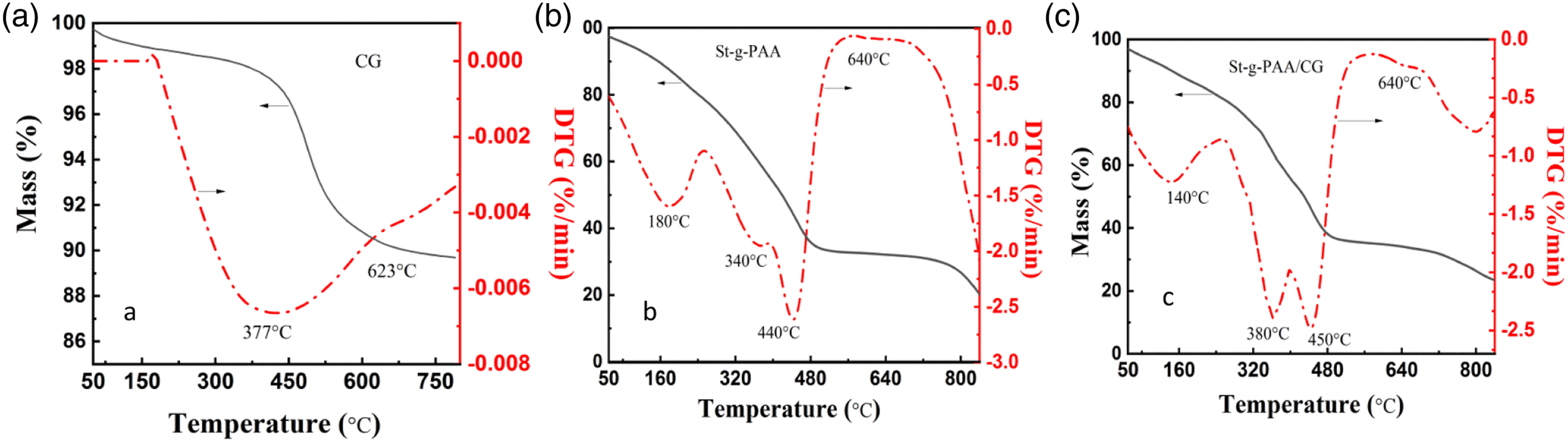
Both St-g-PAA and St-g-PAA/CG (Figures 5(b) and 5(c)) showed three weight loss stages from 100°C to 800°C. The majority of the weight loss of the superabsorbents occurred in the first stage within the temperature range of 180°C to 340°C for St-g-PAA and 140°C to 380°C for St-g-PAA/CG, thus leading to a 37.9% and 30.5% reduction in the weight of St-g-PAA and St-g-PAA, respectively, as the bonded water was lost in the network. 34 The second mass loss stage happened within the temperature ranges of 340°C to 440°C and 380°C to 450°C, which led to a mass loss of 26.3% and 24.5%, respectively, as the adjacent carboxyl groups on the graft chain of starch and St-g-PAA were dehydrated. 35 Finally, a weight loss of 17.45% and 16.10% occurred in St-g-PAA and St-g-PAA/CG within the temperature range of 550°C to 800°C and 640°C to 800°C, respectively, as the crosslinking structures were oxidated and decomposed and the residual organics were broken down. 36
Compared with St-g-PAA, the temperature required for complete carbonization of St-g-PAA/CG was higher, indicating the St-g-PAA/CG exhibited lower mass loss and thermal decomposition rates; this was mainly because CG addition strengthened the crosslinking of the network structure. 37 Furthermore, the residual weight of St-g-PAA/CG was 1.34% higher than that of St-g-PAA, indicating that CG addition slightly improves thermal stability. On the other hand, St-g-PAA was grafted with CG via the crosslinking network, which was formed through the hydrogen bonding between the hydroxyl groups on the CG surface and the hydrophilic groups in St-g-PAA; this led to a slight interaction between St-g-PAA and CG. 38 These above-mentioned effects slightly improve thermal stability.
Water absorbency studies
Effect of starch content on water absorbency
Figure 6(a) shows the effect of starch content on water absorbency in distilled water. The water absorbency of the superabsorbent composites elevated with an increase in the starch content and had an optimum value of 560 g·g−1 when the starch content was 30 wt.%. Further elevating the starch content decreased water absorbency. This may be because a lower starch content leads to lower viscosity, which allows AA to easily penetrate starch, improve graft efficiency, and increase water absorbency. Excessive starch can hamper the graft reaction due to steric hindrance, thereby reducing water absorbency.
39
Effects of (a) starch content, (b) KPS content, (c) MBA content, (d) neutralization degree, (e) CG content, and (f) pH on water absorbency.
Effect of KPS content on water absorbency
Figure 6(b) shows the effect of the KPS content on the water absorbency of the superabsorbent composites in distilled water. It is clear that water absorbency was enhanced with an increase in the KPS content, and an optimum value of 560 g·g−1 was achieved after the KPS content was elevated to 1.0 wt.%.
The high water absorbency indicated the active participation of the initiator to form many reactive free-radical sites on the starch backbone, which was beneficial to both the chain transfer reaction and the growth of the grafting polymerization chain. Consequently, when the KPS content was lower than 1.0 wt.%, more free-radical sites were generated. Increasing the KPS content led to formatting more three-dimensional networks, which could absorb and hold more water. However, water absorbency began to decline after further elevating the KPS content. Such a result is attributed to the rapid termination of macroradicals due to their excess formation; thus, the average length of the water absorption chains became short, which reduced the mass of the water absorbed. 40
Effect of MBA content on water absorbency
Figure 6(c) shows the effect of MBA content on the water absorbency of the superabsorbent composites in distilled water. As long as the MBA content was lower than 0.18 wt.%, the water absorbency increases with a rise in the MBA content d. When the MBA content was 0.18%, the water absorbency reached its maximum value. After the MBA content was increased from 0.18 wt.% to 0.24 wt.%, the water absorbency expeditiously declined from 560 g·g−1 to 201 g·g−1. This is because excess crosslinker content produced more crosslinking points and increased the crosslinking density, declined the space available in the macromolecular network for water to enter, and inhibited the stretching of the polymer chain.41,42 Consequently, water absorbency was decreased.
Effect of neutralization degree on water absorbency
Figure 6(d) shows the effect of neutralization degree on the water absorbency in distilled water. When the neutralization degree reached approximately 60%, the water absorbency reached its maximum value. With a decrease in the neutralization degree, AA dissociation became extremely weak. Consequently, this made it difficult to control the polymerization reaction. Besides, during the cross-linking reaction of other components, acrylic acid can also undergo a self-cross-linking reaction, which results in the formation of a highly self-cross-linking polymer and thus decreases water absorption. 43 When the neutralization degree rose, the number of –COO− groups in the macromolecular network also increased. This led to a rise in the difference in the osmotic pressure observed inside and outside the network. Furthermore, –COO− attached to the polymer chains and led to an electrostatic repulsion, which was beneficial for the network expansion of swollen superabsorbent composites. These effects helped improve water absorbency. 44 However, with a further increasein the neutralization degree, the water absorbency was reduced. Meanwhile, with an increase in the NaOH concentration, the ionic strength of the swollen solution was improved, which decreased the gel phase content; thus, water absorbency declined. Consequently, more sodium ions reacted with -COO−, which shielded -COO− present on the PAA chain and inhibited effective electrostatic repulsion owing to a decline in water absorbency. 45
Effect of CG content on water absorbency
Figure 6(e) shows the effect of CG content on the water absorbency in distilled water. The CG content clearly affected the water absorbency. Wang 46 spotted similar trends for superabsorbent composites with and without SC. The water absorbency exhibited a linear relationship with CG content when the CG content was less than 10 wt.%. When the CG content exceeded 10 wt.%, water absorbency was reduced. An excessive CG content in the superabsorbent composite network significantly inhibited water uptake. This may be because CG participates in the polymerization process and facilitates the formation of a three-dimensional network, which prevents the polymer chains from tangling and weakens the hydrogen bonding among hydrophilic groups including -COOH, -COO−, and -OH. 47 With a decrease in the degree of physical crosslinking, more water molecules could be accommodated by the network sites; thus, the water absorption was improved. However, with an increase in CG content, more crosslinking points were generated, which affected the graft polymerization. It is worth noting that the network voids may be filled by excessive CG content, which blocks the polymer network structure and decreases the water absorbency. 48
Effect of pH on water absorbency
The swelling properties of St-g-PAA/CG (10 wt.%) in different solutions with varying pH are shown in Figure 6(f). The swelling behavior of the samples was significantly influenced by the pH value. The water absorbency of the samples increased in the pH range of 2 to 7; however, it immediately decreased when the pH increased in the range of 7 to 13. Several -COOH groups were formed by the protonated carboxylate groups in an acidic solution. The ionization of -COOH is hindered by external H+ ions, which makes it form hydrogen bond links in the gel; this gel exhibits a contraction state.
Meanwhile, owing to the electrical neutrality of -COOH, the mutual repulsion between groups on the network chain is reduced; this hinders the stretching of the network structure, weakens the elasticity of the network structure, and thus decreases water absorbency.
When the pH value was exceeded 7, -COOH groups in the network structure were converted to -COO− groups. The Na+ cations from NaOH solutions form a counter-ion shielding effect on -COO−, which impeded the anion-anion electrostatic repulsion in the cross-linked network of superabsorbents. These interferences led to the reduction of the network and decreased water absorbency. A similar pH-dependent swelling behavior has been noted in another ionic superabsorbent system. 49
When the pH value reached 7, the hydrogen bond effect disappeared partially and a buffer action of -COO− and -COOH groups was observed. Consequently, a high electrostatic repulsion may increase the St-g-PAA/CG swelling and improve the water absorbency.
Environmental-responsiveness of the superabsorbent composite
Figure 7(a) shows the alternating swelling-deswelling behavior of St-g-PAA/CG (with 10 wt.% CG) in distilled water and 0.9 wt.% NaCl solution. The water-swollen St-g-PAA/CG can shrink and lose the absorbed water after being immersed into the NaCl solution. If the shrunken gel is placed in distilled water again, it absorbs the water and is reswollen. Furthermore, St-g-PAA/CG exhibits a stimuli-responsive property. The intra- and inter-molecular complexes formed between Na+ ions (from the NaCl solution) and carboxylate groups (from St-g-PAA/CG) could be the reason behind the significant decrease in water absorbency. (a) Environmental-responsiveness of St-g-PAA/CG (10 wt. %): swelling in distilled water and deswelling in NaCl (0.9 wt.%). (b) Reversibility of water absorption in distilled water.
After three cycles of swelling and deswelling, St-g-PAA/CG still maintains its water absorbency, thereby demonstrating that swelling is reversible. This environmental-responsiveness has been exhibited by other superabsorbents as well.50,51 The distinct environmental-responsiveness of the superabsorbent composite allows becoming a functional material for many applications, such as controlled systems and soil remediation.
Reversibility of water absorption
Several practical areas, such as architecture and controlled systems, need superabsorbent materials that can exhibit excellent repeated absorbency after they are used a few times to ensure cost-effectiveness. In this work, the water absorption reversibility of St-g-PAA/CG (10 wt.%) and the pure superabsorbent was investigated (Figure 7(b)). For the pure superabsorbent, the water absorbency was 187 g·g−1 after the fifth cycle. Meanwhile, the superabsorbent composite showed good reswelling capability and retained a water absorbency of 437 g·g−1even after the fifth cycle. The enhancement may be a result of incorporating CG, which could effectively prevent the collapse of the inter-network structure in the gel. These results implied that St-g-PAA/CG is a kind of reusable and recyclable water-absorbing material, which can be applied in various fields, such as agriculture, health nursing, and horticulture. 52
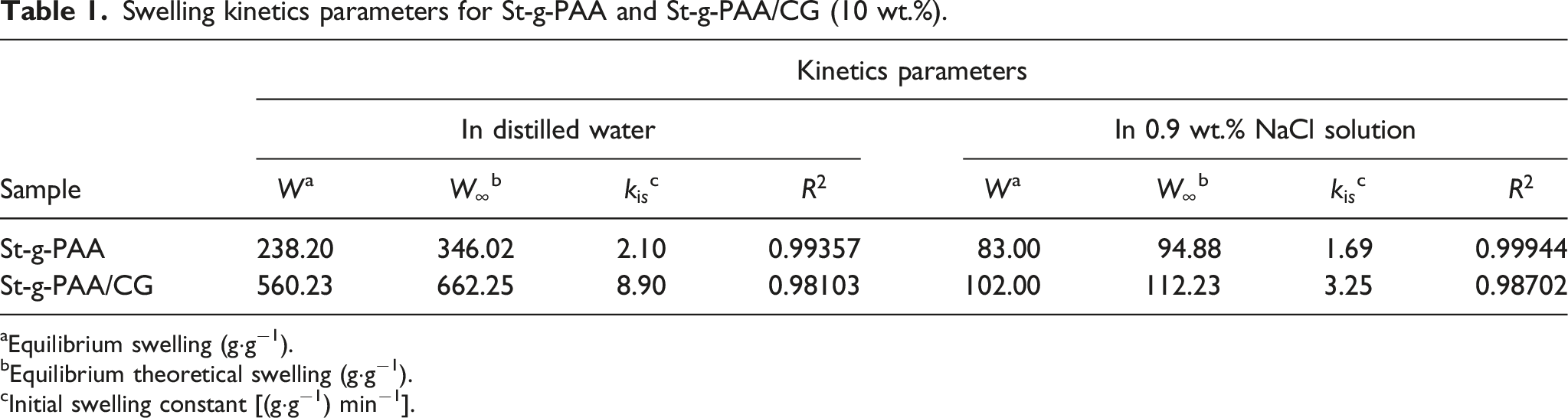
Swelling kinetics
The incorporation of CG into the polymeric system can also influence swelling kinetics. The swelling kinetics of the superabsorbent was determined using Scott’s second-order equation
53
The equation can also be written as (a) Swelling behavior of St-g-PAA and St-g-PAA/CG (10 wt.%) in distilled water; (b) Swelling kinetics parameters for St-g-PAA and St-g-PAA/CG (10 wt.%). aEquilibrium swelling (g·g−1). bEquilibrium theoretical swelling (g·g−1). cInitial swelling constant [(g·g−1) min−1].
The swelling properties of St-g-PAA/CG (10 wt.%) and St-g-PAA in distilled water (Figure 8(a)) and 0.9 wt.% NaCl solution (Figure 8(c)) improved during the initial 400 min, and then steadily deteriorated until the equilibrium swelling capacity was accomplished.
As shown in Figures 8(b) and 8(d), for the samples that swelled in distilled water and 0.9 wt.% NaCl solution, the fitting curves were linear (correlation coefficients R2 > 0.98); this indicates that the swelling kinetics followed the Schott’s second order kinetic model. In addition, the results of swelling kinetic parameters (including
Conclusions
To diminish environmental pollution and maximize the application potential of CG, a series of St-g-PAA/CG superabsorbent composites were synthesized through aqueous solution polymerization using starch, AA, and CG. Meanwhile, KPS and MBA were used as the initiator and cross-linker, respectively. The analyses conducted using FTIR, SEM, and EDS show that the superabsorbent composites have been successfully prepared, and that CG has been incorporated into the polymeric matrix of the composites. Meanwhile, the factors affecting water absorbency (starch content, initiator content, crosslinker content, CG content, and neutralization degree) were investigated. Adding CG improved the swelling capabilities of St-g-PAA/CG (the maximum water absorbency is 560 g·g−1 and 102 g·g−1 in distilled water and 0.9% NaCl solution, respectively, when 10 wt.% CG is incorporated). The water absorbency of St-g-PAA/CG is affected by pH, which allows them to be used in different applications. The swelling kinetics of St-g-PAA/CG was in accordance with the Scott’s kinetic model. The superabsorbent composite displayed notable environmental-responsiveness when the swelling medium was changed from distilled water to 0.9 wt.% NaCl solution, while showing excellent water absorbency (437 g·g−1) in distilled water even after the fifth cycle of swelling and reswelling. This study not only achieved high value-added utilization of waste CG, but also effectively reduced production cost of superabsorbent composites, that exhibit excellent water absorbing performance and water absorption reversibility. The useful characteristics of the novel St-g-PAA/CG superabsorbent composite can be employed in different fields, such as horticulture, agriculture, health care, and controlled drug delivery.
Footnotes
Acknowledgements
This research was financially supported by Yulin National High Tech Industrial Development Zone Project of China (Grant No. CXY-2021-24); Yulin Municipal Science and Technology Bureau Project, P.R. China (Grant No. CXY-2021-101-03); Technological Innovation Guidance of Shaanxi Province, P.R. China (Grant No. 2020QFY05-03); Engineering and Technology Research Center for the reuse of LAN charcoal wastewater in Yulin, P. R. China (Grant No. CXY-2021-142).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Engineering and Technology Research Center for the reuse of LAN charcoal wastewater in Yulin, P. R. China (CXY-2021-142), Yulin Municipal Science and Technology Bureau Project of China (Grant No. CXY-2021-101-03), Technological Innovation Guidance of Shaanxi Province, P.R. China (2020QFY05-03), Yulin National High Tech Industrial Development Zone Project of China (Grant No. CXY-2021-24).




