Abstract
Starch-g-poly(acrylic acid)/organo-mordenite (St-g-PAA/OMOR) superabsorbent composites were prepared via inverse suspension polymerizationutilizing potassium persulfate as an initiator and N, N’-methylene bisacrylamide as a crosslinker. Different dosages of cetyltrimethylammonium bromide were used to obtain organo-mordenite (OMOR) samples with different organification degrees. The effects of the organification degree of OMOR on the water absorbency, swelling behavior, and reswelling properties of St-g-PAA/OMOR were studied. The results from both FTIR and XRD analyses indicate that the Si–OH group of OMOR was involved in the formation of the St-g-PAA/OMOR composite. SEM analyses showed that the organification degree of OMOR influenced the morphology of the St-g-PAA/OMOR composite. The water absorption absorbency, swelling rate, and reswelling capability of the St-g-PAA/OMOR composite prepared using the OMOR with the optimal organification degree were superior to those of the St-g-PAA/mordenite composite. The swelling behaviors of the St-g-PAA/OMOR composites, which were measured in distilled water, obeyed Schott’s second-order kinetics model. The St-g-PAA/OMOR composite prepared using the OMOR with the 8 wt.% organification degree had the highest water absorption capacity and the highest swelling rate. Moreover, after five swelling–shrinking cycles, the composite retained its excellent water absorption capacity.
Introduction
Superabsorbent, cross-linked hydrophilic polymers, can absorb water, saline solution, or other solutions. Due to its unique three-dimensional network structure and large functional groups, superabsorbent has been an exciting sort of functional candidate for various kinds of applications, such as horticulture, 1 agriculture, 2 effluent treatment, 3 and other applications.4,5 However, the superabsorbent originated from petroleum products are costly and poorly degradable, which limits the applications of the polymer in many fields. Currently, it is imminent to research superabsorbents based on eco-friendly materials. The new material from naturally available raw materials is much more demanding. The research and development of superabsorbent composites with both inorganic and organic material have attracted impressive attention. For example, researchers have prepared superabsorbent composites based on natural polysaccharides (starch, chitosan, chitin, and cellulose) and inorganic materials (clay and zeolites).6–9
Starch is an abundant polymer that has excellent biodegradability and biocompatibility. Being derived from plants, starch consists of 10–20% linear amylose macromolecules and 80–90% with branched amylopectin macromolecules, respectively. The position of the branch on the primary substrate is seemly to locate on the C6 forming–D (1,6)-glycosidic linkages, which generate high solubility, excellent chemical reactivity, and strong hydrophilicity. 10 Hence, starch is an economical and attractive feedstock to produce superabsorbent.11,12
Due to their low manufacturing costs and improved performance, superabsorbent composites based on inorganic clay have received an extensive interest.13,14 Mordenite (MOR) is a zeolite that has a robust three-dimensional network. It consists of silica and alumina tetrahedral units by shared oxygen atoms (T-O) linkage. Due to the exchangeable cations and reactive OH groups on MOR’s structural sites and surface, the graft copolymerization makes possible by grafting hydrophilic Si-OH bond on hydrophilic vinyl monomers. 15 However, because they are small, MOR particles easily aggregate because of their high specific surface energy. Therefore, without effective pretreatment, MOR particles may disperse homegeneously, which causes their poordispersion in the polymeric composites and compromises the properties of the composites. Consequently, it is necessary to treat MOR effectively before incorporating it into polymeric matrix.
Assisted by the isomorphic substitution of the cations in its lattice, MOR has a cation-exchange capacity that allows cationic surfactants, such as cetyltrimethylammonium bromide (CTAB) to improve its dispersibility.16,17 However, due to its the long chain, CTAB is too large to enter the pores of MOR. Therefore, CTAB absorbs on the external surface of MOR particles. When the surface of MOR particles is progressively modified by CTAB, the organification degree of the resulting organo-mordenite (OMOR) particles is expected to affect the properties of the OMOR-based polymeric composites. Wang et al. 18 reported that the organification degree of attapulgite (APT) influences the swelling behaviors of an APT-based absorbent polymer: the 8.02% organification degree of APT maximizes the water absorption capacity of the absorbent polymer. Zhang et al. 19 prepared a novel superabsorbent PAA-AM/SH/organo-APT and found that the organification degree of APT influences the swelling rates of the composites in different saline solutions.
Our previous study focused on optimizing the synthesis of the starch-g-poly(acylic acid)/organo-mordenite(St-g-PAA/OMOR) superabsorbent composite and the investigating the water absorption and swelling properties of the superabsorbent composite. 20 In this study, the influence of the organification degree of organo-mordenite (OMOR) on the swelling behavior of the St-g-PAA/OMOR superabsorbent composite was investigated.
Materials and methods
Materials
Corn starch ((C6H10O5)n) was obtained from Xiang Zailai Co. Ltd. (Xi’an, China). MOR whose cation-exchange capacity (CEC) of 154 mmol/100 g was obtained from NanKai University Catalyst Company (Tianjin, China). The MOR was treated with sodium hydroxide solution (NaOH) for 24 h and then was dried at 100°C for 12 h. Cetyltrimethylammonium bromide (CTAB) was purchased by Guangfu fine Chemical Research Institute (Tianjin, China). Potassium persulfate (KPS) as reaction initiator and sodium hydroxide (NaOH) were obtained from Sheng’ao Chemical Reagent Co., Ltd (Tianjin, China). N, N’-methylene bisacrylamide (MBA) as a crosslinker purchased from Kermel Chemical Reagent Factory (Tianjin, China). Acrylic acid (AA) as a reactive monomer was obtained from Da Mao Chemical Reagent Factory (Tianjin, China). Polyglyceryl fatty acid ester (PGFE) as suspending stabilizer was employed from the Da He Foods and Technologies Co., Ltd. (Zhengzhou, China). n-Hexane was obtained by Tian Li Chemical Reagent Factory (Tianjin, China). Other chemicals were overall analytical grade.
Organo-mordenite (OMOR)
Five dosages of CTAB (50%, 100%, 150%, 175%, and 200% of the CEC value of MOR) were prepared to produce OMOR samples with various organification degrees. Each of the five dosaes of CTAB was added to 50 ml of distilled water. Then, 10.0 g of MOR was suspended in each of the five CTAB solutions. Each suspension was ultrasonically treated on a 70°C water bath for 60 min to obtain an OMOR sample with a particular organification degree. The OMOR sample was filtered and then washed with aqueous ethanol to remove the bromide ion (Br-). Afterwards, the OMOR was dried in a 70°C oven to constant weight before being through a 30-mesh sieve. The OMOR samples prepared using the CTAB dosages that were 50%, 100%, 150%, 175%, and 200% of the CEC value of MOR referred to as OMOR1#, OMOR2#, OMOR 3#, OMOR 4#, and OMOR 5#, respectively.
The organification degree of MOR was expressed in terms of the weight percent of the CTAB-derived organic cations on the OMOR surface and was measured according to equations (1) and (2).
21
Synthesis of St-g-PAA/OMOR superabsorbent composite
The serial superabsorbent composites based on starch (St), AA and OMOR with various organification degrees were prepared via inverse suspension polymerization according to the procedure reported in our previous article. 22
In a 500-ml four-neck flask connected to a condenser, a stirrer, a thermometer, and a nitrogen source, 4.8 g of starch was solubilized at 95°C in 50 mL of distilled water for 30 min. Then, the temperature of the starch solution was reduced to 35–40°C, and n-hexane (300 mL) and PGFE were added to the solution. The resulting slurry was stirred for 15 min, and KPS was added to it. The solution was stirred for 15 min. Then, partially neutralized AA and 10 wt.% of each of OMOR samples with different organification degrees (0%, 4%, 8%, 19%, 20%, and 25%) were added to the solution. Afterwards, MBA (5 g/L) was added to the solution, and the polymerization reaction was conducted under mechanical stirring (400–600 rpm) for 3 h on a 70°C water bath. The reaction mixture had been washed several times with distilled water and ethanol to remove residual reactants before being dried to constant weight in a 70°C oven. The flowchart for synthesis of St-g-PAA/OMOR superabsorbent composites is illustrated as Scheme 1. Preparation flowchart of St-g-PAA/OMOR superabsorbent composites.
Assessment of water absorbency and swelling rate for the superabsorbents
The water absorbency was decided at normal temperature. 0.1 g samples were submerged in an excess amount of distilled water for 12 h to attain the swelling equilibrium. Residual water was removed from the swollen composit by passing the composite through a 200-mesh sieve. The water absorbency of the St-g-PAA/OMOR was calculated according to equation (3):
The kinetics of the swelling of St-g-PAA/OMOR superabsorbent composites in distilled water was assessed according to a e method described in the previous report. 22
Characterizations
The fourier transform infrared (FTIR) of samples were obtained by using a Bruker Tensor 27 FTIR spectrometer. The X-ray diffraction patterns (XRD) of samples were obtained by using a Bruker D8 XRD pectrometer and a Cu Kα radiation source,. The surface morphology and structure of samples were characterized using a Zeiss Sigma 300 scanning electron microscope.
Results and discussion
FTIR analysis
The FTIR spectra of MOR and OMOR are illustrated in Figure 1. The absorption bands at 3488, 1631, 1236, 824, 655, and 462 cm−1 are prominent in the FTIR spectrum of MOR (Figure 1(a)). The absorption bands are attributed to the stretching vibration of the –OH bond, bending vibration of the –OH bond, stretching vibration of the Si-O(Al) bond, symmetric stretching of the Si-O-Si bond, and bending vibration of the (Si-O- Al), respectively.
23
The FTIR spectra of: (a) MOR, (b) OMOR (1#), (c) OMOR (2#), (d), and (f) OMOR (5#).
Compared with the FTIR spectrum of MOR, the FTIR spectra of OMOR samples (Figures 1(b)-(f)) new absorption bands at 2922 cm−1 and 2848 cm−1 (asymmetric and symmetric stretching vibration of C-H bonds). The appearance of the new bands suggests the replacement of the exchangeable cations on the MOR surface with the organic cations of CTAB 24 and the position of these peaks might be related to the increase in the disordered tail (hexadecyl) group –CH2 of the surfactant molecules caused by nonsaturation of zeolite’s surface by CTAB. 25
The infrared spectra of St-g-PAA/MOR and St-g-PAA/OMORs are shown in Figure 2. The absorption peaks of starch at 1466 cm−1 (bending vibration of C-OH) disappeared, and the new-found peaks at 1716 cm−1, 1566 cm−1, 1448 cm−1,1412 cm−1 (asymmetric and symmetric stretching vibration of -COO-)
26
existed in Figure 2. This result proved that polyacrylic acid chains had been grafted on the starch backbone. The characteristic absorption peaks of MOR at 650 cm−1 and 510 cm−1 (internal vibration of Si-O symmetric stretching and internal vibration of Si-O) also appeared after the polymerization process. 27,28 IR spectra of: (a) St-g-PAA/MOR (10 wt.% MOR) and (b-d) St-g-PAA/OMOR (10 wt.% OMOR) composites with different organification degree of OMOR (1#, 2# and 5#).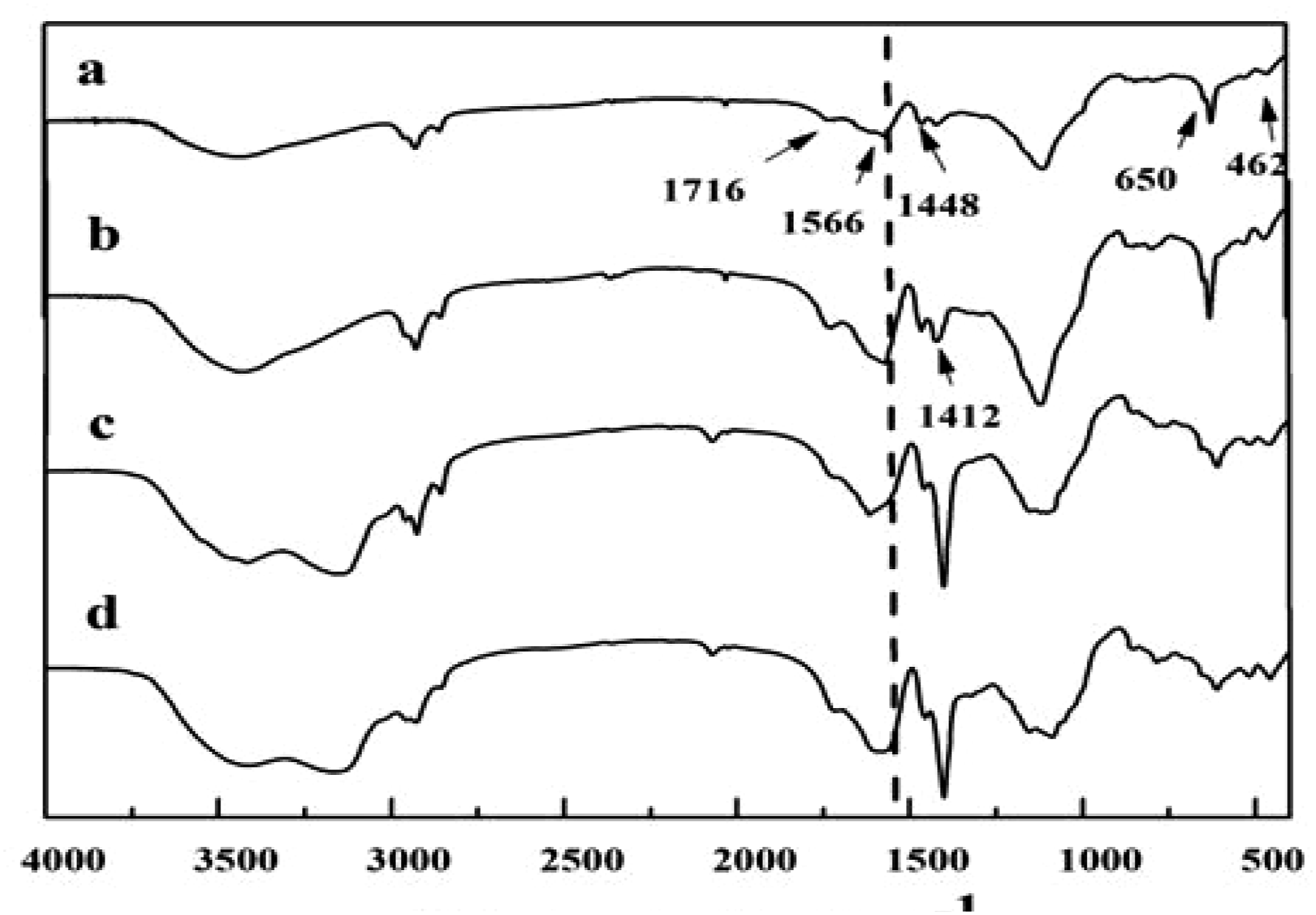
Additionally, the Si-OH deformation at 998 became weak in the spectra of St-g-PAA/MOR and St-g-PAA/OMORs. This result revealed that MOR and OMOR also took part in the reaction by their Si-OH groups. 29
What’s more, compared with St-g-PAA/MOR, the absorption peaks of St-g-PAA/MORs at 1568 cm−1 (Figure 2(b)), 1573 cm−1 (Figure 2(c)) and 1579 cm−1 (Figure 2(d))) attributed to –COO- shifts to higher wavenumber with the organification degree of MOR raised. This result may result from the effect of the organification degree of MOR on the interaction between AA and MOR.
XRD analysis
The typical diffraction peaks of MOR appeared at 6.5°, 8.6°, 9.7°, 13.5°, 15.1°, 21.5°, and 24.6°(Figure 3(a)). The diffraction peaks corresponding to the Miller indices of [110], [020], [200], [111], [310], [330], and [420],respectively.
30
After the organification of MOR, the intensities ofthe typical diffraction peaks increased (Figure 3(b)), which indicates that organificiation of MOR increasethe crystallinity of MOR compared to that of MOR. In addition, in the XRD spectrum of OMOR (Figure 3(b)), there were no the diffraction peakscorresponding to CTAB, which indicates that CTAB scatters well on the surface of OMOR. The results of XRD and FTIR analyses suggested that CTAB was absorbed on the surface of MOR through cation-exchange reactions. The adsorption of CTAB has no adverse influence on the crystallinity of the resulting OMOR. The XRD powder patterns of (a) MOR, (b) OMOR (2#), (c) St-g-PAA/MOR (10 wt.% MOR) and (d) St-g-PAA/OMOR (2#,10 wt.% OMOR).
After polymerization with starch and AA, the typical diffraction peaks of MOR are not present in the XRD powder pattern of St-g-PAA/MOR (Figure 3(c)). This may be because MOR is embeded in the polymeric network of the composite. On the other hand, as shown in Figure 3(d), some of the typical diffraction peaks of the MOR, i.e.,the peaks at 9.7°, 13.5°, 21.5°, and 24.6° are present in the XRD powder pattern of the St-g-PAA/MOR composite indicates that the interaction between AA and OMOR occurs mainly on the surface of OMOR.
Effect of MOR degree of mordentie on water absorbency of the superabsorbent composites
Variation of water absorbency for St-g-PAA/MOR (10 wt.% MOR) and St-g-PAA/OMOR (10 wt.% OMOR) with different organificaiton degree.


The effcet of organification degree on the superabsorbent composites.
However, when the organification degree of OMOR increases above 8 wt.%, the water absorbency of the St-g-PAA/OMOR composite decreases. The higher organification degree of OMOR, the higher the amount of the negative long alkyl chain of CTAB that is absorbed on the OMOR surface. This may leas to excess hydrophilic groups that promote the termination of the polymerization reaction or shrinkage of the polymeric network and impede the expansion for the network.This result are in agreement with the ones obtained by Wang 32 and Zhang. 33
SEM analysis
To clarify the roles of MOR and OMOR in the polymeric networks of samples, the morphology of MOR, OMOR, St-g-PAA/MOR, and St-g-PAA/OMOR was investigated by SEM (Figure 5). As shown in Figure 5(a), MOR particles are and have a scaly and granular surface. In comparison with MOR particles, OMOR particles dispersed favorably (Figure 5(b)).However, there are no noticeable differences between the SEM micrograph of MOR and that of OMOR indicates that the organification of MOR does not alter the structure of MOR with respect to the struture of MOR.This results are in agreement with the results of XRD analysis. SEM images of: (a) MOR, (b) OMOR (2#), (10 wt.% MOR), St-g-PAA/MOR (10 wt.% MOR) and (b-d) St-g-PAA/OMOR (10 wt.% OMOR) composites with different organification degree of OMOR (1#, 2# and 5#).
Figure 5(c) shows that the St-g-PAA/MOR has a dense and corrugated surface. On the other hand, as shown in Figures 5(d)-(f), due to the introduction of OMOR, the surface of the St-g-PAA/OMOR composite is more ordered than that of the St-g-PAA/MOR. Furrows, cavities, analogous pores and gullies structures are present on the surface of the St-g-PAA/OMOR composite. The structural components allow the interaction between the hydrophilic groups of the composite and water from external environments. 34 Thus, they are benefical to the permeation of water into the composite. However, when the organification degree of OMOR is too high, the surface the St-g-PAA/OMOR becomes compact and the number of pores decreas, both of which slowe the permeation of water into the polymeric network of the composite.
Furthermore, compared with OMOR (Figure 5(b)), most of the signal MOR particles cannot be observed on the St-g-PAA/OMOR (Figure 5(d)-(f)), indicating that a good dispersion was achieved during the copolymerization. The analysis of the SEM explained the enhancement of water absorption given by incorporating the OMOR into the St-g-PAA macromolecule network. Increasing the organification degree of OMOR, the micrographs of the samples were changed. This fact was confirmed the organification degree could affect the structure of the composite, and then could change the water absorbency property.
The influences of the organification of MOR on swelling behavior for the superabsorbent composites
Figure 6(a) shows the influence of the organification degree of OMOR on the swelling rate of the St-g-PAA/OMOR. The characterizations of the swelling kinetics for the samples were quite the same. The curves become steeper, accompanied by the increase of the significant degree. Firstly, the speed of water uptake increasd distinctly, which is abided by a slower rate up to equilibrium is achieved. The degree of swelling raises shapely within the first 100 min and 200 min for St-g-PAA/MOR and St-g-PAA/OMORs, after immersion. Based on the superabsorbent, the swelling degree elevates smoothly when equilibrium is achieved. As reported, the swelling rate of superabsorbent dominantly depends on not only surface area and particle size, but also the level of the penetration of water into the polymeric network through diffusion and capillarity.
35
(a) Swelling rate in distilled water for St-g-PAA/MOR (10 wt.% MOR) and St-g-PAA/OMOR (10 wt.% OMOR) composites with different organification degree of OMOR (1#, 2# and 5#) and (b) Wt vs. t plot for St-g-PAA/MOR (10 wt.% MOR) and St-g-PAA/OMOR (10 wt.% OMOR) composites with different organification degree of OMOR (1#, 2# and 5#).
Schott’s pseudo-second-order swelling kinetics model was used to analyze kinetic behaviors.
36
On the ground of the experimental data, the fitting curves of
Swelling kinetics for St-g-PAA/MOR (10 wt.% MOR) and St-g-PAA/OMOR (10 wt.% OMOR)with different organificaiton degree.

aEquilibrium swelling (gwater·gabsorbent−1).
bEquilibrium theoretical swelling (gwater·gabsorbent−1).
cInitial swelling constant [(gwater·gabsorbent−1)·min−1].
Overall, 8 wt.% was the optimum organificant degree of OMOR for improving the swelling rate of the St-g-PAA/OMOR composite. This meant that polymer networks for St-g-PAA/OMOR (1#), St-g-PAA/OMOR (5#), and St-g-PAA/MOR require relatively much more time than that of St-g-PAA/OMOR (2#) to reach the penetration of water through diffusion and capillarity.
At the optimum organification degree of OMOR capillaries were noticeable(Figure 5(e)). The capillaries accelerate the penetration of water molecules into the polymeric network. However, the excessive organification degree made so many CTAB on the surface of MOR during the polymerization procession, which generates extreme crosslink points. It decreased the network voids resulted in the increase of the degree of physical crosslinking. On the other hand, the extreme organification degree may lead to more hydrophobic regions hindering the samples from absorbing and uptaking water molecules. Furthermore, related to the SEM analysis that increased the organificaiton degree, the surface of the samples became less coarse, and many porous decreased where could influence the capillarity of the sample and then slow down the penetration of water molecules into the polymeric network.18,37
Effect of organification degree of MOR on the reversibility of water absorption
Figure 7 shows the reversibility of the St-g-PAA/MOR and St-g-PAA/OMOR superabsorbent composites. The water absorbency is 70 g·g−1 after the fifth repetitive cycle for the St-g-PAA/MOR. On other hand, the St-g-PAA/OMORs composites showe good reswelling capability and retained a water absorbency of 470 g·g−1 (St-g-PAA/OMOR with 10 wt.% OMOR 2#),400 g·g−1 (St-g-PAA/OMOR with 10 wt.% OMOR 1#), and 156 g·g−1 (St-g-PAA/OMOR with 10 wt.% OMOR 5#) even after the fifth cycle. This indicates that the existence of OMOR is also favorable to improving the reversibility of water absorption. Additionally, the St-g-PAA/OMOR (10 wt.% OMOR 2#) superabsorbent composite achieves the highest water absorbency among all of the samples after the fifth reswelling. This indicated that optimum organification degree is demands for improving the reswelling capability of the St-g-PAA/OMOR superabsorbent composite to a higher degree. Due to its reswelling capability, the St-g-PAA/OMOR is particularlysuitable for agricultural applications.
38
Reversibility of water absorption for St-g-PAA/MOR (10 wt.% MOR) and St-g-PAA/OMOR (10 wt.% OMOR) composites with different organification degree of OMOR (1#, 2# and 5#) in distilled water.
Conclusions
The effects of the organification degree of OMOR on the water absorption, swelling, and reswelling properties of the St-g-PAA/OMOR superabsorbent composite were investigated. OMOR improved the water absorption capacity of the St-g-PAA/OMOR composite. Moreover, the swelling rate and reswelling capability of the St-g-PAA/OMOR composite were superior to those of the St-g-PAA/MOR composite. The OMOR with the optimal organification degree improved the water absorption capacity, swelling rate, and reswelling capability of the St-g-PAA/OMOR composite: the highest water absorbency, the highest swelling rate, and the highest reswelling capability of the composite were obtained when the organification degree of OMOR was 8 wt.%.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was financially subsidized by National College Student Innovation and Entrepreneurship Training Program of China (Grant No. 202111395024); Yulin Municipal Science and Technology Project of China (Grant No.2019-82-1); Yulin National High Tech Industrial Development Zone Project of China (Grant No.CXY-2021-24); Startup Fund for Distinguished Scholars of Yulin University of China (Grant No. 18GK20).



