Abstract
A series of PVC composites were produced using an in-situ suspension polymerization method with an optimal amount of 5 wt% of MgAl(NO3) layered double hydroxide (LDH) or LDH-MPTMS, which is MPTMS-intercalated Mg-Al LDH. The physical, mechanical, and thermal properties of the composite samples were compared to pure PVC. Results from the Brabender® plastograph showed that the PVC grains produced with LDH-MPTMS had a longer thermal stability time and shorter fusion time. The addition of LDH-MPTMS nanosheets increased the gelation degree of PVC particles, resulting in a lower temperature/time requirement for processing. The thermal stability of the composite material was confirmed through a standard dehydrochlorination test, which demonstrated a 40% improvement in dehydrochlorination rate compared to pure PVC. This improvement was 12% higher than that observed in the PVC/LDH composite. TGA curves indicated a significant increase in the 5 and 50% weight loss temperatures of PVC resins with the addition of 5wt% LDH or LDH-MPTMS, with an approximate growth of 11°C. The glassy state storage modulus and Tg of the PVC/LDH-MPTMS composite were higher than those of pure PVC and the PVC/LDH composite. Mechanical analysis revealed that the PVC/LDH-MPTMS composites exhibited greater stiffness and toughness, as well as significantly higher Charpy notched impact strength, tensile strength, and Young’s modulus compared to both the PVC/LDH composite and pure PVC.
Keywords
Introduction
Polyvinyl Chloride (PVC) is a common thermoplastic polymer widely applied in different applications such as pipes, window and door profiles, wire, medical devices, and cable insulation. After polyethylene (PE) and polypropylene (PP), PVC is the third most widely used thermoplastic in the world. 1 Compared to other conventional plastics such as polystyrene (PS) and PE, PVC has disadvantages such as sightly inferior thermal stability and low processability. 2 Additives such as fillers, lubricants, softeners, thermal stabilizers, and copolymerization with other monomers have been used to improve the properties of the mentioned plastic.3–5 One efficient method to improve this plastic’s mechanical and thermal properties is to compound it with mineral fillers.6–9
Among nanoparticles, LDHs are one of the best fillers, widely and significantly used to increase mechanical properties in polymer composites.10, 11 Although these nanoparticles have shown a great tendency to form micro-scale grains, the homogeneous dispersion of these materials in the polymer field has always been challenging; Still, surface modification of these materials can overcome this problem. The insertion of nanostructured nanomaterials into polymer matrices to form composite polymers can be carried out in one or more of the following three ways; solution intercalation, melt intercalation, and in-situ interactive polymerization. 12 Due to the possibility of wetting LDH layers by monomers, in-situ polymerization can bring a more homogeneous dispersion and an increase in properties compared to the above two methods. The literature review indicates that an exfoliating polymer/organically modified LDH-composite can be prepared by in-situ polymerization of some monomers, such as vinyl and lactides. 13 For this purpose, the nanoparticles are mixed with monomer to obtain a homogeneous dispersion and then polymerized by the in-situ suspension polymerization method.14, 15
Bao et al. developed and investigated composites of PVC/LDH by in-situ polymerization of the monomer of vinyl chloride (VCM), in which the particles of the LDH were intercalated by dodecyl sulfate. They found that the storage modulus of glass area and Tg of PVC composites are larger than pure PVC. Their studies showed that the incorporation of the LDH nanoparticles in the PVC matrix boosted the stiffness and toughness of the matrix. Also, Young’s modulus, tensile strength, and Charpy notched impact strength of this method are more excellent than pure PVC, and PVC composites made by the melt compounding method are prepared. 16 The incorporation of LDH (layered double hydroxide) nanoparticles in the PVC (polyvinyl chloride) matrix can boost both the stiffness and toughness of the matrix simultaneously due to the following reasons: i) Improved dispersion: The LDH nanoparticles have a high aspect ratio and a large surface area, which allows them to disperse uniformly in the PVC matrix. This improves the interfacial interaction between the nanoparticles and the polymer matrix, leading to enhanced mechanical properties. ii) Reinforcement effect: The LDH nanoparticles act as reinforcing agents in the PVC matrix. Due to their high aspect ratio, they provide a large number of nucleation sites for crystallization of the polymer matrix. This results in the formation of a more ordered structure, which increases the stiffness of the matrix. Iii) Toughening effect: The LDH nanoparticles also act as toughening agents in the PVC matrix. They absorb energy during deformation and prevent crack propagation by creating a tortuous path for the crack to follow. This results in an increase in toughness of the matrix. iv) Synergistic effect: The combination of reinforcement and toughening effects of the LDH nanoparticles results in a synergistic effect on the mechanical properties of the PVC matrix. The stiffness and toughness of the matrix are improved simultaneously, leading to an overall improvement in its mechanical performance. Therefore, the incorporation of LDH nanoparticles in the PVC matrix can boost both the stiffness and toughness of the matrix simultaneously, making it a promising material for various applications that require high mechanical strength and durability.
Moreover, LDHs materials can be introduced as thermal stabilizers used in PVC resins. According to a recent study, it has been proven that the chemical composition of LDH thermal stabilizers and their type of dispersion in the polymer matrix significantly affect the performance of the mentioned composite. 17
It has also been proven that the presence of nanoparticles mentioned above increases the activation energy of PVC degradation, increasing the stability of polymer chains against dehydrochlorination. 18
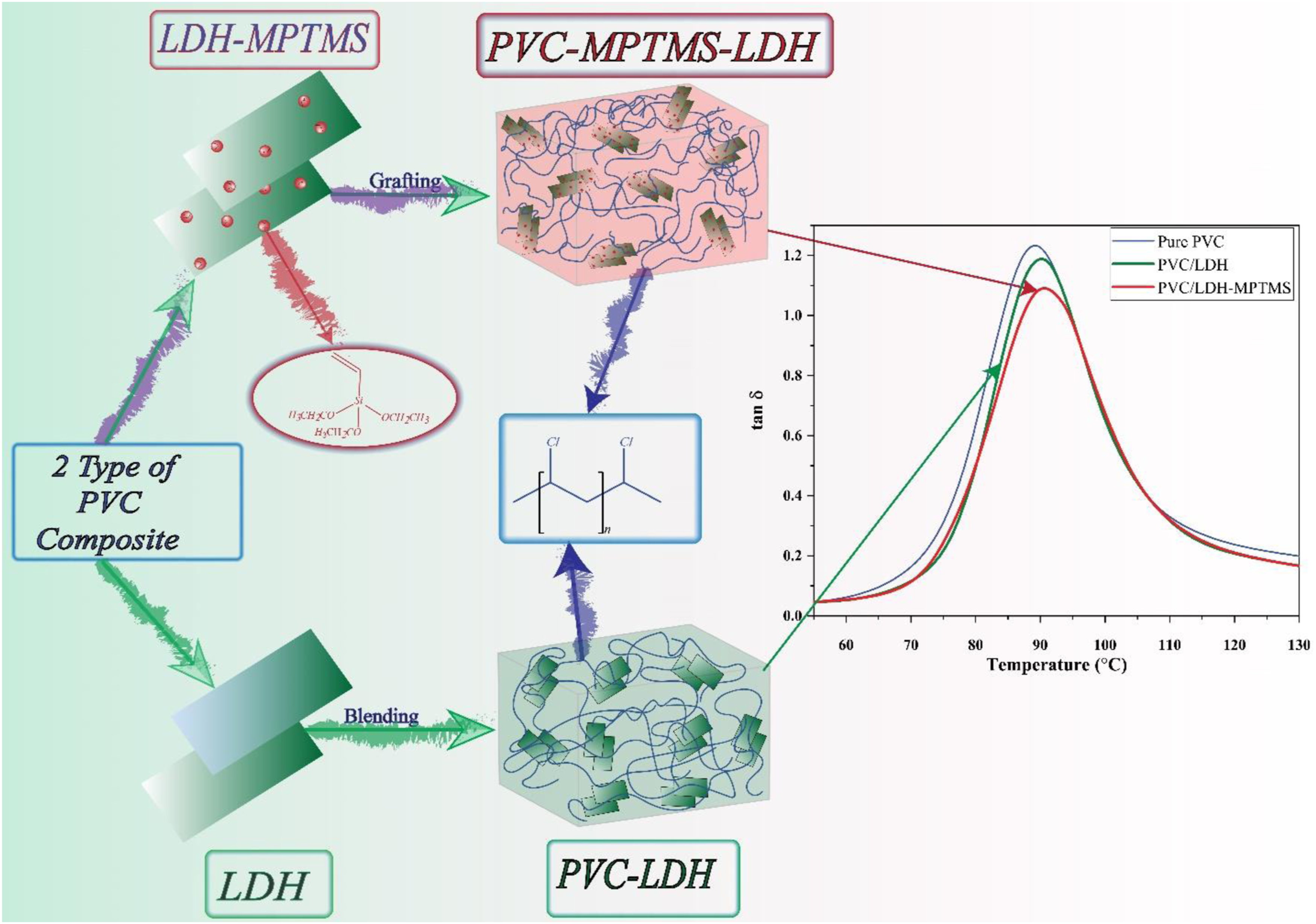
In this work, PVC composites were synthesized by VCM in-situ suspension polymerization with 5 wt% (based on the total VCM initial charge) 3-(trimethoxysilyl) propyl methacrylate (MPTMS)-intercalated Mg-Al-NO3 layered double hydroxide (PVC/LDH-MPTMS) or Mg-Al-NO3 layered double hydroxide (PVC/LDH). Their morphological, thermal, and mechanical properties were compared to pure PVC. It should be noted that the polymerization procedure and how to achieve LDH contents of 5 wt% were presented in detail in earlier work. 19 In our previous work, the surface characteristic of LDH particles before and after the modification with MPTMS was considered, and in addition, the content of particles on molecular properties, including K-value, molecular weight distribution (MWD), and particle morphology such as particle internal structure, primary particles size, and particle size distribution were investigated on PVC grains. The present work studied and evaluated the impact of LDH nanoparticles on thermal stability and fusion. Previous work found that the MWD and K-value of final PVC grains were insensitive to the MPTMS modification of LDH particles, while the effect on morphological characteristics was significant. Therefore, the impact of LDH and its modification on the behavior of dehydrochlorination and fusion is significant, and, interestingly, both nanoparticles can affect the PVC thermal stability. Using the Brabender® plastograph method and differential scanning calorimetry (DSC), the degree of fusion of the prepared samples was compared. 19
Experimental
Materials and methods
PVC compounds recipe.

1parts by weight per hundred parts of resin
2containing or not containing nanoparticles
Fusion Characterization
A non-intermeshing rotors type of Brabender® Plastirecorder EC instrument with an electrically heated mixing head (W 50 EHT mixer) and a volume capacity of 50-cm3 was utilized for assessment of Fusion behavior and dynamic thermal stability. Three different types of PVC with the same composition of material were processed at 197°C and 60°r/min for rotor speed conditions based on ASTM D2538-02.
To determine the degree fusion of the samples, the sample was mixed for 4 min using a Brabender® plastograph and were evaluated by a Netzsh 200 F3 automatic cooling DSC machine. The test atmosphere was chosen N2 and was performed from ambient temperature to 250°C with 10°C/min.
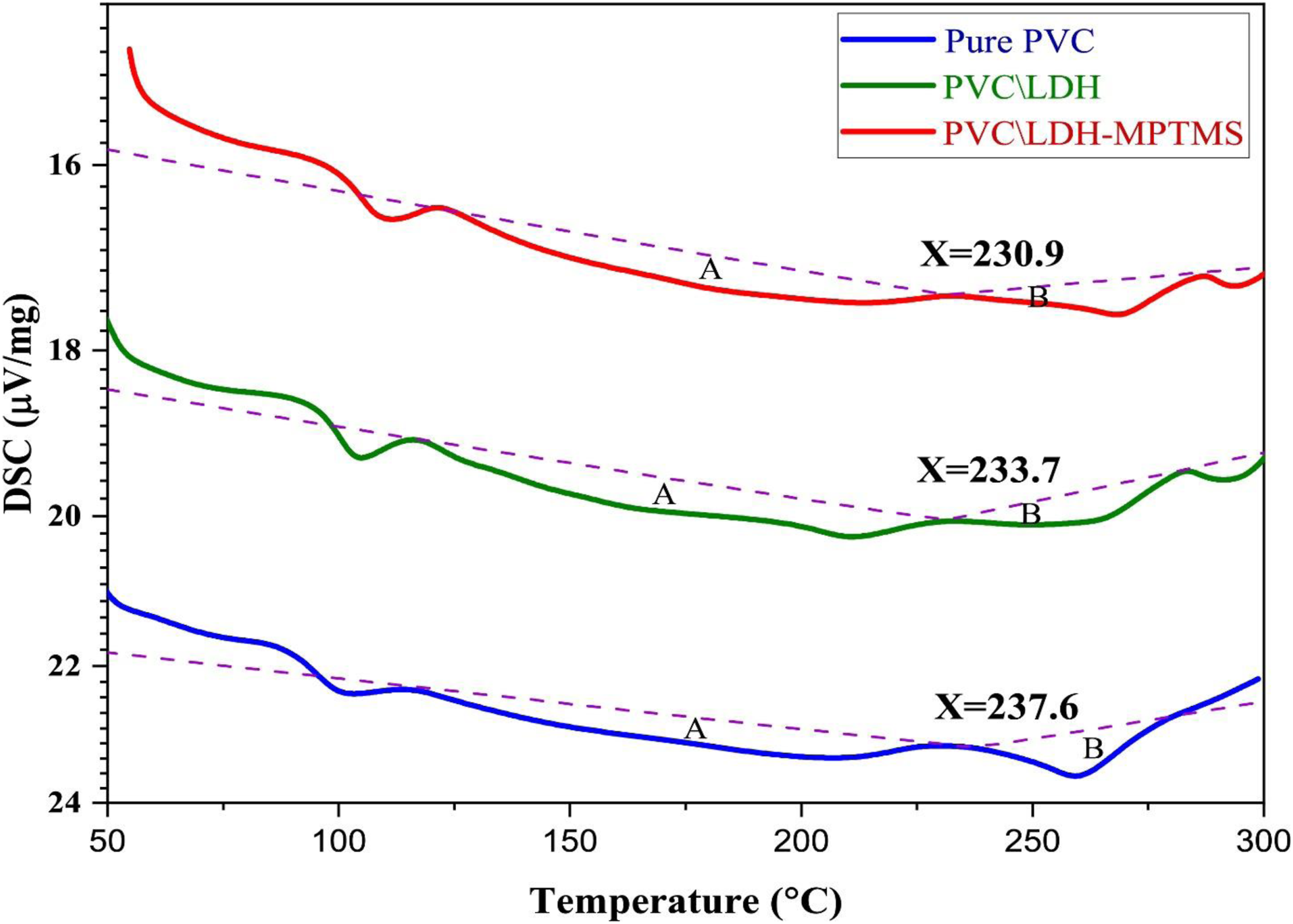
A typical DSC curve for the PVC gelation analyses is pictured in Figure 1. The gelation degree of PVC was computed from the following equation (1).
20
DSC thermogram of PVC samples: X is proposed to be the processing temperature.
The data sets calculated from DSC curves.
Dehydrochlorination Rate
Using the conductimetric measurement technique with ISO 182-3 standard, the dechlorination rate of PVC composites was measured at 180°C under N2 flow. During the decomposition of PVC samples, hydrochloric acid was swept into DI water and the conductivity changes were monitored continuously through a pH/conductometer 914 (Metrohm).
The time required to change the conductivity (∆ts) to the value of 50 μScm−1, which is related to the weight loss of 0.05 wt% by weight of HCl, was considered as the stability time. 21
After that, the rate of dechlorination was obtained according to the following equation 2
Dynamic mechanical analysis (DMA)
DMA was studied with a dynamic mechanical analyzer (DMTA-PL, Polymer Lab) with the oscillation amplitude and frequency set to 0.15 mm and 10 Hz, respectively. The tests were carried out with 1 °C/min heating rate and 0–130°C temperature range.
Thermogravimetry analysis (TGA)
METTLER T 1 OLEDO analyzer (model SSC/5200) with helium environment was used to explore the thermal degradation of PVC samples, and the heating rate and temperature range of the test was 5°C/min and 0–600°C, respectively.
Mechanical characterization
A study of the tensile test was performed using a universal tensile tester (model 4206, Instron) with 10 mm/min test speed. Charpy notched impact samples were investigated by a Ceast pendulum impact tester, and the dimensions of the samples were 65 × 13 × 3.2 mm3. A standard Charpy-V specimen, with 0.25 mm notch tip radius was prepared by a CEAST notch opener. The temperature of all mechanical tests was 23–25°C (room temperature) and the test of each sample was repeated 5 times.
Results and discussion
Morphological effect
Figure 2 shows the torque-time behaviour obtained from Brabender internal mixer for three samples. It is obvious that both LDH-contained PVC samples (PVC/LDH and PVC/LDH-MPTMS) have a longer stability time (the plateau region) and so have higher thermal stability than pure PVC.
Processing characteristics and microstructure of final PVC samples.

Pure PVC typically has larger particle sizes compared to nanocomposites because the addition of nanoparticles to the PVC matrix can act as nucleation sites for the formation of smaller particles. This is due to the fact that the presence of nanoparticles can provide a template for the growth of smaller particles, resulting in a more uniform and finer particle size distribution. In addition, the presence of nanoparticles can also improve the dispersion of the PVC particles, leading to a more homogeneous nanocomposite structure. This can result in improved mechanical properties, such as increased stiffness and strength, as well as enhanced thermal and chemical resistance. Overall, the addition of nanoparticles to PVC can improve its properties and performance, including reducing particle size and improving particle dispersion. Since pure PVC with larger primary particles needs a more temperature and time to melt, so the probability of its degradation is high, compared to PVC/LDH and PVC/LDM-MPTMS samples, because they have a lower time and process, they have more suitable mechanical and degradation properties, which is commercially viable, therefore, it can be economical. As explained in a previous work,
23
in-situ polymerization in the presence of nano-particles creates a 3D network of finer primary particles which are collapsed more easily than that formed in conventional polymerization. From the results of the present work and the data given in the previous work,
23
it can be concluded that one of the important parameters in determining the temperature processing of thermal-sensitive PVC grains is morphology. Torque/time curve for PVC samples prepared in a Brabender® Plasticorder.
Dehydrochlorination Rate
The effects of LDH and its modification on the dehydrochlorination rate of PVC grains prepared by in-situ polymerization are illustrated in Figure 3. In PVC/LDH-MPTMS composites, it is observed up to two times less than pure PVC resins, also these composites show better thermal stability and longer discoloration time than PVC/LDH. Very important is the fact that the PVC nanocomposites have a significant effect on the fusion behavior of the PVC compound so it needs a very short time to reach the maximum torque in the fusion plot. Commercially, this would have a significant influence on the degradation and mechanical properties as PVC resin with larger primary particles in the pure PVC requires a higher temperature and/or processing time to melt and, subsequently, is more likely to degrade. As mentioned in earlier work,
24
in-situ VCM polymerization in the presence of a nanoparticle provides a three-dimensional network of finer primary particles that are destroyed more readily than that formed under normal conditions. From these results and those reported in earlier work,
25
it can be concluded that morphology is an important parameter in determining the temperature processing of thermal-sensitive PVC grains. Generally, the presence of nanoparticles makes the nucleation and nanodomains of primary particles increase and 3D network of primary particles happens in an earlier state of polymerization. This phenomenon is more pronounced for MPTMS intercalated LDH than pure LDH. The dehydrochlorination rate for PVC samples.
This superiority may be due to chemical contribution and microstructure. The exfoliation extent of LDHs in PVC/LDH-MPTMS was significantly higher than that of PVC/LDH. Moreover, PVC/LDH-MPTMS due to having MPTMS molecules a superior and intrinsic HCl absorption capability compared to PVC/LDH within the process of dehydrochlorination. In general, it can be also noted that the dehydrochlorination rate of LDH-contained PVC composite is lower than pure PVC because LDH and LDH-MPTMS have some un-reacted hydroxyl groups in their molecular structures, which can react with the released HCl molecules from degradation, thereby resulting in further increase of the thermal stability time.
Thermal Stability and Mechanical Properties
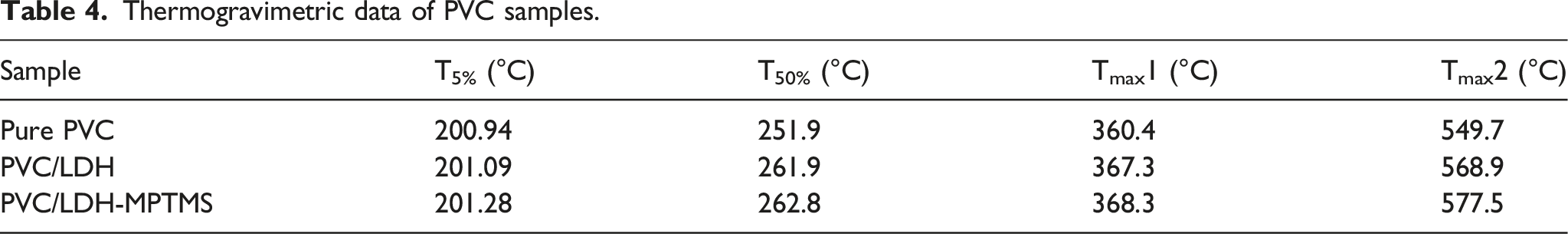
Figure 4 shows the TGA and DTG curves for the synthesized samples. The data obtained from this curve are the onset of degradation (T5%), half decomposing temperature (T50%), and maximum decomposition temperature (TMax), and are summarized in Table 4. These data clearly show that the three temperatures introduced above in composites containing LDH are higher than pure PVC. In three tested samples, PVC/LDH-MPTMS composite has higher TMax than the other two samples. From these data, it can be concluded that the presence of LDH can improve PVC resistance to thermal degradation. This thermal stability can be described by the good dispersion of LDH particles, especially LDH-MPTMS in the PVC matrix, the good interfacial adhesion between PVC and LHD particles, as well as the synergistic effect between LDH and MPTMS, because the MPTMS have double bond could act as a start point in polymerization process. When LDH modified with MPTMS, the grain PVC chain cause the nano sheet to intercalate and consequently nano composite properties improved. Thermogravimetric (TGA) curves and DTG thermograms of the PVC samples. Thermogravimetric data of PVC samples.
The good dispersion of LDH nanosheets in the PVC matrix can enhance the immobilization of the chains; LDH fillers act as physical barriers that can prevent mass and heat transfer, and on the other hand, MPTMS can catalyze charring, which leads to increasing the stability of the residue polymer chains. Consequently, by protecting the underlying PVC, the thermal stability of the PVC Composite increases.
Dynamic mechanical properties
Variations of storage modulus with temperature for PVC and PVC composites are given in Figure 5. The results showed that the storage modulus of pure PVC in the temperature range of 120°C is lower than PVC composites containing LDH. At lower temperatures, the storage modulus of the PVC/LDH and PVC/LDH-MPTMS samples are, respective, about 7% and 12.5% more than that of the pure PVC sample. It confirms that well-dispersed LDH and, especially LDH-MPTMS, particles stiffen the PVC matrix. Nevertheless, when using pure LDH as a nanoparticle in in-situ polymerization (PVC/LDH), the stiffening effect is gradually reduced with the increasing temperature, most likely due to the agglomeration form of the LDH particles. Evolution of storage modulus with temperature of PVC samples.
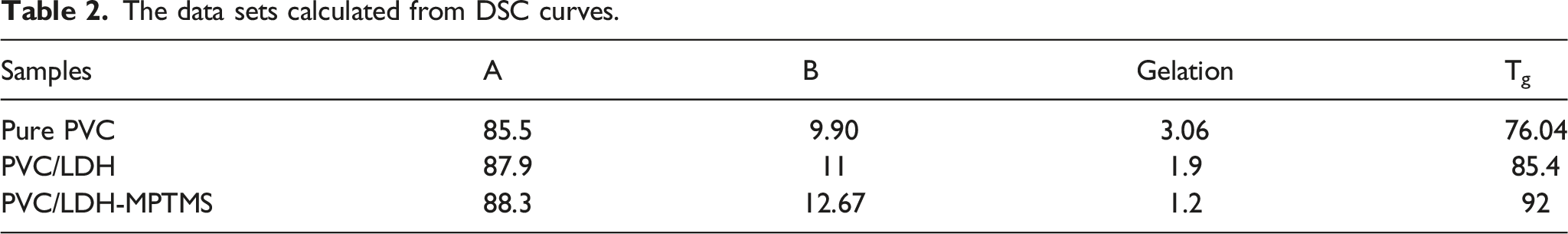
The loss factor (tanδ), a criterion of the damping ability of a polymer material, against temperature is plotted in Figure 6. The values of glass transition temperatures, Tg, of the pure PVC and LDH-filled-PVC nanocomposites, are summarized in Table 2. The Tg peak of PVC in LDH-filled-PVC composites tends to shift towards larger temperature, especially when the LDH particles intercalation and well-dispersion are increased. Eventually, these results support the fact that well-dispersed nanoparticles limit the chain segmental motions which is more pronounced in the case of PVC/LDH-MPTMS. In the case of PVC/LDH, however, agglomeration of LDH particles weakens the contact and, subsequently, the interaction between the chains of PVC and the nanosheets in such a way the Tg declines to 87.9°C which is lower than PVC/LDH-MPTMS Changes of tanδ with temperature of PVC samples.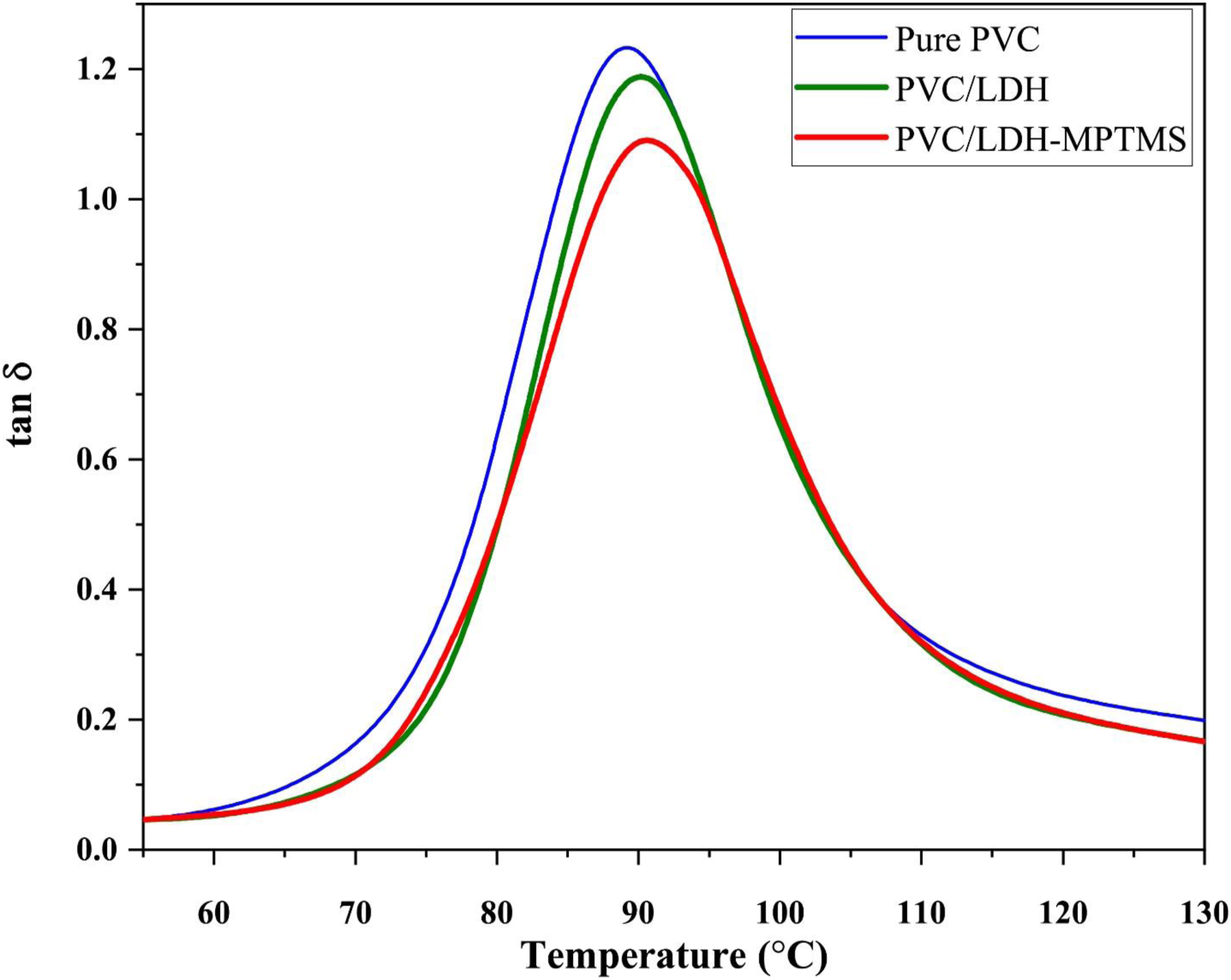
Figure 7(a) indicates the tensile stress–strain behavior of PVC samples. As can be seen in the figure, pure PVC exhibits brittle behavior, but when LDH particles are incorporated in the PVC matrix during in-situ polymerization, the behavior caused by composites is a ductile behavior, such as necking and stress whitening. The mechanism by which LDH change the brittle behavior of PVC to ductile behavior is known as the “particle-induced plasticity” mechanism. When LDH are added to a PVC matrix, they create regions of stress concentration at their interfaces with the polymer chains. These regions of stress concentration act as nucleation sites for the formation of shear bands, which are regions of localized deformation in the polymer matrix. As a result, the addition of LDH increases the mobility of the PVC chains and enhances the plasticity of the material. This leads to a transition from brittle to ductile behavior in the PVC. The exact mechanism by which nanoparticles induce plasticity depends on the size, shape, and surface chemistry of the particles, as well as the nature of the polymer matrix. Their tensile yield strength, young’s modulus, elongation at break (λmax), and Charpy notched impact strength are evaluated and plotted in Figures 7(b)–(d). Mechanical analysis of PVC samples (a) Stress-Strain curve (b) Young’s modulus value (c) elongation at break value (d) value of Charpy notched impact strength.
In Figure 7(b), Young’s modulus and yield strength of the PVC composite is more than pure PVC and it shows that the module increases with the loading of LDH particles and this is more pronounced for LDH-MPTMS. The presented results show that LDH nanoparticles lead to the hardening of PVC.
The large interfacial areas and subsequent strong interaction between LDH particles and PVC matrix achieved by the in-situ polymerization process of VCM in the attendance of LDH and especially, LDH-MPTMS causes to much higher λmax, in composite samples as shown in Figure 7(c).
Figure 7(d) shows the variation of Charpy notched impact strength of PVC samples, prepared by the in-situ suspension polymerization method. The impact strength of the PVC/LDH-MPTMS composite is higher than the PVC/LDH and pure PVC.
Conclusions
In the present work, we have prepared two types of PVC nanocomposites named PVC/LDH and PVC/LDH-MPTMS by using the in-situ suspension polymerization method. The results indicate enhanced thermal stability with the incorporation of LDH and especially LDH-MPTMS into the PVC matrix. The rate of dehydrochlorination in comparison with the pure PVC and the PVC/LDH composite. The weight loss temperatures (T5%, T50%) for pure PVC experienced an increase after composite with LDH and modified LDH. The storage modulus of the glassy state and Tg of the PVC/LDH-MPTMS composites obtained from DMA analysis are larger than that of the pure PVC and the PVC/LDH composite that can approve the stiffening and toughening of PVC by LDH nanoparticles. The tensile strength, young’s modulus, and Charpy notched impact strength have the maximum amount for PVC/LDH-MPTMS, among the three prepared samples, including pure PVC and the PVC/LDH composites.
Footnotes
Author contribution
Reza Mohammadi Berenjegani, Reza Darvishi and Ghasem Payam contributed to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data availability statement
All findings and data supporting figures included in this paper are available from the corresponding author upon reasonable request.
