Abstract
A heterostructure of diamagnetic magnesium–aluminium layered double hydroxides (Mg–Al LDHs) and photomagnetic cobalt–iron Prussian Blue analogue (Co–Fe PBA) was designed, synthesized and then designated as
Keywords
1. Introduction
As the most promising groups of molecule-based magnets, PBAs are cyano-bridged three-dimensional face-centred cubic structures, which have received much attention due to their various interesting properties. For example, high TC value magnets of V[Cr(CN)6]086 2.8H2O (TC = 315K), [1] electrochemically switchable magnets of [Cr212(CN)6] 2.8H2O [2] and magnetic pole inversion in (NixIIMn1-xII)15[CrIII(CN)6] [3, 4] have been reported. Furthermore, photoswitchable magnetic compounds have especially been developed after first discovering photo-induced magnetization in the cobalt–iron PBA (Co–Fe PBA), [5] which is derived from the electron transfer of FeII to CoIII ions upon visible light illumination at low temperature. [6]
Moreover, magnetism on low-dimensional cyanide-bridged compounds has also attracted much attention due to limited magnetic interaction compared to the three-dimensional network. For example, a cyanide-bridged Co–Fe chain displaying magnetic and electric bistabilities arising from electron-transfer-coupled spin-transition, [7] a series of cyanide-bridged Co–Fe squares exhibiting two-step charge-transfer-induced spin-transition in the solid state, [8] and a layered Ni–Cr bimetallic compound expressing metamagnetic behaviour from the antiferromagnetic to the ferromagnetic phase under an applied field of 1,200 Oe [9] have been reported. As seen, various approaches, such as capping stronger ligands [10] or using the Langmuir–Blodgett technique, [11] have been applied in order to explore the properties of low-dimensional cyanide-bridged magnetic compounds.
On the other hand, layered double hydroxides (LDHs) are inorganic layered compounds consisting of positively-charged brucite-like host layers and charge-balancing interlayer anions. The general formula is [MII1-xMIIIx(OH)2, (An−)x/n.mH2O], where MII and MIII are respectively divalent and trivalent metal ions, while An− are interlayer anions with water molecules located interlayer. LDHs have been widely studied owing to their application in catalysts, anion exchangers, separation, electrochemistry, biotechnology, photochemistry and so on. [12-16] Recently, the magnetic properties of LDHs have been reported. [17-19] For example, the magnetism of Ni–Mn LDH has shown competing interactions: in-plane (ferromagnetic) and out-of-plane (antiferromagnetic). The spontaneous magnetization at low temperature was observed in Ni–Cr and Ni–Fe LDHs. [17-20] Ni–Mn LDH has shown that the magnetism was driven by the competition between the ferromagnetic in-plane (due to Ni ions spin state) and the antiferromagnetic inter-plane. [21]
Intercalation of molecule-based magnetic materials into LDH layers might realize novel magnetic materials with a low-dimensional structure. So far, the insertion of Mn–Cr oxalate-bridged molecule-based magnets into Zn–Al LDH hosts has exhibited ferrimagnetic ordering due to the presence of dominant antiferromagnetic interactions between metallic centres. [21] Moreover, the growth of two-dimensional ferromagnetic Ni–Cr PBA into diamagnetic Zn–Al LDH has exhibited long-range magnetic ordering. [21] In addition, the controlled growth of M–Fe PBA (M = Ni, Co) into diamagnetic Mg–Al LDH and magnetic Ni–Al LDH has exhibited spin-glass behavior. [22]
Therefore, in order to understand the photomagnetic properties of low-dimensional Co–Fe PBA, it was two-dimensionally intercalated into the MgAl LDH template by using the stepwise anion exchange method. The obtained compounds were characterized by X-ray diffraction (XRD), infrared (IR) spectroscopy, 57Fe Mössbauer spectroscopy, scanning electron microscopy (SEM) and magnetic measurement.
2. Experimental Section
2.1 Synthesis
All reagents used were of analytical grade and not further purified. Potassium hexacyanoferrate (III), magnesium, aluminium, ammonium and cobalt nitrate, urea and ethylene glycol were purchased from Wako. In order to minimize the contamination of carbonate ions, Milli-Q water and ethylene glycol were bubbled with nitrogen gas before the experimental process. The nitrogen flow was continued for 30 minutes at room temperature. In order to prepare the

The intercalation process of CoFe PB into LDH. The first step was a synthesis of LDH containing nitrate anions by the homogeneous precipitation method. The second step was the replacement of the nitrate anions (NO3−) with hexacyanoferrate (III) anions ([Fe(CN)6]3−) by the anion exchange method. The last step was the formation of interlayer CoFe PB in a two-dimensional network.
2.1.1 MgAl–NO3 LDH (LDH–NO3)
The white product MgAl–NO3 LDH was synthesized in line with the homogeneous precipitation method. [18] Typically, 0.33 M of magnesium nitrate hexahydrate (Mg(NO3)2.6H2O), 0.165 M of aluminium nitrate (Al(NO3)39H2O), 1.65 M of urea and 1 M of ammonium nitrate (NH4NO3) were dissolved in 100 ml of Milli-Q water. The mixed solution was refluxed for one day under continuous magnetic stirring and nitrogen protection. The resulting precipitate was centrifuged, washed with Milli-Q water and anhydrous ethanol a few times, and finally dried in a vacuum at room temperature (
2.1.2 Intercalation of the hexacyanoferrate (III) anion (LDH–Fe(CN)6)
The intercalation process was carried out by the anion exchange method. [19] 0.25 g of
2.1.3 Formation of Co–Fe PBA in the LD Hinterlayer (LDH–PB)
113 mg of
2.2 Physical measurements
Structural information was characterized by an X-ray powder diffraction (XRD) and Fourier transform infrared spectroscopy. XRD patterns were collected on a D8 Discover (Bruker) using Ni-filtered Cu Kα radiation (1.5418 Å, 40 kV and 40 mA). The data were collected with a step size of 0.01° in the 5°< 2θ< 75° range. The counting time was 0.42 °/min. Infrared spectra were recorded with an FT-IR 660 Plus spectrometer (JASCO, Japan) in the 4,000–400 cm− 1 range using powdered samples diluted in KBr pellets. The 57Fe Mössbauer spectra were measured using a Wissel MVT-1000 Mössbauer spectrometer with a 57Co/Rh source in the transmission mode. Surface morphology was examined with a field emission scanning electron microscope (FE-SEM) using SIRION(FEI). The homogeneous nature of the hybrid materials and their morphology were studied at a voltage of 5 keV. Elemental maps were obtained using an energy-dispersive X-ray spectrometer (EDX). Magnetic measurements were performed with a superconducting quantum interference device (SQUID) magnetometer (Quantum Design MPMS-XL-5). The susceptibility data were recorded in the temperature range of 2–20 K at a constant field of 100 Oe for zero-field-cooled (ZFC) and field-cooled (FC) curves, as well as under the magnetic field range of −5 T – 5 T at 2 K. The Xe lamp was used as the light source. The samples were put onto transparent tape, which was then surrounded with light fiber. The samples were illuminated at 5 K with the filtered visible light for one day.
3. Results and Discussion
3.1 Characterization of LDH–NO3 and LDH–Fe(CN)6 during the intercalation process
Figure 1A showed XRD patterns of
The powder XRD data and calculated parameters of the compounds (a) LDH–NO3, (b) LDH–Fe (CN)6, and (c) LDH–PB. BS is basal spacing.


(A) XRD patterns of (a)
Figure 1B shows the IR spectra of
SEM images of the
3.1.1 Characterization of LDH–PB
XRD patterns showed that the diffraction peaks at 8.21° (003), 16.47° (006), 24.80° (009) and 60.81° (110) in
Furthermore, an SEM image of
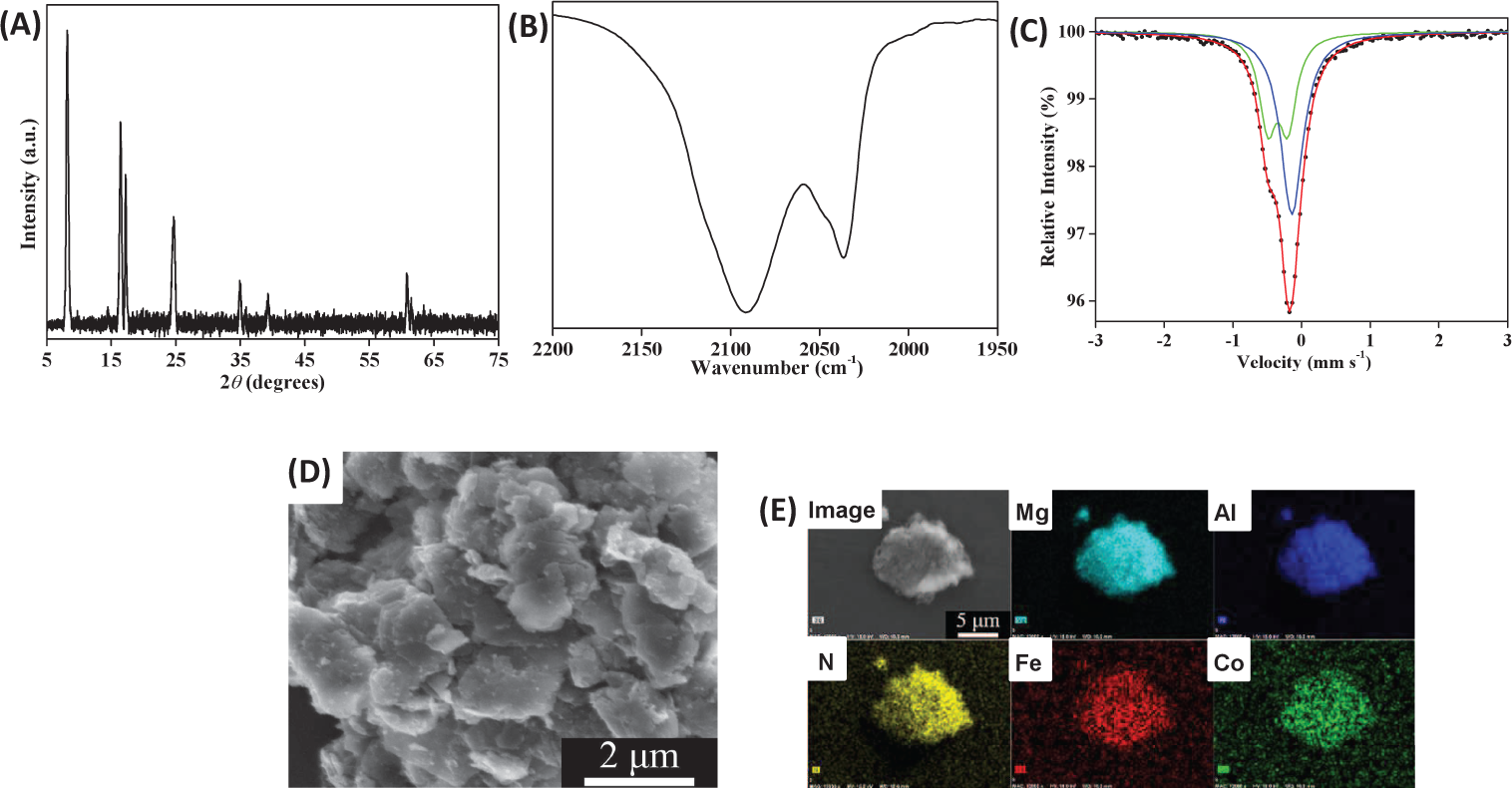
Characterizations for
In order to compare the properties,
3.1.2 Magnetic properties of LDH–PB
Figure 3A shows the ZFC and FC curves of
3.1.3 Photomagnetic properties of LDH–PB
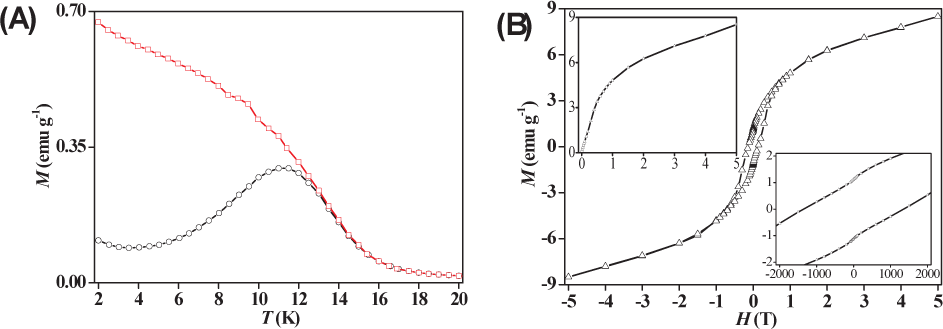
(A) ZFC (black circle) and FC (red square) magnetization curves versus temperature in the region from 2 K to 20 K under a magnetic field of 100 Oe for

Photomagnetic properties for
4. Conclusion
Co–Fe PBA was intercalated into what seems to be a two-dimensional framework of the Mg–Al LDH template by the stepwise anion exchange method. The obtained
Footnotes
5. Acknowledgements
This work was supported by the China Scholarship Council (C.-J. Z.).
